Chemistry 1.1 - 1.2 Test (Definitions Only)
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
Beaker
Holds liquids, may be graduated, white spot for labeling

Erlenmeyer flask
Holds liquids, shape avoids loss due to splashing, used for titration

Florence Flask
Heats liquids, Shape allows even distribution of heat while boiling, never graduated

Test tubes
hold liquids or solids, can be heated directly or in water bath, may be used to mix small quantities of chemicals, large variety of sizes

Fluted funnel
Funneling liquids, useful for pouring liquids through small openings, can contain filter paper for separating solids from suspensions by filtration

evaporating dish
evaporating solvent, can be used to dry a damp product, ceramic material allows direct heat to high temperatures

watch glass
holding or covering, useful for holding a sample of chemical, may cover a beaker or flask to prevent evaporation, hold chemicals while drying

crucible
heating to high temperatures, heating covered partially covered samples, ceramic material may be directly heated until read hot

pipe stem triangle/clay triangle
provides a base to hold a crucible, sits atop a wrought-iron ring, stems are made of ceramic material

graduated cylinder
measuring volumes of liquids, sizes vary

burette
measuring volumes of liquids, delivers various volumes through a valve called a stop cock, more precise (exact) than the graduated cylinder

pipette
measuring volumes of liquids, may be graduated, may be volumetric, liquid is drawn up with a pipette bulb or suction device
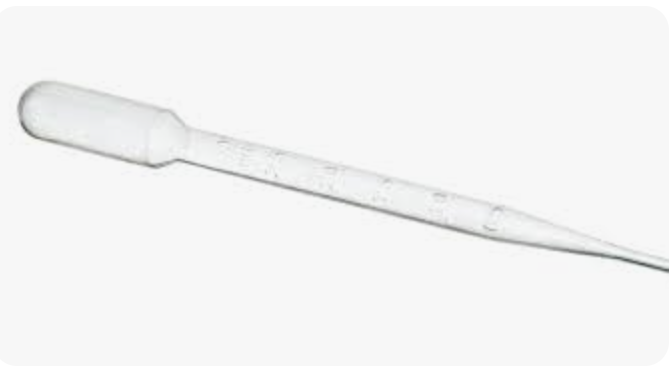
thermometer
measuring temperatures, commonly measure temperature in degrees Celsius

Ring stand
providing a post to attach, can attach ring clamps, burette clamps extension clamps etc ,also called a utility stand

Ring clamp
Attaching to rind stand, supports a ceramic pads, pipe stem triangle, or an evaporating dish, may surround a baker as a safety ring

Burette clamp/utility clamp
attaches to ring stand, may hold a test tubes in a station position, may support the neck of a flask, holds a burette

flint striker
lights bunsen burner, provides a spark by moving a flint across a file

Bunsen burner
provides heat, adjusts flame temperature by addition of air through the barrel, adjust flame height by turning the regulator valve

test tube holder
holds test tubes, used for heating tubes over flames, used for removing tes tubes from water baths
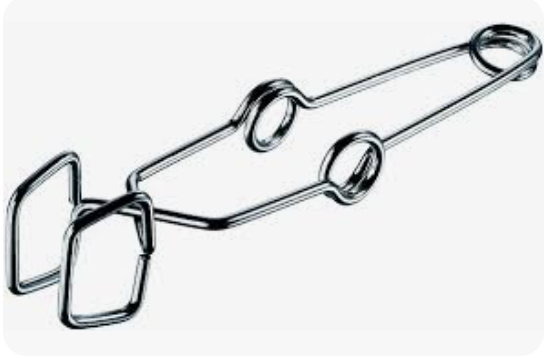
beaker tongs
lifts hot beakers, rubber cover allows thongs to firmly grasp and move beakers of all sizes
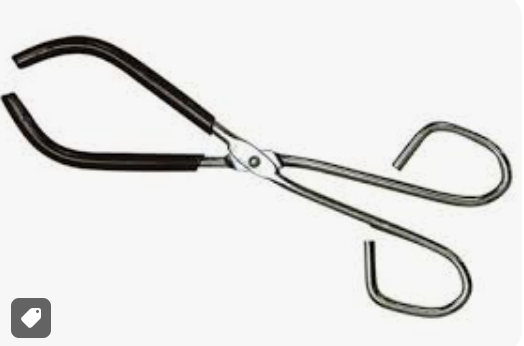
crucible tongs
holds hot crucibles, may remove or adjust crucible lid, hold hot evaporating dishes, NOT designed for lifting beakers or test tubes

ceramic pad
provides base for holding glassware, sits atop a wrought-iron ring, provides a flat surface for beakers or flasks, sometimes called a wire guaze

scoopula
moving samples of solids, sometimes called spatula, should NOT be used as a stirring rod

oxidizers
A chemical symbol indicating a substance that can cause another substance to lose electrons (e.g., in a combustion reaction)

Flammable/combustible
A chemical symbol indicating a substance that can easily ignite and burn rapidly

Explosive material
A chemical symbol indicating a substance that can explode violently when subjected to heat, shock, or friction

Acute toxicity
A chemical symbol indicating a substance that can cause severe immediate health effects via a single exposure (e.g., poisoning)

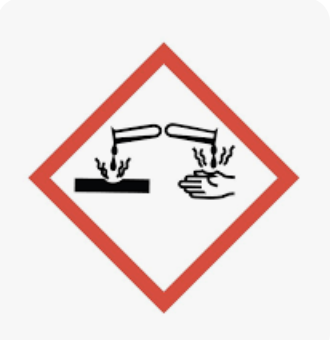
Corrosion
A chemical symbol indicating a substance that can cause visible destruction or irreversible alterations to living tissue by chemical action at the site of contact
Gas under pressure
A chemical symbol indicating a gas stored under pressure which may explode if heated

Carcinogen toxic to humans
A chemical symbol indicating a substance that can cause cancer, reproductive harm, or damage to specific target organs

Irritant or narcotic effects
A chemical symbol indicating a substance that can cause skin/eye irritation, respiratory irritation, or drowsiness/dizziness (narcotic effects)

Aquatic toxicity
A chemical symbol indicating a substance that is toxic to aquatic life, potentially causing long-lasting harmful effects to marine or freshwater ecosystems

Scientific Method
true method to solving problems
Observation
First step of scientific method, involves collecting data
Quantative
numbers or quantities
qualitative
qualities or changes in the quality of matter
hypothesis
second step of scientific method,“if…then” statement that explains observations
Experimentation
third step of scientific method, determines whether hypothesis is accurate or not
sources of error
errors that we have no control over
mistakes
errors humans do
Theory
fourth step of scientific notation, cohearent set of explanations
model
explains the collection of obervations
law
different from a theory, general statement of fact, related to math
Robert Boyle
pioneered the use of the scientific method
scientific notation
method of representing numbers in exponential form,
standard scientific notation
one non-zero digit to the left of the decimal
mantissa/decimal portion
first portion of the scientific notation, should always be expressed as a number between 1 and 10
ordinate/exponential portion
second potion of scientific notation, the base 10 raised to some power
positive exponent
indicated large number in scientific notation, the number of places the decimal must be moved to the right
negative exponent
indicated a small number in scientific notation, the number of places the decimal must be moved to the left
dependent variable
value that responds to the variation
independant variable
value that is not affected
“Time”
nearly always independent
fume hood
enclosed area equipped with fans to draw vapors out of the hood and vent them outside, contains gas jets, sinks, lights, electrical outlet, and maybe chemicals emitting toxic fumes
eyewash station
used to wash chemicals out of eye, eyes should be held open and rinsed continuously for 10-15 mins, used by pushing on a hand bar or foot petal
safety shower
spills a large portion of the body require removal of clothing and washing of the entire region for 10-15 mins, used by pulling ring and will poor 200L of water
Fire extinguisher
sets out fires, used by pulling safety pin from the top, should never be used on a person, contain carbon dioxide and ar good for class A,B, and C fires
Type A fire
wood or paper
Type B fire
oil or grease (most Chemicals)
Type C fire
electrical equipment
Fire blanket
used to put out a fire on a person, may be enclosed in a box
emergency gas shut off
allows all gas out kets in laboratory to b shut off, turn handle perpendicular to the gas line or push large red button
Spill control station
may contain, absorbent pillows to soak up spills, safety goggles, and gloves, and chemicals to neutralize acid and base spills
acid spills
should be neutralized with sodium bicarbonate o baking soda
base spills
should be neutralized with acetic acid or vinegar
First Aid Kit
all labs should have this, should contain antibiotic cream or ointment and plenty of bandages
Glass disposal container
a plastic bucket or specially designated recyclables box for disposal of glass
chemical disposal
clear containers marked for disposing solutions or precipitates containing heavy metals or any toxic chemical
Fire Alarm
Must know where this is