CH 302 - Exam 3 Review Facts
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Boron
usually forms only three covalent bonds (contrary to the octet rule)
found in boric acid which is used to kill ants
found in borax which is used as detergent
nitrogen
can assume many oxidation states (from -3 to +5)
found in explosives, fertilizers, and as an oxide in laughing gas, Viagra, smog, and preservatives
made with the Haber process from H2 and N2 at low temperatures
carbon
has three allotropes
C60 - forms interesting tubular + spherical structure
Graphite - sp2 hybrodized
Diamond - sp3 hybrodized
CO3- - inorganic form of carbon that is an essential partof the cycle involving greenhouse gas
CO2 - a diprotic acid
CaCO3 - limestone
phosphorous
found in soaps, toothpaste, fertilizer, and pesticides
Is extracted from Ca3(PO4)2 rock
Phosphate is the backbone of nucleic acids like DNA
Phosphates cause algae to bloom which means their use is discouraged for environmental reasons
halides
have small radii, high ionozation energy, high electronegativity, and form -1 anions
oxides and hydrides are acidic
fluoride
F- inserts instead of OH- in tooth enamel to protect from decay
Chlorine
manufactured as Cl2, a strong oxidizing agent
used in disinfection and sanitation
used to make PVC tubing
noble gases
inert with 2 or 8 electrons in filled shells
specialty uses include Cryogens (He), inert gases (Ar), and lights (Ne)
Wet Method
produces H3PO4 (phosphoic acid)
Claus Process
a two step oxidation produces elemental sulfur from H2S, which is contaminant in natural gas, methane
Contact Process
A four step oxidation process that produces H2SO4 from elemental sulfur, S
claus + contact processes
combine to produce H2SO4 through a series of oxidation and acid/base reactions
Sulfuric acid
is the most manufactured chemical in the world
about half of all sulfuric acid is used to solubilize phosphate in rocks by the wet method (the solubilized phosphate is used in fertilizers)
it is also a strong oxidizing agent and a strong dehydration agent
Batteries (aka voltaic/galvanic cells)
Delta G is negative
K is greater than 1
E is positive
Reduction occurs at the cathode
Oxidation occurs at he anode
e- flow Anode to Cathode
The sign of the cathode is positive
the sign of the anode is negative
Electrolytic cells
Delta G is positive
K is less than 1
E is negative
Reduction occurs at the cathode
oxidation occurs at the anode
e- flow anode to cathode
the sign of the cathode is negative
the sign of the anode is positive
Nernst equation theory
Q < 1 means more reactants (E>E knot)
Q = 1 means equal ratio of products to reactants (E=E knot)
Q >1 means more products (E<E knot)
Q = K means the system is at equilibrium (E=0)
What is the value of a Faraday?
9.65 × 10^4 C
aluminum
lightest metal commonly used in commercial applications
obtained from the energy intensive Bayer Process (bauxite rocks —> aluminum oxide) and the Hall Process (aluminum oxide —> aluminum)
the oxite forms ruby, sapphire, and topaz gemstones
alkali metals
have large radii, low ionzation energy, low electronegativity, and form +1 cations
oxides and hydrides are basic
metal displacement reaction with water: M + H2O —> M^+n + OH- + H2
react vigorously in water (coinage metals [Cu, Ag, Au, Pt] do not)
beryllium
forms covalent bonds because of its small size and ability to attract electrons
calcium
in building materials from concrete to teeth
Mg2+
in chlorophyll
makes the molecule more rigid to permit electron transport for photosynthesis instead of energy loss through vibrations
Group 1 Metals
react in cold water (explode)
Group 2 metals:
React in hot water
Transition metals
react in acid
Coinage metals
do not react
displacement reactions of metals
most reactive metals are in the lower right corner (Li is the strongest reducing agent)
least reactive metals are in the top right corner (Au is the weakest reducing agent)
General displacement reaction: M1 + M2X —> M2 + M1X — metal 1 is more active and replaces metal 2
What are the demands of a modern battery?
small
cheap
high power
long life
reversible
environmentally safe
how are secondary batteries built with consideration?
minimize liquid or gas production, so contents of the reaction are contained in the battery casing for recharging
What is wrong with lithium-ion batteries?
they catch on fire — ex) they’re not allowed as commercial cargo on places
Li-ion batter parts
From left to right
Cathode (+)
Electrolyte (contains everything)
Lithium-metal oxide
porus seperator
lithium ion + lithium-carbon (graphite)
anode (-)
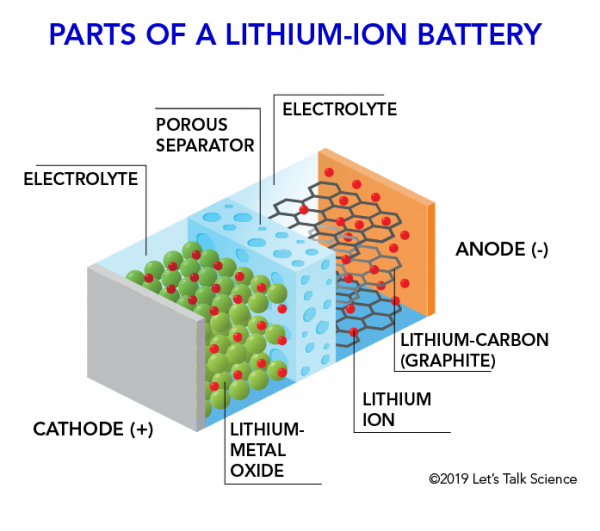
battery design
modern batteries are made with solids and pastes rather than liquids and gases to avoid losses of material
primary batteries are single use. Rechargeable = secondary batteries are environmentally superior and convenient
inefficient batteries produce heat, not work. Controlling heat dissipation is important practically and for safety
Lightweight batteries use less dense, higher charge density materials (like lithium)
Primary Batteries
disposed of after one cycle
famous batteries include:
common alkaline battery (ex: Duracell) — used in household appliances
Zn-C battery — used in hearing aids
Secondary barreries
reversible (reusable/rechargeable)
built with consideration
famous batteries include:
Lithium-ion batteries — used in electronics and electric cars
lead-acid batteries — used in gas-powered vehicles, recharged by an alternator
NIMH (replacing NiCd [know both]) — used in household appliances
rust in the real world
Rust is made from the oxidation of iron in the presence of water and air for a long period of time
Fe + H2O + O2 —> Fe(OH)2 + Fe(OH)3
Rusting can be prevented by adding metals that are easier to oxidize through specific processes:
Galvanization with Zn
Sacrificial electrodes with Mg, Al
Stainless steel with Cr and V added through Fe
Waxes and coating