U2 Chem
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Observations from combustion of alcohols
Blue flame (complete combustion), less clean the longer the chain
Observations from the oxidation of an alcohol using KCr2O7
The reaction mixture turns from orange to green
Conditions for oxidation of alcohols
Oxidising agent like KCr2O7, dilute H2SO4, heat under reflux/ distillation with addition
What oxidation reactions do alcohols undergo
1* alcohols → aldehyde (and water) → carboxylic acid
2* alcohols —> ketone
3* alcohols —> no reaction
Conditions for the dehydration of alcohols
Concentrated acid -phosphoric acid and heat
Conditions for the chlorination of a 1* or 2* alcohol
PCl5 (white solid)
Conditions for the chlorination of a 3* alcohol
Concentrated HCl and shaking
Conditions for the bromination of an alcohol
Potassium Bromide and sulfuric acid at 50%
Conditions for the iodination for alcohols
Red phosphorous and iodine
Test for a hydroxyl group
PCl5, if the mixture releases misty fumes, a chlorination reaction has taken pace
Test for aldehydes after distilling it from oxidising an alcohol
Benedict’s/ Fehling’s solution (blue —> orange)
Test for carboxylic acids
Base (NaCO3 or NaHCO3) reacts to form CO2 which can be bubbled through lime water and should turn cloudy
Addition reaction general equation
A + B = C
General elimination reaction
2 atoms/ groups attached to carbons are removed
The carbons forms a double bond
Substitution reaction general equation
A + B = C + D
What is a hydrolysis reaction
A type of substitution reaction where the OH replaces an atom/ group
Reaction of hallogenoalkanes with water or alkalis
An alcohol is produced
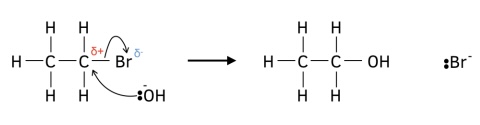
Mechanism of hallogenoalkane with ethanolic potassium hydroxide

Conditions for the elimination reaction of a hallogenoalkane with an OH- group
Ethanolic KOH with hear under reflux
Test for C-Hallogen bond length and strength
Hydrolysis reaction: water and heat/ NaOH is added to displace the halide ions
AgNO3 is added to form a silver halide precipitate
Silver halide precipitate colours
Silver chloride: White
Silver bromide: Cream
Silver iodide: Yellow
Bond test speed for Cl, Br, I and 1*, 2*, 3* hallogenoalkanes from fastest to slowest
I, Br , Cl and 3*, 2*, 1*
Conditions for nucleophilic substitution of ammonia with a hallogenoalkane
concentrated alcoholic ammonia
Sealed test tube (pressure)
Heat
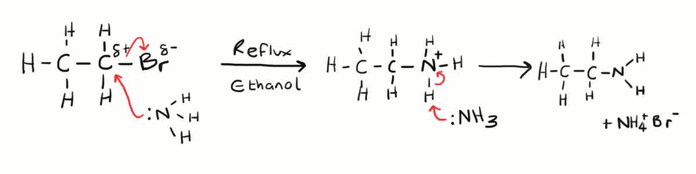
Mechanism for reaction of ammonia with halogenoalkane
(Chlorine may also take a hydrogen to form HCl instead of ammonia in the intermediate)
Reaction mechanism for cyanide ion with hallogenoalkane
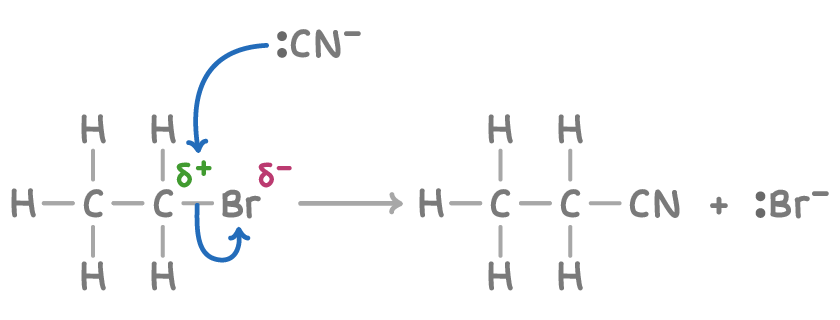
Conditions for reaction of cyanide ion with hallogenoalkane
KCN
Heat under reflux
Pattern in oxidising and reducing powers of halogens
Oxidising: high and decreases down the group
Reducing: low and increases down the group
Colours of halogens in solutions
Chlorine: Pale green
Bromine: Yellow/ orange
Iodine: Brown
Colours of halogens in non polar/ organic solutions
Bromine is a brighter orange and iodine is purple
Cyanide ion
CN-
Colours of halogens in standard conditions
Fluorine: yellow gas.
Chlorine: pale green gas.
Bromine: orange-brown liquid
Iodine: grey-black solid with purple vapour.
Test for halide ions
-Nitric acid is added to remove other ions like carbonates
-Silver nitrate solution is added
-Once the precipitates are formed if they are difficult to differentiate, NH3(aq) is added to test solubility
Solubility of silver precipitates in aqueous ammonia
Silver chloride dissolves in dilute concentrations
Silver bromide dissolves in concentrated concentrations
Silver iodide does not dissolve
Colours of halides in gases, aqueous solutions or acids
Colourless in all
Rules in oxidation number for oxygen
Usually 2-
With flourine it is 1-
In peroxide it is 1-
Rules in oxidation number for hydrogen
1+ except when combined with a less electronegative element (metals)