29 - intro to a level organic chem
1/38
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
39 Terms
functional group
an atom or group of atoms in an organic molecule, that determines its characteristic chemical and physical properties
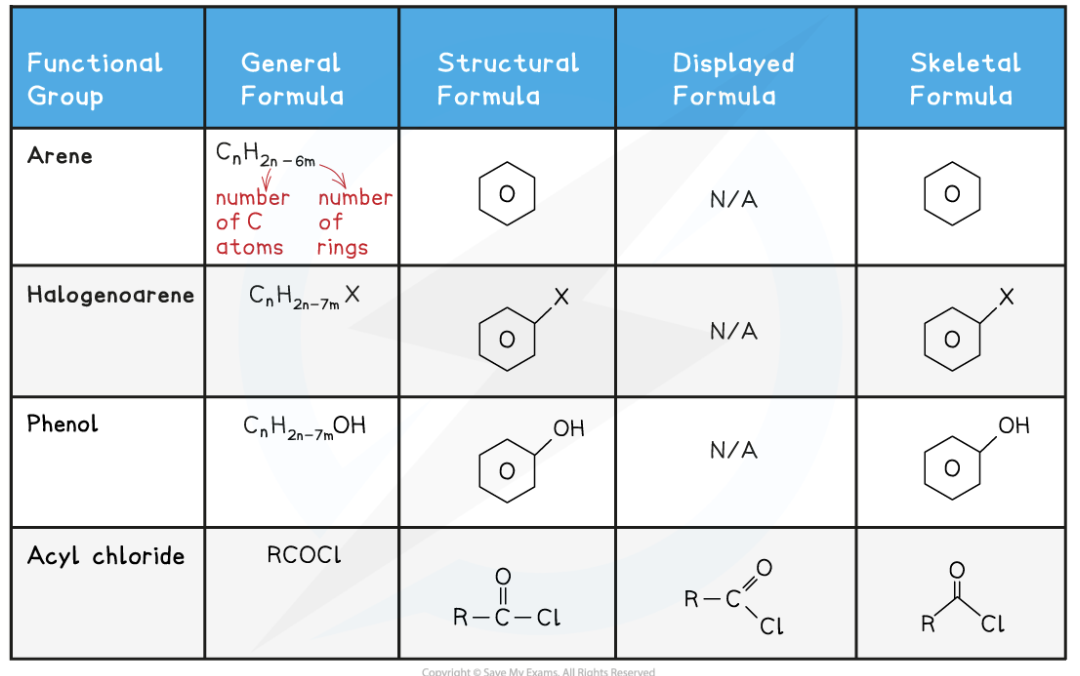
arenes
aromatic compounds that contain a benzene ring
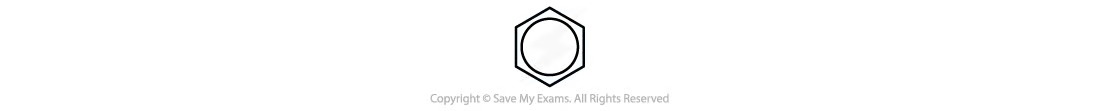
|→ the arena functional group
aromatic compounds
the general term aromatic is used to describe non-hydrocarbon compounds (eg - ketones, carboxylic acids) that contain a benzene ring (some of the earliest discovered aromatic compounds had pleasant odors - hence the name). this is to contrast them from aliphatic compounds (eg - ethanoic acid), that do not contain a benzene ring.
structure of benzene
a ring of six carbon atoms, each with a hydrogen atom attached. each carbon atom uses three of its valence electrons to form σ bonds with adjacent atoms, leaving one electron in a p orbital. rather than overlap in pairs to form normal c=c double bonds, these six p orbitals overlap to form one 6-centered delocalized π orbital, which is represented in the skeletal formula by a circle inside a hexagon.
Benzene has the molecular formula C6H6 and empirical formula CH
kekule structure for benzene
August Kekule , a German chemist proposed the following structure for benzene.
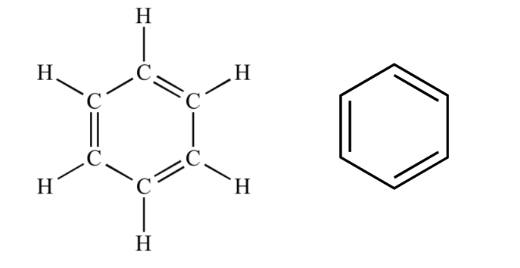
Kekule’s benzene molecule is based on a cyclic structure with alternate double bonds and single
bonds.
However, the Kekulé structure could not account for certain unexplained properties of benzene
The structure has three double bonds, so it should undergo addition reactions like other alkenes
do. However for real benzene, it does not undergo addition reactions and does not decolourise
bromine water.
Single and double bonds have different bond length, therefore Kekulé’s benzene should have a
distorted hexagonal shape. However in real benzene, all the C-C bonds are identical and it is a
perfect hexagon .
Bond lengths C – C : 0.154 nm , C = C : 0.133 nm , (Carbon – Carbon) benzene : 0.139 nm
shape of benzene
Benzene and other aromatic compounds contain sp2 hybridised carbons as two of their p orbitals have mixed with an s orbital
This means that each carbon atom in benzene and other aromatic compounds has one p orbital
sp2 hybridisation

The carbon atoms in aromatic compounds are sp2 hybridised as two of their p orbitals mix with an s orbital
Each carbon atom in the ring forms three σ bonds using the sp2 orbitals
The remaining p orbital overlaps laterally with p orbitals of neighbouring carbon atoms to form a π bond
This extensive sideways overlap of p orbitals results in the electrons being delocalised and able to freely spread over the entire ring
Benzene and other aromatic compounds are regular and planar compounds with bond angles of 120o
The delocalisation of electrons means that all of the carbon-carbon bonds in these compounds are identical and have both single and double bond character
The bonds all being the same length is evidence for the delocalised ring structure of benzene
The planar structure of benzene

Like other aromatic compounds, benzene has a planar structure due to the sp2hybridisation of carbon atoms and the conjugated π system in the ring
chemical and physical properties of arenes
Due to the delocalised electron ring (π system of electrons), these compounds are electron-rich and therefore can undergo electrophilic attack under the right conditions
However, because the delocalised electron ring system makes benzene so stable, it is resistant to addition reactions
This is very different to alkenes, which are very reactive and readily undergo addition reactions
Physical properties
Benzene has van der Waals dispersion forces of attraction between the molecules and has a boiling point of 80 C
The presence of the non-polar hydrocarbon part in the arene functional group means that these compounds are often insoluble in water
Benzene would have to break many hydrogen bonds between the water molecules to be soluble in water, which does not happen as it is not energetically favourable
halogenoarenes
These are aromatic compounds that contain a halogen bonded to a benzene ring
The halogenoarene functional group

They are also known as aryl halides
chemical and physical properties of halogenoarenes
Chemical properties
These compounds are also prone to electrophilic attack because of the π system of delocalised electrons
The halogens can also take part in substitution reactions
Physical properties
Chlorobenzene, bromobenzene and iodobenzene are all liquid at room temperature with an oily texture
As you might expect, the boiling points increase as the size of the halogen attached increases, because the number of electrons within the molecule increases
Like other arenes, halogenoarenes are insoluble in water because of the non-polar hydrocarbon part of the ring
These molecules are large relative to the size of water molecules, and as with the arenes it is not energetically favourable for the halogenoarene molecules to break the hydrogen bonds between the water molecules, so it does not happen
phenols
Phenols are another type of aromatic compounds containing a hydroxide bonded to a benzene ring
The phenol functional group

chemical and physical properties of phenols
Chemical properties
The -OH group in phenols is more acidic than in alcohols as the oxygen donates one of its lone pairs of electrons into the ring system
This causes an increased electron density of the ring, causing it to become much more reactive than benzene itself
It also makes it easier for the hydrogen of the -OH group to be donated
Phenols can also react with reactive metals such as sodium to form alkoxide ions
Physical properties
Phenol is a white, crystalline solid, and it has a disinfectant-like smell
Due to the -OH group in phenols, they can form hydrogen bonds with water molecules, and therefore to a degree phenol is soluble in water
acyl chlorides
cyl chlorides are (carboxylic) acid derivatives containing:
A chlorine atom attached to a C=O group (replacing what would have been the -OH group of a carboxylic acid)
An acyl (hydrocarbon) group attached to a C=O group
The acyl chloride functional group

Acyl chlorides are also known as 'acid' chlorides
chemical and physical properties of acyl chlorides
Chemical properties
They are fuming liquids and are colourless, with a strong smell
Acyl chlorides are extremely reactive and readily take part in substitution reactions in which the chlorine atom is substituted by other species
This reactivity is why they are fuming liquids and why they have such a strong smell - they react with any water vapour in the air
Physical properties
Acyl chlorides react violently with water, so we cannot say whether or not they would be soluble in water
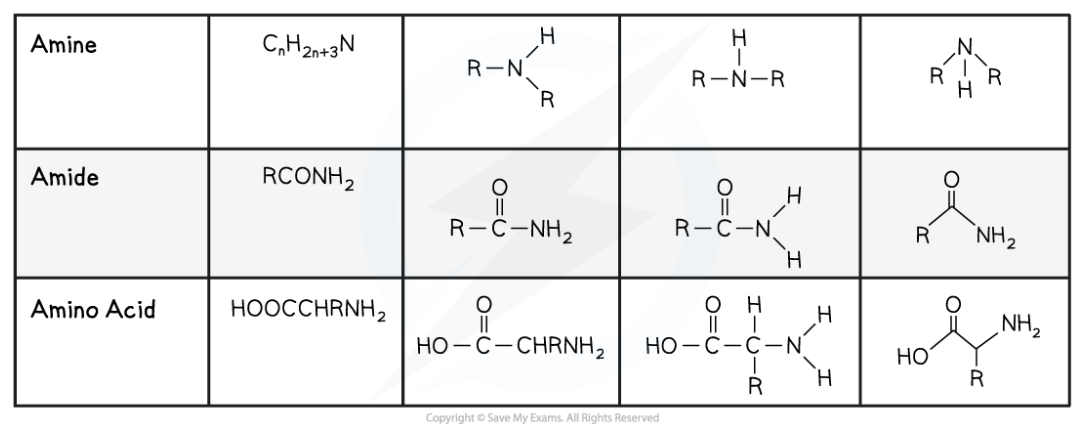
amines
Amines are compounds with the -NH2 (primary amine), -NH (secondary amine) or -N (tertiary amine) group
The amine functional group

Classification of amines
In primary amines, the N of the amine group is bonded to one R group (and two hydrogen atoms)
In secondary amines, the N of the amine group is bonded to two R groups (and one hydrogen atom)
In tertiary amines, the N of the amine group is bonded to three R groups
chemical and physical properties of amines
Chemical properties
Due to the lone pair of electrons on the nitrogen, amines are basic compounds
Physical properties
The lone pair on the N of the amine group means that they can form hydrogen bonds
They are often soluble in water because they form hydrogen bonds with water molecules
The smaller amines are very soluble in water, but their solubility decreases as the non-polar hydrocarbon chain gets longer
They often have a fishy smell, especially as the size of the amines increases
amides
Amides are compounds containing:
An amine (-NH2) group
A carbonyl group (C=O)
The amide group is -CONH2
The amide functional group

chemical and physical properties of amides
Chemical properties
Amides are less basic than amines, as the lone pair of electrons on the nitrogen is delocalised
Physical properties
Amides are often soluble in water as they can form hydrogen bonds with water molecules
The smaller amides are very soluble in water, but their solubility decreases as the non-polar hydrocarbon chain gets longer
amino acids
Amino acids are the building blocks of proteins and consists of:
An amine (-NH2) group
A carboxyl (-COOH) group
The α-amino acid functional group

chemical and physical properties of amino acids
Chemical properties
Amino acids react with bases to form salts
They also react with alcohols to form esters
The reaction of amino acids with amines gives amides
Physical properties
Most of the amino acids are soluble in water but insoluble in organic solvents
Amino acids have chiral centres and exhibit optical isomers (except for glycine)
formulae of organic compounds
Nomenclature of simple aliphatic organic molecules with functional groups table
Functional group | Example | Name |
|---|---|---|
Acyl chloride | 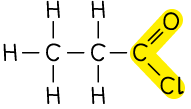 | propanoyl chloride |
Amine | 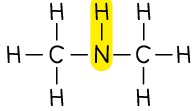 | dimethylamine |
Amide | 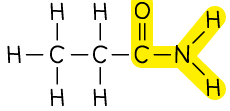 | propanamide |
Amino acid | 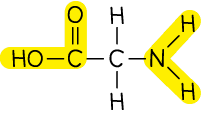 | 2-aminoethanoic acid |
nomenclature of aromatic compounds
Functional group | Example | Name |
|---|---|---|
Arene |

| Propyl benzene |
Chlorobenzene |

| 2-methylchlorobenzene |
Phenol
|  | 2,3-dimethyl phenol |
electrophilic substitution
Electrophiles are species that are electron deficient and can act as an electron pair acceptor
Electrophiles are ‘electron loving’ species
Substitution reactions are reactions that involve the replacement of one atom or group of atoms by another
Electrophilic substitution reactions are therefore reactions in which an atom or group of atoms are replaced by an electrophile after an initial attack by the electron-deficient species
An example of an electrophilic substitution reaction is the reaction of benzene with bromine in the presence of anhydrous aluminium bromide catalysts
The bromine acts as an electrophile and attacks the electron-rich benzene ring
A hydrogen atom is substituted by a bromine atom to form bromobenzene and hydrogen bromide
Benzene undergoes substitution reactions rather than addition reactions because of the stability of the benzene ring
Electrophilic substitution of benzene by bromine

A hydrogen atom in benzene is substituted by a bromine atom, which acts as an electrophile
addition elimination
Acyl chlorides are reactive organic compounds that undergo many reactions such as nucleophilic addition-elimination reactions
In nucleophilic addition-elimination reactions, the nucleophilic addition of a small molecule across the C=O bond takes place followed by elimination of a small molecule
Examples of these nucleophilic addition-elimination reactions include:
Hydrolysis
Reaction with alcohols to form esters
Reaction with ammonia and primary amines to form amides
hydrolysis
The hydrolysis of acyl chlorides results in the formation of a carboxylic acid and HCl molecule
This is a nucleophilic addition-elimination reaction
A water molecule adds across the C=O bond
A hydrochloric acid (HCl) molecule is eliminated
Hydrolysis of propanoyl chloride to form propanoic acid and HCl

Acyl chlorides are hydrolysed to carboxylic acids via nucleophilic addition-elimination
formation of esters
Acyl chlorides can react with alcohols to form esters
The esterification of acyl chlorides is also a nucleophilic addition-elimination reaction
The alcohol adds across the C=O bond
A HCl molecule is eliminated
Esterification of propanoyl chloride to form ethyl propanoate and HCl

Acyl chlorides are esterified with alcohols to form esters via nucleophilic addition-elimination
formation of amides
Acyl chlorides can form amides with primary amines and concentrated ammonia
The nitrogen atom in ammonia and primary amine has a lone pair of electrons which can be used to attack the carbonyl carbon atom in the acyl chlorides
The product is an amide (when reacted with ammonia) or N-substituted amide (when reacted with primary amines)
This is also an example of a nucleophilic addition-elimination reaction as
The amine or ammonia molecule adds across the C=O bond
A HCl molecule is eliminated
Forming amides from propanoyl chloride

Acyl chlorides undergo reactions with ammonia and primary amines to form amides via nucleophilic addition-elimination
shapes of aromatic molecules
Aromatic molecules consist of one or more rings with conjugated π systems
Conjugated π systems arise from alternating double and single bonds in which the electrons are delocalised
Aromatic compounds are called ‘aromatic’ as they often have pleasant odours
Examples of aromatic compounds table
Functional group | Example | Name |
|---|---|---|
Arene |

| Propyl benzene |
Chlorobenzene |

| 2-methylchlorobenzene |
Phenol |

| 2,3-dimethyl phenol |
stereoisomers
Stereoisomers are molecules that have the same structural formula but have the atoms arranged differently in space
There are two types of stereoisomerism
Geometrical (cis / trans)
Optical
optical isomerism
A carbon atom that has four different atoms or groups of atoms attached to it is called a chiral carbon or chiral centre
Compounds with a chiral centre (chiral molecules) exist as two optical isomers which are also known as enantiomers
How enantiomers occur

A molecule has a chiral centre when the carbon atom is bonded to four different atoms or group of atoms; this gives rises to enantiomers
The enantiomers are non-superimposable mirror images of each other
Their physical and chemical properties are identical but they differ in their ability to rotate plane polarised light
Hence, these isomers are called ‘optical’ isomers
One of the optical isomers will rotate the plane of polarised in the clockwise direction
Whereas the other isomer will rotate it in the anti-clockwise direction
How a polarizer works

When unpolarised light is passed through a polariser, the light becomes polarised as the waves will vibrate in one plane only
biological activity of enantiomers
Enantiomers also differ from each other in terms of their biological activity
Enzymes are chiral proteins that speed up chemical reactions by binding substrates
They are very target-specific as they have a specific binding site (also called active site) and will only bind molecules that have the exact same shape
Therefore, if one enantiomer binds to a chiral enzyme, the mirror image of this enantiomer will not bind nearly as well if at all
It’s like putting a right-hand glove on the left hand!
Enzymes acting on different biological enantiomer substrates

enantiomers
Enantiomers are optical isomers that are mirror images of each other and are non-superimposable
They have similar chemical properties but differ from each other in their ability to rotate plane polarised light and in their biological activity
optical activity
Let’s suppose that in a solution, there is 20% of the enantiomer which rotates the plane polarised light clockwise and 80% of the enantiomer which rotates the plane of polarised light anticlockwiseThere is an uneven mixture of each enantiomer, so the reaction mixture is said to be optically active
The net effect is that the plane of polarised light will be rotated anticlockwise
Similarly, if the percentages of the enantiomers are reversed, the reaction mixture is still optically active but now the plane of polarised light will be rotated clockwise
In this case, there is 20% of the enantiomer, which rotates the plane anticlockwise
And 80% of the enantiomer, which rotates the plane clockwise
racemic mixtures
A racemic mixture is a mixture in which there are equal amounts of enantiomers present in the solution
A racemic mixture is optically inactive as the enantiomers will cancel out each other's effect and the plane of polarised light will not change
How the percentage of enantiomers affects plane polarised light

When one of the enantiomers is in excess, the mixture is optically active; when there are equal amounts of each enantiomer the mixture is optically inactive
effect of optical isomers on plane polarized light
Molecules with a chiral centre exist as optical isomers
These isomers are also called enantiomers and are non-superimposable mirror images of each other
The major difference between the two enantiomers is that one of the enantiomers rotates plane polarised light in a clockwise manner and the other in an anticlockwise fashion
The enantiomer that rotates the plane clockwise is called the R enantiomer
The enantiomer that rotates the plane anticlockwise is called the Senantiomer
These enantiomers are, therefore, said to be optically active
Therefore, the rotation of plane polarised light can be used to determine the identity of an optical isomer of a single substance
For example, pass plane polarised light through a sample containing one of the two optical isomers of a single substance
Depending on which isomer the sample contains, the plane of polarised light will be rotated either clockwise or anti-clockwise
No effect will be observed when the sample is a racemic mixture
Using polarized light to distinguish between R and S enantiomers

chirality and drug production
Most of the drugs that are used to treat diseases contain one or more chiral centres
These drugs can therefore exist as enantiomers which differ from each other in their ability to rotate plane polarised light
Another crucial difference between the enantiomers is in their potential biologicalactivity and therefore their effectiveness as medicines
Drug compounds should be prepared in such a way that only one of the optical isomers is produced, in order to increase the drugs’ effectiveness
Some drug enantiomers can have very harmful side effects
potential biological activity of enantiomers
If conventional organic reactions are used to make the desired drug, a racemic mixture will be obtained
In a racemic mixture, there are equal amounts of the two enantiomers
The physical and chemical properties of the enantiomers are the same, however, they may have opposite biological activities
For example, the drug naproxen is used to treat pain in patients who suffer from arthritis
One of the enantiomers of naproxen eases the pain, whereas another enantiomer causes liver damage
One enantiomer of a drug used to treat tuberculosis is effective whereas another enantiomer of this drug can cause blindness
Thalidomide is another example of a drug that used to be used to treat morning sickness, where one of the enantiomers caused very harmful side effects for the unborn baby
separating racemic mixtures
Due to the different biological activities of enantiomers, it is very important to separate a racemic mixture into pure single enantiomers which are put in the drug product
This results in reduced side-effects in patients
As a result, it protects pharmaceutical companies from legal actions if the side effects are too serious
It also decreases the patient’s dosage by half as the pure enantiomer is more potent and therefore reduces production costs
A more potent drug has better therapeutic activity
chiral catalysts
In order to produce single, pure optical isomers, chiral catalysts can be used
The benefits of using chiral catalysts are that only small amounts of them are needed and they can be reused
For example, an organometallic ruthenium catalyst is used in the production of naproxen which is used in the treatment of arthritis
Using catalysts to direct the production on one enantiomer

The organometallic ruthenium catalyst is a chiral catalyst which ensures that only one of the enantiomers is formed which can be used in treating arthritis
Enzymes are excellent biological chiral catalysts that promote stereoselectivity and produce single-enantiomer products only
Stereoselectivity refers to the preference of a reaction to form one enantiomer over the other
Due to the specific binding site of enzymes, only one enantiomer is formed in the reaction
The enzymes are fixed in place on inert supports so that the reactants can pass over them without having to later separate the product from the enzymes
The disadvantage of using enzymes is that it can be expensive to isolate them from living organism
Therefore, more research has recently been carried out into designing synthetic enzymes
Although using enzymes to produce pure enantiomers in drug synthesis takes longer than conventional synthetic routes, there are many advantages to it in the long run
For example, using enzymes to synthesise drugs is a greener process as fewer steps are involved compared to conventional synthetic routes