Genomes
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
Genomics
The study of whole genomes
genomics involves the development and application of more effective mapping, sequencing and computational tools
used to predict existences and functions of previously undefined genes
verify predictions using molecular biology techniques
sequencing the (human) genome reveals the blueprint for our form and functions
Human Genome Project
first formally discussed in 1985
discovery science: identification of all elements in a biological system
officially began in 1990, estimated 15 years, $3 billion
3-5% of budget committed to studying the ethical, legal, and social implications (ELSI) of human genome mapping. Social and personal repercussions are generating new areas of biolocial concern
2001: draft version of human genome published
2003: “completion” of project, covered 92% of genome
remaining 8% contains highly repetitive DNA, hard to read and to place into order
2022: complete human genome sequence published by the Telomere-to-Telomere (T2T) Consortium
used newer sequencing technologies that are capably of long sequence reads (able to sequence large stretches of repetitive DNA)
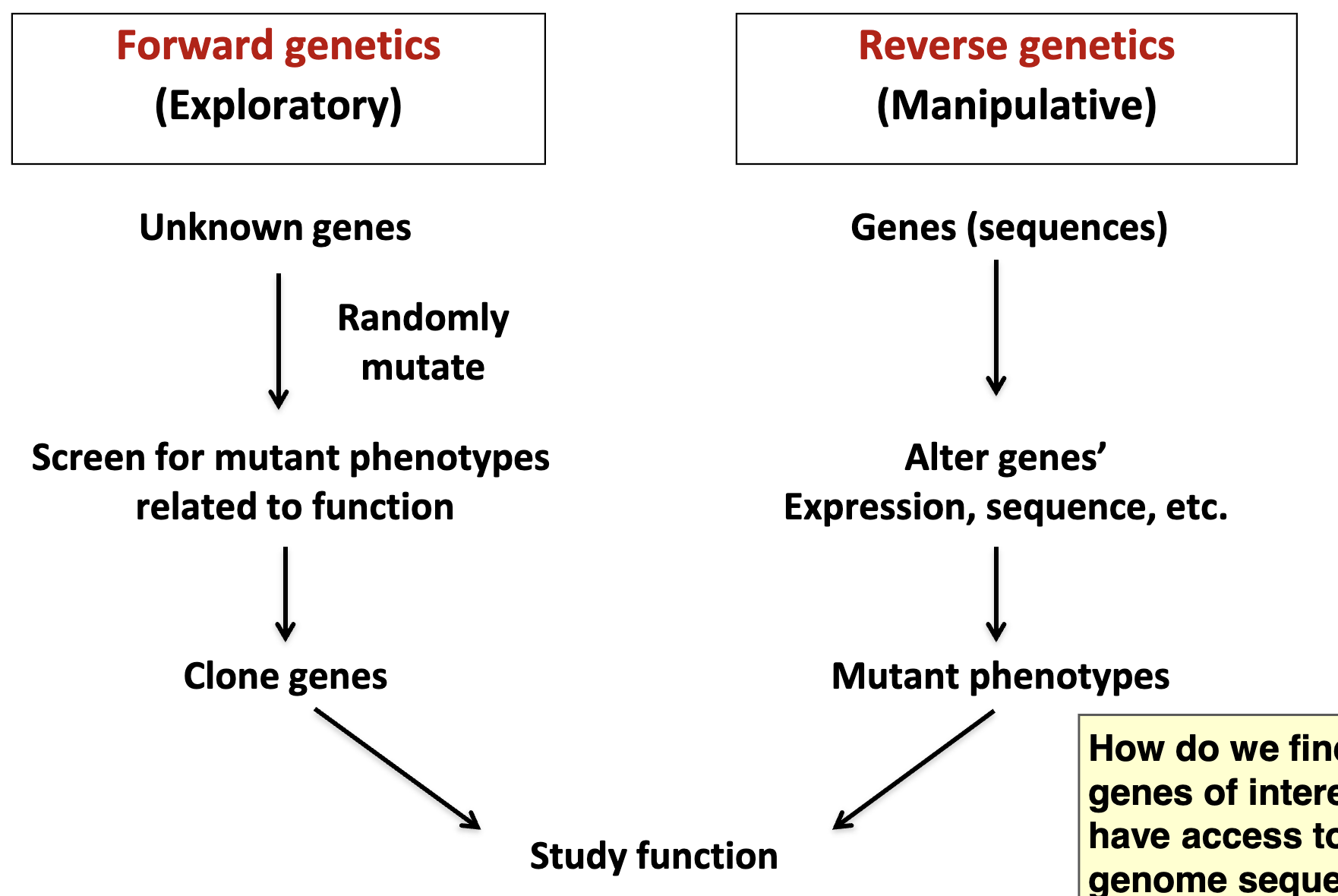
Recall: Two approaches to using genetics to study development
genome sequencing projects and the universality of biological processes lend themselves to reverse genetics
GWAS (genome-wide association studies) can be used to identify genes of interest (forward genetics)
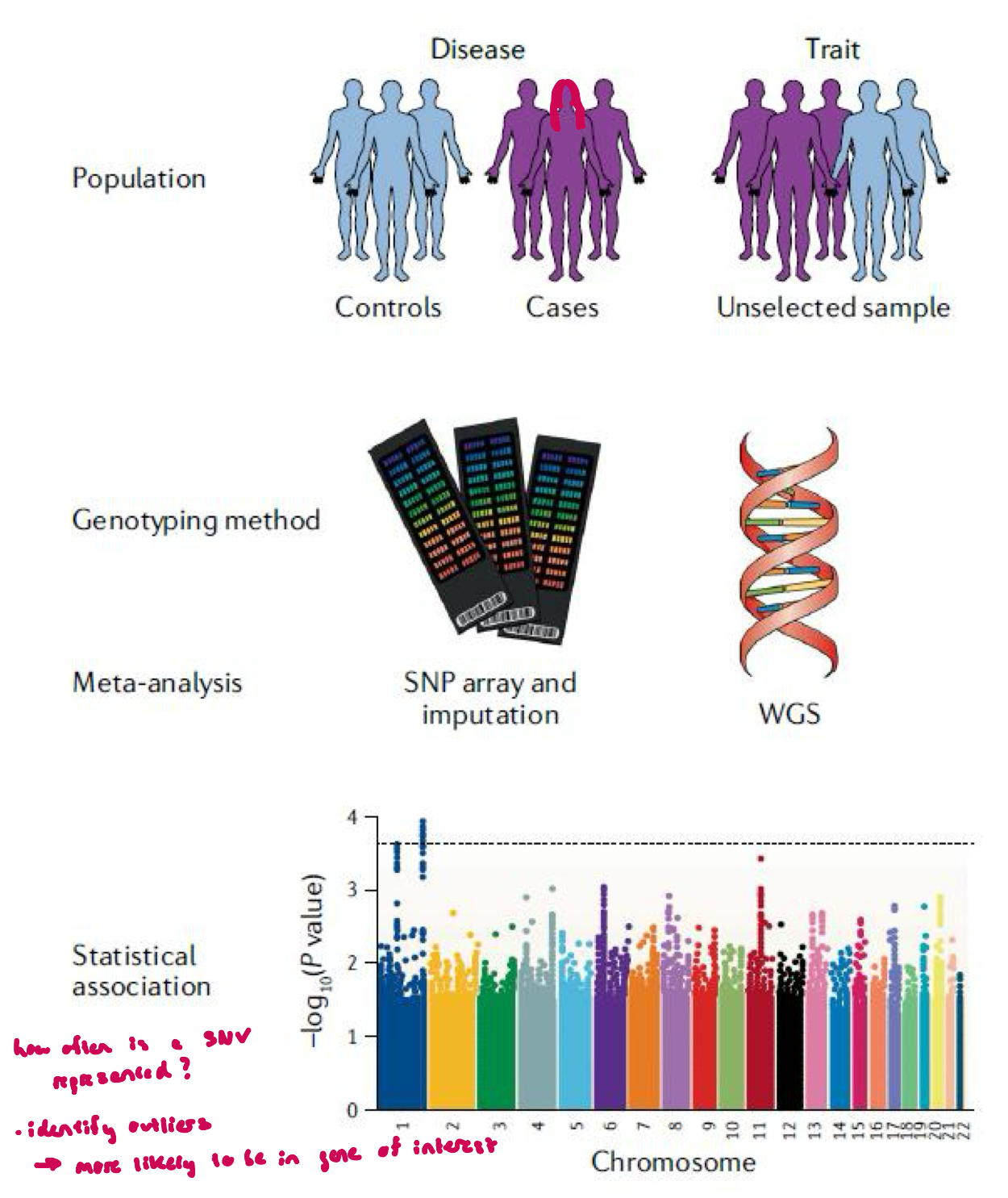
GWAS
large-scale approach to identifying genes and gene variants of interest
thousands of SNVs (single nucleotide variants) analyzed across the genomes of many individuals to identify possible genotype/phenotype associations
analysis done thru whole genome sequencing (WGS) or genome-wide SNP arrays
association confirmed thru more conventional studies
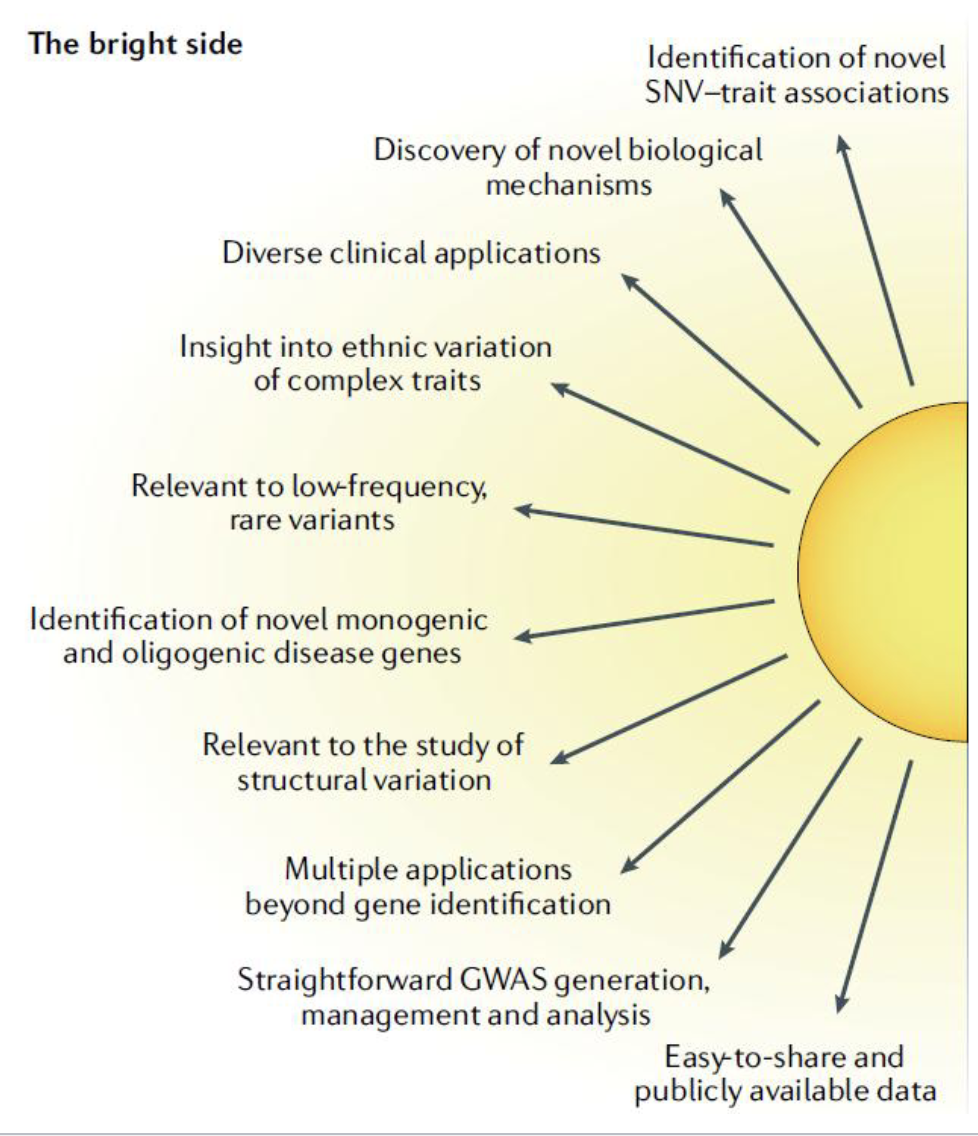
Benefits of GWAS
identification of thousands of novel variant/trait associations (as of 2019, there are 52416 such associations)
identifcation of risk loci for a broad range of medical issues
can identify novel genes of unknown function, or identiy a previously unknown role of a gene is disease pathology
can used identified associations to screen people who are at risk of developing certain diseases
can identify new target genes for drug development
Benefits of GWAS Figure
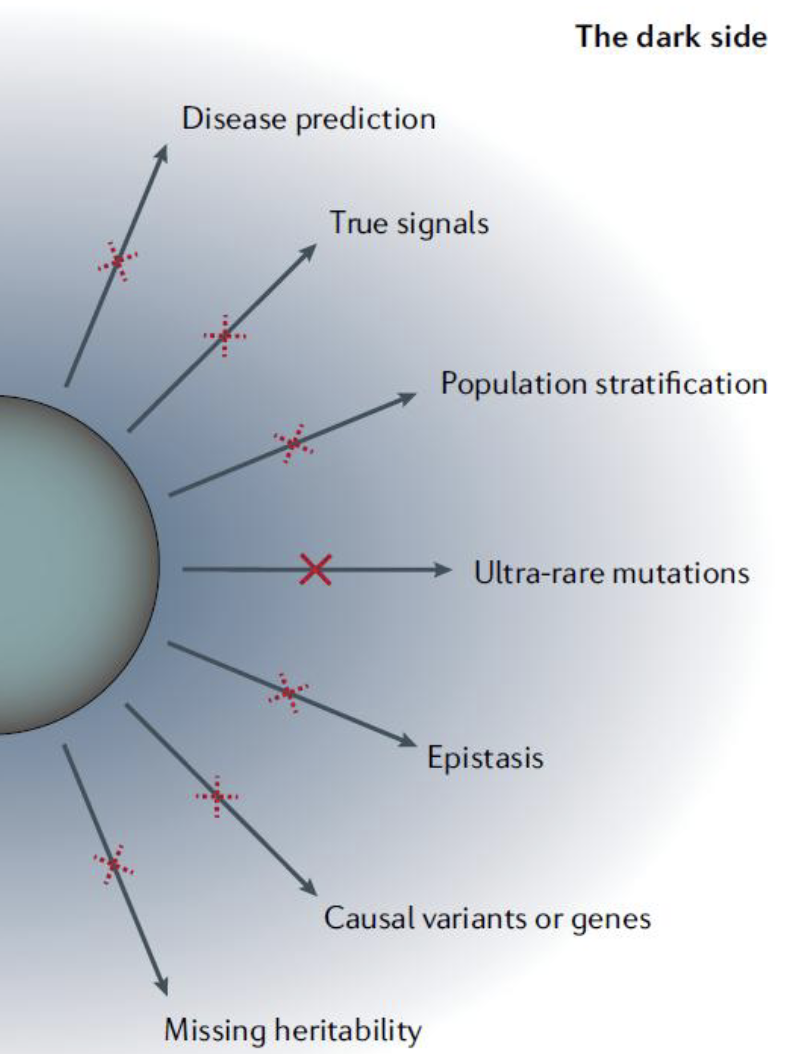
Limitations of GWAS
There are ethnic differences in risk variants–was the study sufficiently broad to capture these?
Much of this work have been done on populations of European origin – other populations are underrepresented.
genetic variants can only explain a small fraction of the heritability of a disease trait– it misses environmental factors
Difficulty in analysis e.g. Which SNV actually maps to a gene that causes the trait, and which are merely linked to the gene? Which SNVs are in noncoding sequences?
Cannot identify all genes responsible for complex traits. Likewise, has limited predictive value
Limitations of GWAS Figure
Example of GWAS Limitation
Mis-diagnosis of hypertrophic cardiomyopathy due to mismatched reference pool
Mis-diagnosis of hypertrophic cardiomyopathy due to mismatched reference pool
the Human Genome Mutation Database is a database with human mutations that have been associated with disease. It is based on data from published studies.
a number of allelic variants identified in this database were associated with hypertrophic cardiomyopathy
these genetic variants originally identified because they appeared more frequently in tested patients than in unrelated control populations
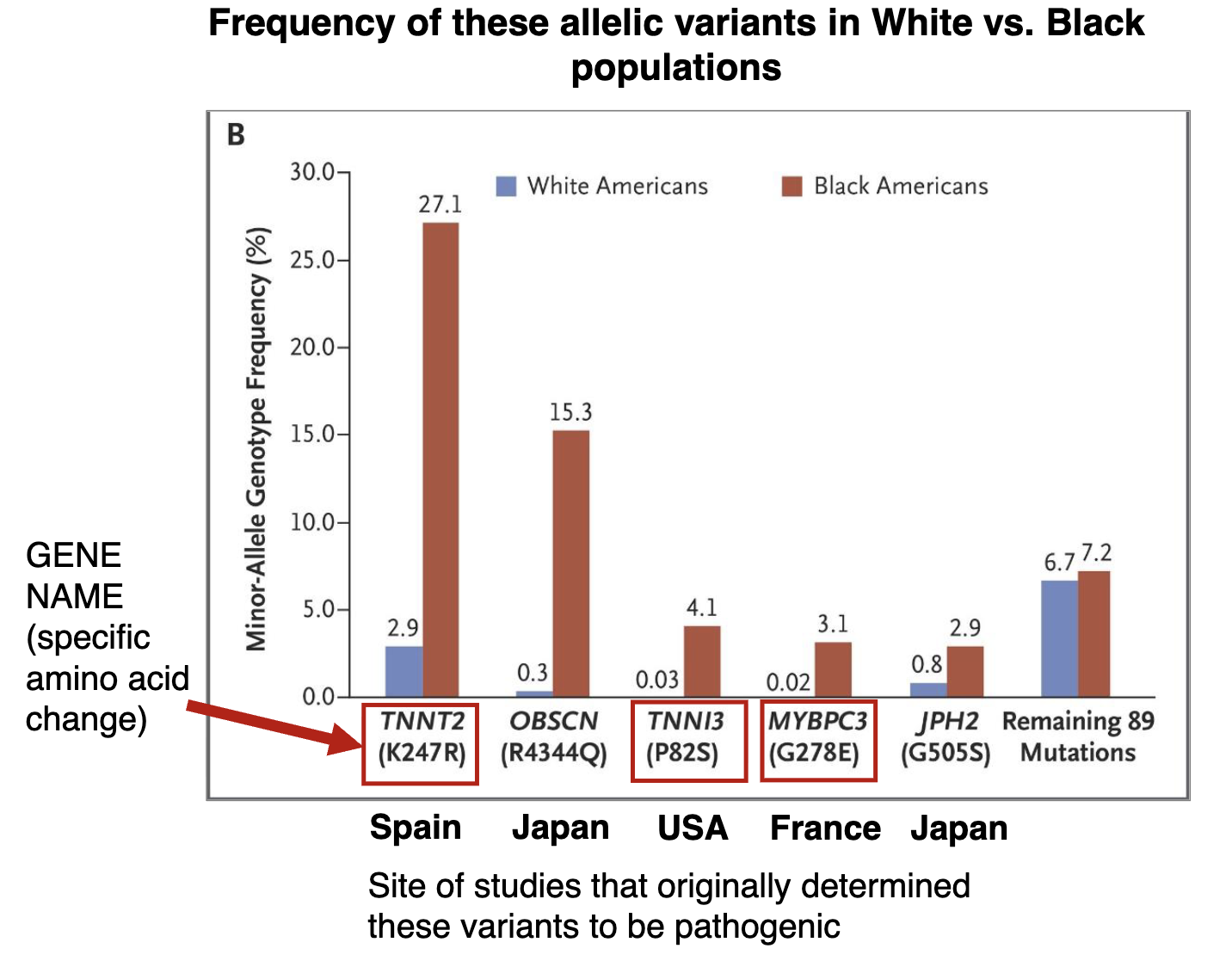
TNNT2, variant K247R: gene studied in Spain
TNNI3, variant P82S, gene studied in U.S
MYBPC3, variant G278E: gene studied in France
mainly based on European individuals, but these allelic variants have been used for genetic tests that screen for disease
How prevalent are these disease-associated allelic variants?
Manrai et al., (2016) used 3 other databases to check the allelic frequencies of these variants in the greater population (note the diversity)
NHLBI Exome sequencing project (4300 White Amer. + 2203 Black Amer.)
1000 Genomes Project (phase 1: 1092 individuals across 14 populations worldwide)
human genome diversity project (938 individuals across 51 populations worldwide)
Manrai et al. also looked at clinical records of patients w/ hypertrophic cardiomyopathy, including info about which allelic variants they have
How prevalent are these disease-associated allelic variants? Figure
Consequence of a mismatched reference pool
Misdiagnosis of hypertrophic cardiomyopathy
these 5 allelic variants had previously been identified to be associated with hypertrophic cardiomyopathy.
While these variants are rare in White Americans (ie. European ethnicity), they are more frequent in Black Americans (ie. African ethnicity).
Mis-classified as disease-causing variants, leading to misdiagnoses of Black Americans
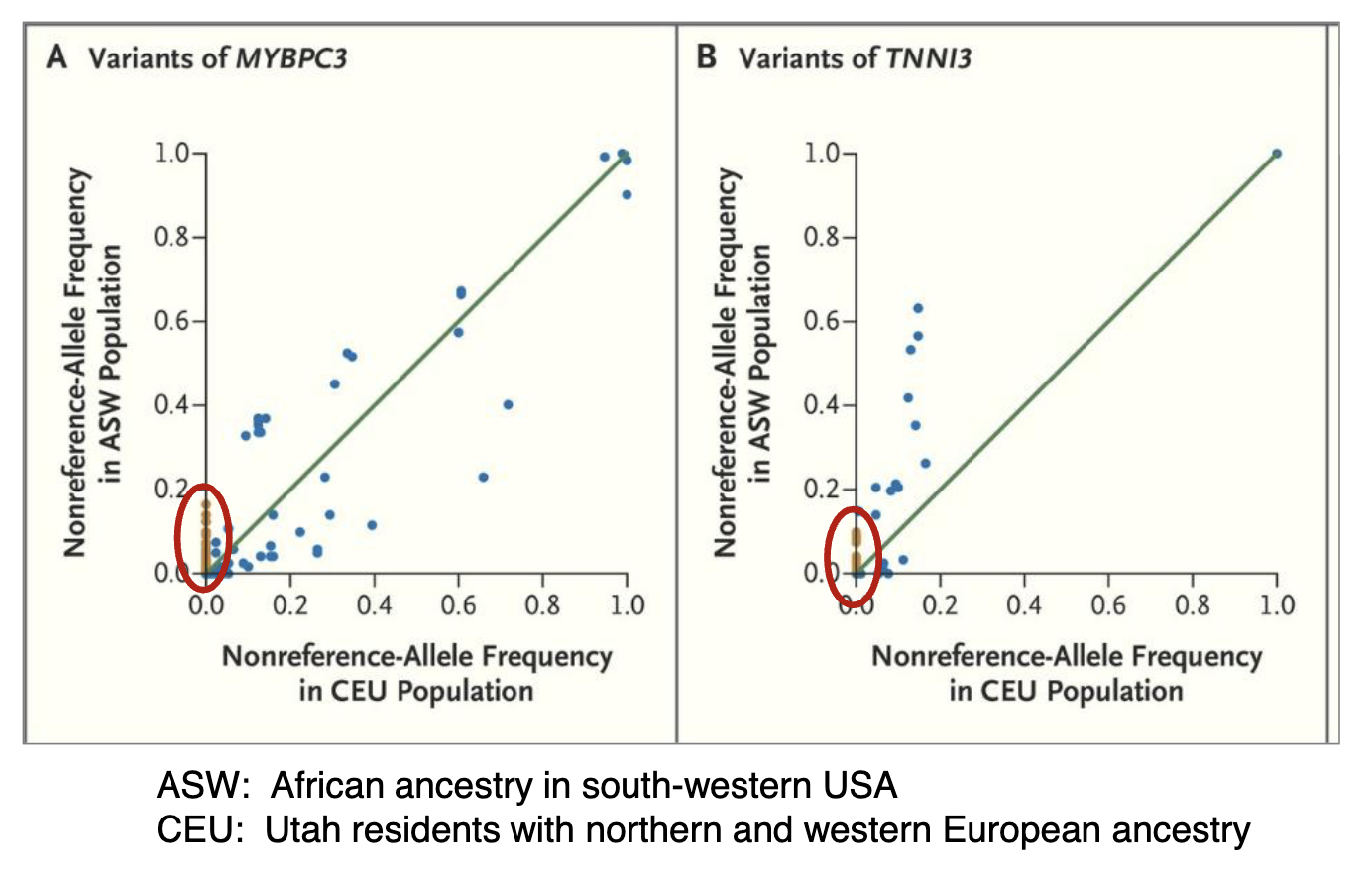
Allelic Variants of African-American Genomes
African-American genomes contain more “private sites” i.e allelic variants that are only present in one population but not the other
Genome Projects Targeting Other Populations
Human Heredity and Health in Africa (H3Africa): “H3Africa empower African researchers to be competitive in genomic sciences, establishes and nurtures effective collaborations among African researchers on the African continent, and generates unique data that could be used to improve both African and global health”
GenomeAsia 100k: “GenomeAsia 100K plans to create reference genomes for Asian population as well as identify rare and frequent alleles associated with these populations”
SIlent Genomes Project: genome project targeting Indigenous populations in Canada
Concerns about Commercial DNA Tests
what did you gain?
who own the data after you have submitted your biological sample?
how else might that data be used? how has it already been used? did you anticipate that application or consent to it? was your consent properly informed
“DNA on loan”
Tri-council policy on human research includes provisions for working with genetic material and creation of biobanks
this contains specific policies on working with Indigenous populations, b/c of past abuses of such research:
DAN samples remain the property of the individual. The researcher has only the permission to work with these samples for a specific purpose
if a new research objective can be carried out, consent must be given again for this new use
Social and Ethical Concerns of Genomic Research
which populations are represented in the Human Genome sequence? Who decides which population is the normal reference group?
Who decides what questions get asked, or what diseases get studied?
who owns the genetic data? who benefits
what impact does this have when genomic studies are carried out?
importance of diversity and representation in research subjects, researchers and those who fund the research and make the policy decisions
importance of treating research subjects and their genetic data with resect