Qualitative Analysis of Metallic and Non-metallic Ions
1/21
Earn XP
Description and Tags
CHEM182 - Analytical Chemistry (Lab)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Sodium ion, Na+
Yellow flame

Calcium ion, Ca2+
Brick red or Orange flame

Cupric ion, Cu2+ in CuSO4
Green flame

Cupric ion, Cu2 + in Cu(NO3)2 or CuCl2
Blue flame

Strontium ions, Sr+2
Red flame

Potassium ion, K+
Purple or lavender flame

Magnesium ion, Mg2+
White flame
Barium ion, Ba2+
Light green flame

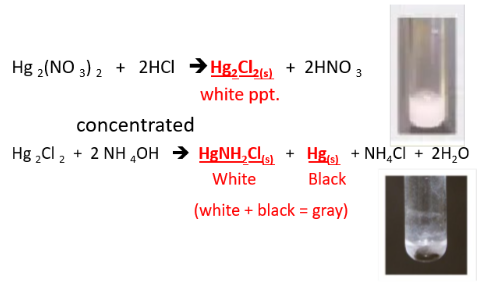
METALLIC IONS: Hg1+ (mercurous ion)
REAGENTS USE:
- HCl
- (concentrated) NH4OH
POSITIVE RESULTS:
- white ppt.
- white & black ppt/ gray ppt
SUBSTANCE FORM:
- HgCl or Hg2Cl2 mercury (I) chloride
- HgNH2Cl (white), diamine mercury (I) chloride, Hg (black) mercury metal
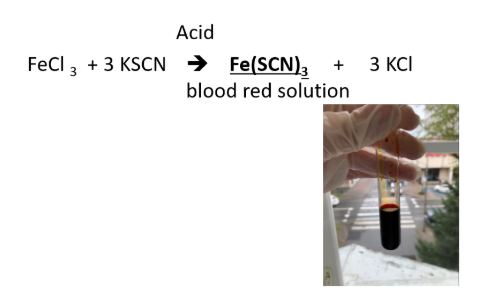
METALLIC IONS: Fe3+ (ferric ion)
REAGENTS USE:
- KSCN
POSITIVE RESULTS:
- blood red solution
SUBSTANCE FORM:
- Fe(SCN)3, ferric thiocyanate

METALLIC IONS: Cu2+ (cupric ion)
REAGENTS USE:
- NH4OH
- (concentrated) NH4OH
POSITIVE RESULTS:
- light blue ppt
- dark blue solution
SUBSTANCE FORM:
- Cu(OH)2, copper (II) hydroxide
- Cu(NH3)4(OH)2, tetraammine copper (II) hydroxide
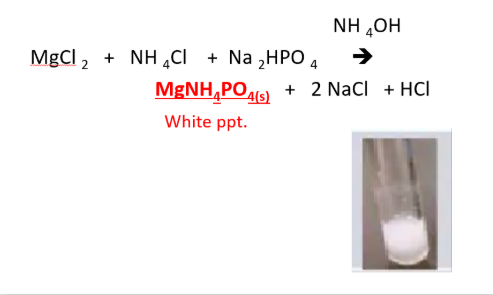
METALLIC IONS: Mg2+ (magnesium ion)
REAGENTS USE:
- NH4Cl
- Na2HPO4
POSITIVE RESULTS:
- white ppt
SUBSTANCE FORM:
- MgNH4PO4, magnesium ammonium phosphate

METALLIC IONS: Ca2+ (calcium ion)
REAGENTS USE:
- (NH4)2C2O4
POSITIVE RESULTS:
- white precipitate (most white precipitate forms)
SUBSTANCE FORM:
- CaC2O4, calcium oxalate

METALLIC IONS: Ag1+ (silver ion)
REAGENTS USE:
- HCl
- (concentrated) NH4OH
POSITIVE RESULTS:
- white precipitate
- colorless (soluble in NH4OH)
SUBSTANCE FORM:
- AgCl silver chloride
- Ag(NH3)2Cl, diammine silver chloride
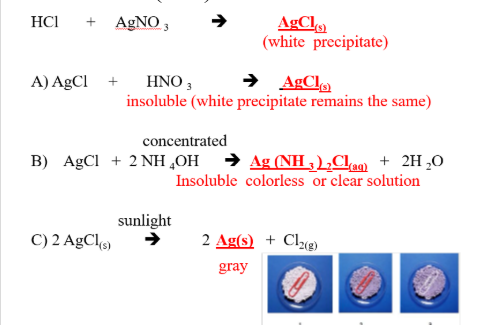
NON-METALLIC IONS: Cl–1 (chloride)
REAGENTS USE:
- AgNO3
- HNO3
- concentrated NH4OH
- Sunlight
POSITIVE RESULTS:
- white precipitate
- white precipitate (Insoluble in nitric acid)
- Soluble in ammonium hydroxide
- color of precipitate changed to gray
SUBSTANCE FORM:
- AgCl (s), Silver chloride
- AgCl (s), Silver chloride
- Ag(NH3)2Cl, diamino silver chloride
- Ag (s), silver metal
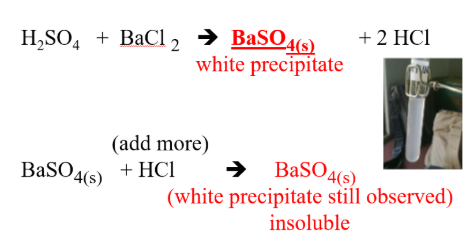
NON-METALLIC IONS: SO4–2 (sulfate)
REAGENTS USE:
- BaCl2
- HCl
POSITIVE RESULTS:
- white precipitate
- insoluble in hydrochloric acid
SUBSTANCE FORM:
- BaSO4(s), barium sulfate
- BaSO4(s), barium sulfate
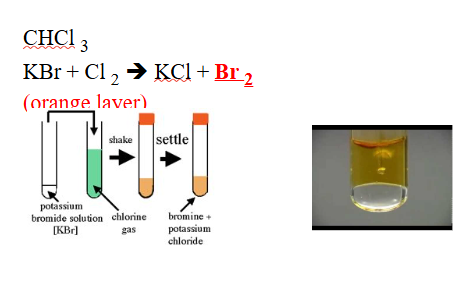
NON-METALLIC IONS: Br–1 (bromide)
REAGENTS USE:
- Cl2
- CHCl3
POSITIVE RESULTS:
- Soluble in Cl2
- Formation of an orange layer
SUBSTANCE FORM:
- Br2, bromine
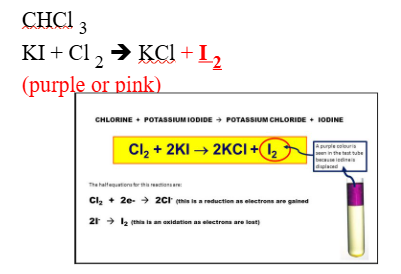
NON-METALLIC IONS: I–l (iodide)
REAGENTS USE:
- Cl2
- CHCl3
POSITIVE RESULTS:
- Soluble in Cl2
- Formation of purple layer
SUBSTANCE FORM:
- I2, iodine

NON-METALLIC IONS: PO4–3 (phosphate)
REAGENTS USE:
- (NH4)2MoO4 + HNO3
POSITIVE RESULTS:
- yellow precipitate
SUBSTANCE FORM:
- (NH4)3PO4 • 12MoO3, ammonium phosphomolybdate
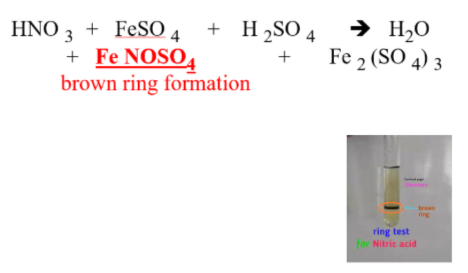
NON-METALLIC IONS: NO3–1 (nitrate)
REAGENTS USE:
- FeSO4 + H2SO4
POSITIVE RESULTS:
- brown ring formation
SUBSTANCE FORM:
- FeNOSO4, ferrous nitrosyl sulfate
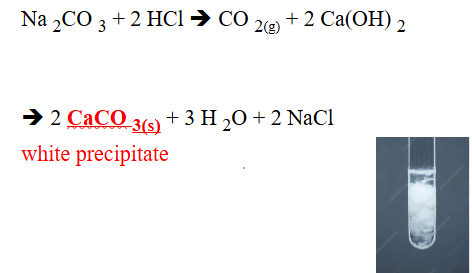
NON-METALLIC IONS: CO3–2 (carbonate)
REAGENTS USE:
- HCl
- Ca(OH)2
POSITIVE RESULTS:
- Evolution of CO2 gas
- white precipi
SUBSTANCE FORM:
- CO2, Carbon dixide
- CaCO3, calcium carbonate

NON-METALLIC IONS: C2O4 –2 (oxalate)
REAGENTS USE:
- CaCl2
POSITIVE RESULTS:
- white ppt
SUBSTANCE FORM:
- CaC2O4, calcium oxalate