ANS Exam 3
1/148
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
149 Terms
Division of the Estrous Cycle
Based on Ovarian Structures:
Follicular Phase
Proestrus
Estrus
Luteal Phase
Metestrus
Diestrus
Follicular Phase
Growth of Follicles
Dominant structure- preovulatory follicle
Dominant hormone- Estradiol
Starts with regression of corpora lutea
Ends with ovulation
Relatively short (23-33% of the cycle)
In a 21-day cycle → 5-7 days
Proestrus + Estrus
Luteal Phase
Growth of corpus luteum (corpora lutea)
Dominant structure - Corpus luteum
Dominant hormone- Progesterone
Prepares uterus for pregnancy
Supports embryo development
Starts with ovulation
Ends with luteal regression
Longer phase (66-75% of cycle
In a 21 days cycle → 14-16 days
If not pregnant:
CL undergoes luteolysis (regression)
Cycle restarts with new follicular phase
Metestrus + Diestrus
Proestrus
Increased estrogen
Follicles growing
1st stage of follicular phase
Progesterone decreasing
Due to CL regression
Estrus aka “heat”
Sexual receptivity
Peak estrogen production
Lowest Progesterone
Maximum follicle size and estrogen
LH & FSH surges
Ovulation
Mare: ovulates during estrus (estrus lasts 4-9 days)
Cow/Sheep: ovulate after estrus, in metestrus (last 8 hours)
High estrogen stimulates GnRH surge → LH surge → ovulation
Behavioral sign
Affectionate behavior
Increases body temp
Metaestrus
Increasing progesterone production
Estrogen decreasing (follicle ruptured)
Formation of the CL following ovulation
Ovulation - Cow & Ewe
LH and Estradiol- low
Diestrus
Sustained progesterone production
Maximum progesterone
Estrogen= lowest
Fully functional CL
CL dominant structure
Follicles continue to grow in waves
Shift in LH & FS
Longest period of cycle
Why estrogen stays low
Progesterone blocks the surge center in the hypothalamus
→ Prevents LH surge
Only the tonic center releases small pulses of LH + FSH
→ Not enough to cause ovulation
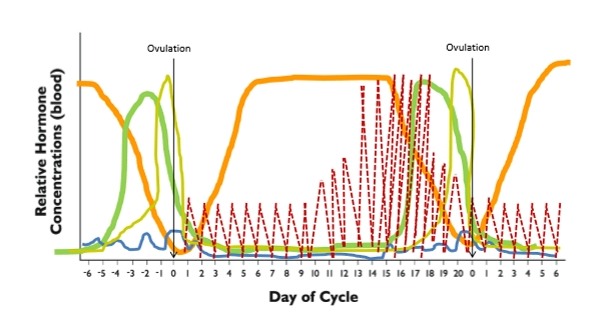
Differences between Estrous and Menstrual Cycles
Cycle based on what we can SEE
Menses’ vs. Estrus
Menstrual 50 follicular: 50 luteal
Estrous cycle: 25 follicular: 75 luteal
Ovulation in the middle (day 14)
Menstrual cycles occur in primates
Ovulation is silent (no behavioral signs)
Menstrual cycle sequence
Consist of six events:
Menstruation: Sloughing of endometrial lining, low estrogen + progesterone
Follicular growth: Follicle develop, estrogen rises, Endometrium begins rebuilding
Ovulation (day 14): Triggered by LH surge, Marks end of follicular phase
Luteinization: CL forms, progesterone rises, Endometrium thickens further
Endometrial growth
Luteolysis
Estrous vs. Menstrual Cycle: Key Comparison
Estrous Cycle
Seen in livestock
Defined by estrus (heat)
Ovulation often tied to estrus
Follicular phase = 25%
Luteal phase = 75%
No menses
Sexual receptivity is obvious
Menstrual Cycle
Seen in primates
Defined by menses
Ovulation is silent
Follicular = 50%
Luteal = 50%
Endometrium sloughs if no pregnancy
No clear behavioral estrus
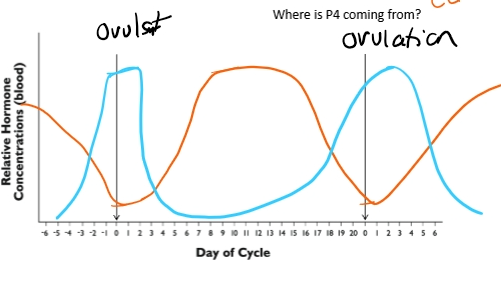
Progesterone + Estrogen
Progesterone
Produced by CL
At ovulation → low
After ovulation → increases CL forms
Mid cycle → Peak
Before next ovulation → decreases due to PGF2a
P4 blocks the surge center
Estrogen
Produced by growing follicles
just before ovulation → high
Mid cycle → low
End of cycle → rises again as follicles regrow
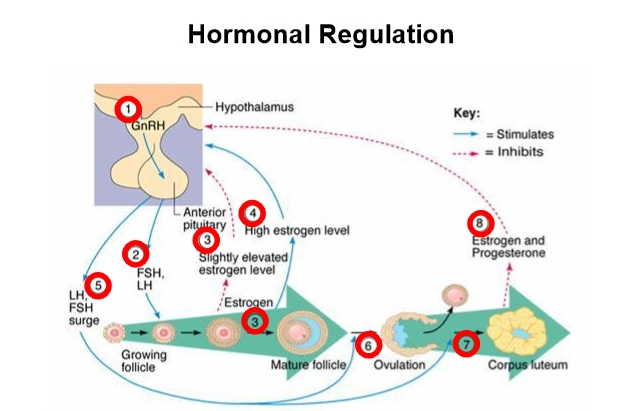
Add the Gonadotropins (Anterior Pituitary)
Follicle Stimulating Hormone (FSH)
Stimulates follicle growth
Follicles → produce estrogen
FSH rises early in the cycle
Small “blip” at ovulation
FSH pattern:
Small rise → supports follicle waves → small bump at ovulation
Luteinizing Hormone (LH)
Controlled by:
Tonic center (low pulses)
Surge center (big spike)
Estrogen stimulates the surge center
→ LH surge → ovulation
LH pattern:
Low pulses → massive spike → back to low pulses
Add the Uterine Hormone
Prostaglandin F₂α (PGF₂α)
Produced by: Uterine endometrium
Function: Kills the corpus luteum (luteolysis)
Begins pulsing around day 10
Pulses increase in:
Amplitude
Frequency
Causes progesterone to drop
PGF₂α pattern:
Low → pulses begin → pulses intensify → CL dies → drops again
Full hormone sequence
1. Follicular Phase Begins (P4 low)
CL regresses → progesterone drops
Low P4 removes block on surge center
FSH rises → follicles grow → estrogen increases
2. Estrus + LH Surge
Estrogen peaks → positive feedback to surge center
Surge center releases GnRH
Anterior pituitary releases LH surge
Ovulation occurs (if P4 is low)
3. Luteal Phase Begins
Ovulation → CL forms → progesterone rises
High P4:
Blocks surge center
Prevents LH surge
Prevents ovulation
Supports uterine lining
4. Diestrus (Longest Phase)
CL fully functional → max progesterone
Follicles grow in waves but undergo atresia
Estrogen stays low
5. Luteolysis
Uterus releases PGF₂α pulses
Pulses intensify → CL dies
Progesterone drops sharply
6. Cycle Restarts
Low progesterone → surge center unblocked
FSH rises → follicles grow → estrogen rises
New estrus approaches
Estrous vs. Menstrual Cycle (Final Comparison)
Estrous Cycle (Livestock)
Defined by estrus (heat)
Follicular = 25%
Luteal = 75%
No menses
Ovulation tied to estrus (species‑dependent)
Menstrual Cycle (Primates)
Defined by menses
Follicular = 50%
Luteal = 50%
Ovulation ~day 14
Endometrium sloughs if no pregnancy
Ovulation Occurs when progesterone is LOW
Progesterone must be low for the LH surge to occur.
When P4 is low → surge center is unblocked → LH surge → ovulation.
After ovulation:
Follicle ruptures
Corpus luteum (CL) forms
Progesterone begins to rise
If the female is NOT pregnant
The uterine endometrium detects no embryo.
It releases prostaglandin F₂α (PGF₂α).
PGF₂α:
Kills the CL (luteolysis)
Causes progesterone to drop
Allows the cycle to restart
PGF₂α pattern:
Begins pulsing around day 10
Pulses increase in amplitude + frequency
High pulses → CL death → P4 drops → follicular phase begins again
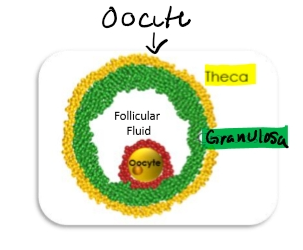
Parts of the Follicle
Oocyte
Oogenesis- development of the oocyte= able to fertilize
Granulosa cells
Inner cell layers
Two types:
Mural granulosa
Cumulus granulosa
Functions:
Covert testosterone → estrogen
Support oocyte development
Communicate via follicular fluid
Thecal cells
Outer yellow layer
Produces testosterone
Testosterone diffuses into granulosa cells → converted to estrogen
This is the two-cell, two- gonadotropin model
Follicular fluid
Fills the antrum
Allows communication between granulosa cells
Stores nutrients, growth factors
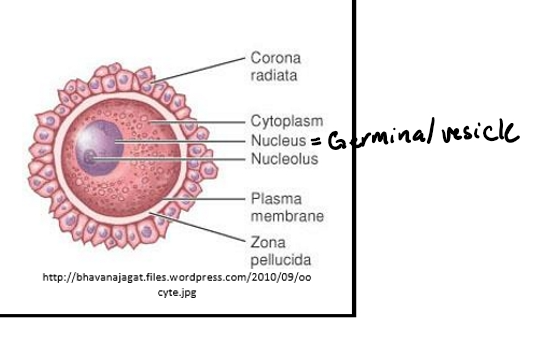
Components of oocyte
Zona Pellucida- assists fertilization and may provide protection
Thick, protective glycoprotein shell
Functions:
Protects the oocyte
Sperm bind here during fertilization
Vitelline membrane- surface layer of the oocyte
Immediately beneath the zona pellucida
Called the oolemma
Perivitelline space
Space between zona pellucida and vitelline membrane
Provides cushioning and structural separation
Cytoplasm (yolk)- contains many proteins, enzymes, nutrients needed for survival of the zygote
Supports embryo until it reaches the uterus
Germinal vesicle- nucleus containing genetic information
The nucleus of the oocyte
Contains the genetic material
Will undergo meiosis during maturation
Oogenesis
Oogenesis- formation and development of the ovum (egg) - gametes fixed number of germ cells
It begins in fetal life, pauses for years, and only resumes after LH surge during reproductive cycle
Oogonium- the primordial cell which develops into the oocyte
Oocyte- the early, not yet fully developed ovum
Ovum- the egg- the cell that is capable of developing into a new individual
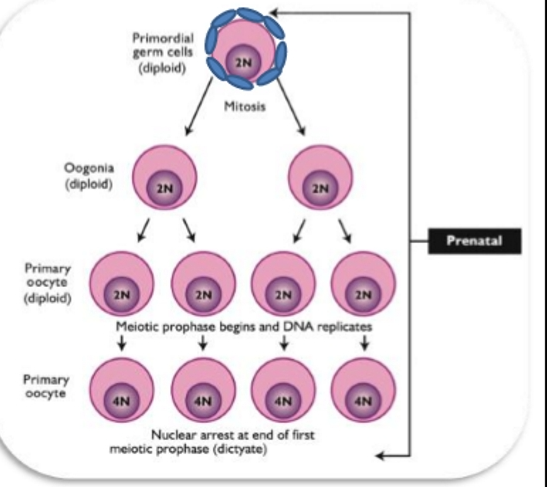
Primordial Germ Cells → Oogonia → Oocytes
Primordial Germ Cells
Originate in the yolk sac
Migrate to the gonadal ridge
Undergo mitosis to increase in number
Fill the sex cords
Oogonia
Earliest form of female germ cells
Undergo mitosis in fetal ovary
After mitosis, they begin meiosis I
Primary Oocyte
Formed in fetal life
Frozen in meiosis I (prophase I)
This is the stage found in:
Primordial follicles
Primary follicles
Secondary follicles
Tertiary (antral) follicles
Even preovulatory follicles
Key point:
The oocyte stays frozen in meiosis I for YEARS until the LH surge.
Oocyte Formation & Atresia
Primary oocytes formed from mitotic divisions of oogonia
Primary oocytes enter MEIOSIS 1
Become dormant
Maximum number formed during mid to late fetal life
Once max number is attained, atresia or natural degeneration begins and continues for life
Decrease in number of gamete’s
Atresia (Programmed cell death)
After peak numbers in mid‑late fetal life, oocyte numbers decline.
Atresia = apoptosis‑like death of germ cells.
Continues throughout life.
Reason for reproductive senescence (menopause):
→ Females eventually run out of oocytes.
Follicle Stages vs. Oocyte Stages
Follicle Stage | Oocyte Stage |
Primordial | Primary oocyte (meiosis I arrest) |
Primary | Primary oocyte |
Secondary | Primary oocyte |
Tertiary (antral) | Primary oocyte |
Preovulatory (Graafian) | Primary oocyte |
Important:
The oocyte does NOT complete meiosis I until the LH surge.
What triggers meiosis to resume? What happens? Why polar bodies
What triggers meiosis to resume?
LH surge (caused by high estrogen)
What happens?
Primary oocyte completes meiosis I
Produces:
Secondary oocyte
First polar body (genetic trash can)
Why polar bodies?
Oocyte must go from diploid → haploid
Polar body = “genetic dumpster” to discard extra chromosomes
Cumulus granulosa cells:
Surround the oocyte
LH surge disrupts their tight communication
This loss of inhibition allows meiosis to resume
Oocyte Activation & Development
Meiosis resumes only in preovulatory follicle(s)
LH & FSH remove inhibitors
Activation occurs at estrus
Only a select few are stimulated to develop
Repeats every cycle
Oocyte Activation & Development continued
Fertilization → Completion of Meiosis II
If sperm penetrates the zona pellucida:
Secondary oocyte completes meiosis II
Produces:
Ovum (haploid)
Second polar body
Result:
Haploid ovum + haploid sperm → zygote
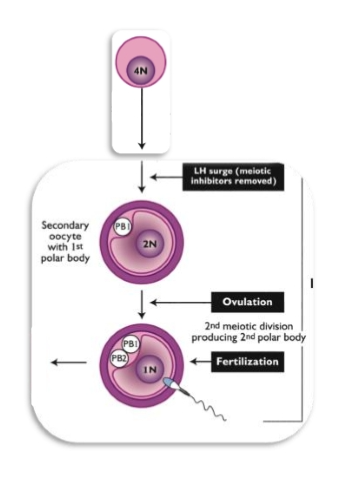
Secondary oocyte → Ovulation
The secondary oocyte is ovulated
It does not finish meiosis II unless fertilization occurs.
Ovulated oocyte= secondary oocyte, not an ovum
Oogenesis Timeline
Fetal life:
Oogonia → primary oocytes → freeze in meiosis IPuberty onward:
Follicles recruited each cycle; oocyte still frozenLH surge:
Meiosis I completes → secondary oocyte + polar bodyOvulation:
Secondary oocyte releasedFertilization:
Meiosis II completes → ovum + second polar body
Why this matters:
Only oocytes that reach the LH surge can be ovulated.
Most oocytes die via atresia.
Meiosis is tightly controlled by granulosa cells and LH.
Oocyte numbers over the female’s lifetime
Oocyte Numbers Over the Female’s Lifetime
Key Concept:
Females are born with all the oocytes they will ever have, and the number only decreases from fetal life onward.
Human Oocyte Numbers
Fetal Life
Peak number of oocytes in mid–late fetal development
~6–7 million oogonia at maximum
Begin meiosis → become primary oocytes
Many die via atresia
At Birth
Only 1–2 million oocytes remain
All are primary oocytes frozen in meiosis I
At Puberty
Only ~300,000 remain
Still primary oocytes in primordial/primary/secondary follicles
During Reproductive Life
Only ~400–500 oocytes are ever ovulated
The rest die via atresia
At Menopause (45–50 years)
Oocyte pool is essentially depleted
Reproductive senescence occurs because no viable gametes remain
Cattle Oocyte Numbers
Fetal Life
Peak: ~2.7 million oocytes
At Birth
Only ~70,000 remain
At Puberty
~24,000 oocytes remain
Lifetime Ovulations
Cattle ovulate far fewer oocytes than humans
Only ~500 ovulations in a lifetime (depending on lifespan)
Gamete Wastage (Atresia)
Your professor emphasized this:
From fetal peak → ovulated oocytes
99.98% of oocytes are lost
Only 0.02% ever reach ovulation
From puberty → ovulated oocytes
Only ~2.8% of remaining oocytes are ovulated
97% die via atresia
Why so much loss?
Atresia = programmed cell death
Occurs continuously from fetal life → menopause
Ensures only the healthiest follicles reach ovulation
Why Females Undergo Reproductive Senescence
They run out of oocytes
Once the ovarian reserve is depleted → cycles stop → menopause
Primordial follicles
Primordial Follicles (fetal life → Puberty)
Earliest follicles present in the ovary during fetal life.
Found in nests/clusters
Structure
Primary oocyte inside (arrested in meiosis I).
Single layer of squamous epithelial cells.
These follicles remain present through childhood and puberty
After puberty, some primordial follicles begin to activate and grow.
Activation of Primordial Follicles
Growth is stimulated by ovarian growth factors, especially:
Members of the TGF-B family
Anti-mullerian hormone
Activin and inhibin
Primary follicles
Primary Follicle
Structural Changes
Oocyte enlarges
Follicular cells change from:
Squamous → cuboidal/columnar
Oocyte gains the zona pellucida:
A hardened glycoprotein layer surrounding the oocyte
Oocyte is still a primary oocyte, still arrested in meiosis I.
Single layer follicle cells
Primary oocyte but starts to expand
Follicle cells expand (cubodial)
Zona pellucida forms (harden layer outside)
Secondary Follicles
Secondary Follicle
Follicular cells proliferate → multiple layers
First appearance of two distinct cell types:
Granulosa cells (inner)
Theca cells (outer)
Still pre antral
Oocyte remains a primary oocyte
Primary oocyte
Multiple layers of granulosa cells
Thecal layers forms
Pre-antral (no follicular fluid)
Start to see division of granulosa & Theca)
Tertiary Follicle
This is where follicuogensis becomes fully active
Development of the antrum (fluid-filled cavity)
Oocyte is still a primary oocyte
Follicle now has distinct functional regions:
Oocyte- Still arrested in meiosis I
Germinal vesicle- nucleus of the oocyte
Mural granulosa cells- Produce estrogen; more cells= more estrogen
Cumulus granulosa cells- Surround oocyte; provide meiotic inhibition
Theca cells- produces testosterone (precursor for estrogen
Follicular fluid- Reservoir for hormones, nutrients, growth factors; enables communication across granulosa layers
Folliculogenesis
Classification and regulation of follicle growth after they get to tertiary follicle
Parts of follicle
Granulosa cells
Thecal cells
Follicular fluid
Theca= testosterone able to transition to granulosa to produce E2
Two cell- Two gonadotropins Model
Theca Cells
Have LH receptors.
LH binding stimulates:
Cholesterol → pregnenolone → progesterone → testosterone.
Testosterone diffuses into granulosa cells.
Granulosa Cells
Have FSH receptors.
FSH binding increases aromatase.
Aromatase converts:
Testosterone → Estrogen.
Changing hormone Dependency during Follicle growth
Small follicles → highly FSH-dependent.
As follicles grow:
They become increasingly LH-dependent.
This shift is essential for:
Sustained estrogen production.
Preparing for the LH surge and ovulation.
Granulosa & Theca cells
Granulosa
From cortex
Responsive to FSH
Convert testosterone to estradiol
Theca
From stroma
Responsive to LH
Convert Cholesterol to Testosterone
Follicular Fluid (Antral Follicles)
Components
Hormones
Steroids
Gonadotropins
Prostaglandins
Proteins
Enzymes
Carbohydrates
Functions
Supports follicle growth
Aids in oocyte development and health
Mediates granulosa cell functions
What is inside follicular fluid and Function?
Gonadotropins:
Luteinizing hormone (LH)
Follicle‑stimulating hormone (FSH)
Steroid hormones:
Estrogen
Progesterone
Testosterone
Prostaglandins
Nutrients:
Essential proteins
Enzymes
Carbohydrates
Growth factors
Functions of follicular fluid
Supports follicle growth and development.
Maintains communication between granulosa cells across the follicle.
Keeps the oocyte alive and healthy.
Provides nutrients in a hypoxic environment (granulosa cells lack direct blood supply).
If follicular fluid deviates from normal → follicle loss.
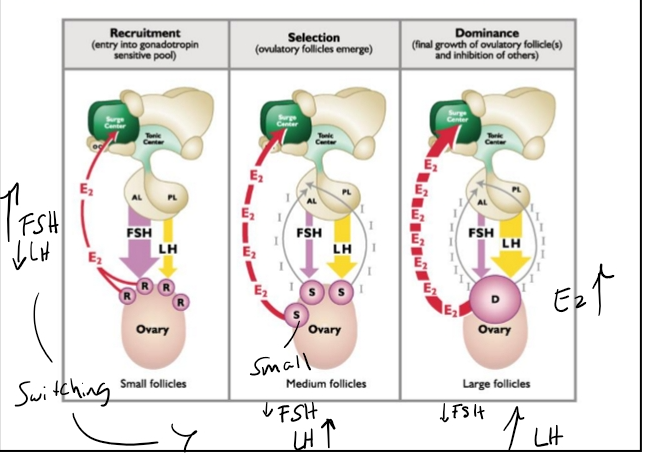
Stages of follicular growth
Stage 1: Recruitment
A large number of small antral follicles are recruited.
These follicles produce low estrogen.
Many follicles enter this stage, but most will not continue.
🔹 Stage 2: Selection
A refinement process.
Some follicles continue growing; many undergo atresia (cell death).
Selected follicles:
Increase in size.
Increase in granulosa cell number.
Produce more estrogen.
Atresia reduces the number of follicles but increases the size and estrogen output of survivors.
🔹 Stage 3: Dominance
Only one dominant follicle in nonovulatory species (humans, cattle, mares).
Litter‑bearing species may have co‑dominance.
Dominant follicle characteristics:
Largest follicle on the ovary.
Produces a tremendous amount of estrogen.
Reaches maximum size.
Has the most granulosa cells and the most follicular fluid.
Early Antral (tertiary)
Primary oocyte
Oocyte reaches maximum size
Granulosa cells expand
Antrum forms
Thecal layer more prominent
Interna
Externa
Follicular pool and timeline
The follicular pool includes:
Primordial follicles
Primary follicles
Secondary follicles
Transition from primordial → tertiary follicle takes 45–60 days (species‑dependent).
Once in folliculogenesis, growth occurs much more rapidly.
Regulation of Folliculogenesis
What determines which follicles advance?
Exact mechanisms are still unknown.
But we know it depends heavily on gonadotropin support:
Which follicles receive FSH?
Which receive LH?
Follicles that fail to receive adequate support undergo atresia.
Graafian Follicle (Preovulatory)
Last stage of tertiary follicle
Primary oocyte
Features:
Maximum granulosa cell number.
Maximum follicular fluid volume.
Oocyte is still a primary oocyte, arrested in meiosis I.
Contains the cumulus–oocyte complex (COC).
This is what is expelled at ovulation.
Follicular Development
How do follicles grow?
Waves
Number of follicles change throughout estrous cycle
Stimulated or regulated by?
Gonadotropins
Growth factors
Activin or inhibin
Fates?
Ovulation
Atresia
Cysts
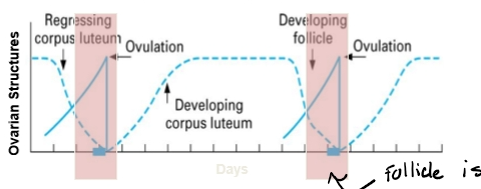
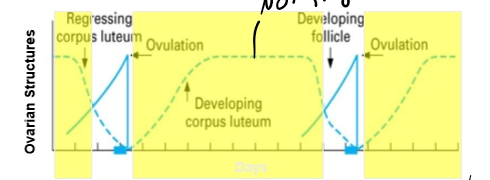
Follicular Waves During the Estrous Cycle
Follicles grow in waves.
During the luteal phase, high progesterone suppresses follicular progression.
During the follicular phase, progesterone drops → follicle growth increases.
Activin & Inhibin: Regulation of FSH
Produced by granulosa cells
Activin
Produced by small follicles
Increases FSH from the anterior pituitary
“Activin activates”
Inhibin
Produced by large follicles
Decreases FSH
“Inhibin inhibits”
Why this matters
Helps regulate which follicles continue to grow.
Prevents too many follicles from becoming dominant.
Coordinates follicle fate with the hormonal environment.
Follicle Fate
Possible outcomes
Ovulation
Requires low progesterone
Must occur during the correct stage of estrus
Only the dominant follicle has this opportunity
Atresia (death)
Most follicles undergo this
Occurs at all stages of folliculogenesis
Ensures only the best‑supported follicle survives
Most follicles undergo Atresia
The majority of follicles will not ovulate.
They undergo atresia (programmed cell death).
Atretic follicles are removed from the ovary.
Atresia can occur at any stage of folliculogenesis.
Cystic Follicles (Pathological Fate)
A. Cystic Follicles in Cattle
Occur when follicles stop responding to normal hormonal signals.
Common in high‑producing dairy cows due to disrupted hypothalamic–pituitary–ovarian signaling.
Cystic follicles:
Do not ovulate.
Do not undergo atresia.
Persist on the ovary.
Hormone profile:
Low estrogen
Low progesterone
Defined as >25 mm in diameter.
A major reproductive management challenge in cattle.
B. Polycystic Ovarian Syndrome (PCOS) in Humans
Different phenotype from cattle.
Characterized by:
Many small recruited follicles that remain on the ovary.
Follicles fail to progress beyond early antral stage.
Strongly associated with metabolic dysregulation.
Results in irregular cycles and impaired ovulation.
Folliculogenesis Review (Recruitment → Selection → Dominance)
Follicular Pool
Includes:
Primordial follicles
Primary follicles
Secondary follicles
Once follicles reach the tertiary (antral) stage, they enter folliculogenesis:
Recruitment
Many follicles enter.
Low estrogen production.
Selection
Some follicles continue; many undergo atresia.
Selected follicles grow larger and produce more estrogen.
Dominance
In monovulatory species (humans, cattle, mares): one dominant follicle.
In litter‑bearing species: multiple co‑dominant follicles.
Dominant follicle produces very high estrogen.
Dominant Follicle & Ovulation Conditions
Dominant follicle can ovulate only if progesterone is low.
If progesterone is high (luteal phase):
Dominant follicle undergoes atresia.
A new wave of recruitment → selection → dominance begins
Follicle growth
Many recruited
Few selected
One dominant follicle
Where do the rest of the follicles go?
What about litter bearers?
Increase progesterone dominant follicle undergo atresia
Litter-Bearing Species
Species like pigs, dogs, cats:
Recruit more follicles.
More follicles survive selection.
Multiple follicles become co‑dominant → multiple ovulations.
Oocyte Status in Tertiary Follicles
Even in large tertiary follicles:
The oocyte is still a primary oocyte.
Still arrested in meiosis I.
Oocyte reaches maximum size in the tertiary stage.
Surrounded by the cumulus–oocyte complex (COC).
This entire complex is expelled at ovulation.
Waves of follicular growth
Occur in patterns
Ovulatory vs. non-ovulatory
Dominant vs. subordinant
Signals
LH
FSH
Inhibin
Follicles grow in waves throughout the cycle.
Luteal phase:
High progesterone suppresses follicular progression.
Follicular phase:
Progesterone drops → follicle growth accelerates.
Waves repeat until:
A dominant follicle ovulates
ORProgesterone rises again and the dominant follicle undergoes atresia.
Follicular Waves Occur Only in Tertiary (Antral) Follicles
At this point, we are focusing only on tertiary follicles.
These follicles grow in waves throughout the estrous cycle.
Waves occur in two types of periods:
Obligatory period
Non‑obligatory period
Obligatory vs. Non‑Obligatory Periods
Non‑Obligatory Period
Occurs when progesterone is high (luteal phase).
High progesterone:
Blocks the surge center in the hypothalamus.
Prevents the LH surge.
Without an LH surge:
Dominant follicles cannot ovulate.
They undergo atresia.
Obligatory Period
Occurs when progesterone is low.
Low progesterone:
Removes inhibition on the surge center.
Allows estrogen from the dominant follicle to activate the surge center.
Leads to the LH surge → ovulation.
First wave
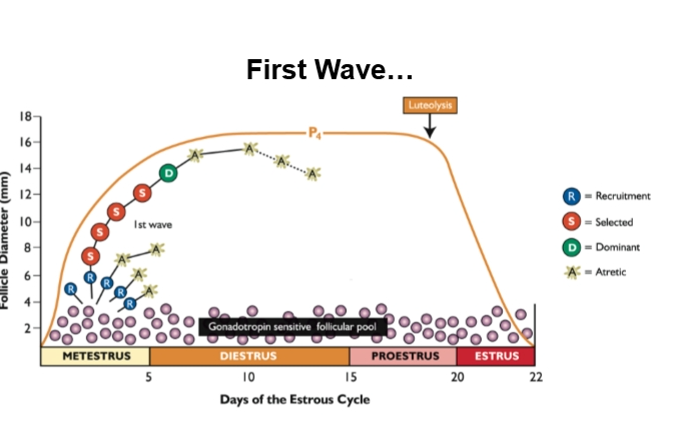
Second Wave
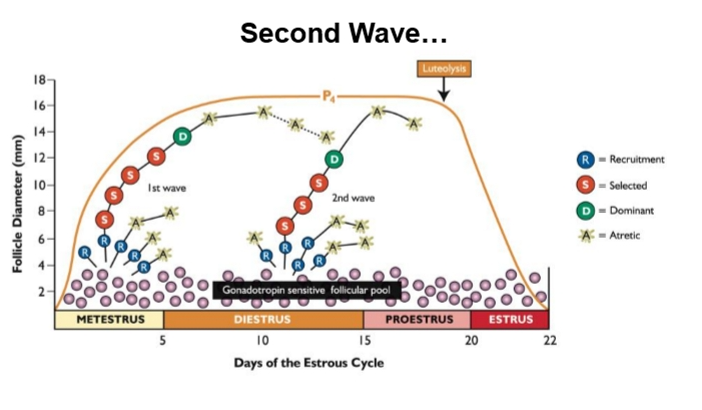
Follicle Categories Within a Wave
Recruited follicles
Many enter; most die.
Selected follicles
Survive initial refinement; grow larger.
Dominant follicle
One per wave in monovulatory species.
Suppresses subordinate follicles.
Only ovulates if progesterone is low.
Subordinate follicles = all recruited/selected follicles that are not dominant.
Hormonal Regulation During Waves
Key regulators
FSH
LH
Inhibin (from dominant follicle)
Why no Activin here?
Dominant follicles produce inhibin, not activin.
Inhibin suppresses FSH, preventing new follicles from rising to dominance.
First Follicular Wave After Ovulation
Ovulation → corpus luteum forms → progesterone rises.
High progesterone = non‑obligatory wave.
Follicles still undergo:
Recruitment
Selection
Dominance
BUT the dominant follicle cannot ovulate → undergoes atresia.
Real Estate Effect
Dominant follicles take up physical space on the ovary.
When a dominant follicle undergoes atresia:
Space opens up.
Allows new recruited follicles to grow.
This is why waves repeat.
Second Follicular Wave
Same pattern:
Recruitment → selection → dominance.
Progesterone is still high.
So this wave is also non‑obligatory.
Dominant follicle again undergoes atresia.
Signals for follicle growth
Follicle stimulating Hormone
Granulosa cell mitosis
Increases LH receptors
Steroidogenesis (increase E2)
Effects preantral & antral follicles
Luteinizing Hormone
Steroidogenesis
Effect antral follicles
Surge starts ovulation
Resumption of meiosis -oocyte
How FSH and LH Drive Follicle Growth
FSH
Stimulates mitosis in granulosa cells.
More granulosa cells → more estrogen.
FSH also increases LH receptors on granulosa cells.
This is crucial for later LH responsiveness.
LH
Acts on theca cells → produces testosterone.
Testosterone diffuses into granulosa cells → aromatized to estrogen.
LH is also required for ovulation once the surge occurs.
Transition to the Ovulatory Wave
In a three‑wave cow, the third wave occurs when:
Progesterone is falling (luteolysis).
Low progesterone:
Removes inhibition on the surge center.
Estrogen from the dominant follicle activates the surge center.
LH surge occurs.
Dominant follicle becomes pre‑ovulatory (Graafian).
Ovulation happens.
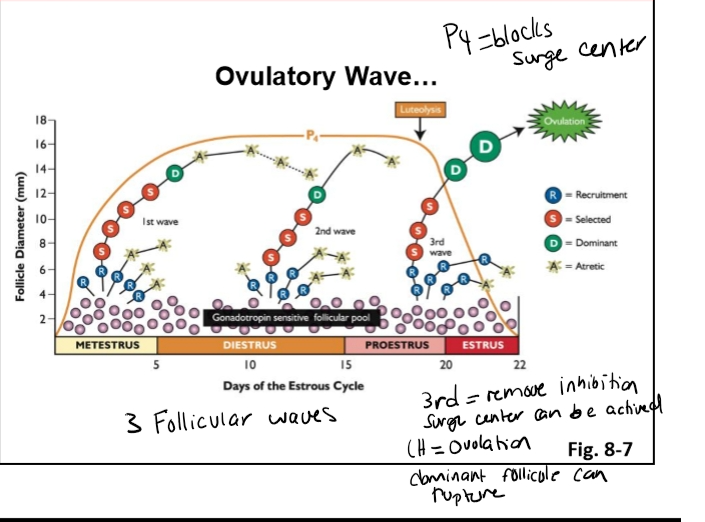
How Many Waves Do Cows Have?
Cows can have 1–6 waves per estrous cycle.
Most commonly: 2 or 3 waves.
Three‑wave cows are more fertile than two‑wave cows.
Why?
In two‑wave cows:
The dominant follicle remains dominant longer.
It is exposed to high progesterone for a longer time.
This prolonged exposure → follicle aging → reduced fertility.
Dominant Follicle Suppression of Subordinates
Dominant follicle produces:
Estrogen
Inhibin
These suppress FSH.
Low FSH prevents subordinate follicles from:
Growing
Becoming co‑dominant
Competing for dominance
This ensures only one follicle reaches ovulatory size in nonovulatory species.
How the Dominant Follicle Suppresses Subordinate Follicles
The dominant follicle actively prevents other follicles from continuing growth. It does this in three major ways:
A. Physical Suppression
The dominant follicle is large and takes up significant space on the ovary.
This physically limits the ability of subordinate follicles to grow.
B. Hormonal Resource Competition
The dominant follicle has:
More granulosa cells
More theca cells
More receptors for FSH and LH
Because of this, it absorbs most of the gonadotropins delivered to the ovary.
Subordinate follicles receive less FSH and LH, so they cannot continue developing.
C. Inhibin Production
Dominant follicles produce high levels of inhibin.
Inhibin suppresses FSH at the anterior pituitary.
FSH is critical for early recruited follicles.
Low FSH → subordinate follicles undergo atresia.
Follicular Waves in a Three‑Wave Cow
Wave 1 (Yellow Wave)
Recruitment → selection → dominance.
Progesterone is high → dominant follicle cannot ovulate.
Dominant follicle undergoes atresia.
Wave 2
Same pattern: recruitment → selection → dominance.
Progesterone still high → dominant follicle again undergoes atresia.
Wave 3 (Pink Wave)
Recruitment → selection → dominance.
Progesterone decreases due to luteolysis.
Dominant follicle now has the opportunity to:
Activate the surge center
Trigger the LH surge
Ovulate
Inhibit follicle growth
Presence of a dominant follicle
Inadequate production of gonadotropins
Steroids
Estrogen and androgens
Inhibin
Protein produced by follicles which inhibits other follicles from developing by decreasing FSH release
Follicular growth
growth
plateau
regress?
ovulate?
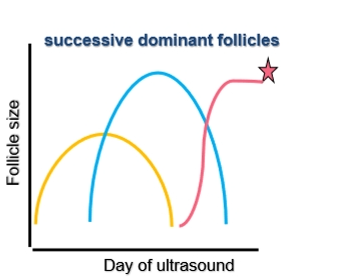
Endocrine Control of Follicular Waves
A medical‑textbook style diagram summarizes the logic:
Step 1: Hypothalamus
Releases GnRH.
Step 2: Anterior Pituitary
Releases FSH and LH in response to GnRH.
Step 3: Ovary
FSH and LH stimulate follicular growth.
Granulosa cells proliferate → estrogen increases.
If progesterone is low, high estrogen activates the surge center → LH surge.
Step 4: Ovulation
LH surge triggers:
Rupture of the dominant follicle
Release of the cumulus–oocyte complex (COC)
Remaining cells differentiate into the corpus luteum (CL)
Hormonal regulation
Hormonal Dependency Across Follicular Stages
A. Recruited Follicles
Highly dependent on FSH
FSH stimulates:
Granulosa cell mitosis
Aromatase expression
Early estrogen production
LH is less important but still needed for testosterone production.
B. Selected Follicles
Begin shifting from FSH‑dependent → LH‑dependent
Why?
FSH increases LH receptors on granulosa cells.
Estrogen production increases.
Inhibin begins to rise → suppresses FSH.
C. Dominant Follicle
Strongly LH‑dependent
Minimal reliance on FSH.
Produces:
Very high estrogen
Very high inhibin
Inhibin suppresses FSH → prevents new follicles from rising.
Application: Superstimulation / Superovulation
If you want to increase the number of oocytes a prized animal produces:
Use exogenous FSH
Why?
FSH keeps recruited follicles alive.
Prevents atresia.
Allows more follicles to continue through selection and dominance.
This is the basis of superstimulation protocols used in:
Cattle
Sheep
Goats
Embryo transfer programs
Why not use LH?
LH targets later‑stage follicles.
It does not rescue early recruited follicles.
To increase oocyte number, you must support the recruited pool, not the dominant follicle.
Transition to Ovulation
The pre‑ovulatory (Graafian) follicle contains:
Cumulus–oocyte complex
Mural granulosa cells
Theca interna + externa
LH surge triggers:
COC expulsion
Collapse of remaining follicular cells
Luteinization → formation of the corpus luteum
Question
What would you do if you were a producer and you wanted to increase the number of ova produced by your prize cow?
Ovulation Requires an LH Surge (Across All Mammals)
All mammalian species—spontaneous ovulators (humans, cows, mares) and induced ovulators (cats, rabbits)—require a luteinizing hormone (LH) surge for ovulation.
LH surge = universal trigger for follicular rupture.
What about FSH?
FSH also increases during the surge.
Its exact role in ovulation is not fully understood.
It may help prime small follicles in earlier waves.
But LH is the true driver of ovulation.
Ovulation = Pressure + Wall Weakening
Your professor’s analogy:
Ovulation is like popping a pimple.
You need:
Pressure pushing outward
Weakening of the follicular wall
The LH surge initiates both processes.
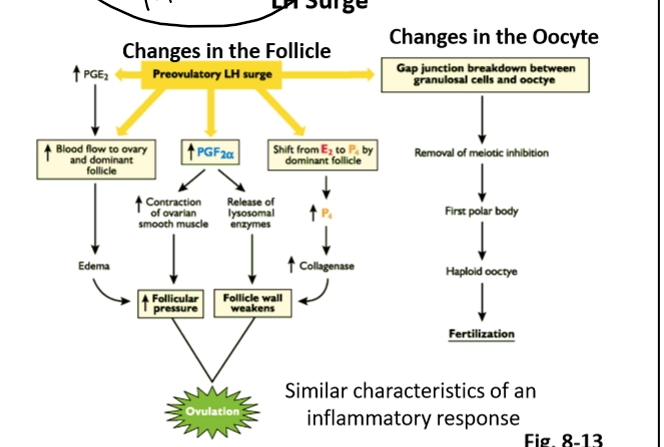
Mechanism 1: Increasing Pressure on the Follicle
A. Prostaglandin E2 (PGE2)
LH surge → ↑ PGE2 (from the ovary).
PGE2 is a vasodilator.
Effects:
↑ blood flow to the ovary
↑ fluid accumulation
Edema forms around the follicle
Pressure increases on the follicular wall
This swelling helps “push” the follicle toward rupture.
B. Prostaglandin F2α (PGF2α)
LH surge → ↑ ovarian PGF2α (not uterine).
PGF2α causes smooth muscle contractions in the ovary.
These contractions:
Squeeze the follicle
Add mechanical pressure
Help force the oocyte outward
PGF2α also has a second role (see below).
Mechanism 2: Weakening the Follicular Wall
A. PGF2α → Lysosomal Enzyme Release
PGF2α triggers release of lysosomal enzymes from:
Theca cells
Granulosa cells
These enzymes:
Digest components of the follicular wall
Break down cellular junctions
Soften the tissue
This makes the follicle easier to rupture under pressure.
B. LH Surge → ↑ Progesterone → ↑ Collagenase
LH surge stimulates granulosa + theca cells to produce progesterone.
Progesterone increases collagenase activity.
Collagenase:
Breaks down collagen in the extracellular matrix
Weakens the follicular wall
Helps create the rupture point
Ovulation Resembles an Inflammatory Response
Ovulation shares features with inflammation:
Increased blood flow
Edema
Tissue breakdown
Enzyme release
Structural remodeling
This is why the ovary becomes red, swollen, and structurally altered at the ovulation site.
LH Surge Effects on the Oocyte
The LH surge also acts directly on the cumulus–oocyte complex (COC):
Disconnects cumulus granulosa cells from the oocyte.
Removes meiotic inhibition.
Allows the oocyte to transition:
Primary oocyte → Secondary oocyte (completes meiosis I)
If fertilization occurs:
Secondary oocyte completes meiosis II → haploid ovum
This prepares the oocyte for fertilization.
The Physical Act of Ovulation
The follicle ruptures at the weakened spot.
Follicular fluid + COC are expelled into the peritoneal cavity.
The fimbriae of the oviduct sweep the COC into the oviduct.
A 2008 surgical observation captured this process in real time:
The “egg” seen was actually follicular fluid, not the oocyte (too small to see with the naked eye).
After Ovulation: Luteinization
Remaining granulosa + theca cells collapse inward.
They undergo terminal differentiation.
This forms the corpus luteum (CL).
CL produces progesterone, which:
Supports pregnancy
Suppresses new LH surges
Prevents additional ovulations
A preovulatory follicle has several key structural components:
Cell Layers & Their Functions
Structure | Color (from lecture) | Function |
Theca cells | Light green | Produce testosterone (precursor for estrogen). |
Granulosa cells (mural) | Natural/regular green | Convert testosterone → estrogen. |
Cumulus granulosa cells | Red | Surround the oocyte; form the cumulus–oocyte complex (COC). |
Oocyte | Center | The egg cell that will be ovulated. |
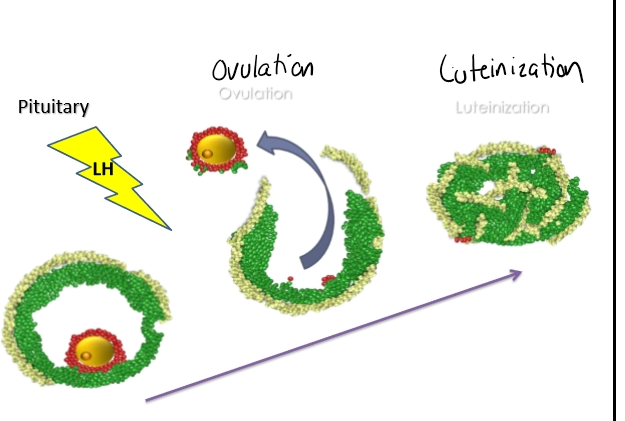
Ovulation
Stimuli
LH surge
FSH
Specific cascade of events
Degradation of follicle wall
Extrusion of oocyte
Resumption of meiosis
Collapse of the follicle
LH Surge
Hormonal Conditions Leading to Ovulation
High estrogen + decreasing progesterone → positive feedback on the surge center.
This triggers the LH surge.
LH surge = the key signal that initiates follicular rupture.
Events Required for Ovulation
To ovulate, the follicle must:
A. Increase pressure inside the follicle
Fluid accumulation in the antrum.
Swelling pushes outward on the follicular wall.
B. Weaken the follicular wall
Enzymatic digestion.
Breakdown of connective tissue.
Lysosomal enzymes from theca + granulosa cells help digest the wall.
Outcome
→ Rupture of the follicle
→ Expulsion of the cumulus–oocyte complex (COC)
After Ovulation: What Happens to the Follicle?
The remaining cells (theca + granulosa + some cumulus cells) collapse inward.
This process forms the corpus luteum, but it happens in stages:
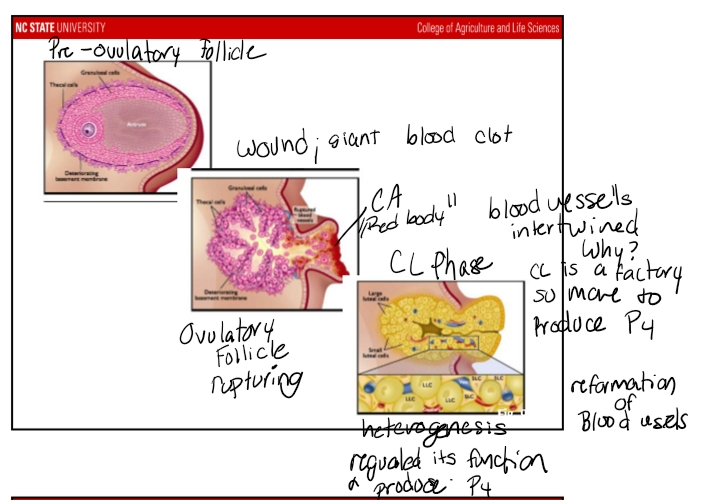
Stage 1: Corpus Hemorrhagicum (CH)
Immediately after rupture, blood vessels tear.
Creates a wound-like structure filled with blood.
Appears as a red body protruding from the ovary.
Duration: hours to 1–2 days, depending on species.
Luteal Phase
Corpora lutea formation
Luteinization
Progesterone production
Longer phase
66-75% of cycle
Ends with luteolysis
Work with uterus to aid with embryo
Luteolysis= CL dies and decrease in P4
Luteinization
Process that transforms the granulosa and theca cells in luteal cells. Undergo terminal differtiation → into luteal cells
Triggered by the surge of LH
Causes profound changes in the follicles that become corpora lutea: Disruption the tissue of ovary is damaged.
Definition:
The process where theca + granulosa cells undergo terminal differentiation into luteal cells.
Key points:
Triggered by the LH surge.
Terminal differentiation = once they become luteal cells, they cannot revert.
Cells reorganize, blood vessels regrow, and the structure becomes the corpus luteum (CL).
The CL
A fully formed CL is:
A heterogeneous tissue containing:
Large luteal cells
Small luteal cells
Endothelial cells (forming a dense capillary network)
Fibroblasts
Mast cells
Immune cells
All of these contribute to CL function and maintenance.
Pre-ovulatory Follicle- → Ovulatory → CL
🔬 6. Origin of Large vs. Small Luteal Cells
Based on a classic (and rarely replicated) antibody‑labeling study:
Luteal Cell Type | Origin |
Small luteal cells | Theca cells |
Large luteal cells | Granulosa cells |
This remains the accepted dogma, though the original study had limitations and has not been repeated due to difficulty.
🌙 7. Function of the Corpus Luteum
The primary function of the CL:
→ Produce progesterone
Progesterone:
Maintains pregnancy (if it occurs)
Suppresses LH/FSH
Prevents new follicular waves from ovulating
Prepares the uterus
📈 8. The Luteal Phase in the Estrous Cycle
The luteal phase begins at metestrus, right after ovulation.
Key features:
Progesterone rises as the CL forms.
The process of CL formation = luteinization.
The longest phase of the estrous cycle:
66–75% of the cycle.
CL remains until it receives the signal for luteolysis (death of the CL).
After luteolysis:
Progesterone drops sharply.
The cycle transitions back toward the follicular phase.