Chapter 3 Enzymes
0.0(0)
0.0(0)
Card Sorting
1/21
Study Analytics
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
22 Terms
1
New cards
Enzymes
enzymes or biological catalyst are proteins that speed up chemical reactions.
enzymes help to increase the rate of the reaction without being used up.
they are sensitive molecules, specific, are not used up in reactions, can sometimes work in both directions of a reaction and can catalyst each step of entire metabolic pathways.
enzymes help to increase the rate of the reaction without being used up.
they are sensitive molecules, specific, are not used up in reactions, can sometimes work in both directions of a reaction and can catalyst each step of entire metabolic pathways.
2
New cards
substrate
the reactant of a reaction catalysed by an enzyme.
* Enzymes and substrate (reactant) bind via the active site
* Substrate then undergoes conformational change to become the product and is released
* Enzyme is unchanged and can be reused
* Enzymes and substrate (reactant) bind via the active site
* Substrate then undergoes conformational change to become the product and is released
* Enzyme is unchanged and can be reused
3
New cards
Features of enzymes
1. reusable - when an enzyme catalyses a reaction the enzyme is not used up, they can then catalyse future reactions.
2. specific - most enzymes only bind to one specific substrate, which means they tend to catalyse just one chemical reaction.
3. reversible
4. speed up, not create
5. have an active site
6. are proteins - most enzymes are proteins, not all.
7. are a subset of catalysts
8. act on entire biochemical pathways
9. end in ‘-ase’ - enzyme names typically end with the suffix ‘-ase’ like catalyse, polymerase, ligase, lactase
10. above the arrow
4
New cards
active site
the part of an enzyme where the substrate binds.
when a substrate binds to an enzyme’s active site, together they form an enzyme-substrate complex (the structure formed when an enzyme and substrate are bound together.)
many chemical bonds including hydrogen bonds or hydrophobic interactions hold the substrate and active site together in the enzyme-substrate complex.
when a substrate binds to an enzyme’s active site, together they form an enzyme-substrate complex (the structure formed when an enzyme and substrate are bound together.)
many chemical bonds including hydrogen bonds or hydrophobic interactions hold the substrate and active site together in the enzyme-substrate complex.
5
New cards
conformational change
a change in the three-dimensional shape of macromolecules such as proteins.
upon binding, the active undergoes a conformational change to accomodate the substrate, and the substrate undergoes a small change in turn.
upon binding, the active undergoes a conformational change to accomodate the substrate, and the substrate undergoes a small change in turn.
6
New cards
lock and key vs induced fit model
* Enzymes are highly specific to what substrates they can attach to and hence which biochemical reactions they can catalyse
* Lock and key model = the substrate exactly fits the active site of the enzyme
* Induced fit = a slight adjustment is needed upon the substrate binding for a better fit
* Induced fit is the currently accepted model
* Induced fit model states that an enzyme undergoes a conformational change to become complementary in shape to the substrate.
* Lock and key model = the substrate exactly fits the active site of the enzyme
* Induced fit = a slight adjustment is needed upon the substrate binding for a better fit
* Induced fit is the currently accepted model
* Induced fit model states that an enzyme undergoes a conformational change to become complementary in shape to the substrate.
7
New cards
activation energy
the energy required by a reaction for it to proceed.
enzymes function by lowering the activation energy required by a reaction
* catabolic reaction = larger molecule breaking down into smaller molecules
* Anabolic reaction = smaller molecules joining to form a larger molecule
enzymes function by lowering the activation energy required by a reaction
* catabolic reaction = larger molecule breaking down into smaller molecules
* Anabolic reaction = smaller molecules joining to form a larger molecule
8
New cards
factors that affect enzymes
* the general factors that impact on enzyme function in relation to photosynthesis and cellular respiration: changes in **temperature, pH, concentration, competitive and non-competitive enzyme inhibitors**
* They operate the best in optimal conditions, which are different for every enzyme
* They operate the best in optimal conditions, which are different for every enzyme
9
New cards
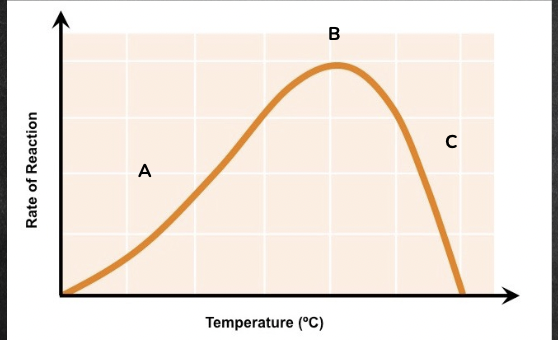
temperature
* A = as the temperature increases, the enzymatic activity also increases (because more heat means more kinetic energy for successful collisions between enzyme and substrate)
* B = optimum temperature (temperature at which the enzyme functions optimally)
* C = when the temperature exceeds the optimum, the enzyme (which is a protein) begins to denature, undergoes a conformational change and is no longer functional, hence the enzymatic activity decreases
the enzymes found within the human body have an optimal temperature range of 36 - 38 degree Celsius, meaning they function most effectively at the human body temperature of 37 degree Celsius.
* B = optimum temperature (temperature at which the enzyme functions optimally)
* C = when the temperature exceeds the optimum, the enzyme (which is a protein) begins to denature, undergoes a conformational change and is no longer functional, hence the enzymatic activity decreases
the enzymes found within the human body have an optimal temperature range of 36 - 38 degree Celsius, meaning they function most effectively at the human body temperature of 37 degree Celsius.
10
New cards
denaturation
the disruption of a molecule’s structure by an external factor such as heat.
* Broken bonds result in a conformational change
* Active site is no longer specific, enzyme reaction cannot occur
* Broken bonds result in a conformational change
* Active site is no longer specific, enzyme reaction cannot occur
11
New cards
What happens when it is too hot?
If the temperature goes beyond its optimal, an enzyme is at risk of denaturing, where the bonds that create its tertiary and quaternary structures are broken down.
denaturation causes a conformational change in the active site of an enzyme, causing the substrate to no longer it. this change is irreversible meaning that a denatured enzyme cannot regain its function even if the temperature decreases.
denaturation causes a conformational change in the active site of an enzyme, causing the substrate to no longer it. this change is irreversible meaning that a denatured enzyme cannot regain its function even if the temperature decreases.
12
New cards
what happens when it’s too cold?
An enzyme’s activity decreases below the optimal temperature, as the enzyme and substrate molecules move slower and collide less frequently.
When it becomes too cold, enzymes experience little to no activity and can freeze.
Enzymes can regain functionality when reheated as significant denaturation does not occur at low temperatures.
When it becomes too cold, enzymes experience little to no activity and can freeze.
Enzymes can regain functionality when reheated as significant denaturation does not occur at low temperatures.
13
New cards
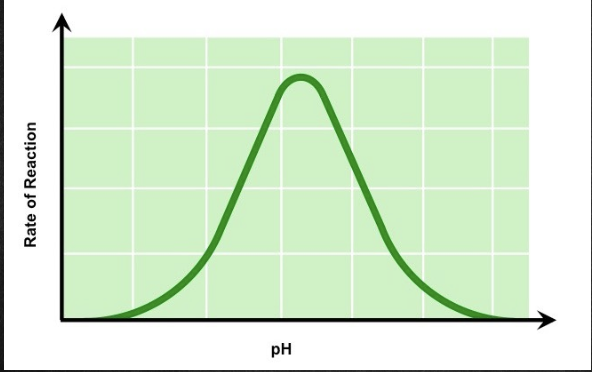
pH
The activity of an enzyme is affected by pH. When the pH becomes too acidic or basic for an enzyme, it can denature.
* Denaturation due to pH occurs when the pH is too high AND too low, which results in a bell curve
* Optimum pH for enzymes in the human body can be very different
* Denaturation due to pH occurs when the pH is too high AND too low, which results in a bell curve
* Optimum pH for enzymes in the human body can be very different
14
New cards
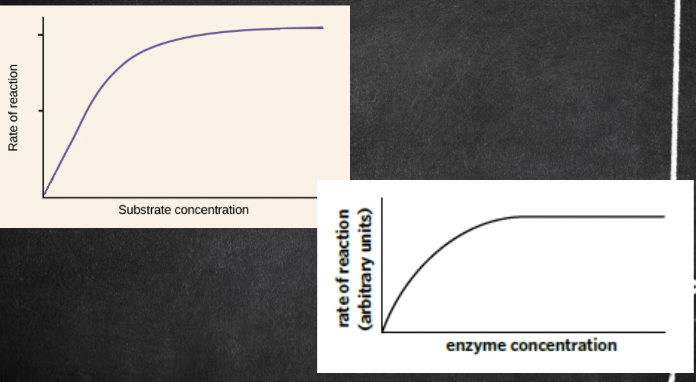
Concentration
The concentrations of both substrate and enzyme molecules influence the overall reaction rate.
15
New cards
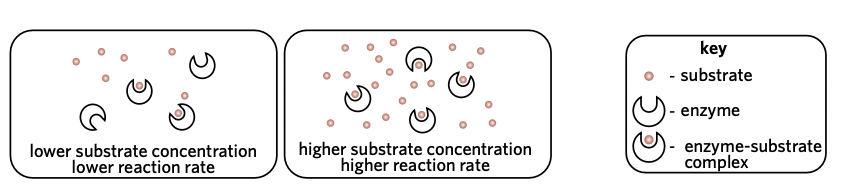
Substrate concentration
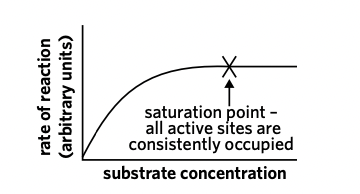
If the enzyme concentration remains constant while the substrate concentration increases, then the reaction rate will increase.
* As substrate concentration increases, the rate of reaction will also increase UP TO A CERTAIN POINT (saturation point)
* At the saturation point, all enzyme active site are occupied by the substrate, so the rate cannot increase any more
* This is shown by the graph plateauing/flattening
* As substrate concentration increases, the rate of reaction will also increase UP TO A CERTAIN POINT (saturation point)
* At the saturation point, all enzyme active site are occupied by the substrate, so the rate cannot increase any more
* This is shown by the graph plateauing/flattening
16
New cards
Saturation point
the point at which a substance cannot receive more of another substance.
17
New cards
enzyme concentration
* As enzyme concentration increases, the rate of reaction will also increase, as there is the large number of active sites available for the substrate to bind in.
* Once again, only up to a certain point
* Eventually there will be so many enzymes that they will be in excess and the graph plateaus
* Once again, only up to a certain point
* Eventually there will be so many enzymes that they will be in excess and the graph plateaus
18
New cards
Inhibitions
* Enzyme inhibitors are molecules that bind to an enzyme and prevent it from performing its function.
* When an inhibitor is bound to an enzyme, the enzyme can either no longer catalyse its specific reaction, or its functioning is greatly reduced.
* Can be competitive or non-competitive
* When an inhibitor is bound to an enzyme, the enzyme can either no longer catalyse its specific reaction, or its functioning is greatly reduced.
* Can be competitive or non-competitive
19
New cards
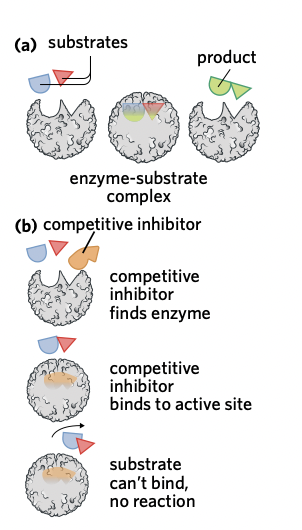
Competitive inhibition
Competitive inhibition occurs when an inhibitor molecule binds to an enzyme’s active site.
This binding directly occupies and blocks the active site, meaning the substrate is now unable to bind with the enzyme and no reaction will occur.
This binding directly occupies and blocks the active site, meaning the substrate is now unable to bind with the enzyme and no reaction will occur.
20
New cards
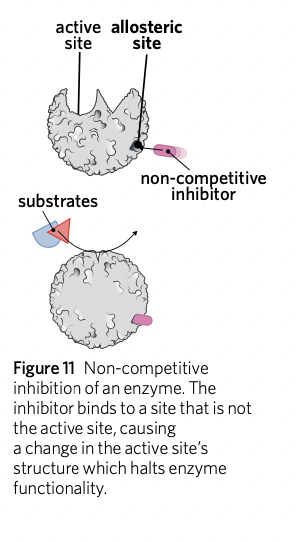
Non-competitive inhibition
Non-competitive inhibition (also known as allosteric inhibition) occurs when an inhibitor binds to an enzyme at a site other than the active site (an allosteric site).
This binding causes a conformational change in the active site of the enzyme. \n The conformational change in the active site’s structure prevents the substrate from binding to it, preventing the reaction from occurring
This binding causes a conformational change in the active site of the enzyme. \n The conformational change in the active site’s structure prevents the substrate from binding to it, preventing the reaction from occurring
21
New cards
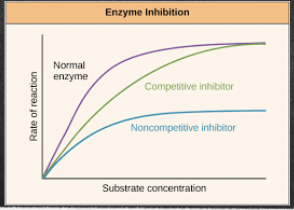
competitive and non-competitive
* both forms of inhibition result in decreased enzyme activity, as the enzyme will no longer be able to catalyse the reaction
22
New cards