Reaction cond summary
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
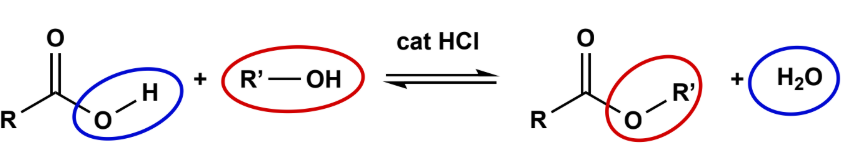
Fisher Esterification
Acid Cat (H2SO4 or ArSO3H or HCl), fully reversible
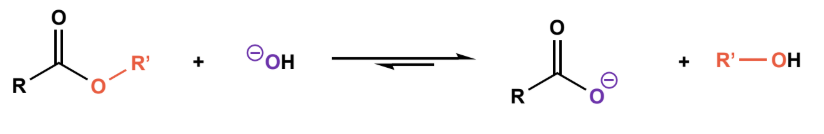
Saponification
excess base
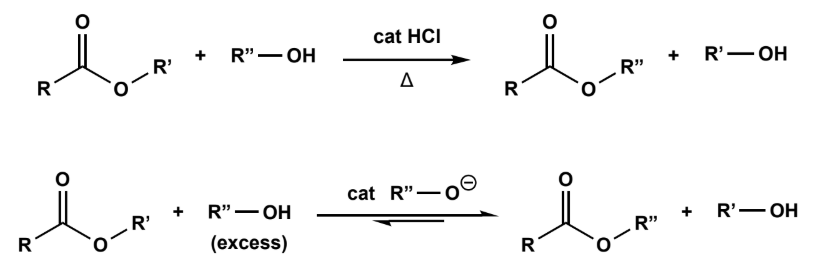
Transesterification
either strongly acidic or strongly basic
Esters to amides
mildly basic conditions (zwitterion interm.)
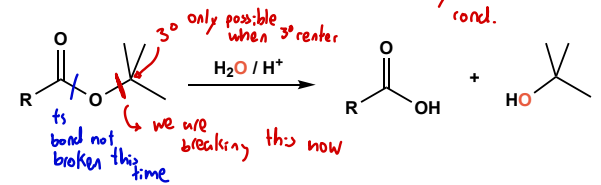
Ter-butyl ester cleavage
acidic condition, with reso interm and carbocation
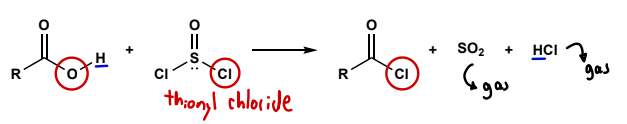
Acid chloride synthesis
mildly basic looking (cause of zwitterion), carbocation interm.
Acid chloride to anhydride
strongly basic conditions (salt of COOH attacks it).
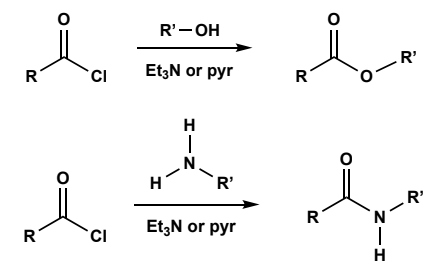
Acid chloride to Esters
mildly basic conditions (zwitterion interm.) with sacrificial base (Et3N or pyr).
Acid chlorides or anhydrides to Amides
mildly basic conditions (zwitterion interm.) with sacrificial alcohol or base (Et3N or pyr).
Schotten Baumann
biphasic mildly basic cond. (base = NaOH or Na2CO3)
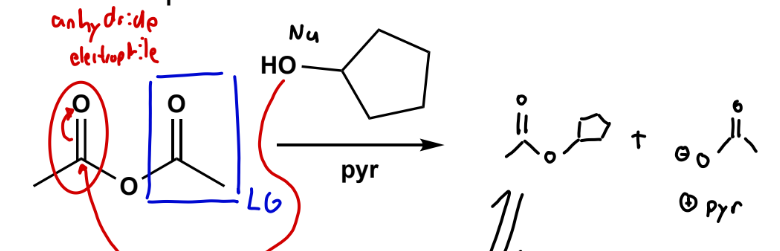
Anhydrides to esters
mildly basic conditions (zwitterion interm.) with sacrificial base (Et3N or pyr).
Amide hydrolysis
under strongly acidic conditions or strongly basic, and heat
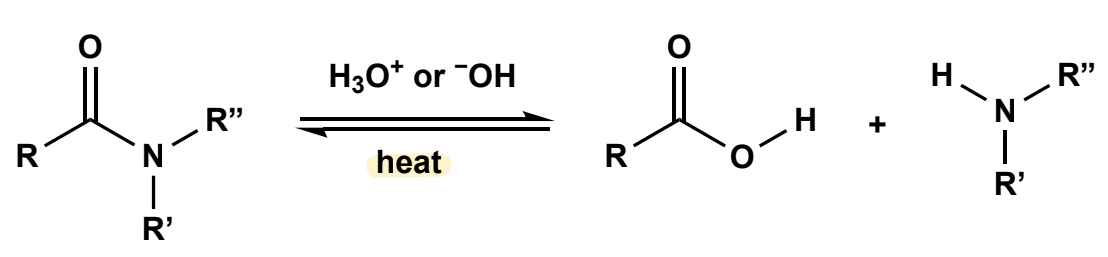
Amides to nitriles
mildly basic looking (cause of zwitterion) (resembles Acid chloride synthesis). Uses base: pyr or Et3N
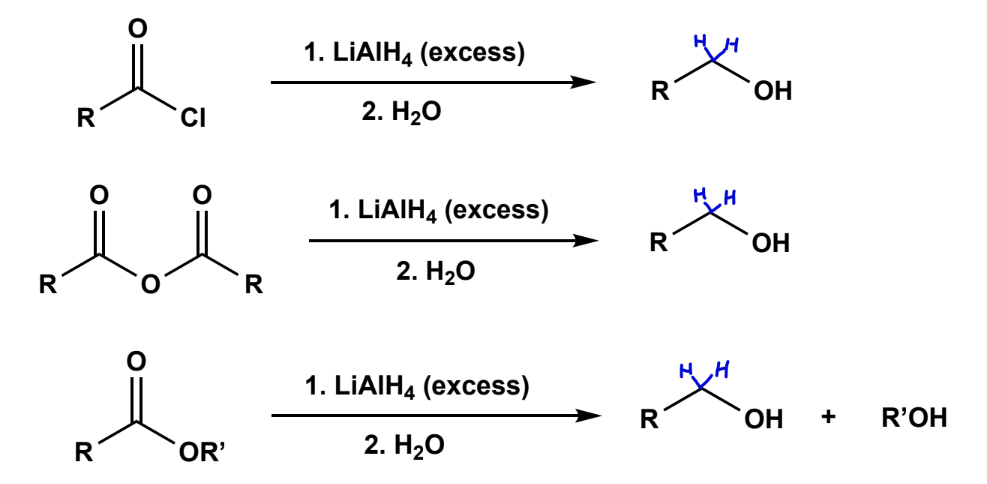
Carbon derivative reduction using LiAlH4
strongly basic looking, with acidic workup.
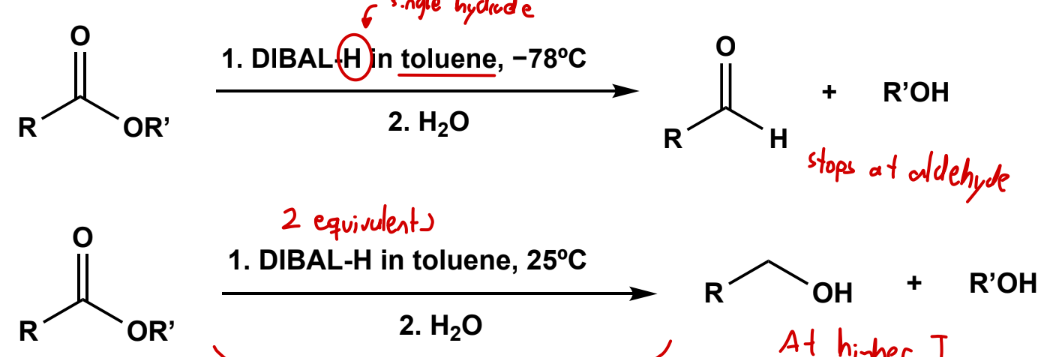
Reduction using DIBAL-H
in toluene, -78 C to stop at aldehyde, 25 C for full reduction
Gabriel Amine synthesis
1 equiv of Base for deproto, alkyl halide, and either hydrolysis or hydrazine
Enolization
base (strength depending on product pka)
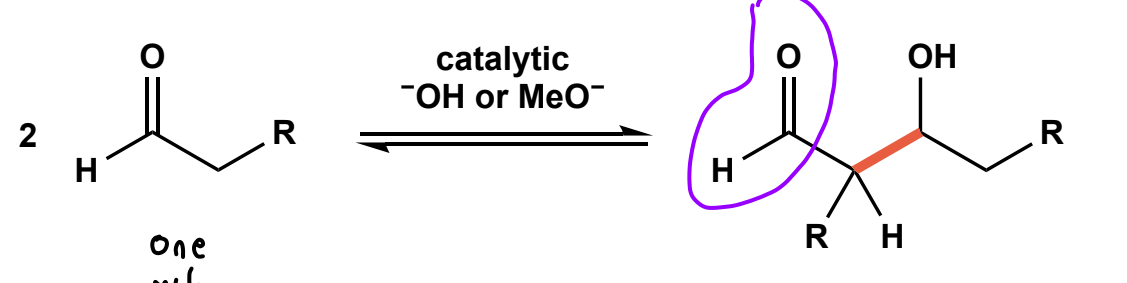
Aldol addition
2 carbonyls, and catalytic OH- or OMe- (cause we using aldehydes or ketones), polar protic solvent, followed by workup.
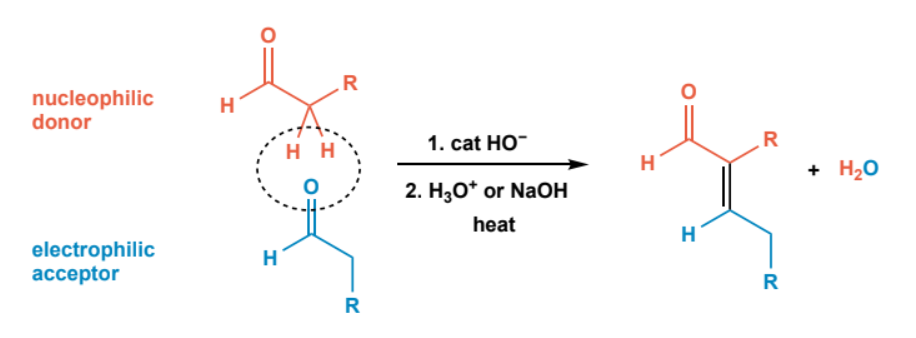
Aldol condensation
aldol addition followed by catalytic acid or base (H3O+ or NaOH) and heat.
Knoevenagel reaction
donor with very acidic proton + mild base (e.g. Et3N).
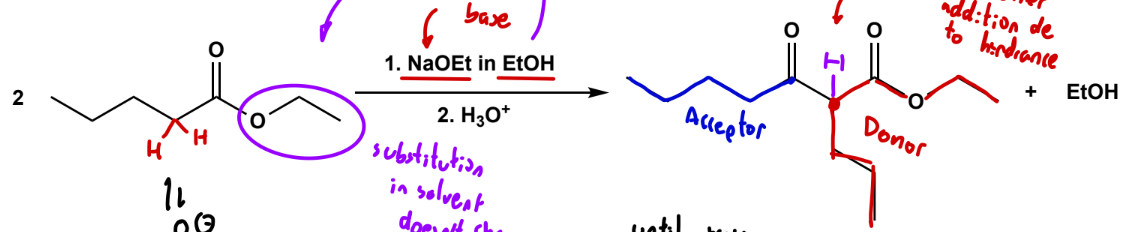
Claisen condensation
NaOR in ROH, H3O+ w.u. (notice how the R are the same, that's so if substitution with solvent occurs, the group doesn't change). Here we do have an -OR leaving group.
Dieckman condensation
Intramolecular claisen,1,6 or 1,7 diester, NaOR and H3O+ w.u.
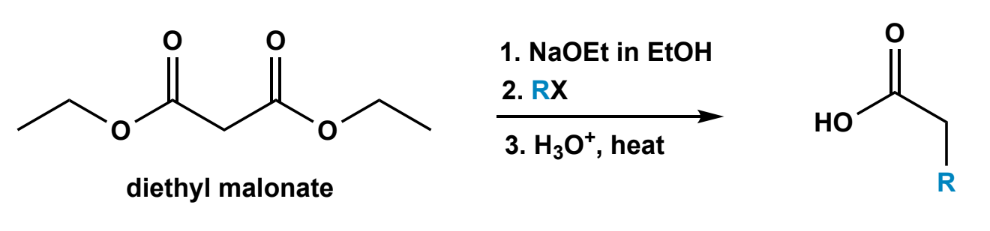
Malonic Ester and Acetoacetic synthesis
either malonic ester or acetoacetate ester, NaOR in ROH, RX (for SN2), followed with H3O+, heat (ester hydrolysis and decarboxylation).
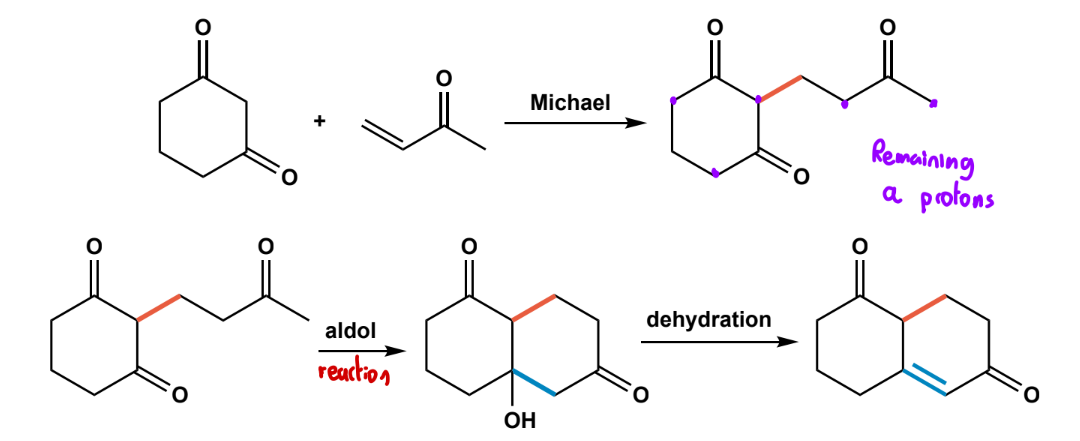
Michael Addition
a-b unsaturated carbonyl (acceptor), and double stabilized enolate (donor), catalytic base (e.g. KOH) in ROH solvent.
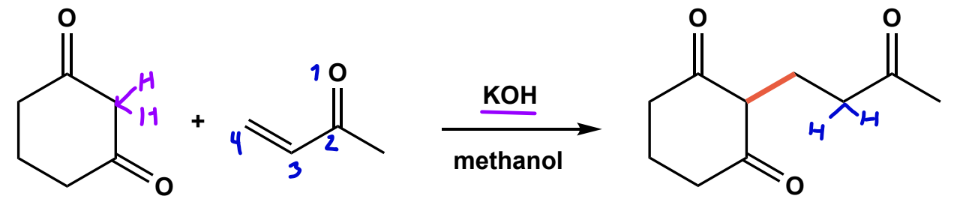
Robinson annulation
michael addition + intramolecular aldol + dehydration to from 2-cyclohexenone.