Bio Exam 2: Chemistry and Macromolecules
1/105
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
Protons
define an element (the same element cannot have a different number of protons), has mass, has positive charge
Neutrons
neutral, same element can have different numbers of neutrons
Electrons
negative charge, virtually no mass, number changes
Atomic mass
number of protons and neutrons (weight)
Atomic number
number of protons
Electrons in each orbital
2 (think box diagram in 103)
1st shell holds - electrons
2
2nd shell holds - electrons
8
Valence electrons
electrons that fill outermost shell (trying to get to 8 by forming bonds)
Covalent bonds
2+ atoms shares electrons
Ionic bonds
atoms steals/gives electrons
Nonpolar bonds/ diatomic molecules
sharing is equal (neither side is stronger than the other) (Hydrogen and Carbon are very nonpolar molecules)
Polar bonds
sharing is not equal (one atom holds bonds closer to themselves)
Two elements that mainly form polar bonds (bullies)
Nitrogen and oxygen
Ionic bonding
stealing/giving of electrons, always results in a charge (molecule that gain electron has negative charge and molecule that gave electron has a positive charge)
Water is (non/polar)
POLAR
Hydrophilic (polar)
attracted to water (because water is also polar and polar and polar attract)
Hydrophobic (nonpolar and nonionic)
Repels water (because polar does not like nonpolar)
How can you have polar bonds but a nonpolar molecule
If the polar bonds equal out on both sides EX: O=C=O the oxygen pulls the carbon towards it equally so the force equals out and makes the molecule SYMMETRICAL
Polar molecules are
asymmetrical EX h2o is bent because oxygen has a strong pull on both hydrogens
Ionic bonds are different from covalent bonds because
ionic bonds do not share electrons like covalent, they steal them
How are hydrogen bonds different from covalent and ionic bonds
hydrogen bonds form when a hydrogen gains a slightly positive charge from being attracted to a very negative element (like O or N). Hydrogen bonds are weaker than covalent or ionic bonds
Water properties
cohesion, surface temp, density, solvent
Water cohesion
Hydrogen bonds form between water molecules causing water to “stick together” which allows for water to travel up the roots and stems of plants
Water density
density decreases as water freezes (turns to ice) because of the crystalline lattice formed between molecules having a lot of space between them. This makes ice float which means things living in water bodies do not freeze when the temperature drops
Water as a solvent
the charges of water molecules allow for them to dissolve many substances helping with transport of nutrients (transported as aqueous solutions)
Water surface tension
water b
Hydrophobic molecules in water
group together so hydrophilic heads are exposed but hydrophilic tails are not (think circle or double layered line of molecules)
Ionization
when water molecules break apart H2O←→
Acid
High H+ concentration
Base
OH- concentration (combines w H+)
pH scale
scale from 1-14 that measures how basic/how many OH- (8-14), neutral (7), or acidic/how much H+ (1-6) something is/has
pH equation
-log[H+]
A lower pH means
Higher [H+] EX: 10^-3 is a low, acidic pH of 3
A higher pH means
Lower [H+] EX: 10^-8 is the high, basic pH of 7
A solution with pH 6 has how many more H+ ions than a pH of 7
10 times as many
Dehydration synthesis (condensation rxn)
removal of water molecules to bring other molecules together/form bond
Hydrolysis
breaks bonds using water (reverse of dehydration synthesis)
Hydroxyl group
OH EX: -OH
Methyl Group
CH3 EX:
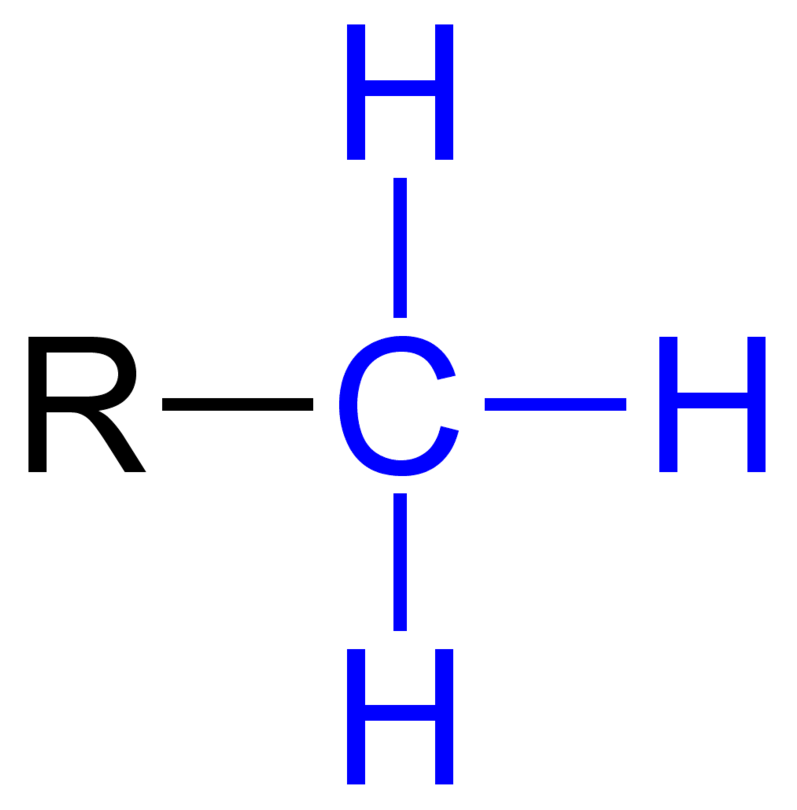
Carbonyl
C=O EX:

Carboxyl
O-C-OH EX:
Amino Group
NH2 or NH3 (one H will have positive charge (H+)) EX:
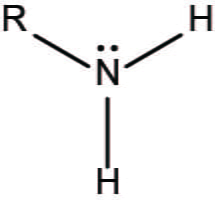
Phosphate
PO3-?
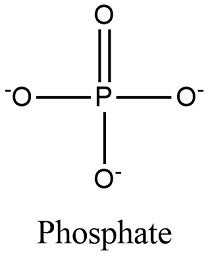
Sulfhydryl
-SH EX:

Hydrogen v.e.-
1
Oxygen v.e.-
2
Nitrogen v.e.-
3
Carbon v.e.-
4
Carbohydrates characteristics
C and H2O, polar, dissolves in water
Monosaccharides
quick energy and used in DNA or RNA
Disaccharide
quick energy in diet EX: glucose
Polysaccharides
energy storage EX: glycogen or starch
Nucleic Acid (nucleotide)
contains phosphate group, sugar, and NITROGEN
Lipids (fats and steroids)
energy storage, long chains of hydrophobic regions (H and C) EX cell membrane
Saturated fat (lipid)
all bonds are H-C (no bend)
Unsaturated fats (lipids)
has double bonds (=)
Phospholipid
C and H tail (nonpolar and saturated) but polar and unsaturated head
Proteins (amino acids)
1) Central carbon 2) amino group 3) carboxyl group 4) side chain (r group)
How to classify protein hydrophilic of hydrophobic
look at side chain
Triglycerides
type of lipid involved in energy storage (1 molecule of glycerol and 3 fatty acids)
Steroid hormones
type of lipid that can pass through membranes formed by phospholipids
Nucleic acids
long chains of nucleotides that contains phosphate group, sugar, and nitrogen
Similarities between ATP and nucleic acids
ATP has a single nucleotide but is a protein and nucleic acid contains multiple nucleotides
Proteins role
found in cell membrane and is responsible for transport across membranes, structure, signaling, and storage
protein synthesis
DNA → mRNA → Amino acids → Protein
DNA contains the instructions.
mRNA carries the instructions.
tRNA brings the building blocks (amino acids).
Ribosomes assemble the amino acids into a protein.
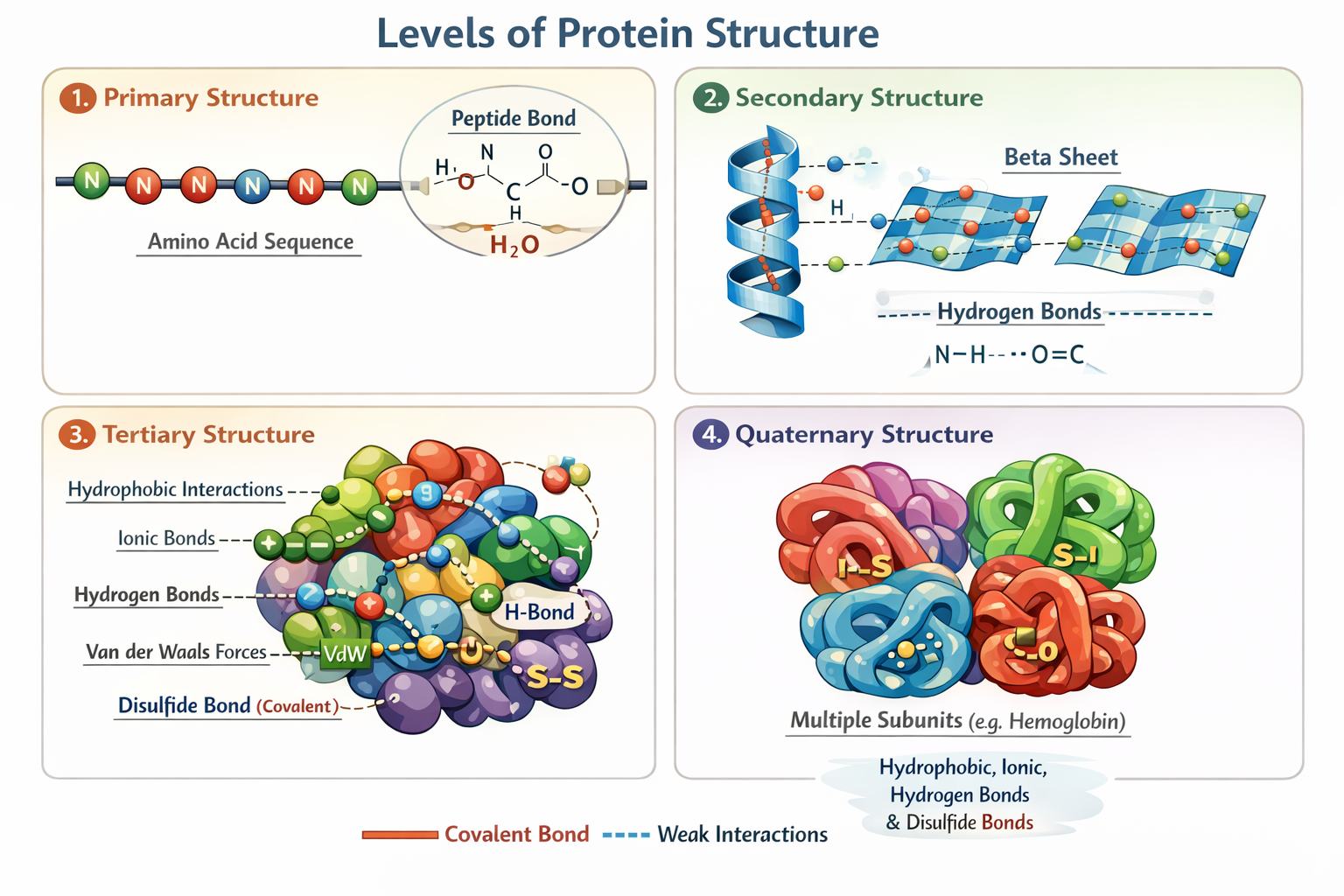
Primary structure of protein
covalent peptide bonds between a carboxyl and an amino group formed through dehydration synthesis (chain of amino acids)
Secondary structure of protein
folding of the polypeptide backbone due to hydrogen bonds (causes sprials)
tertiary protein structure
overall shape of polypeptide chain (includes all the sidechains and has a nonpolar inside polar outside) because side chains are included the spirals fold into different shapes
quaternary protein structure
multiple tertiary structures together as one
Sketch of protein structures
Why cells need a biological membrane
controls what enters and exits cell, protects cell, maintains internal balances (pH, concentration, etc.), holds cell together, allows for communication between other cells
How do lipids come together to form membranes
polar heads come together to encase nonpolar tails and protect them from water (circle or bilayered) because of 2nd law of thermodynamics
Weak interactions between lipids in membranes
hydrophobic sticks with hydrophobic (tails by tails) and hydrophilic wants to be by other hydrophilic (heads by heads) which causes water and the hydrophilic parts of the cell to be by each other forming H bonds. because most of cell has weak bonds or attraction it remains fluidlike
Water excludes lipids by
forcing lipids (which are typically nonpolar) to group together (imagine oil in water) reducing amount of space lipids take up so it does not decrease entrophy
membrane fluidity
the ability of molecules to be able to flow through or within the lipid bilayer
unsaturated lipids and fluidity
unsaturated lipids have double bonds, causing their tails to have bends, which means they are not able to pack as closely together, causing them to be more flexible/fluidlike
cholesterol and fluidity
at high temps cholesterol reduces the movement of phospholipids so the membrane is not too fluid and at low temps it prevents them from packing too close together and causing the membrane to be too rigid
membranes are semi permeable meaning
they allow some molecules to pass through while others cannot depending on things like shape, size, polarity, and charge
Simple diffusion
molecules pass straight through membrane needing no energy (passive) and no protein transporter (nonpolar molecules and very small polar molecules)
facilitated diffusion 1
molecules pass through a channel protein but require no extra energy (passive) or interaction from protein (ions)
facilitated diffusion 2
molecules (polar) pass through a transporter protein but still require no extra energy to do so
Active transport
polar molecules pass through transporter protein to go AGAINST concentration gradient (so they require extra energy to do so) (ions or polar)
passive transport
no extra energy required
active transport
requires extra energy
facillitated diffusion
requires protein channel
simple diffusion
molecules pass directly through membrane
osmosis is
diffusion of water down its concentration gradient (moves to high solute concentration so it can dilute more)
tonicity is
comparison of hypotonic (less concentrated) hypertonic (more concentration) and isotonic (equal concentration) inside and outside of cells
prokaryotic cell
bacteria cells, NO nucleus or membrane bound organelles, small, binary fusion reproduction
eukaryotic cell
has membrane bound organelles and nucleus, plants and animal cells
Plant cells specific
cell wall for support, choloroplasts (photosynthesis), central vacuole (storage)
animal cell specific
lysosomes (break down)
cytoplasm
fluid and organelles
cytosol
fluid component of cytoplasm
cytoskeleton
proteins in cytoplasm that have structural support (animal cell specific (plant have cell wall))
nucleus
stores DNA and has pores to let out RNA
mitochondria
powerhouse-performs respiration, breaks down glucose/metabolism; has its own DNA (mom), double membrane, own ribosomes
chloroplast (PLANTS ONLY)
performs photosynthesis, green, has own DNA and ribosomes
bacteria and mitochondria+chloroplasts
double membrane, free ribosomes+DNA, independent