Bio 110 Exam 2 Matkin
1/234
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
235 Terms
What is a enzyme?
Part of catalysts in living organisms which help speed up chemical reactions
what is ribozymes?
catalytic RNA
what does enzyme made up of?
protein catalysts
why are catalysts necessary to speed up a reaction?
enzyme helps stretching or straining of the bonds so the 2 molecules can interact with each other
activation energy
energy required to start a reaction by breaking bonds in the reactant molecules
transition state
unstable state where bonds are stretched (activation energy lowered)
how to overcome activation energy?
heat and enzymes by stretch and strain
what type of reaction is burning glucose?
exergonic reaction
what are the products of burning glucose?
CO2 and H2O
does burning of glucose required additional energy?
no
how does enzyme speed up chemical reaction?
by lowering the activation energy
does enzymes affect free energy?
no
enzyme only speed up reactions that would occur without them
true
how does enzyme lower activation energy?
1. positioning reactants together to facilitate bonding
2. straining bonds in reactants to achieve transition state
enzymes are ________ for the reactions they catalyze
specific
substrates
The reactants that bind to the enzyme
active site
Region on enzyme where substrate binds.
enzyme + substrate =
enzyme-substrate complex
results from the fit between the shape of the active site and the substrate
specificity
enzyme only bind with the right shape and fit with the substrate
lock and key metaphor
can enzyme changes shape? why?
yes, because of the chemical interactions with the substrate.
induced fit
confirmational changes and bring chemical groups of the active site together due to enzyme and substrate
catalyst reaction
stretching and straining on the bond
steps of an enzyme-catalyzed reaction
1. Substrate Binds
2. enzyme undergoes conformation change that binds the substrates more tightly
3. substrates are converted to products
4. products are released
affinity
attraction of enzyme
enzyme with high affinity
conduct catalyst
enzyme with low affinity
more substrate needed
velocity
amount of product produced per second
what would happen if the velocity of the reaction is high?
the substrate will be higher. producing more product (with max velocity)
Saturation
all active site is filled and cannot go faster. enzymes have to wait in line till its turn
V max
Enzyme is working at it's maximum rate.
What is the Michaelis constant (Km)?
The concentration of substrate at which an enzyme works at half its maximum velocity.
What does the Michaelis constant (Km) measure?
It measures how tightly the substrate is bound to the enzyme.
competitive inhibition
Inhibitor competes with substrate for active site.

competitive inhibitor enzyme reaction
Km increase = more substrate needed
noncompetitive inhibition
binds at allosteric site.
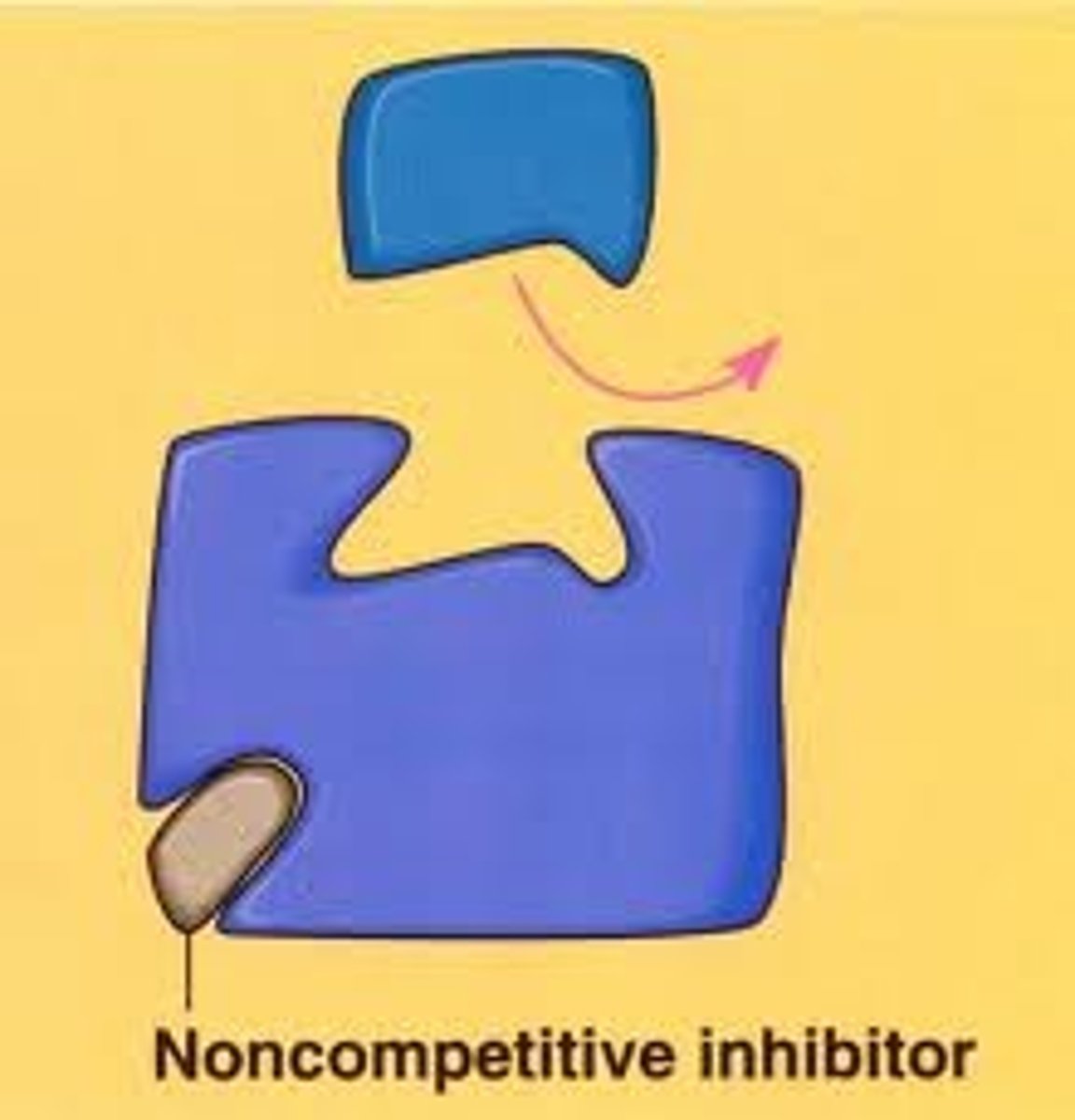
noncompetitive inhibitor enzyme reaction
lowers V max without affecting Km
factors that affect enzyme activity
temperature and pH
metabolism
All of the chemical reactions that occur within an organism
metabolic pathway
begins with a specific molecule and ends with a product
what does each step of metabolic catalyzed by?
a specific enzyme
catabolic reaction
exothermic reaction in organisms
how does catabolic pathway release energy?
by breaking down complex molecules into simpler compounds
Anabolic reaction
endothermic reaction in organisms
why does anabolic pathway consume energy?
to build complex molecules from simpler ones
does anabolic pathway must coupled to exergonic reaction?
yes
what does building blocks use to recycle building blocks
catabolic reaction
what does catabolic reaction use to store energy?
ATP and NADH
how to make ATP?
substrate level phosphorylation and chemiosmosis (oxidative phosphorylation)
how substrate-level phosphorylation make ATP?
- enzyme directly transfers phosphate from one molecule to another molecule
- oxygen not needed
how does chemiosmosis create ATP?
energy stored in an electrochemical gradient is used to make ATP from ADP and P
what reaction transfer electrons between reactants?
redox
Oxidation
loss of electrons
reduction
gain of electrons
how are energy intermediates create?
by the oxidation of organic molecules
NAD+
oxidized
NADH
reduced
where does electrons from organic compounds usually transferred to first?
NAD+
what are the functions of NAD+ during cellular respiration?
oxidizing agent
what does NADH do during synthesis reaction? Why?
donate electrons to energize them
what is another name for anabolic reactions?
biosynthetic reactions
what happen in anabolic reactions?
make large macromolecules or smaller molecules not available from food
does anabolic reaction require energy?
yes, from catabolic ATP
gene regulation
the turning on and off of genes
Cellular Regulation
cell-signaling pathways like hormones
biochemical regulation
feedback inhibition- product of pathway inhibits early steps to prevent over accumulation of product
what does feedback inhibition do?
prevents a cell from wasting chemical resources by synthesizing more product that it is needed
allosteric regulation
a regulatory molecule binds to a protein at one site and affects the protein's function at another site, may either inhibit or stimulate an enzyme activity
half life
time it takes for 50% of the molecules to be broken down and recycled
what does the expression of genome allow cells to do?
respond to changes in their environment
why is mRNA degradation important?
Conserve energy by degrading mRNAs for proteins no longer required;
Remove faulty copies of mRNA;
Proteasome
complex that breaks down proteins using proteases enzyme
large proteases ______ bonds between _____ _____
cleave; amino acids
Ubiquitin
tags target proteins to the proteasome to be broken down and recycled
what does ubiquitin do?
- degrade improperly folded proteins
rapidly degrade proteins to respond to changing cell conditions
lysosomes contains what and what does it do?
hydrolases to breakdown proteins, carbohydrates, nucleic acids, and lipids
Autophagy
recycling worn out organelles using an autophagosome. breakdown into macromolecules
cellular respiration
- process by which living cells obtain energy from organic molecules and release waste products
- make ATP
- aerobic respiration
- O2 consumed and CO2 released
when glucose is broken down, energy is ________
released
Glycolysis
breakdown of glucose (6c) into 2 molecules of pyruvate (3c)
stage 1 of cellular respiration
glycolysis
Where does glycolysis occur?
cytoplasm
does glycolysis need oxygen to function?
with or without oxygen
how many steps is in glycolysis and how many phase?
10 and 3 phases
3 phases of glycolysis
1. energy investment
2. cleavage
3. energy liberation
Energy Investment Phase
requires 2 ATP in order to create fructose-1, 6 bisphosphate
what are the molecules produced during glycolysis
- 2 pyruvate (3c)
- 2 net of ATP
- 2 molecules of NADH
enzymes used in glycolysis
- hexokinase
- aldolase
- isomerase
hexokinase (glycolysis)
- transfer phosphate from ATP to glucose = more reactive
- trap atp
Aldolase (glycolysis)
- split sugar molecule in 2 diff. 3c sugars
- create DHAP & G3P
isomerase (glycolysis)
conversion between DHAP and G3P to energy liberation phase
Cleavage Phase
Splitting of glucose into two 3-carbon molecules.
Energy Liberation Phase
2 G3p molecule broken down into 2 pyruvate = 2 NADH and 4 ATP
how does ATP created in glycolysis?
substrate level phosphorylation
How does ADP become ATP?
It gains a phosphate from enzyme (2 to 3)
inputs and outputs of glycolysis
Inputs: Glucose, ATP; Outputs: Pyruvate, ATP, NADH.
second stage of cellular respiration
breakdown of pyruvate
where does breakdown of pyruvate occur?
mitochondrial matrix
Inputs and outputs of breakdown of pyruvate
input: pyruvate (3c)
Outputs: 2NADH, 2 CO2, 2 Acetyl CoA
enzyme used in breaking down of pyruvate
pyruvate dehydrogenase