chp 25 nitrogen acquisition and amino acid metabolism
1/152
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
153 Terms
microorganims acavenge amino acids from their __________ for fuel when needed
environment
herbibores require ________ amount of energy from amino acids
small
plants and usage of amino acids as fuel source
do not use them for fuel but degrade them to form other metabolites
carnivores and amino acids as fuel
get 90% of energy needs from AA’s
under what 3 circumstances do amino acids undergo oxidative catabolism
leftover amino acids from normal protein turnover are degraded
amino acids from diet that exceeds body’s needs are degraded
proteins in body broken down to supply amino acids for energy when carbs are scarce
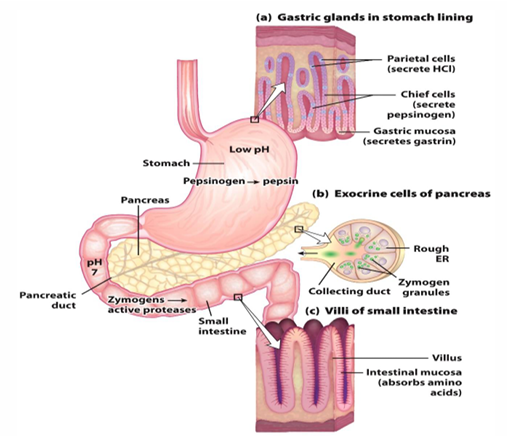
where does digestion of proteins begin
stomach
protein entering stomach stimulates secretion of what
gastrin what
gastrin stimulates secretion of what 2 things
HCL to decrease pH
pepsinogen
pepsinogen is converted into what
pepsin
what does pepsin do
cuts protein into peptides in the stomach
in the stomach we go from proteins to
peptides
where are peptides then hydrolyzed into amino acids
small intestines and pancreas
how is acidic content from stomach neutralized
secretion of secretin which stimulates bicarbonate release from pancreas
presence of AA in small intestines causes what to be released
cholecystokinin (CCK)
CCK does what
stimulates pancreatic enzyme release (trypsinogen, chymotrypsinogen, procarboxypeptidases A and B)
(also secretes bile for lipids)
what other macro also causes CCK to be released
lipids
what converts trypsinogen into trypsin
enteropeptidases (as they do proteolytic cleavage)
trypsin activation activates what enzymes
chymotrypsinogen, procarboxytidases, prelastasem and more trypsin
function of pepsin, trypsin and chymotrypsin, and aminopeptidases and carboxypeptidases
-pepsin will break down proteins into peptides
- trypsin and chymotrypsin will break peptides into small peptides
-aminopeptidases and carboxypeptidases will break down the small peptides into amino acids
the free amino acids go where (from IS to what organ)
into SI epithelial cells —> enter capillaries—> and are taken to liver
why do we have zymogens
because pancreas holds most of our enzymes and if they were active would cause harm to pancreas
what is pancreatic trypsin inhibitor
released by pancreas to prevent autodigestion of pancreas
BECAUSE TRYPSIN ACTIVATES MOST OF THE PANCREATIC ENZYMES
by what are zymogens activated in SI
trypsin
protein degration from stomach to SI cells
Parietal cells release HCL, chief release pepsinogen, and gastric mucosa release gastrin all to start the digestion. In the stomach, the pepsinogen is turned into pepsin (most important) to begin the digestion. Then these will go to the duodenum, and will have bicarbonate released to raise the pH. Then pancreas releases zymogens to continue digestion which are activated. Then when we have amino acids, we absorb them
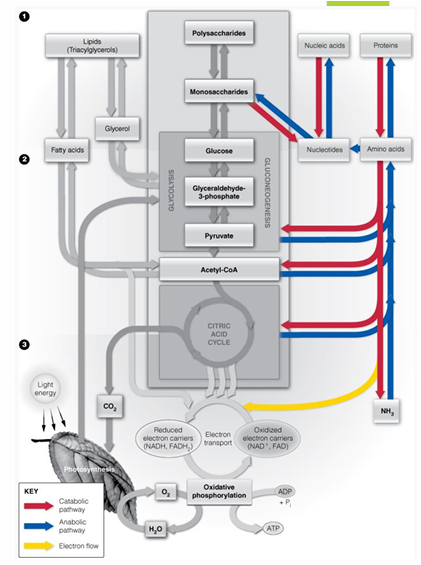
pathway of nitrogen metabolism picture
how are amino acids stored in body
they are not, so proteins are continuously undergoing protein turnover (biosynthesis and degradation)
since amino acids not really stored, how do animals get nitrogen
continuously replenish supply through diet, but if not enough then will use muscle protein (but wont be replenished)
fates of nitrogen in organisms (plants, aquatic vertebrates, terrestial vertebrates and sharks, birds and reptiles, HUMANS )
plants- conserve almost all nitrogen
aquatic vertebrates release ammonia to environment
aquatic vertebrates, terrestrial vertebrates and sharks excrete ammonia as urea
birds and reptiles excrete nitrogen as uric acid (pasty)
humans excrete urea (from AA) and uric acid (from purine catabolism)
humans do what to nitrogen (how excreted)
urea and uric acid
ammonia is what are high levels
toxic
all organisms assimilate NH3 via reactions yielding (what 4 molecules is NH3 possibly turned into
glutamate
glutamine
asparganine
carbamoyl phosphate
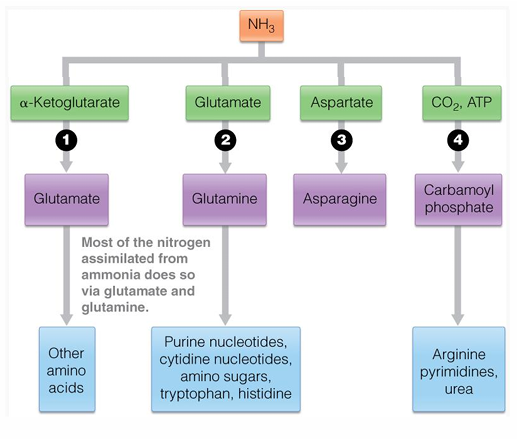
what 2 amino acids are the collecting point for NH2
glutamine and glutamate
in most tissues, what occurs to NH4+
is transferred to glutamate to form glutamine which can then enter the liver and be turned into glutamate again and release NH4+ to urea cycle
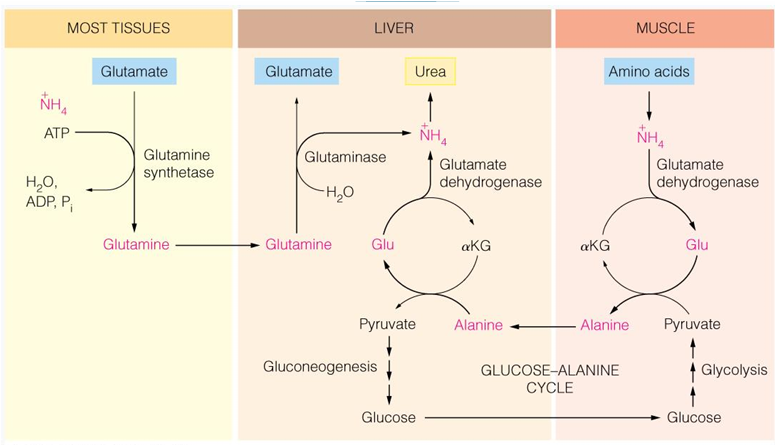
in skeletal tissue (muscle) what occurs to NH4+
NH2 is transferred to pyr to form ala, which is transported to the liver and the amino group is transferred to αKG to form glu
the glu can then release NH4 for urea
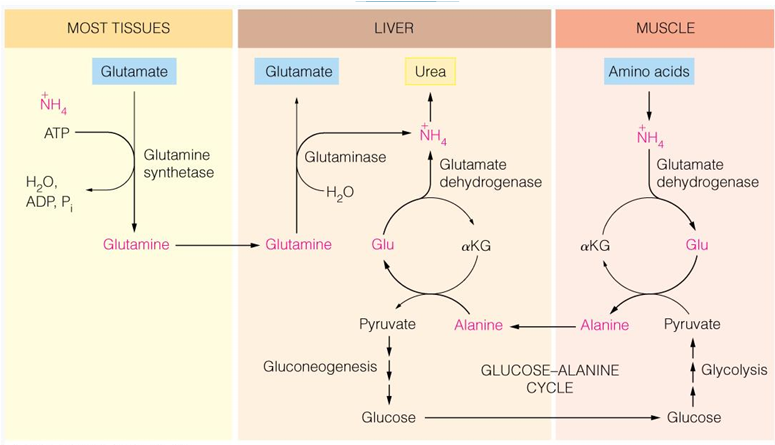
in liver, what occurs to NH4+
AA transfer of NH2 to αKG to form Glu,
Enters the mitochondria to release NH4+ (enzymatic transamination)
what is the first step in degradation of amino acid
removal of amino group
how is the amino group of AA first removed
through enzymatic transamination
what is transamination
removal of amino group from amino acid and transferring it to keto acid, which will make the amino acid a keto acid and the keto acid an amino acid
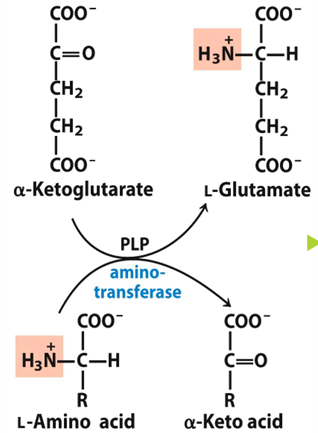
what enzymes do transamination
aminotransferases
what do aminotransferases require
cofactor PLP (piroxal phosphate)
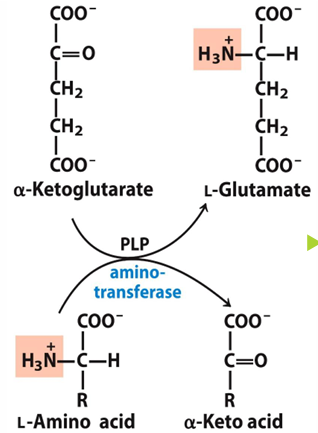
PLP is derived from
B6 vitamin
what typically accepts the amino group
alphaKG (because when it accepts amino group it becomes glutamate)
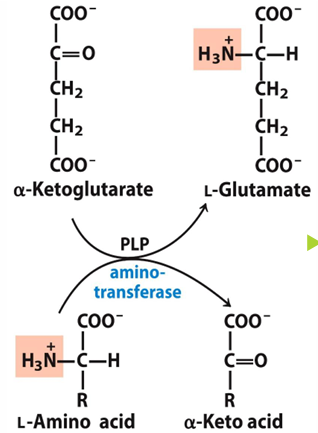
what can act as temporary storage of nitrogen
L-glutamine (can donate the Amino group when needed for AA biosynthesis or amino group can be converted to urea)
aminotransferases are high in what tissue
liver
L-glutamate can function as what for what 2 pathwaysb
amino group donor for biosynthesis pathway OR excretion pathways
because it carries an amino group that can be used to make amino acid or released to go to urea cycle
PLP serves as what
coenzyme for aminotransferases (from B6)
what are the 3 metabolic fates of ammoniun (3 enzymes perform the 3 reactions that could occur)
carbamoyl phosphate dehydrogenase (ammonium—> carbamoyl P)
glutamate dehydrogenase (glutamate to alphaKG by removal of NH4 and usage if NAD or NADP+)
glutamine synthetase (glutamate to glutamine by adding NH4)
carbamoyl phosphate synthetase 1 (does what, ATP needs)
will convert ammonia into carbamoyl phosphate (prep of urea cycle)
2 ATP needed (one to activate bicarbonate and one to phosphorylate carbamate)
what does glutamate dehydrogenase do
reductive amination (add amine) to alpha ketoglutarate to form glutamate OR remove amino group from glutamate to form alpha-ketoglutarate and ammonia (NH4+)
-uses NAD or NADP
what does glutamine synthetase do
ATP dependent amination glutamate to glutamine
after enzymatic transamination in liver, what must happen to ammonia (what amino acid must do so)
Glutamate must dispose of it (since glutamine is carrying the ammonia group as NH3+)
amino acids are converted into what amino acid during transamination to be able do urea cycle
glutamate
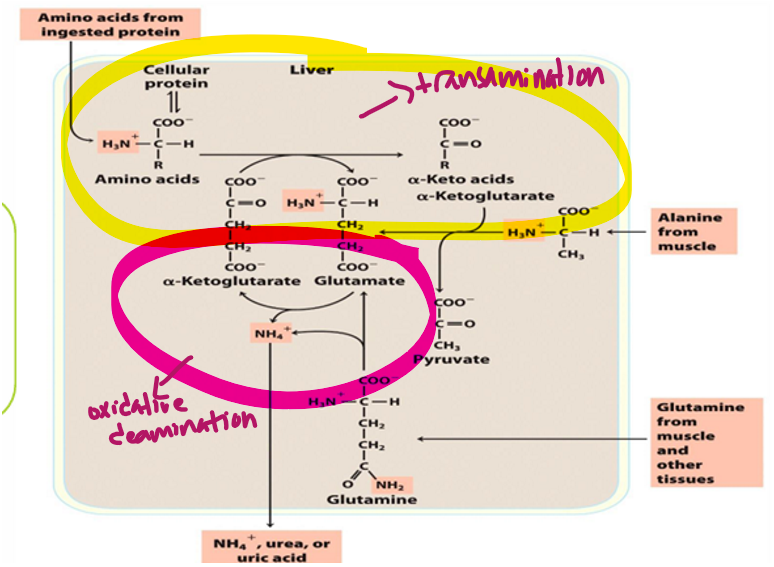
ammonia collected in glutamate is removed by what enzyme
glutamate dehydrogenase
the process of removing the ammonia from glu is what process
oxidative deamination
glu dehydrogenase uses what
NAD+ or NADP+ as electron acceptor (to remove a H and make intermediate)
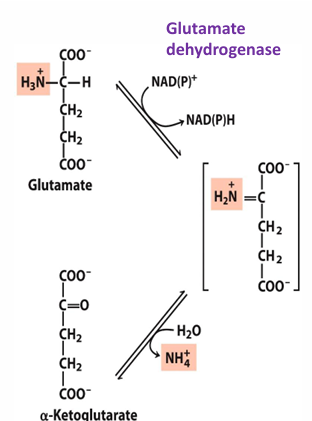
the ammonia released is then processed into what
urea
the pathway for ammonia excretion therefore includes what 2 processes
transdeamination= transamination + oxidative deamination
Transamination to get amino acid into glutamate
Oxidative deamination to remove ammonia from glutamate and make alpha-ketoglutarate
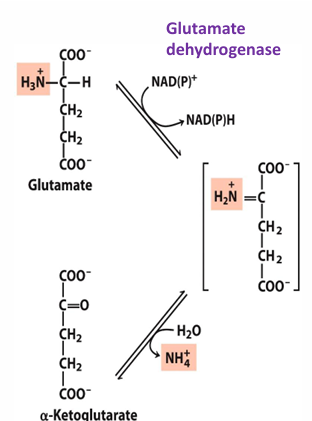
where does oxidative deamination occur in liver cell
mitochondrial matrix
how is ammonia transported from tissues into the liver
in glutamine
the ammonia (NH4+) from intestines and kidneys can go directly to what organ
liver
other tissues have ammonia added to what to form what to be transported
add ammonia to glutamate to make glutamine (by glutamine synthetase)
rmb that glutamine has 2 amino groups
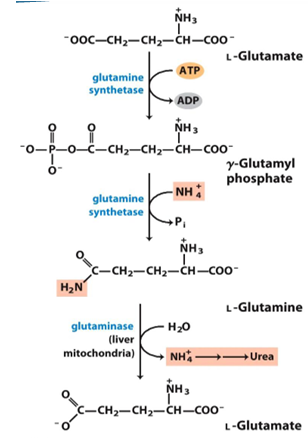
what enzyme makes glutamine
glutamine synthetase (adds amino group)
what happens to the glutamine
it is deaminated to make glutamate and release ammonia
where is glutamine deaminated (what tissues)
intestines, kidneys, and liver
what enzyme is the enzyme that does oxidative deamination
glutamate dehydrogenase
once we have glutamate, it undergoes what (process and enzyme)
further deamination to release the other amino group ads ammonia by glutamate dehydrogenase
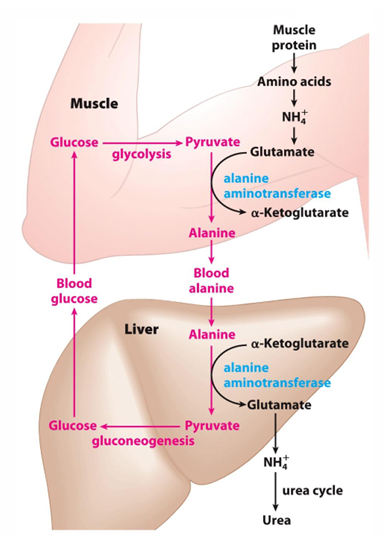
glutamate can also donate ammonia to what molecule when exercising in muscle
pyruvate (to make alanine)
vigorous exercise operates using what respiration type and relies on what pathway for energy
anaerobic
glycolysis
glycolysis yields what compound
pyruvate
during exercise, pyruvate is converted into what
lactic acid
to prevent build up of lactic acid in muscle, we can turn pyruvate into what
alanine to be taken to liver
the NH2 from muscle is transported to the liver in what form (what molecule)
alanine
what is the glucose alanine cycle
cycle where we have muscle protein broken down to give ammonia which then is made to glutamate —> glutamate will donate the amino group to pyruvate to make alanine—> alanine can go to liver to be made back into pyruvate—> into glucose—> back to muscle—> undergo glycolysis to make pyruvate and cycle restarts
what cycle removed ammonia from body
urea cycle
urea cycle turns ammonia into what
urea
where does the ammonia enter from originally
from glutamate, alanine, or glutamine —> but all is converted into glutamate to start process
glutamate in liver releases ammonia how (process and enzyme)
oxidative deamination by glutamate dehydrogenase
what is the only tissue that can produce urea
liver
where in cell does urea cycle occur
some in mitochondria and most in the cytosol
mitochondria is prep and 1
citosol is step 2-4
when glutamate is deaminated by glutamate dehydrogenase, what happens to the ammonia
it will react with bicarbonate to form carbamoyl phosphate
since glutamate was made through transamination and oxidative deamination, collectively the formation of deaminated glu is called what
transdeamination
glutamate dehydrogenase can use what electron acceptors
NAD+ or NADP+
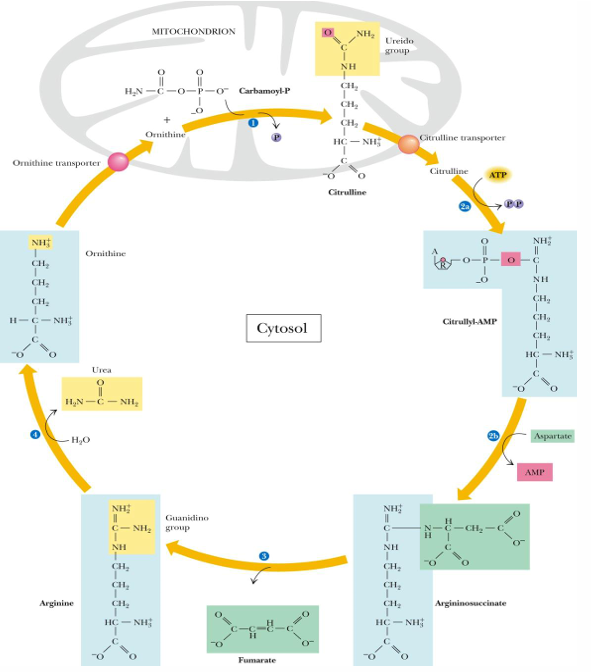
steps of the urea cycle (substrates, products, enzyme if known)
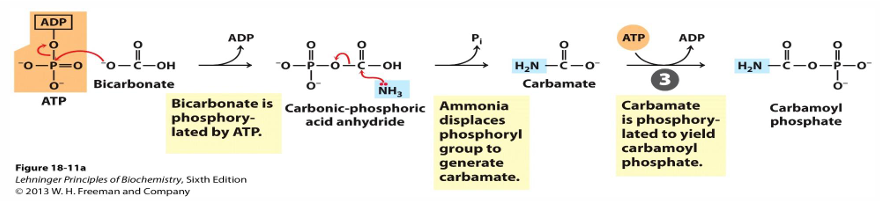
prep: ammonia + bicarbonate —> carbamoyl phosphate
by carbamoyl P synthetase (requires 2 ATP and occurs in mitochondria; first amino group entering cycle)
1. ornithine + carbamoyl P —> citrulline (in mitochondria)
2. citrulline undergoes 2 part reaction
citrulline —> citrullyl AMP (use ATP—> PPi= equal to 2 ATP)
citrullyl AMP —→ arginosuccinate (by adding aspartate with our seconds amino group)
3. arginosuccinate —> arginine (releasing fumarate)
4. arginine —> ornithine (RELEASES UREA)
ornithine taken into mitochondria to start cycle again
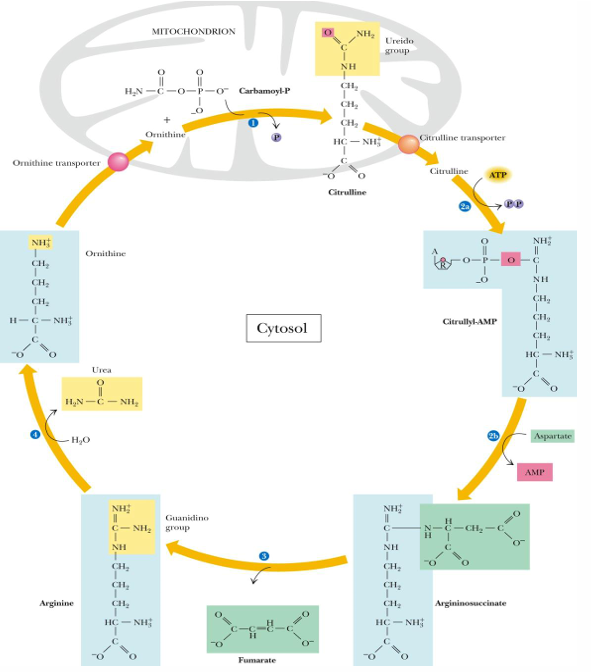
before we can begin the urea cycle, the prep stage does what (enzyme, reaction, ATP)
carbamoyl P synthase recaptures the ammonia released by glutamate and adds it to bicarbonate to make carbamoyl phosphate
this requires 2 ATP
is carbamoyl phosphate synthase regulated or not
yes
after we make carbamoyl phosphate what is the next reaction
carbamoyl phosphate will combine with orthenine to make citrulline in the mitochondria still
after citrulline is taken to the cytosol, what reactions occurs next in the urea cycle
2 part reaction
citrulline —> citrullyl- AMP (uses 2 ATP)
citrullyl- AMP —> arginosuccinate (has addition of aspartate with 2nd amino group)
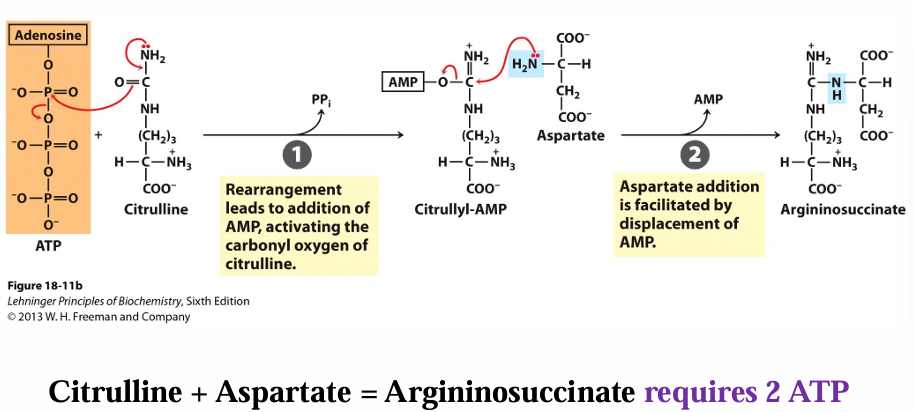
so far, how many ATP have been used in the urea cycle
4 ATp
2 from prep (carbamoyl P synthase)
2 from step 2 (citrulline —> arginosuccinate)
at what points does amino groups enter the urea cycle
carbamoyl phosphate
aspartate
after we get arginosuccinate, what occurs in urea cycle
has fumarate taken off and becomes arginine
after we have arginine, what occurs
we release UREA and make ornithine
in what step do we make urea to be excreted
step 4 (arginine —> ornithine by releasing urea)
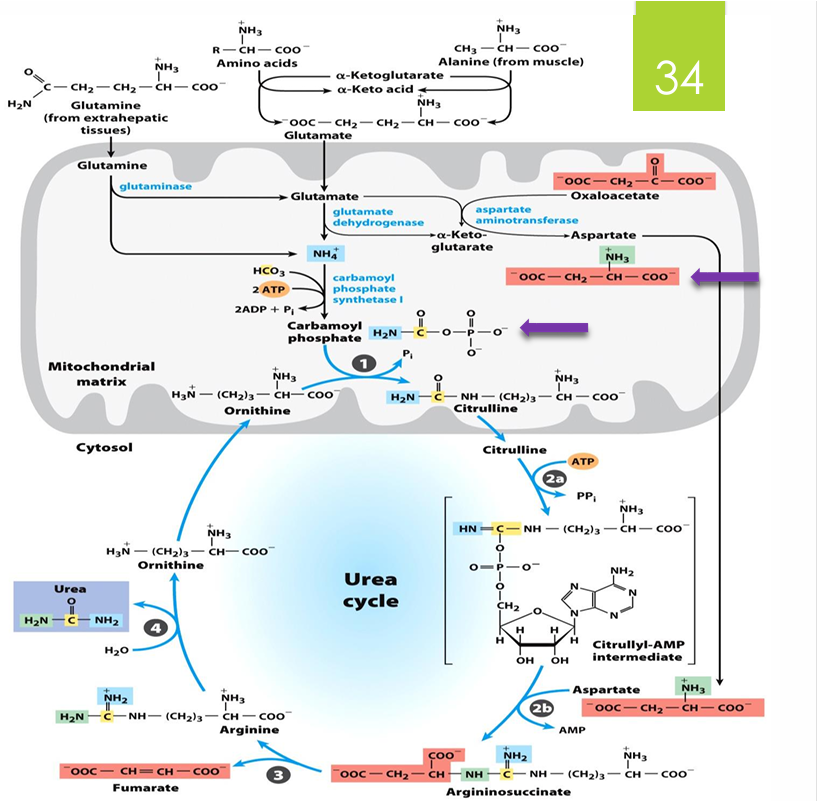
what is the TOTAL energy cost of urea cycle
4 ATP
2 ATP required for the formation of carbamoyl P
2 ATP required for the formation of argininosuccinate
BUT, what is also made in the urea cycle that can provide energy
fumarate
fumarate ends up going to ETC and provides how much ATP
2.5
So what is NET ATP required for urea cycle
1.5 ATP
what allosterically activates carbamoyl phosphate synthetase
N-acetylglutamate (made from glutamate and acetyl CoA)
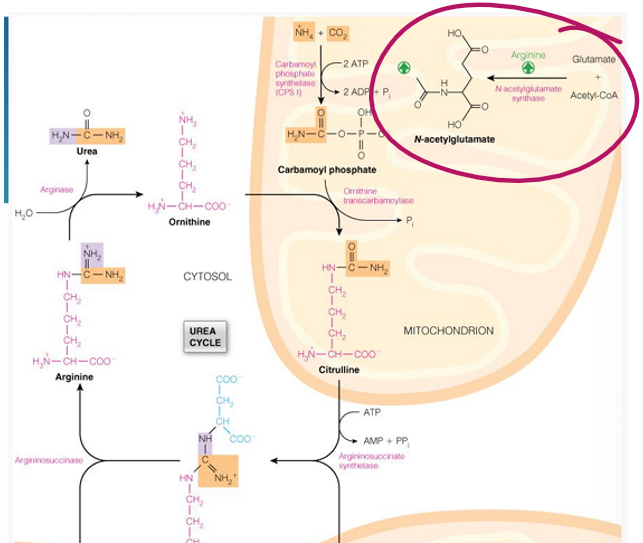
regulation of the urea occurs in the _______ and ________ term
long and short
what molecule does short term regulation
N-acetylglutamate (by activating carbamoyl phosphate synthetase 1
how is N-acetylglutamate made
by N-acetylglutamate synthetase in the liver
done by putting glutamate and acetyl CoA together