RADR 1213 rod exam 1( principles of radiographic imaging)/ spring
1/90
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
91 Terms
An electron is ____ charged and has a mass of ____ amu(s).
A)negatively; 1.673 10−27
B)negatively; 0.000548
c)neutral; 1
D)positively; 0.000548
B) negatively; 0.000548
The unit of mass is the
A.megabyte.
B.kg.
C.liter.
D.cc
B)kg
The orderly arrangement of elements in the periodic table is based upon
A.atomic weights.
B.mass number.
C.neutron number.
D.atomic number.
Atomic number
Photon wavelength is
A.inversely proportional to photon velocity.
B.directly proportional to photon frequency.
C.inversely proportional to photon frequency.
D.is not related to frequency.
C.inversely proportional to photon frequency.
All of the following are true of electromagnetic energies EXCEPT:
A.They are illustrated as sinusoidal waves.
B.They possess both wave and particle characteristics.
C.The relationship between frequency and wavelength is direct and proportional.
D. They are arranged in an orderly spectrum according to frequency and wavelength.
C.The relationship between frequency and wavelength is direct and proportional.
The maximum number of electrons found in any energy level (shell) at any point in time is calculated by the formula
N = Z x e2.
W = F D.
N = amu Z.
2n2
2n2
The sum of protons and neutrons in a nucleus is called the
A.atomic mass unit.
B.atomic weight.
C.quantum number.
D. mass number
D. mass number
Groups of the periodic table?
A.represent elements with the same outer electron configuration
B.are horizontal.
C.represent elements with the same atomic density.
D.all have the same electron binding energies.
A.represent elements with the same outer electron configuration
Carbon has an atomic number of 6. One of its isotopes has a mass number of 14. The number of neutrons in this isotope is
6.
8.
14.
19
8
A period in the periodic table is
A.represented by a column.
B.vertical.
C.the principal quantum number.
D.determined by the valence electrons.
C.the principal quantum number.
In the excitation process, electrons in an atom are moved to a/an ____ energy state.
lower
higher
incomplete
ionization
higher
The maximum number of electrons in a period with a principal quantum number of 4 is
6.
8.
18.
32.
32
Isotopes have ____ mass numbers and ____ atomic numbers.
A) the same; the same
B) the same; different
C) different; the same
D) different; different
c. different; the same
In a vacuum, electromagnetic radiation
A. has a velocity equal to the speed of light.
B. x-ray energy may be bent by a crystalline lens.
C. causes ionizations of air molecules.
D.is altered by a strong magnetic field.
A.has a velocity equal to the speed of light
If the photon frequency of electromagnetic radiation is increased tenfold, then the
A.velocity will increase times 10.
B. velocity will decrease to 10.
C.wavelength will increase times 10.
D.wavelength will decrease to 1/10.
D.wavelength will decrease to 1/10.
The atomic number is the number of ____ contained in the nucleus.
A.neutrons
B.electrons
C.protons
D.positrons
C.protons
An isotope of boron has 5 protons and 6 neutrons. The atomic number of boron is
11.
10.
6.
5
5
An element with an atomic number of 22 has how many electrons in the second principal quantum number level (L shell)?
22
10
8
2
8
The nuclear components that distinguish one element from another are the
A.neutrons.
B.protons.
C.gamma particles.
D.beta particles.
B.protons
An atom has an atomic number of 18 and a mass number of 38. Letting P = the number of protons and N = the number of neutrons, which of the following atoms is the isotope of this atom?
P = 18, N = 20
P = 18, N = 21
P = 20, N = 18
none of the above
P = 18, N = 21
According to the "rule of octets,"
eight energy levels have electrons.
eight protons occupy the outermost shell.
the valence shell is chemically inert.
both a and c.
both a and c.
Which of the following is not a basic characteristic of the wave equation?
velocity
wavelength
frequency
energy
energy
If an electron is gained or lost from an atom, that atom becomes
an ion.
a new element.
an isotope.
radioactive
an ion
All of the following are energy forms EXCEPT
sound.
thermal.
electrical.
nuclear.
Sound
Quarks may behave according to the ____ theory.
string
quantum
octet
electromagnetic
String theory
All of the following are true of the substance glucose (C6H12O6) EXCEPT:
The smallest subdivision of this molecule is glucose.
It is a compound.
It consists of three elements.
It may be broken down into one atom of glucose.
.The smallest subdivision of this molecule is glucose.
When comparing iodine (Z = 53) with barium (Z = 56),
barium has a lower k-shell binding energy.
iodine has more neutrons.
iodine has five (5) orbitals with electrons.
barium has fewer proton
iodine has five (5) orbitals with electrons.
All of the following are related except one. Select the choice that is NOT RELATED.
a source of electrons
a vacuum
thermionic emission
a large potential difference
a large potential difference
The function of the cathode is to produce
a focal point.
an electrical connection to the rectifier.
a thermionic cloud.
a grounding effect.
a thermionic cloud
All of the following are part of the cathode assembly EXCEPT
filament.
stator.
focusing cup.
associated wiring.
stator
All of the following are preferred cathode filament materials EXCEPT
copper.
tungsten.
rhenium.
molybdenum.
Copper
Filament materials are chosen primarily for their
atomic number.
high melting points.
ability to vaporize easily.
voltage tolerance.
high melting points
Which of the following would affect the production of a thermionic cloud?
filament diameter
filament length
filament temperature
all of the above
All of the above
A filament may break due to
evaporation.
deposition.
Vaporization
Vaporization
Driving the thermionic cloud from the cathode to the anode requires
a grid biased anode.
a small voltage.
anode rotation.
a small amperage.
a small amperage
In order to time the x-ray exposure with rapid inspiration on an infant,
three-phase, high-frequency generators are preferred.
the exposure button and prep are pressed simultaneously.
exposure may be initiated as fast as 0.10 sec.
pulsating DC current should pass through the x-ray tube.
three-phase, high-frequency generators are preferred.
The cathode focusing cup has a/an ____ charge.
neutral
isomeric
positive
negative
negative
As more and more electrons build up in the area of the filament, their negative charges oppose the emission of additional electrons. This phenomenon is known as
thermionic emission.
biased cloud formation.
space charge effect.
current saturation.
space charge effect
X-ray photon production occurs at the
anode.
cathode.
filament.
stator windings.
anode
Tungsten is the material of choice for the source of x-ray photons primarily because of its
high atomic number.
low melting point.
low mass number.
G-force tolerance.
high atomic number
Pitting of the anode commonly occurs
when the unit is not warmed up properly.
when the anode is off-focus.
with extended use.
when the kilovoltage is too high.
with extended use
Melting of the anode commonly occurs
when the unit is not warmed up properly.
with extended use.
when the kilovoltage is too high.
when the stator fails and the rotor ceases to turn..
when the stator fails and the rotor ceases to turn
Cracking of the anode typically occurs
with extended use.
when the anode is off-focus.
when a cold anode is hit with a high kVp exposure.
when the cathode cup touches the anode, causing a short.
When a cold anode is hit with a high kVp exposure
The exact point on the anode where the x-ray beam emanates is the
focusing cup.
focal spot.
anode heel.
rotor.
focal spot
The physical area of a rotating anode focal track, affected by the cathode electron beam, is the
effective focal spot.
effective focal track.
actual focal spot.
projected focal track.
actual focal spot
The area of the focal spot that is projected out of the tube toward the object being radiographed is the
effective focal spot.
line focus.
actual focal spot.
thermionic stream.
effective focal spot
The actual focal spot is controlled by the
thickness of the anode disc.
length of the filament.
anode rotation speed.
angle of the cathode focusing cup.
length of the filament
The most common diagnostic radiography x-ray tube target angle is ____ degrees.
12
24
45
54
12
As the actual focal spot size increases, the effective focal spot size
increases.
decreases.
remains the same.
doubles proportionally
increases
As the angle of the anode increases and the incident electron beam size remains the same, the effective focal spot size
increases.
decreases.
remains the same.
cannot be determined.
increases
The use of the line-focus principle causes a problem known as the ____ effect.
space charge
quantum mottle
anode heel
saturation current
anode heel
The only portion of the anode assembly placed outside the vacuum of the glass envelope is the
stator.
rotor.
anode stem.
molybdenum neck.
stator
The function of the stator and rotor assembly is to
change the anode angle.
rotate the anode.
insulate the anode.
disrupt the electromagnets of the induction motor.
rotate the anode
The protective housing controls leakage and scatter radiation through
evaporation.
vaporization.
absorption.
condensation.
absorption
Insulation of the high-voltage components of the tube and absorption of the heat produced by x-ray production are achieved through the use of
a small fan.
a vacuum.
a stator.
dielectric oil.
dielectric oil
What type of radiation is composed of photons not produced at the actual focal spot?
off-focus
extra-focal
both a and b
remnant
Both A and B
When radiographing a wedge-shaped anatomical structure such as the femur, the thicker portion of the anatomy should be positioned
under the cathode end of the tube.
in the exact center of the beam, as it is most intense in the center.
off center laterally to negate the heel effect.
under the anode side of the tube.
under the cathode end of the tube
Leakage radiation from the lead-lined housing must not exceed
10 mR/hr measured at 40 inches.
100 mR/min measured at the underside of the collimator.
10 mR/min measured at the tube head.
100 mR/hr measured at 1 meter.
100 mR/hr measured at 1 meter
To determine the allowable combination of exposures factors, thus preventing x-ray tube damage,
a tube rating chart is referenced after exposure to check correctness.
the automatic exposure circuit is activated.
an x-ray tube rating chart is checked prior to exposure.
an anode cooling chart is referenced prior to exposure.
An x-ray tube rating chart is checked prior to exposure
All of the following would lengthen x-ray tube life EXCEPT:
minimize anode rotation times and prepping.
avoid sudden movements of the tube head assembly.
use low-mA selections whenever clinically possible.
use high-speed anode rotation on distal extremity studies.
use high-speed anode rotation on distal extremity studies.
What is the function of the filament?
The function of the filament is to produce thermionic emission/source of electrons. The filament is heated up before exposure so its warmed up. While you are exposing the rotor is activated by pressing down the exposure and it produces thermionic emission.
List the three parts of the anode assembly.
anode, stator and rotor
In which direction should the anode be placed for an AP projection of the Abdomen? Why?
Because of the anode heel effect the cathode side of the x-ray tube produces more x-ray photons therefore the anode side of the tube should always be positioned according to the body part you are exposing side that is less thick (easier to move through). Therefore the anode should be placed toward the more superior side of the abdomen. And the cathode towards the thicker side (mid to lower abdomen).
How many heat units are generated by an exposure of 70 kVp, 300 mA, and 0.1 second on a single phased rectified unit?
2100 HU
How many heat units are generated by an exposure of 70 kVp, 400 mA, and 0.1 second on a high frequency unit?
3920 HU
During x-ray production, almost all of the kinetic energy of the incident electrons is converted to
a bremsstrahlung photon.
a characteristic x-ray photon.
light.
heat.
heat
What percentage of target interaction results in the production of x-ray photons?
1 percent
50 percent
99 percent
100 percent
1 percent
The types of target interaction that occur in the production of x-ray photons in the diagnostic range are dependent upon the
incident electron kinetic energy.
nuclear binding energy.
incident electron potential energy.
band gap of the valence electrons.
incident electron kinetic energy
The transfer of the incident electrons' kinetic energy to the outer-shell electrons, causing vibration of the outer-shell electron, results in the emission of
infrared radiation.
ultraviolet radiation.
light.
electricity.
Infrared radiation
The energy of a bremsstrahlung photon is
equal to the kinetic energy of the entering electron.
equal to the kinetic energy of the exiting electron.
the sum of the entering and exiting kinetic energies of the incident electron.
the difference between the entering and exiting kinetic energies of the incoming electron.
the difference between the entering and exiting kinetic energies of the incoming electron.
Bremsstrahlung interactions may occur only when the incident electron interacts with
an inner-shell electron.
an outer-shell electron.
the force field around the nucleus.
the nuclear force within the nucleus.
the force field around the nucleus
Characteristic interactions may occur only when the incident electron interacts with
an inner-shell electron.
an outer-shell electron.
the force field of the nucleus.
the band gap of the atom's valence band.
an inner-shell electron.
During a characteristic interaction, the kinetic energy of the incident electron must be ____ of the electron it knocks from its orbit.
less than that
greater than that
proportional to that
characteristic
greater than that
During a characteristic interaction, the dropping of a higher-energy state electron into a lower-energy state "hole" results in the emission of
a photon of electromagnetic energy.
a high-energy electron.
a low-energy electron.
thermal energy.
a photon of electromagnetic energy.
The energy of a characteristic photon is
equal to the binding energy of the entering electron.
equal to the binding energy of the exiting electron.
the sum of the binding energy of the outer and inner shells between which the electron dropped.
the difference between the binding energy of the outer and inner shells between which the electron dropped.
the difference between the binding energy of the outer and inner shells between which the electron dropped
At the end of the characteristic cascade, the ____ shell is missing an electron.
innermost
K
outermost
characteristic
outermost
The K shell can hold a maximum of ____ electrons.
1
2
6
8
2
X-rays are characterized as
charged particles.
electromagnetic waves of short wavelength.
electromagnetic waves with high frequencies.
both b and c.
Both B and C
The emission spectrum of an x-ray beam from a tungsten anode consists of
a characteristic spike at 69 keV.
a significant portion due to bremsstrahlung interactions.
a mixture of brems and characteristic interactions.
all of the above.
All of the above
Electrons traveling from the cathode filament to the anode are called
incident electrons.
primary electrons.
scatter electrons.
secondary electrons.
incident electrons
When comparing the x-ray emission spectrum between beams produced at 70 kVp and 110 kVp,
the 110 kVp beam would demonstrate less brems and a higher characteristic spike.
the 110 kVp beam would demonstrate more brems and a higher characteristic spike.
the 70 kVp beam would demonstrate less brems and a higher characteristic spike.
the 70 kVp beam would demonstrate an equal amount of brems and characteristic radiation.
the 110 kVp beam would demonstrate more brems and a higher characteristic spike.
When the x-ray emission spectra between single- and three-phase generators are compared,
three-phase beams would have a higher average energy.
single-phase beams would be more intense.
less exposure would be required for the same part thickness, using single phase.
the keV of the characteristic spike would be significantly less with the single-phase generator.
Three phase beams would have a higher average energy
Tungsten is the anode material of choice for x-ray production because of its
ability to expand with high speed rotation G-forces
ability of liberating electrons as secondary x-ray sources
creation of off-focal radiation
excellent k-edge value for x-ray production
excellent k-edge value for x-ray production
Molybdenum is an excellent target material for mammography studies due to its
light composite weight during rotation and quite operation.
excellent heat tolerance with high kVp exposures used in mammography.
k-edge production of characteristic radiation, to match soft tissues of the breast.
ability to suppress brems x-ray production and heighten characteristic production.
k-edge production of characteristic radiation, to match soft tissues of the breast.
Draw the Brems Interaction on your scratch paper and turn it in.
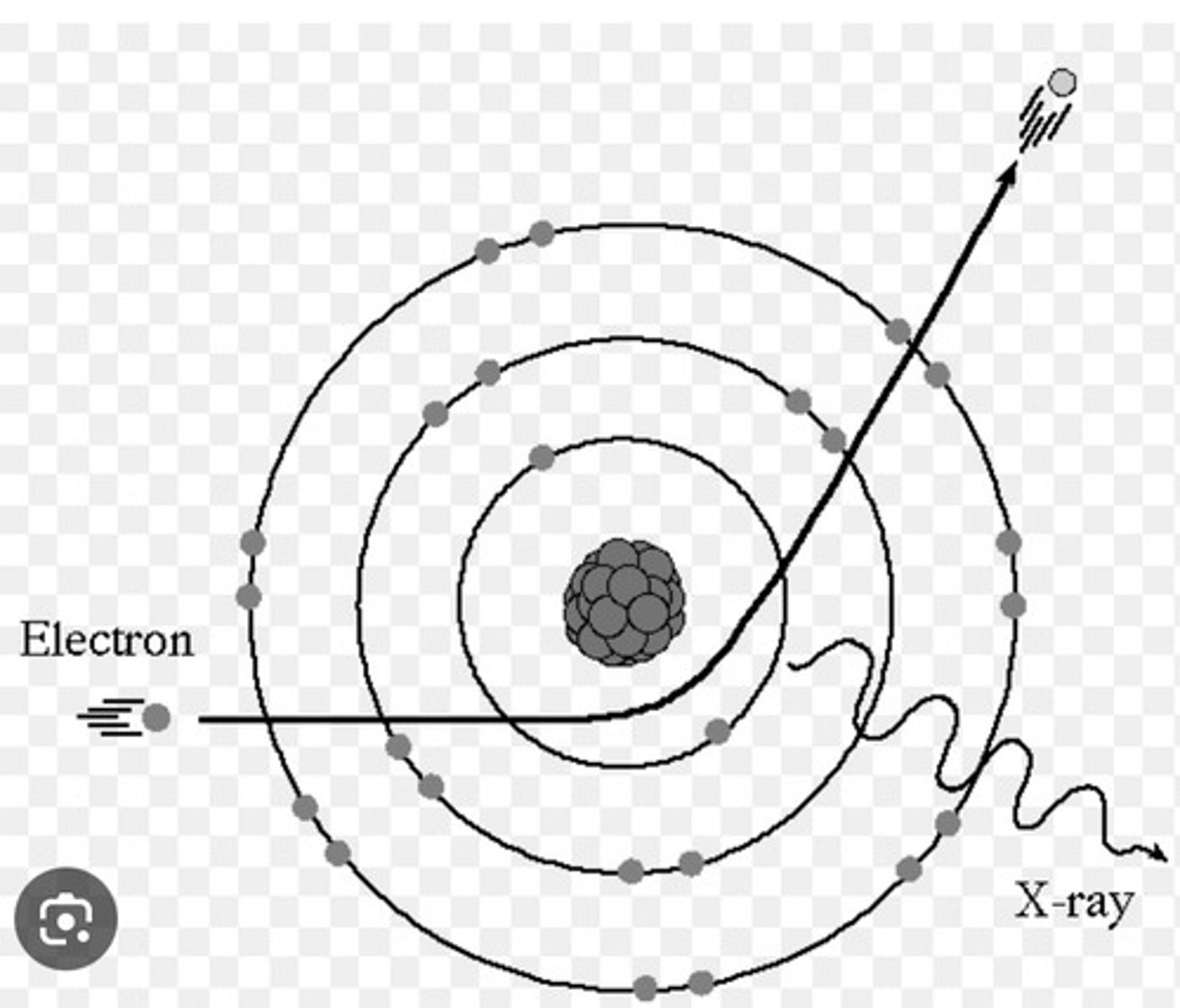
Draw the Characteristic Interaction on your scratch paper and turn in. Be specific.
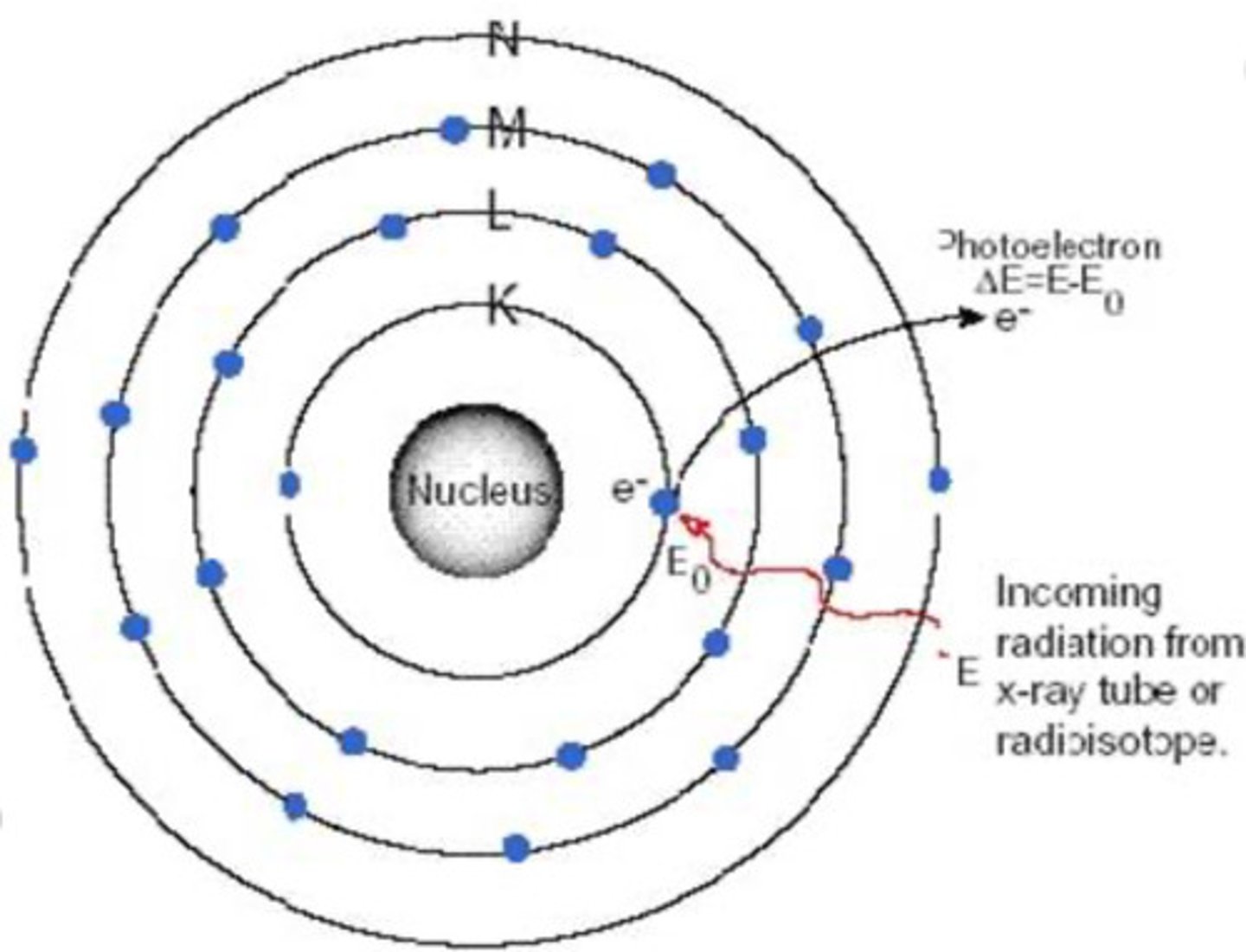
Draw the EM Spectrum showing the correlation between wavelengths and frequencies on your scratch paper.
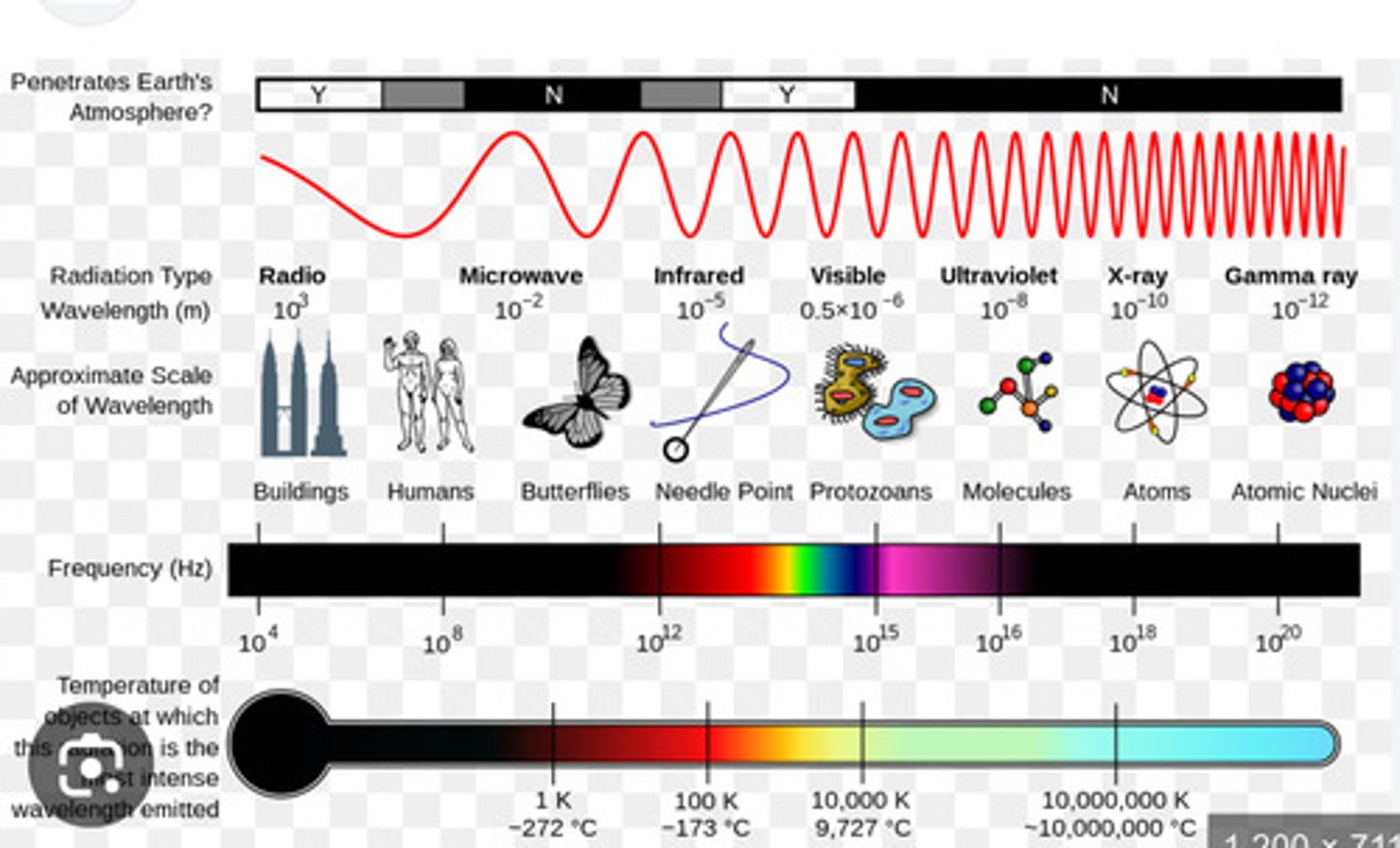
What is the function of the filament?
The function of the filament is to produce thermionic emission/source of electrons. The filament is heated up before exposure so its warmed up. While you are exposing the rotor is activated by pressing down the exposure and it produces thermionic emission.
List the three parts of the anode assembly.
anode, stator, and rotor
How many heat units are generated by an exposure of 70 kVp, 300 mA, and 0.1 second on a single phased rectified unit?
2,100 HU
How many heat units are generated by an exposure of 70 kVp, 400 mA, and 0.1 second on a high frequency unit?
3920 HU