Matter & Energy
Matter:
- Anything that has mass and takes up space.
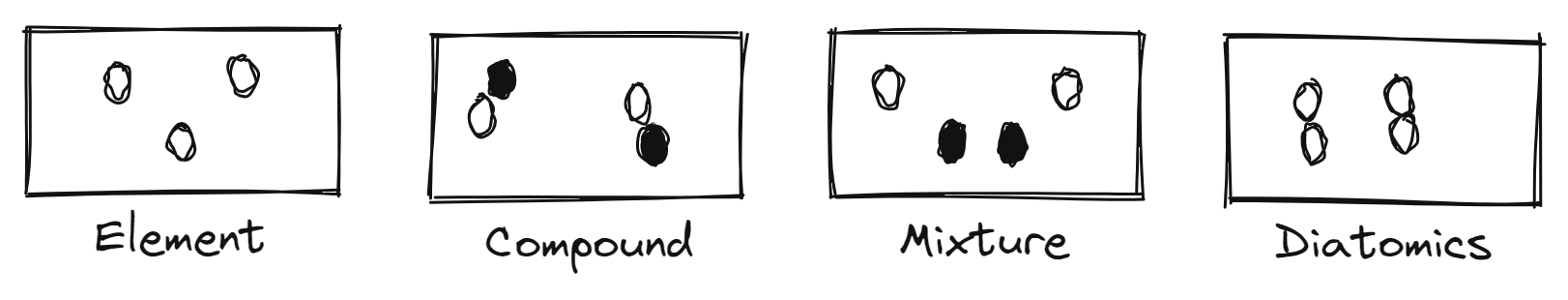
Pure Matter
- substance * definite
- elements/atom type * cannot be broken down
- compounds (2+ atoms) * can be broken down
Impure Matter
- mixtures * indefinite * Homogenous * uniform * Heterogeneous * not uniform
Models
 Properties
Properties
- Physical * 5 senses * mass * solubility * density * volume * odor * phase
- Chemical * behavior * flammability * acidity * radioactivity
Separating Mixture
- Filtration * heterogenous * different particle size

- Distillation * homogenous * different boiling point
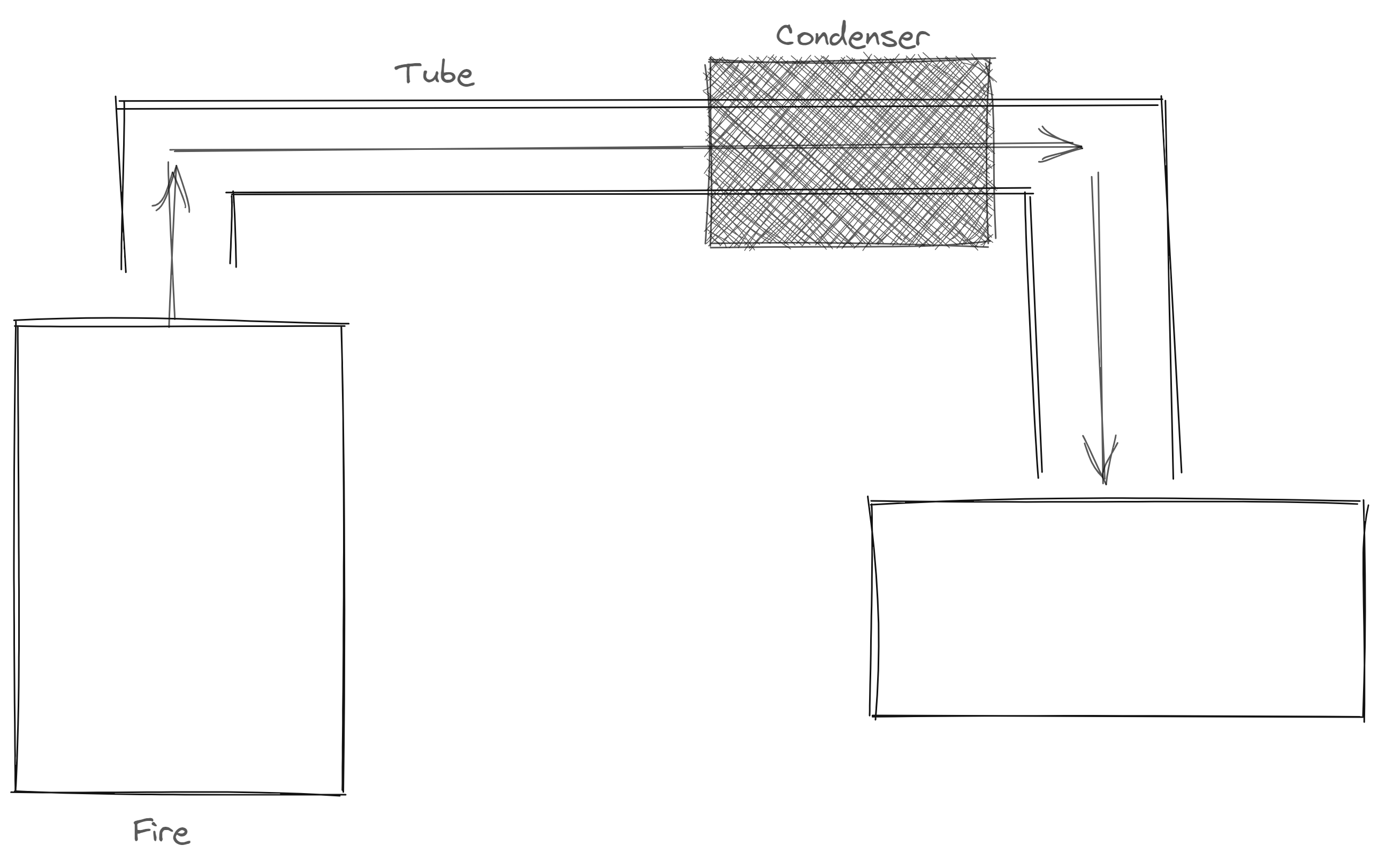
- Chromatography * different molecules in a mixture * solubility

Phases of Matter
- Solid * definite volume & shape * crystal & lattice structure
- Liquid * definite volume * no definite shape
- Gas * chaotic
- no definite shape & volume
Energy
- ability to do work
- The Law of Conservation of Energy: energy cannot be created nor destroyed.
- endothermic - absorbed
- exothermic - released
Temperature
- Q=MCΔT * Q - Heat (Joules) * M - Mass * C - Specific Heat * Water: 4.18 J/g*k * ΔT - Change in Temperature * final temp minus initial temp (Tf - Ti)
- High Temperature → More Kinetic Energy → More Movement
- Heat travels from High → Low
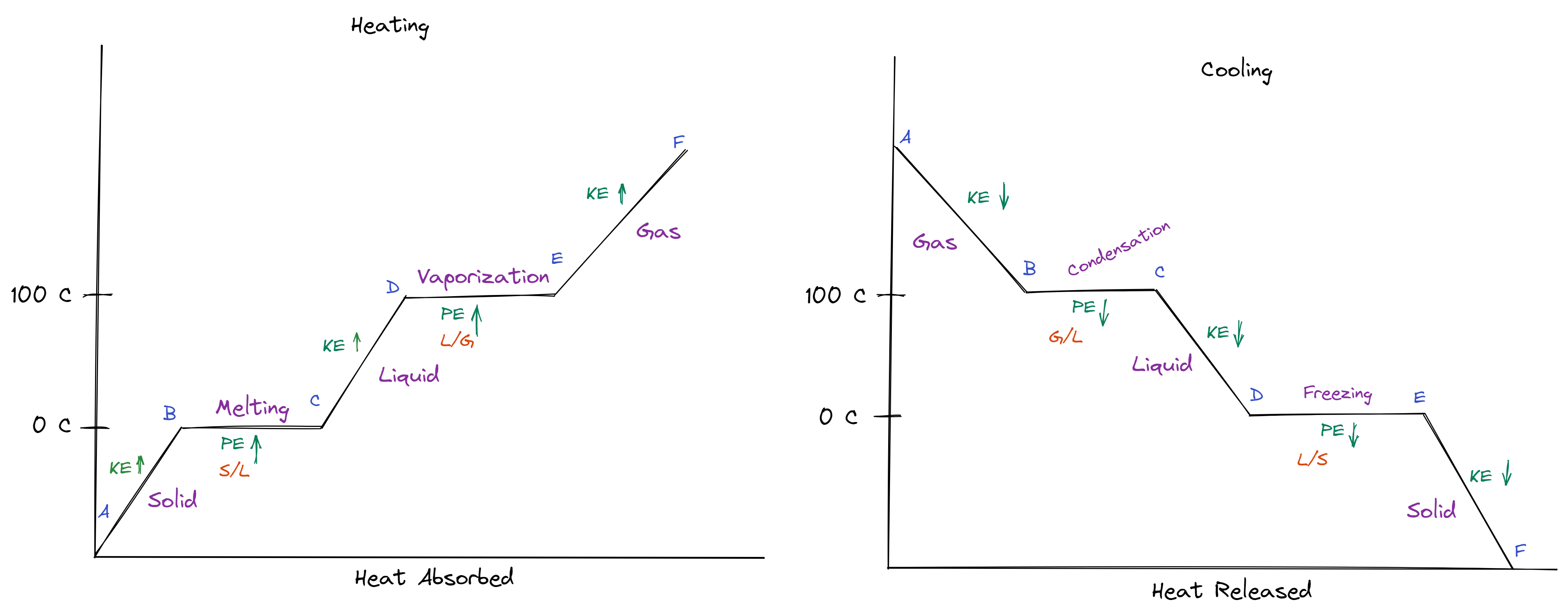
Heating & Cooling Curve
- Heating: endothermic * Heat of Fusion: Melting * Q=MhF * hF = 334 J/g * Heat of Vaporization: Vaporization * Q=MhV * hV = 2260 J/g
- Cooling: exothermic
- Diagrams:
Changes
- Physical * change in appearance * does not change in identity * phase changed (solid → liquid → gas)
- Chemical * identify
\