4.8 - Organic Synthesis and Analysis
1/27
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
28 Terms
Mass spec
Highest m/z = Mr
Use others to find fragments using parts of Mr value
Add a positive charge to all species
Give species and value
Infrared spectroscopy
Give bond and frequency
Focus on bonds in functional group
Absence of a bond can be useful
C NMR
Number of peaks => number of carbon bonding environments
Shift value => nature of carbon bonding environment
Low resolution H NMR
Number of peaks => number of H bonding environments
Shift value => nature of H bonding environment
Ratio of peak areas => relative number of equivalent hydrogen nuclei in each bonding environment
Reflux
Heating volatile organic compounds
Needed as reactants would evaporate before reaction is complete + products (if volatile) would also be lost
Filtration
Filtration under suction or reduced pressure = better at removing an insoluble solid from a liquid than paper filtration
Distillation
Simple distillation = separate a mixture of a soluble solid from the solvent
Fractional = separate a mixture of miscible liquids with different boiling temperatures
Steam = seperate insoluble liquids from a reaction mixture
Reduced pressure can be used if the substance decomposes when heated. This is used to lower the Tb
Simple distillation
Simple distillation = separate a mixture of a soluble solid from the solvent
Fractional distillation
Fractional = separate a mixture of miscible liquids with different boiling temperatures
Steam distillation
Steam = seperate insoluble liquids from a reaction mixture
Distillation at a reduced pressure
Reduced pressure can be used if the substance decomposes when heated. This is used to lower the Tb
Explain the process of recrystallisation
Purify a solid. Solvent chosen is one that dissolves more of the solid when it is hot than when it is cold
Dissolve the solid in the minimum volume of hot solvent
Filter hot if required to remove any in dissolved solid which would be an impurity
Cool the hot solution
Filter under suction/reduced pressure to remove the solid
Wash the solid with ice cold solvent
Dry to a constant mass
Determine the purity of the solid by testing its melting temperature
Melting temperature and purity
Pure = has an exact melting temperature
Impurities = melting temp is lowered and solid will melt over a range of temps below the one given
Addition polymerisation
One type of unsaturated monomer
Polymer is only product
Condensation polymerisation
Two types of monomer
Polymer and small molecules produced
2 types
2 examples of condensation polymers
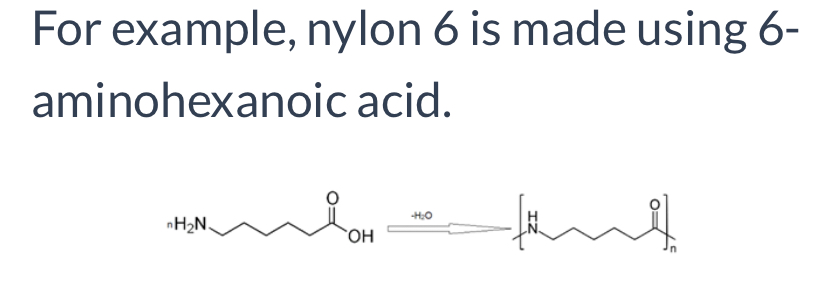
Polyamides
Polyesters
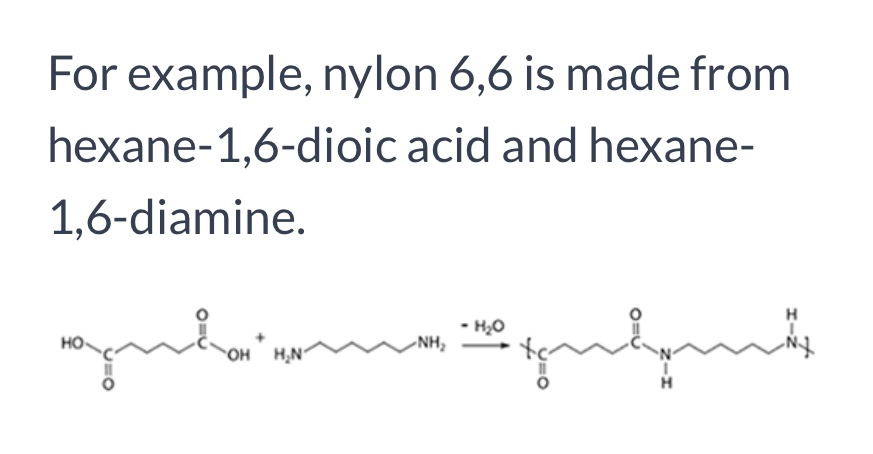
Polyamides
Diamine + dicarboxylic acid
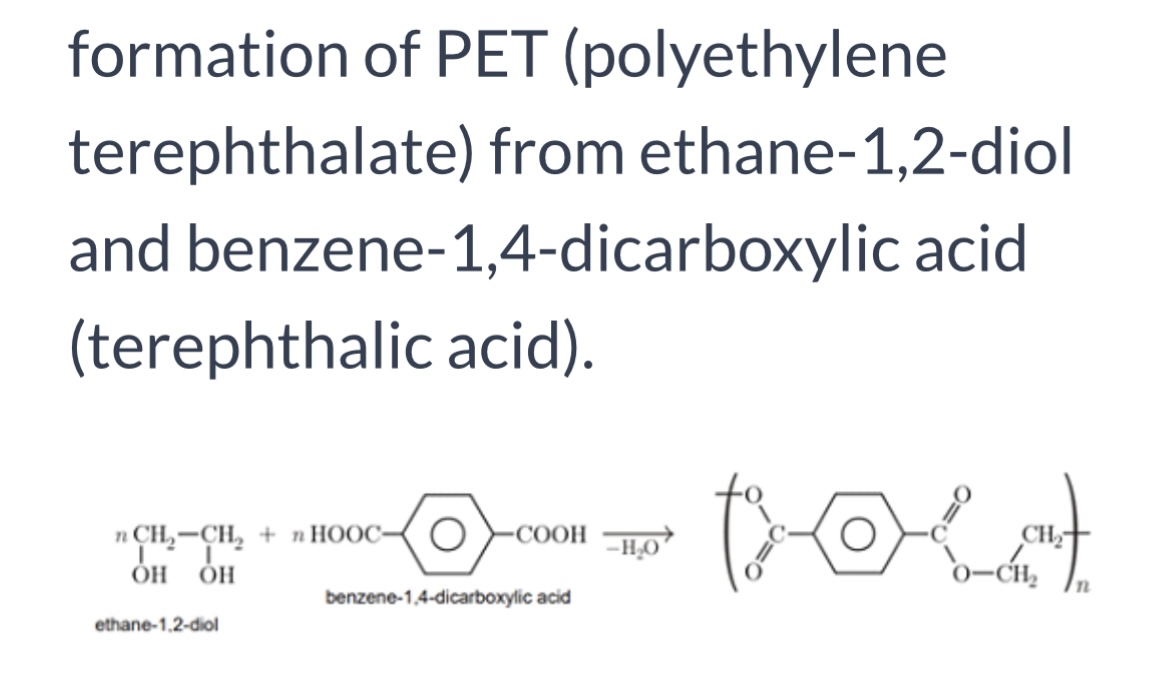
Polyesters
Diol + dicarboxylic acid
Formation of PET
Formation of Nylon-6
Formation of Nylon-6,6
High Resolution H NMR
Number of peaks => number of H bonding environments
Shift value => nature of H bonding environments
Ratio of peak areas => relative number of equivalent H nuclei in each bonding environment
Splitting pattern of the peaks => (n+1) rule where a peak splits into n+1 parts where n= total number of H nuclei on neighbouring C atoms
0 atoms = singlet peak
1 = doublet
2 = triplet
3 = quartet
Thin layer chromatography
SP = surface of a thin glass/plastic plate
Spot of mixture placed about 10mm from bottom edge
Place stood or suspended in about 5mm of solvent
Solvent travels up plate
Components separate
Components identified from Rf (retardation factor)
Stationary phase
The stationary phase is the part of the chromatography that remains in place as the process occurs. In paper chromatography, this is the paper, and in thin layer chromatography, this is the silica or aluminium oxide on the TLC plate.
Mobile phase
The mobile phase is the part of the chromatography that moves as the process occurs. In both paper and thin layer chromatography, this is the solvent being used.
Rf value
Distance moved by component/distance moved by solvent front
Gas liquid chromatography
Organic mixtures
Components identified from time taken for component to reach detector (retention time)
Integrate area under pan in the recording of the detector = estimate of amount of component present
High performance liquid chromatography
Mixture forced through a column of the SP (small particles in a small diameter tube)
Pump a liquid (MP) through
Components identified by retention times
Areas under peaks by integration