BIOS 301 - Membrane Transport
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
Use of SNARE proteins in membrane fusion
Local increase in calcium ion signals release of neurotransmitter
t-SNARE (target membrane), v-SNARE (vesicle membrane), and SNAP-25 (target membrane) intertwine to form a coiled bundle of 4 alpha-helices
Two membranes are drawn together and fused
ATP is not required for SNARE coiling, but is required for SNARE disassembly
Passive transport (general)
Movement of a substrate down its concentration gradient
Active transport (general)
Movement of a substrate against its concentration gradient or electric potential
Simple passive diffusion
Cell membranes are permeable to small, nonpolar molecules that diffuse across the membrane to achieve concentration/charge equilibrium
Delta G equation for membrane transport
Cin, Cout = concentration of substrate inside vs. outside cell
Z = charge of ion
F = Faraday’s constant
Delta psi = membrane potential
R = ideal gas constant
T = temp in Kelvin
If Cout > Cin, 1st term makes delta G more negative (favorable)
If delta psi is negative and Z is positive, 2nd term makes delta G more negative (favorable)
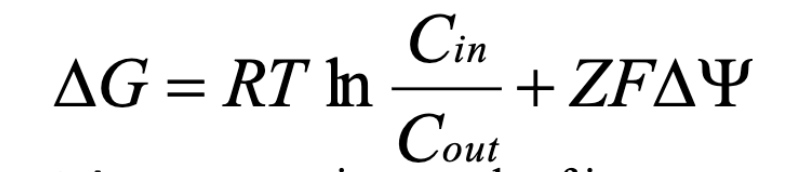
Flux equation for passive membrane transport
J = flux of particles passing through the membrane
Cin-Cout = concentration gradient (steepness)
l = thickness of membrane (distance)
D = diffusion coefficient across membrane (composition of medium)
K = partition coefficient (molecule identity)
Very small for most polar molecules
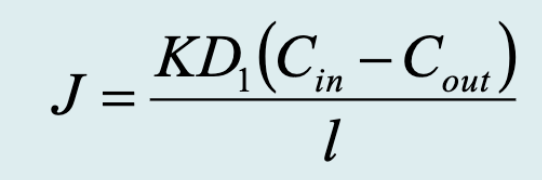
Permeability coefficient
Measures the rate at which a substance passes through the membrane during passive diffusion
Higher value means greater permeability
Is an experimentally measured lumped parameter of l, K, D
Facilitated diffusion
Involves passive diffusion of polar/charged molecules, facilitated by transporters
Transporter is lined with hydrophilic amino acid side chains, allows substrate to be solvated in channel
Transporter binds substrate through many weak, noncovalent interactions (facilitates dehydration)
Solutes are surrounded by shell of water molecules before they pass through membrane
Three classes of transport systems
Uniport, symport, antiport
Can be either active or passive transport
Uniport (definition, example)
A single solute moves in one direction across the membrane
Ex: glucose transporter in erythrocytes (GLUT1)
Moves glucose (down its concentration gradient) from blood plasma into erythrocytes
Glucose transporter (structure/function)
Has 12 transmembrane domains
Transmembrane domains consist of amphipathic alpha helices
Polar residues interact with glucose
Nonpolar residues interact with membrane lipids
Aquaporin selectivity
Determined by amino acid properties within the protein channel
Size exclusion - His narrows pore to sterically block larger molecules
Electrostatic repulsion - Arg provides electrostatic barrier that repels cations (e.g. protons, H3O+)
Water dipole reorientation - NPA motifs flip water’s dipole to make it energetically impossible for proton travel through channel
Ensures only neutral water passes
K+ ion channel selectivity
To enter the channel, solvated K+ must shed its H2O shell
Backbone carbonyl oxygens of channel residues orient to perfectly mimic geometry of H2O molecules surrounding solvated K+
Means minimal energy penalty for K+ to enter channel
Na+ is smaller than K+ - carbonyl oxygens cannot replace its hydration shell
Energetically costly
Antiport (definition, example)
Moves two different substrates in opposite directions
Ex: chloride bicarbonate exchanger
CO2 produced by respiring tissues is converted to blood soluble HCO3- by carbonic anhydrase
As HCO3- enters erythrocytes, Cl- exits
Keeps cell equilibrated (no net charge transfer)
HCO3- is converted back to CO2 by carbonic anhydrase and released into lung space for excretion
Primary active transport
Solute transport is coupled directly to an exergonic reaction (e.g. ATP hydrolysis)
Secondary active transport
Uphill transport of solute 2 is coupled to downhill flow of solute 1 (originally pumped uphill by primary active transport)
Na+/K+ ATPase
Primary active antiporter
Facilitates movement of Na+ and K+ against their electrochemical gradients
Contains three domains:
N (nucleotide binding domain) binds ATP/Mg2+ and phosphorylates Asp in P domain
P (phosphorylation domain) contains key Asp residue
A (actuator domain) removes phosphate from Asp with each pump cycle
Mechanism
Transporter binds 3 Na+ from inside of cell
Phosphorylation alters enzyme shape/affinity - releases Na+ and binds 2 K+ from outside of cell
Dephosphorylation alters enzyme shape/affinity - releases K+
Symport (definition, example)
Moves two different substances in the same direction
Ex: Na+/glucose symporter (secondary active transport)
Na+ concentration is high extracellularly (gradient set by Na+/K+ ATPase) → draws Na+ inward
Provides energy needed to transport glucose from gut to epithelial cell (against concentration gradient)
Glucose uniporter moves glucose from epithelial cells → blood (via passive diffusion)
ABC transporter
ABC = ATP Binding Cassette
Hydrolyze 2 ATP to transport a specific substrate (primary active transporters)