BI101: Exam 2 Topics
1/51
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
Which of the following is true of a solution in equilibrium?
Molecules will move randomly, but not directionally
___________ is the movement of solutes (i.e. particles) down a concentration gradient, while ___________ is the movement of water down a concentration gradient
Diffusion, Osmosis
If a cell is hypertonic to it's environment...
Animal cells that remain in hypotonic environments are in danger of....
In a saline solution, ______ is the solute and ______ is the solvent.
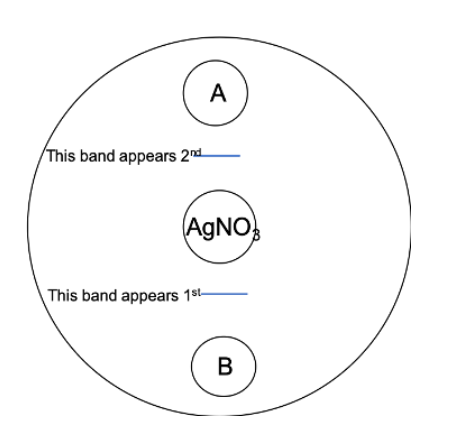
The diagram below illustrates an experimental set-up similar to what was conducted in class. Suppose the large circle represents an agar dish, and the smaller internal circles represent wells punched into the agar (all equidistant from one another). Silver nitrate (0.1 M AgNO3 ) was placed in the center well, while 0.1 M of two other solutions, potassium iodine (KI) and potassium chloride (KCl) were placed in the two exterior wells (labeled A and B). These chemicals form a white band when they meet and react with the silver nitrate. The order in which the bands (blue lines) appeared are labeled on the diagram. The molecular weight of the ion traveling for KI is 126g/mol while the molecular weight of the ion traveling for KCl is about 35g/mol.
Given this information, which chemical was in well A?
0.1 M KI (Potassium Iodide)
[Use the information and scenario presented in question 1, when answering this question]: Without changing the types of chemicals (i.e. you still need to add KI and KCl to the exterior wells), list two ways you could decrease the amount of time it would take for both bands to appear (i.e. make it go faster).
Note: You also can't change the shape of the plate or move the wells closer together.
1. Temperature- Warming the plate would cause all the solutions to move faster
2. Concentration- the molecules would move faster if a higher concentration was added into each well.
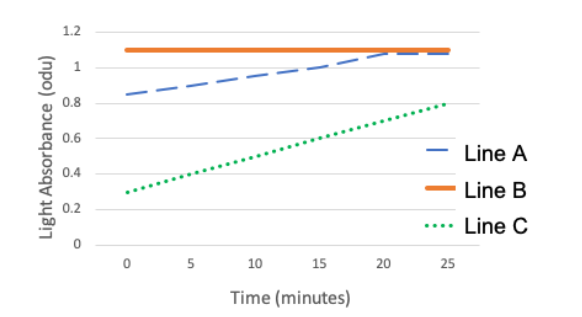
Suppose 3 dialysis bags are filled with 1 M of phenol red (the red dye we used in lab). One bag is placed in each of the 3 solutions below:
1 beaker contains 0.8 M phenol red
1 beaker contains 1 M phenol red
1 beaker contains 0.2 M phenol red.
The dialysis bags are permeable to phenol red, meaning the dye can enter and leave the bag. After the bags are submerged in their respective beakers, a sample of the surrounding water is transferred to a cuvette and the light absorbance is measured with a spectrophotometer every 5 minutes for 25 minutes. The spectrophotometer readings were used to generate the graph below, but the lines were not labeled to indicate the solution they represent. Note: The absorbance reading at time 0 represents the absorbance of the dye in the beaker, before the dialysis bag was added to it.
Based on this information, which phenol red concentration is associated with line B (0.2 M, 0.8 M, or 1 M)? How did you determine this? Note, to earn full credit you need to reference the what is happening in the beakers. Specifically, you need to answer the following:
1. Relatively, how much dye is entering the surrounding water (from the bag) in the beaker illustrated by line B compared to line A and C?
2. What does this indicate about the phenol red concentration in the bag, relative to the starting phenol red concentration of the water in the beaker?
3. Given what you said above, which phenol red concentration (0.2 M, 0.8 M, or 1 M) must be illustrated by Line B in the graph and why?
1. The fact that the line is completely horizontal means the absorbance isn't changing, which indicates that the dye is not leaving the bag into the surrounding water. Dye is leaving the bag in the lines illustrated by A and C.
2. The only reason dye wouldn't leave, is if the bag were already at equilibrium with the surrounding water from the beginning. Therefore, the concentration of the bag and water must have been equal from the start.
3. Since we know all the bags has 1 M Phenol Red to start with, this must mean that line B illustrates the bag in the 1 M phenol red solution.
In lab, we added various concentrations of salt water to test tubes. We then added a couple of drops of sheep's blood to the tube, which gave the tube a cloudy red appearance. We timed how long it took for the tube to become transparent.
We found that the tube with distilled water (no salt added), become transparent fastest. Explain why. To earn full credit, you need to explain:
1. What causes the liquid to go from opaque (i.e. cloudy) to transparent
2. Why the distilled water became transparent faster than all the other saline concentrations.
Cells being lysed
The distilled water tube became transparent quicker, because water was entering the cells quicker, causing them to lyse faster

Suppose you look at a plant cell under a microscope, and see the following:
These plant cells are in what type of solution, relative to the cell?
Hypertonic
Which of the following best explains why animal lyse in certain solutions, but plant cells do not.
Plants cells will simply utilize excess water for photosynthesis, while animal cells are unable to undergo photosynthesis | |
Plant cells naturally have a faster rate of water loss compared to animal cells | |
Plant cells possess rigid cell walls, while animal cells do not | |
Plant cells have special contractile vacuoles that remove excess water, while animal cells do not | |
Plant cells are not permeable to water, while animal cells are | |
Animal cells possess plasma membranes, but plant cells do not |
Plant cells possess rigid cell walls, while animal cells do not

Suppose you create 3 semipermeable bags and fill them each with a 5% glucose solution. The material the bags are made out of allow the movement of water, but are not permeable to glucose. After filling all the bags, you weigh them and find them all to weigh 5 grams. You then submerge each of the glucose bags in the same volume of water, but the water temperature varies (see diagram below).
All bags were allowed to sit in their respective beakers for 10 minutes, and were then removed to be reweighed. After the 10 minute period, which bag should weigh the least (i.e. it has the least amount of liquid in it)?
Beaker A
In all the beakers, water will move from the surrounding water into the bag, in an attempt to dilute the more concentrated solution on the inside. Note that the gradient is constant across all the beakers, but the temperature is not. We know that osmosis occurs faster in water temperatures, so we would expect water to enter the bag quickest in beaker C and slowest in beaker A. This means beaker A should take on the least amount of water in the 10 minute time frame, and should therefore be lightest.
Suppose a cell with an internal glucose concentration of 2% is placed in a 4% glucose solution. If the cell in question were permeable to glucose, glucose would move via _________________. If the cell in question were NOT permeable to the cell, water would move via _____________________.
diffusion into the cell; osmosis out of the cell
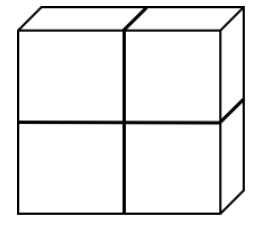
Suppose you have four perfect cubes, each with the following dimensions: 3 cm x 3 cm x 3 cm. You arrange the cubes in a sheet, 2 cubes by 2 cubes (image below). For this object, calculate the surface area to volume ratio. You must show your work to receive any credit.
Total surface area= surface area of 1 cube side # of surfaces exposed - (3 cm 3 cm) * 16 sides= 144 cm2
Total volume= volume of 1 cube # of cubes - (3 cm 3 cm* 3 cm) * 4 cubes= 108 cm3
Surface area to volume ratio = Surface area/ volume - 144 cm2/ 108 cm3= 1.33
You decide to bake a circular cake for your friend's birthday. You buy a cake mix, and mix the ingredients. You are getting ready to choose the pan you want to bake your cake in, so you pull out the two circular pans you own. One pan is 6 inches in diameter, and the other is 12 inches in diameter. You look at the instructions on your cake mix and see that it will take 42 minutes to bake the cake if you use the 6 inch pan, but only 38 minutes if you use the 12 inch pan. Given what we learned in class, explain why it would take different amounts of time, to cook the same amount of batter, in the different pans.
When the surface area is larger, a greater amount of the cake batter is in contact with the environment (heat). Since the more batter will be exposed with the bigger pan, it'll bake faster
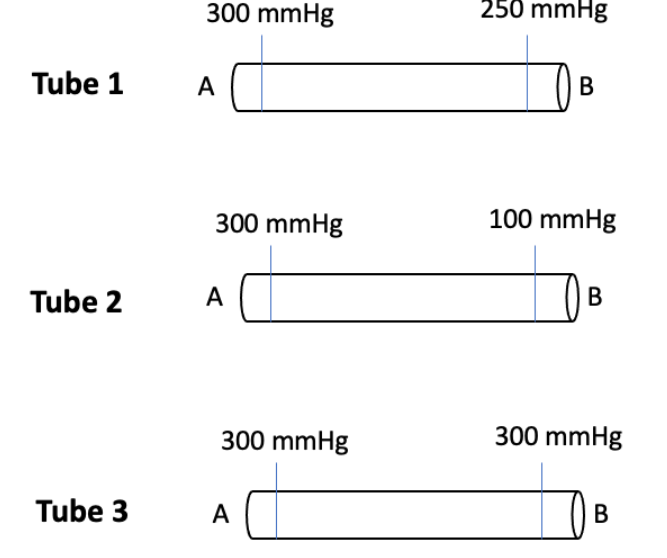
Shown below are 3 different vessels, all equal in diameter. The pressure of two locations is indicated for each vessels (expressed as mmHg). Consider these tubes and then answer the following questions.
What direction would liquid move in Tube 1 (towards side A or B)? How did you determine this?
In which tube (Tube 1, 2, or 3) would liquid move the fastest (i.e. have highest flow rate)? How did you determine this?
What direction would liquid move in Tube 1 (towards side A or B)? How did you determine this?
Side B; liquids and gases move from areas of high pressure to areas of low pressure.
In which tube (Tube 1, 2, or 3) would liquid move the fastest (i.e. have highest flow rate)? How did you determine this?
2; Flow rate is directly proportional to the pressure gradient. The larger the pressure gradient (i.e. pressure difference) the faster the liquid will move (i.e. faster flow rate).
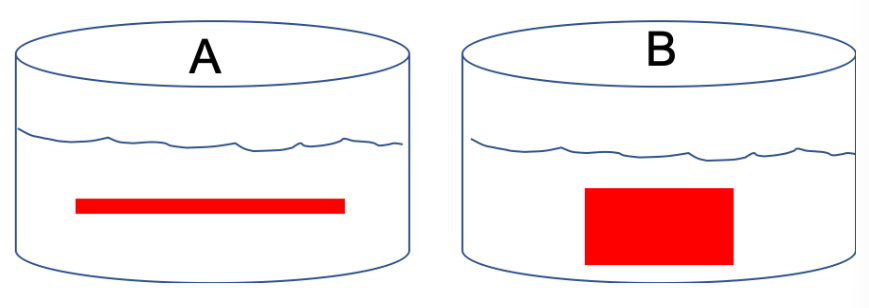
Suppose two dialysis bags (like what we used in lab 4) were filled with equal volumes and equal concentrations of a red dye called phenol red. Although the amount and concentrations of the dye solution are the same, the overall shape of the bags differ (see image below) due to the width of the tubing used to make the bag.
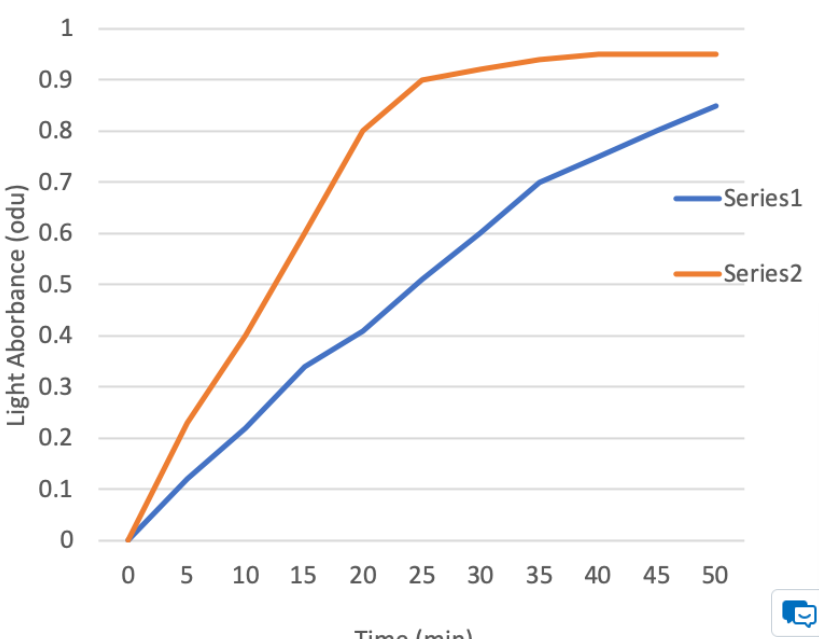
In both beakers, phenol red dye will diffuse out of the bag, into the surrounding solution. The relative amount of dye in the solution surrounding the bag was determined by measuring the light absorbance of the solution every 5 minutes, for 50 minutes, with a spectrophotometer and the data was used to generate the following graph:
Given this information, answer the following questions:
1. Which line (series 1-blue or 2-orange) on the graph is associated with the fastest rate of diffusion? How can you tell?
2. Which bag (A or B) would you expect to be associated with the fastest rate of diffusion? Why?
1. The series 2 line, is experiencing the fastest rate. We can tell because it has the steepest slope, and it reaches equilibrium the quickest (hits plateau fastest).
2. Bag A should be experiencing the faster rate of diffusion, because it has the higher surface area to volume ratio.
As the diameter of a tube increases, what happens to the following factors? Assume all other tube factors (such as length, etc.) are held constant.
|
|
1
2
2
2
Suppose you take two equal quantities of play-doh (malleable clay). You roll one set of play-doh into a ball and the other you flatten into a sheet. Which of the following is true in this situation? Choose all options that are true (there may be more than one).
The play-doh rolled into a ball has the greater surface area.
The flattened play-doh has a greater volume.
The flattened play-doh has the greater surface area
The play-doh rolled into a ball has a greater volume.
The flattened play-doh has the greater surface area to volume ratio.
The play-doh rolled into a ball has the greater surface area to volume ratio.
1, 2, 3, 4, 6, 7
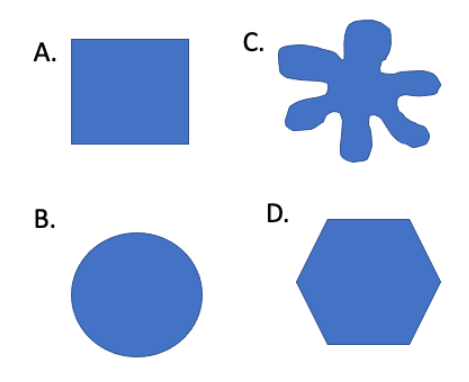
Assume the shapes below have been made out of an agar infused with a pH indicated (like we did in Exercise 2). Although the shape differs, assume that all the molds were made with an equal volume of agar.
Which mold (if any) would be yellow throughout the quickest, if placed in a beaker of vinegar?
Mold C
Suppose you take three fermentation tubes, and add the following to all tubes: 25 mL of live yeast and 10 mL of 10% glucose. Although all the tubes have the same things added, they are placed in different temperatures, as shown below:
Tube 1: Placed in 0°C temperature
Tube 2: Placed in 24°C temperature (roughly room temp.)
Tube 3: Placed in 40°C temperature
Which tube (1, 2, or 3) would you expect to experience the greatest amount of displacement in the fermentation tube after 90 minutes, and why? To earn full credit, your answer needs to indicate what causes the displacement of yeast in the tube, as well as why you would expect this in the tube you chose.
Which tube (1, 2, or 3) would have the greatest amount of displacement?
Tube 3 (40 degrees)
What causes the displacement in the tubes?
Carbon dioxide gas that is produced via cellular respiration
Why would you expect the greatest amount of displacement in the tube you selected?
Warmer temperatures experience faster rates of cellular reactions. In this case, the warmer temperature led to faster rates of cellular respiration, which led to greater accumulations of carbon dioxide gas (hence larger displacement).
Artificial sweeteners (such as Splenda) are similar in taste to glucose, but are not a suitable substitute reactant for cellular respiration. Answer the following in connection to this statement:
-Why do they taste similar?
-Why is glucose is an ideal reactant for cellular respiration (ie. what role does it ultimately play) and why can't artificial sweeteners be used as a substitute reactant? Note: you will get zero credit for saying, because artificial sweaters aren't glucose and you need glucose as a reactant.
Why do they taste similar?
They are similar in 3-D shape, and can therefore trigger the same taste buds. -1 pt.
Why is glucose is an ideal reactant for cellular respiration?
It cannot be digested by enzymes we produce, so we can't access the stored energy in the bonds.
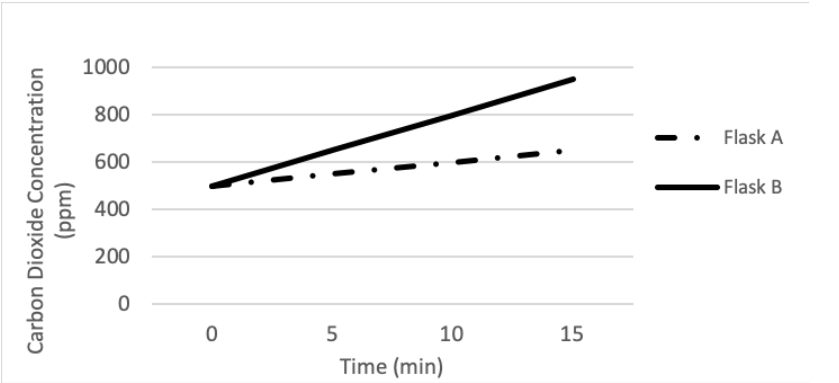
Suppose a probe capable of measuring carbon dioxide concentration was added to two flasks filled with respiring yeast. One flask of yeast had Magnesium Sulfate (MgSO4) added to it, while the other had Sodium Fluoride (NaF) added to it. The carbon dioxide output from both flasks was recorded, and used to generate the graph below:
Based on the graph, we can say that ______ had the sodium fluoride (NaF) added to it and ______ had the fastest rate of cellular respiration.
Flask A; Flask B
Kyle just returned from his daily 5 mile run. Immediately after returning from his run, he measured the following: heart rate, respiration rate, time it took to turn bromothymol blue yellow, and body temperature. He then sits on the couch and watches a two hour movie. After the movie is finished, he once again measures the vitals listed above. Given what you know about cellular respiration, indicate whether you expect the indicated vitals to increase, decrease, or stay the same from from reading 1 (after his run) to reading 2 (after watching the movie).
|
|
2
2
1
2
An increase in which of the following variables would indicate an increased rate of cellular respiration? Select all that apply (there may be more than one right answer; when grading, you lose credit for either selecting wrong answers or leaving correct answers un-selected).
Amount of Glucose
Amount of starch
Amount of ATP
Temperature
Amount of Oxygen
2, 3, 4
Suppose you take two fermentation tubes and add the following: 25 mL of live yeast, 10 mL of 10% glucose; and 5 mL of Magnesium Sulfate (MgSO4 ). Although both fermentation tubes contain the same substances, they are placed in different temperatures, show below:
Tube 1: Placed in 40°C
Tube 2: Placed in 100°C
After sitting in these respective temperatures for 90 minutes, tubes were removed and the displacement of yeast measured. Describe the amount of displacement you would expect in tube 2, relative to tube 1. Why would you expect this relative displacement?
Describe the amount of displacement you would expect in tube 2, relative to tube 1.
We would expect there to be no displacement in the tube 2, but there should be ample displacement in tube 4.
Why would you expect this relative displacement?
The extreme heat caused the enzymes associated with cellular respiration to denature, and therefore, cellular respiration did not occur in tube 2.
Which of the following is false regarding aerobic respiration?
It requires oxygen
It can be completed by facultative anaerobes
It produces carbon dioxide as a by-product
It produces less ATP than anaerobic respiration
It produces less ATP than anaerobic respiration
In lab 6, what was our dependent variable for all three lab exercises?
Amount of Carbon Dioxide
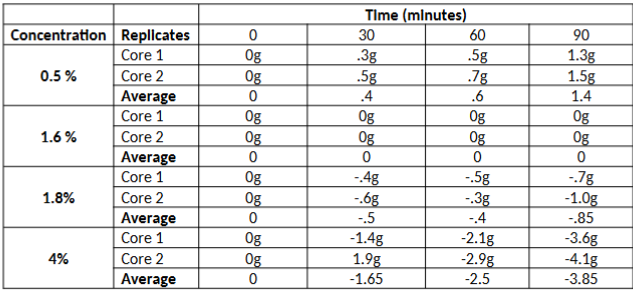
A scientist wants to explore how different saline (i.e. saltwater solutions) concentrations will
influence weight gain or loss of potato cores. To do this, the scientist sets up the following
treatment groups:
Group 1: 2 potato cores subjected to a 0.5% saline solution
Group 2: 2 potato cores subjected to a 1.6% saline solution
Group 3: 2 Potato cores subjected to a 1.8% saline solution
Group 4: 2 Potato cores subjected to a 4% saline solution.
All potato cores are initially the exact same weight and were taken from the same species of
potato. The cores were submerged in a beaker full of 500mL of the respective treatment
concentration, and were removed every 30 minutes, for 90 min, to be weighed.
Suppose the change in weight (Weight at each time point- Weight prior to submersion) was
calculated, and recorded in the table below:
Given this information, what is the natural internal saline concentration of potato cells? How can you tell?
1.6%; the potato core placed in this saline had no net change in weight over the duration of the experiment, indicating they neither gained or lost water. The net exchange of water in the core would only occur if it were in an isotonic solution (i.e. internal concentration of solute is equal to external concentration).
Given what you know about osmosis, explain why the 1.8% and 4% values become increasingly more negative
They are becoming more negative water is leaving the potato cells in the core. Water is
leaving because the cells are in a hypertonic solution (i.e. solute concentration is higher
in solution than it is inside the cells). The higher solute concentration draws water out of
the cells, in an attempt to reach equilibrium.
Given what you know about osmosis, explain why the 0.5% treatment values become increasingly more positive
The cores in the 0.5% treatment are gaining water weight, which is why the values are
becoming more positive. In this case, the cells are gaining weight because the solute
concentration (which we established in question 1 is about 1.6%) inside the cells is
higher than the concentration of the solution (i.e. the solution is hypotonic relative to
the cell). As a result, water is entering the cells causing the cores to gain weight
Would the 1.8% & 4% treatment continue to become more negative indefinitely? Why or why not?
No. Net loss of water would stop when either equilibrium is reached, or when there is no more intracellular water left to leave
[Consider the following for questions 5-7]: You take a dialysis bag filled with 1 M of glucose and place it in a beaker containing .8 M of glucose. The dialysis bag is permeable to the glucose
In this situation, what direction would glucose move?
Out of the cell
What kind of movement is this
Diffusion
Assuming you cannot change the contents of the dialysis bag (i.e. it has to be filled with the same amount of glucose solution) what are two things you could do to make the movement of glucose faster?
Warm beaker - increased temperature = more kinetic energy = faster movement
Add water to the solution (0.8 M) in beaker
[Consider the following for questions 8-11]: Suppose you have a perfect cube with the following dimensions: 4 cm x 4 cm x 4cm
Calculate the cube’s surface area:
L x W of 1 side x # of sides = SA → (4 cm x 4 cm) x 6 = 96cm2
Calculate the cube’s volume:
L x W x H = volume → 4cm x 4cm x 4xm = 643
Calculate the cube’s surface area to volume ratio
SA/V → 96/64 = 1.5
If this cube, and a cube measuring 8 cm X 8 cm x 8 cm were both placed in a solution permeable to the cube. Which would experience the fastest rate of exchange, and why?
The 4×4×4 cube would be faster because it has a higher SA/V ratio and the rate of exchange for any entity increases as the SA/V ratio increases
![<p><strong>[Consider the following for questions 12-15]</strong>: Suppose you took 2 of the 4 cm x 4 cm x 4 cm cubes and stuck them together to make the following arrangement:</p><p>Calculate the total surface area of this object:</p>](https://assets.knowt.com/user-attachments/fec8130b-649a-4509-8142-5788089beef6.png)
[Consider the following for questions 12-15]: Suppose you took 2 of the 4 cm x 4 cm x 4 cm cubes and stuck them together to make the following arrangement:
Calculate the total surface area of this object:
SA of 1 cube side x # of sides exposed = Total SA → (4cm x 4cm) x 10 = 1602
Calculate the total volume of this object
V of 1 cube x # of cubes = Total volume → (4cm x 4xm x 4cm) x 2 = 1283
Calculate the surface area to volume ratio of this object
SA/V → 160/128 = 1.25
Suppose the two cubes above were comprised of metal and were heated to a warm temperature. What could you do to the cubes to increases the rate at which they cooled. Assume you can’t put the cubes in a cooler location
You could separate them.
Separating the cubes would expose an additional cube side on each cube, thus increasing the exposed surface area, and thus the SA/V ratio. Rate of heat loss increases as the SA/V ratio of an object increases.
For the factors below, indicate what happens to the factor as the diameter of a tube is increased (assume all other factors are constant)
Resistance to fluid moving in tube:
Flow of fluid moving through tube:
Capillary Action:
Surface Area to Volume Ratio of Tube:
Decreases
Increases
Decreases
Decreases
[Consider the following when answering questions 21-23]: In Lab 7, Exercise 1 we used fermentation tubes to assess the rate of fermentation. Assume you have a positive fermentation tube control, and a negative fermentation tube control.
Why do tubes experiencing faster rates of fermentation have greater liquid displacement?
Because those tubes produce more CO2 gas (a by-product of cellular respiration), and the gas is what displaces the liquid
Would you expect results from a tube containing yeast and an artificial sweetener to more closely resemble the positive or negative control? Why?
More closely resembles the negative control. A negative control indicates the results you would expect if a reaction were not occurring. Artificial sweeteners cannot be digested and can therefore not fuel cellular respiration
Would you expect results from a tube containing yeast, glucose, and a
respiration enzyme activator to more closely resemble the positive or negative
control? Why?
More closely resemble the positive control. A positive control shows what
optimal results for a reaction look like. Enzyme activators stabilize the active site
on enzymes, making it so the intended reactant fits well. This allows the enzyme
to speed up the reaction, in this cause the reaction we are discussing is cellular
respiration
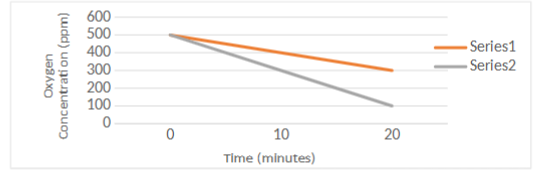
Two flasks of actively respiring peas were placed in the following temperatures: 10C and 40C.
A probe was put in each flask, to measure the relative oxygen concentrations inside the flasks.
Fluctuations in the gas concentration was recorded and used to generate the graph below
Which line on the graph signifies the gas fluctuation of each temperature? How were you able to determine this
We know that rate of cellular reactions increase as the temperature of the system increases,
until the system reaches the such a high temperature that the enzymes facilitating the reaction
denature. The warmest temperature listed here (40C) is right around body temperature
(~37C) and should therefore not be hot enough to denature enzymes. Therefore, we would
expect the warmer to temperature (40C) to have a faster rate of cellular respiration. Oxygen is
a necessary reactant of aerobic respiration, and the rate of consumption is correlated with the
rate of the reaction (faster reaction= faster decline in oxygen). Therefore, we would expect
series 3 (steeper negative slope) to correspond with the warmer temperature
As the rate of cellular respiration increases, what happens to the relative amounts of the following and why?
Oxygen:
CO2:
pH:
H+:
Oxygen: Decrease; oxygen is a reactant of aerobic respiration. Therefore, as the rate of aerobic respiration increases, the amount of available oxygen will initially dip resulting in an increase in respiration rate
CO2: Increase, CO2 is a product of respiration (both aerobic and anaerobic). Therefore, as the rate of respiration increases, the amount of CO2 increases, triggering an increase in respiration rate
pH: Decrease; the CO2 produced via cellular respiration combines with water to form carbonic acid. Acids decrease the pH of solutions
H+: Increase; the CO2 produced via cellular respiration combines with water to form carbonic acid. Acids donate H+ ions into solution