Vaccines
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
What is a vaccine
Protein antigen to stimulate an immune response
What would the perfect vaccine be?
provides an immune response that is strong, lifelong, appropriate and in the right place.
Safe, cheap and easy to administer.
Types of bacterial vaccine
- inactivated/ killed
- attenuated
- subunit
- recombinant
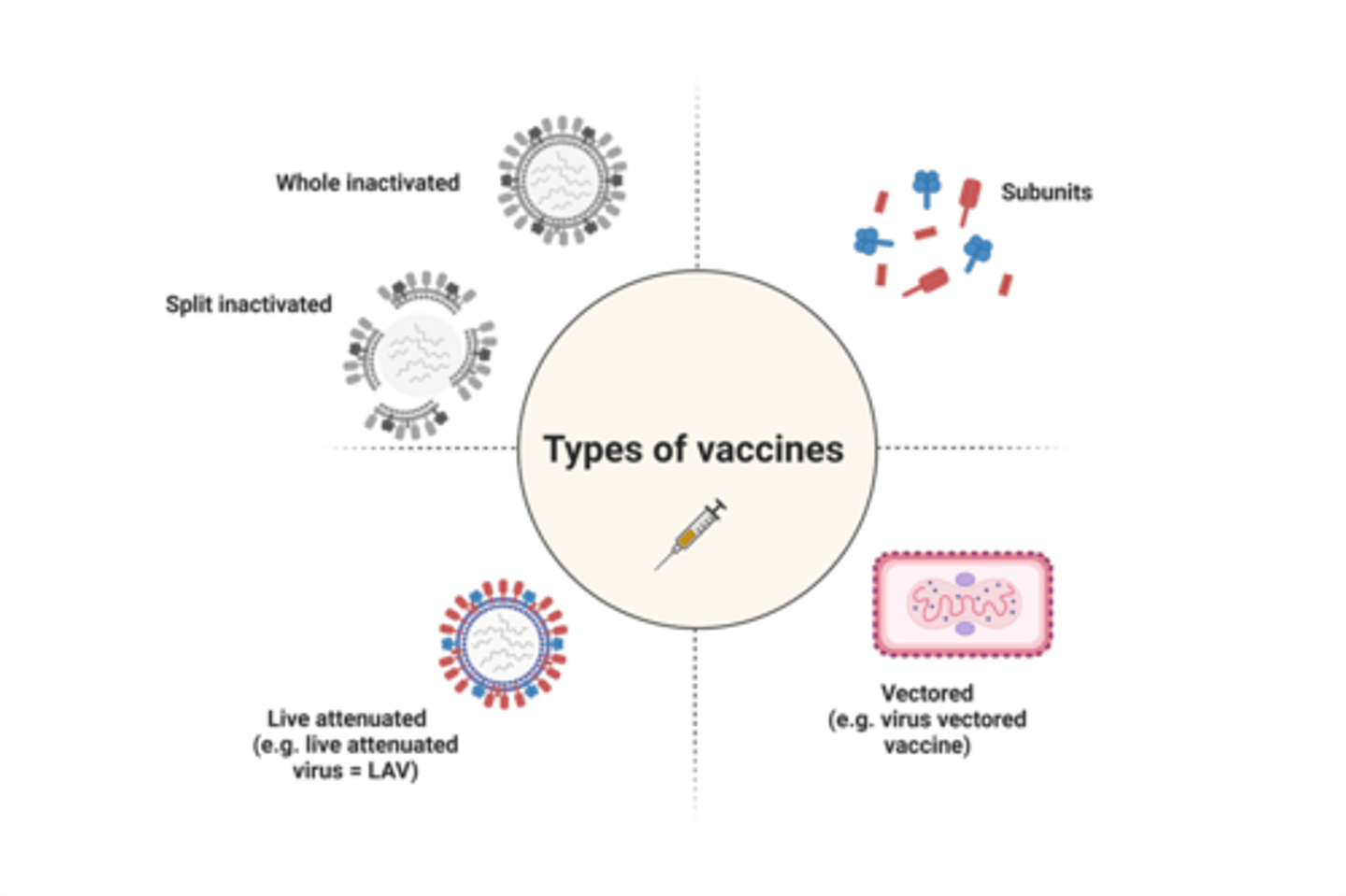
What are the different types of vaccines?
Infectious
Non-infectious
Are inactivated/killed vaccines infectious or non-infectious?
Non-infectious: These vaccines use viruses or bacteria that have been killed or inactivated so that they cannot replicate or cause disease. They still stimulate an immune response because they contain the whole (or split in the case of some virus inactivated vaccines), inactivated pathogen.
Are live attenuated vaccines infectious or non-infectious?
Infectious (but weakened): These vaccines use live viruses or bacteria that have been attenuated (weakened) so that they cannot cause disease in healthy people but can still replicate to some degree in the body. This allows the immune system to recognize and respond to the pathogen as if it were a real infection.
Are subunit vaccines infectious or non-infectious?
Non-infectious: These vaccines contain only specific parts or proteins of the pathogen (like surface proteins) rather than the whole pathogen. Because they don't contain live or inactivated whole pathogens, they are non-infectious.
Are recombinant vaccines infectious or non-infectious?
Non-infectious: These vaccines involve using genetically engineered organisms (like bacteria, yeast, or mammalian cells) to produce specific proteins from the pathogen. These proteins are then used in the vaccine to stimulate immunity. The vaccine does not contain the whole pathogen, and it's non-infectious.
Are nucleic acid vaccines (DNA, RNA, mRNA) infectious or non-infectious?
Non-infectious: These vaccines use genetic material that codes for a protein from the pathogen. The body uses this genetic information to produce the protein, and the immune system generates a response. Since the vaccines do not contain live pathogens, they are non-infectious.
Are vectored vaccines infectious or non-infectious?
Non-infectious: While vectored vaccines use viral vectors (which are viruses different to the target virus of the vaccine), they are non-infectious because the vectors are engineered to be non-replicating and harmless to humans. They do not cause disease, but rather deliver genetic material (for a protein from the target virus) to prompt an immune response. The vector’s only job is to deliver the genetic instructions for producing the target antigen (such as a viral protein) inside the body.
why is it that attenuated vaccines cant replicate fully
got something mutated in them
What are the types of viral vaccine?
- Live non-attenuated/attenuated virus (LAV)
- Inactivated/killed (can be whole virus or split)
- Subunit
- Vectored
- Nucleic acid
describe how a vectored virus vaccine works
Taken from something else
Insert a gene from the thing you are wanting protection from
So vaccine will replicate a tiny bit but will produce lots of proteins to cause an immune response
Bc its not adapted to the animal
(uses a different virus called the vector to deliver genetic material from the target virus so that it can encode a protein from the target virus and trigger an immune response without causing disease = recombinant DNA technology)
how does a viral nucleic acid vaccine work
(directly deliver the genetic material (either DNA or mRNA) into the body, where the cells use it to produce the target antigen)
how does subunit viral vaccines work
(only part of active virus is used e.g. specific proteins or fragments, these can be produced in a lab = recombinant DNA technology)
what are whole viruses
culture the vacccine and then acticate with chemicals
how can we attenuate vaccines
grow them at a low temp
virus adapts to low temp
so when in animal → doesnt replicate as well
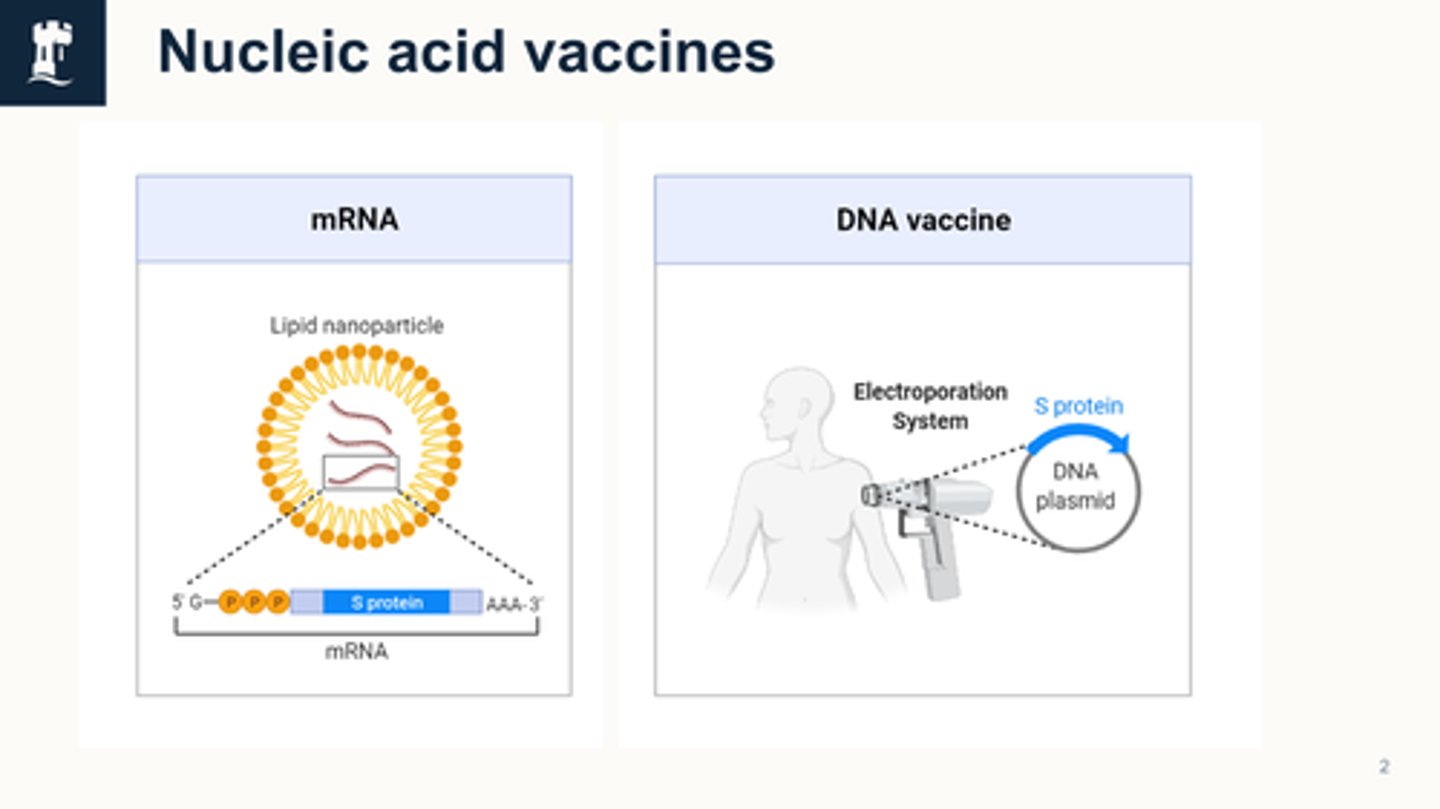
what are the 2 nucelic acid vaccines
mRNA and DNA
explain dna vaccines
inserting a plasmid containing a gene from the target virus into the body.
This plasmid carries the genetic instructions for a viral protein which is produced inside the body to stimulate the immune system = recombinant DNA technology
has to get into the nucelus tho → can be hard to get in
use gene guns to fire with high pressure
can we use nucelic acid vaccines for bact
Can also have the same principle for nucleic bacterial vaccinees that instead use DNA or RNA to deliver genetic material from a bacterial pathogen into the body to stimulate an immune response, similar to how DNA vaccines and mRNA vaccines work for viral infections.
Explain mRNA vaccines
Recombinant DNA technology is used to design the mRNA sequence that codes for a viral protein.
This mRNA is packaged into lipid nanoparticles and injected into the body where it instructs cells to produce the viral protein to trigger an immune response
gets into cytoplasm of cells → template for protein
but rna is fragile → protected by the lipid nanoparticles
require cold storage
What is the difference between recombinant and nuclei acid vaccines?
recombinant vaccines, the antigen (protein) is often produced in a host organism like yeast, bacteria, or insect cells and then purified for use in the vaccine.
- In nucleic acid vaccines, the body itself is used " to produce the antigen, with the genetic material injected directly into the body (not within a something like vectored vaccines).
However the vaccines do rely on recombinant techniques because the genetic material encoding the antigen is not from the natural source. Instead, it is engineered (recombinant) to instruct the body's cells to make the necessary pathogenic protein.
Pros and cons of infectious vaccines
Replication - rapid robust immunity
Single dose may be enough
Potential to cause mild disease (immunocompromised)
Pros and cons of non-infectious vaccines
Less likely to stimulate both cell-mediated immunity and antibodies
Cannot cause signs of disease - safer
Usually requires multiple doses + adjuvant
Duration of immunity generally short (annual)
define adjuvant
an additive which alerts the body to the presence of the vaccine, enhancing the immune response
What is a pro of inactivated/killed vaccines?
Can be made rapidly
Name two cons of inactivated/killed vaccines?
Expensive if high level of containment required and mostly short duration of immunity
What is a limitation of subunit vaccines?
Often poor immune response
What is a potential risk of live attenuated vaccines?
Potential for reversion to virulence
What is a characteristic of vectored vaccines?
Immunity to the backbone
The backbone of a vectored vaccine is the viral vector→ the virus that has been engineered to carry the genetic material (usually DNA or RNA) from the pathogen of interest.
This backbone is typically a harmless virus but this is the potential that if the body has developed immunity to the backbone virus (i.e., the vector virus), it could prevent the successful delivery of the genetic material in subsequent vaccinations, reducing the effectiveness of the vaccine.
What is a requirement for mRNA vaccines?
Need to maintain cold chain
What is an advantage of DNA vaccines?
Good T-cell mediated immunity
What is a challenge associated with DNA vaccines?
Difficult to get strong antibody response
diva=
Differentiation of Infected from Vaccinated Animals
What is the DIVA strategy? How does it work and what are its limitations?
- Use diagnostic test for antibodies to protein/antigen that is only produced by natural infection and is not present in the vaccine
- This allows the distinction between an animal that has been vaccinated (and thus does not have antibodies to the target protein not present in the vaccine) and an animal that is infected (and has antibodies to both the vaccine and the infecting pathogen)
- To make the DIVA strategy possible, vaccines are often designed with genetic modifications such as gene deletions. This means that the vaccine does not contain one or more of the proteins found in the wild-type pathogen.
Example: bovine infectious bovine rhinotracheitis (IBR) caused by bovine herpes virus 1 (BoHV-1): one or more gene is deleted
what are teh control principles like in
all negative herd
low prevalence hered
medium or high prevalence herd
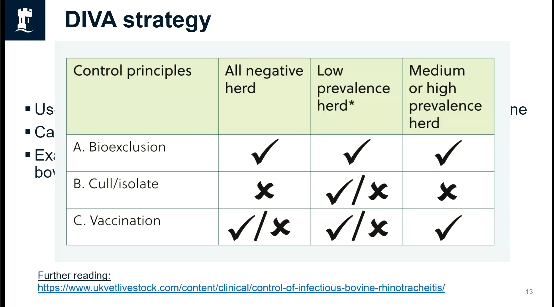
what can you not use diva with
- Cannot use DIVA strategy with vaccine containing whole pathogen as they contain all of the pathogen's proteins, including those involved in natural infection, so will produce all the same antibodies whether naturally infected or given the vaccine
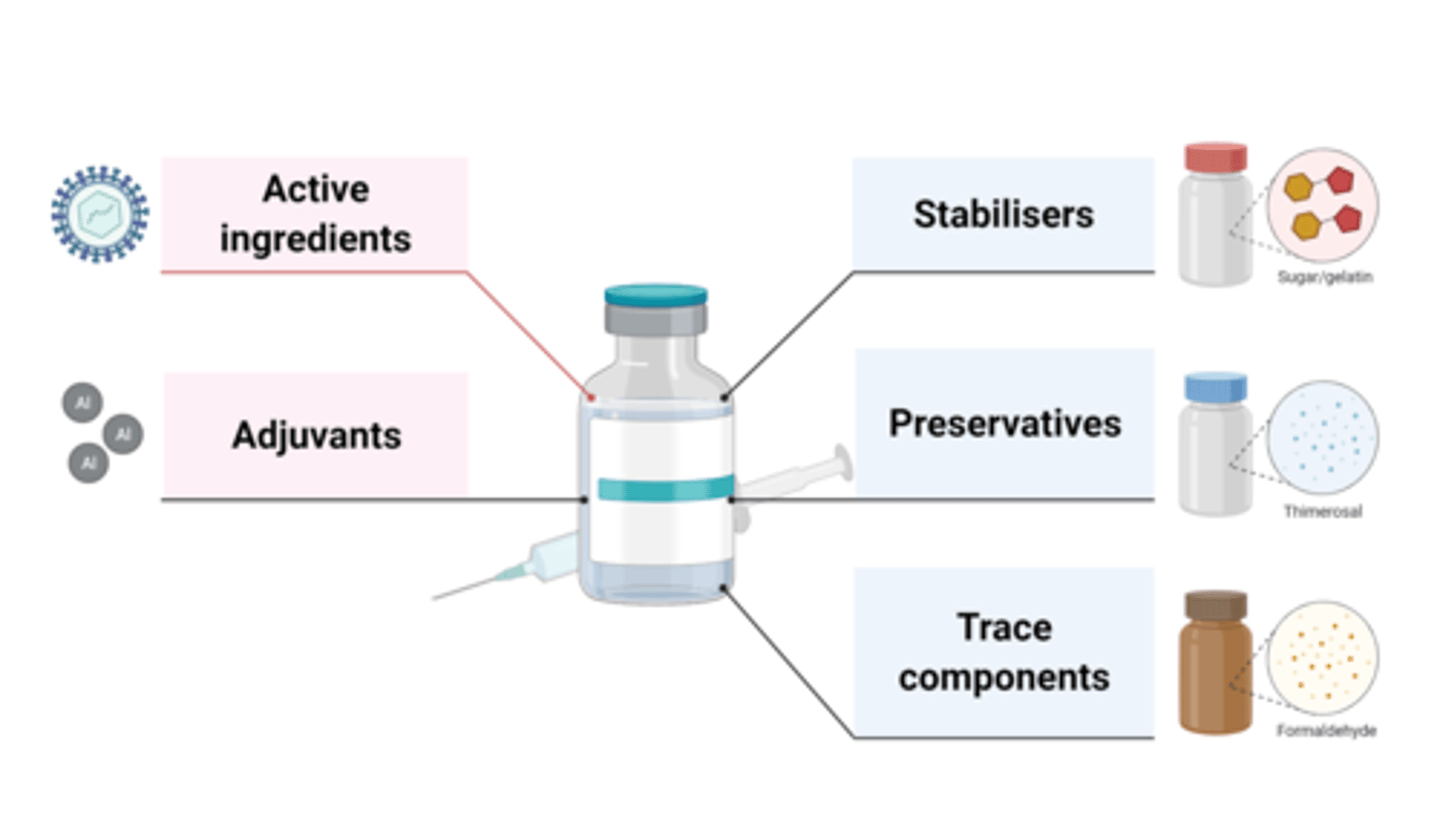
What else is present in a vaccine?
- active ingredients
- adjuvants
- stabilisers
- preservatives
- trace components
What is an adjuvant?
Adjuvants are pharmacological additives (non-antigen component) in a vaccine that enhance the body's immune response to a vaccine's antigen (enhances immunogenicity).
E.g. Chemicals, microbial components, or mammalian proteins.
Give 10 examples of adjuvants
1) Aluminium salts (alum)
2) Oil emulsions
3) Squalene - a triterpene derived from shark liver oil
4) Liposomes
5) Saponins - bitter-tasting, usually toxic plant-derived secondary metabolites
6) Non-ionic block copolymers - surfactants made from ethylene oxide (EO) and propylene oxide (PO)
7) Cytokines
8) Lipopolysaccharide (LPS)
What are the 7 possible mechanisms of action of adjuvants to enhance immunogenicity of a vaccine?
1.Depot formation
2.Secretion of cytokines & chemokines
3.Immune cell recruitment
4.Antigen uptake through antigen-presenting cells (APCs) (then processing and presentation on APC MHC)
5.Maturation and activation of immune cells
6.Activate APCs to traffic to draining lymph nodes (then processing and presentation on APC MHC)
7.Activate antibody-secreting B cells and CD8+ T cell responses
Explain how the depot formation action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Adjuvants can form a localized “depot” at the injection site, where the antigen and adjuvant are slowly released over time. This prolonged release increases the duration of exposure to the antigen, which enhances the immune response.
- Effect: By prolonging the antigen's presence at the site of injection, the immune system has more time to recognize it and initiate a stronger and more sustained immune response.
Explain how the secretion of cytokines and chemokines action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Some adjuvants activate immune cells at the injection site, prompting them to release cytokines (proteins that help communicate between immune cells) and chemokines (proteins that attract immune cells to the site).
- Effect: Cytokines and chemokines help create an inflammatory environment that enhances the immune response by recruiting additional immune cells and signalling pathways that promote immunity.
Explain how the immune cell recruitment action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Adjuvants stimulate the recruitment of various immune cells (such as dendritic cells, macrophages, and T cells) to the site of infection or vaccination.
- Effect: These recruited immune cells help in recognizing the antigen, presenting it to other immune cells, and initiating an immune response. This broadens and strengthens the body’s ability to fight off infection.
Explain how the antigen uptake through APCs action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Adjuvants enhance the ability of antigen-presenting cells (APCs) (such as dendritic cells, macrophages, and B cells) to capture and process the antigen. After antigen uptake, APCs process the antigen and present fragments of it on their Major Histocompatibility Complex (MHC) molecules. The antigen is displayed on either MHC class I (for CD8+ T cells) or MHC class II (for CD4+ T cells).
- Effect: APCs are responsible for presenting antigens to T cells, which is a critical step in activating the adaptive immune response. It allows helper T cells (for MHC II) and cytotoxic T cells (for MHC I) to recognize and respond to the antigen. Therefore efficient antigen uptake by APCs ensures that the immune system can recognize and respond to the vaccine.
Explain how the activation of APCs to traffic to draining lymph nodes action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Adjuvants promote the movement of activated APCs from the injection site to the draining lymph nodes (where immune cells are concentrated).
- Effect: In the lymph nodes, APCs present the processed antigen to naïve T cells, leading to the initiation of an adaptive immune response. This ensures that the immune system is fully engaged and can develop memory for future encounters with the pathogen.
Explain how the maturation of immune cells action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Adjuvants can mature APCs (like dendritic cells) by stimulating them to express costimulatory molecules and cytokines, which are crucial for T cell activation.
- Effect: By maturing APCs, adjuvants ensure that these cells can effectively activate T cells, including both helper T cells (which assist other immune cells) and cytotoxic T cells (which directly kill infected cells).
Explain how the activation of antibody secreting B cells and CD8+ T cell responses action of adjuvants enhances the immunogenicity of a vaccine
- Mechanism: Adjuvants enhance the activation of B cells, which are responsible for producing antibodies, as well as CD8+ T cells, which target and kill infected cells.
- Effect: By promoting both humoral immunity (antibody production by B cells) and cell-mediated immunity (CD8+ T cell responses), adjuvants ensure a comprehensive immune response that can target both the pathogen itself and infected cells.
how are vaccines safe
Pre-licensing testing (laboratory animals and/or target species)
Marketing authorisation licence
Post-licensing testing
What does pre-licensing testing for vaccines (laboratory testing with animals and/or target species) look at?
Safety (produces safety data for vaccine based on the side effects observed in testing), efficacy and effectiveness

What are the two marketing authorisation companies that give out licenses for vaccines?
European medicines agency (pre-brexit) and VMD
what is efficacy
how well it can work under these ideal conditions
One breed of animals
Vaccinate them perfectly
Infect them after a certain time
but real life isnt perfect
What is the post-licensing testing for vaccines?
Batch-release safety tests that are important for post-marketing surveillance of the vaccine's safety and effectiveness
What are the four causes of adverse effects associated with a vaccine?
Vaccine-induced effect (side effect occurs as a result of vaccine)
Vaccine-potentiated effect (side effect occurs as a result of the vaccine but made more severe/more likely to happen due to the presence of other factors like medical conditions or medications)
Programmatic error - 2 doses/wrong vaccine (human error)
Coincident effects (Something else going on and having an influence and happens to be at the same time during the vaccine→ Is not to do with the vaccine)
What are the two key types of adverse effects?
Local:
Heat, swelling, redness at vaccine site
FISS (rare)
Allergic reaction
Systemic:
Lethargy, loss of appetite / fever* (pregnancy loss)
severe allergic reaction/anaphylaxis (rare) Hypersensitivity
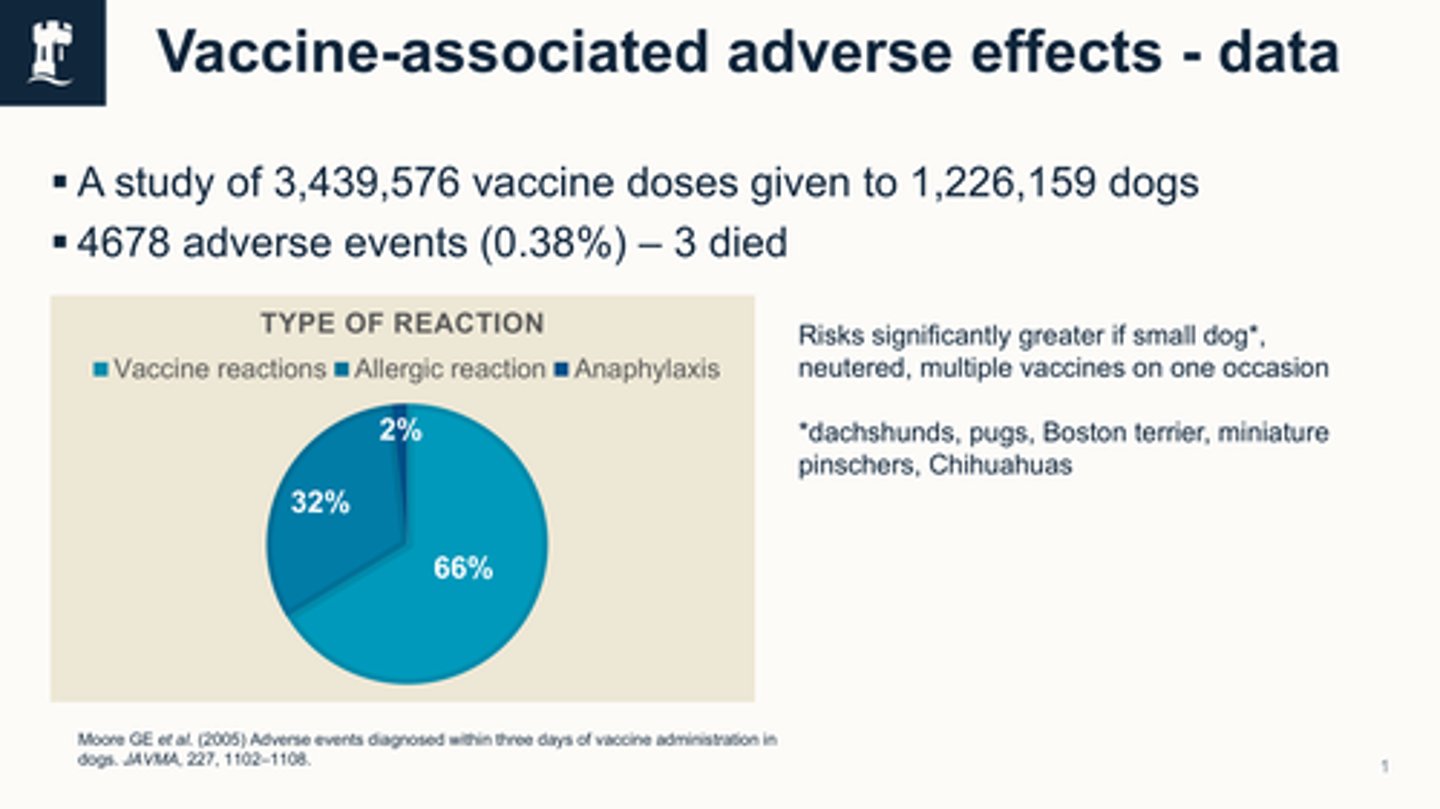
What is a feline injection site sarcoma (FISS)?
high grade sarcoma
Tumours linked to vaccine administration (role of adjuvants and genetics).
Time for development is often 3 months to 4 years.
Recommended treatment is radial excision with radiation therapy.
true or false - FISS are unlikely to reoccur
false - highly likely to reoccur
How do we avoid FISS?
Only administer necessary vaccines.
Avoid adjuvants vaccines - use attenuated or recombinant vaccines.
Follow site recommendations - avoid scruff, use distal limbs, tail and lateral abdomen
Use a different site each year (either record site on vaccination card or adopt a policy practice for each year)
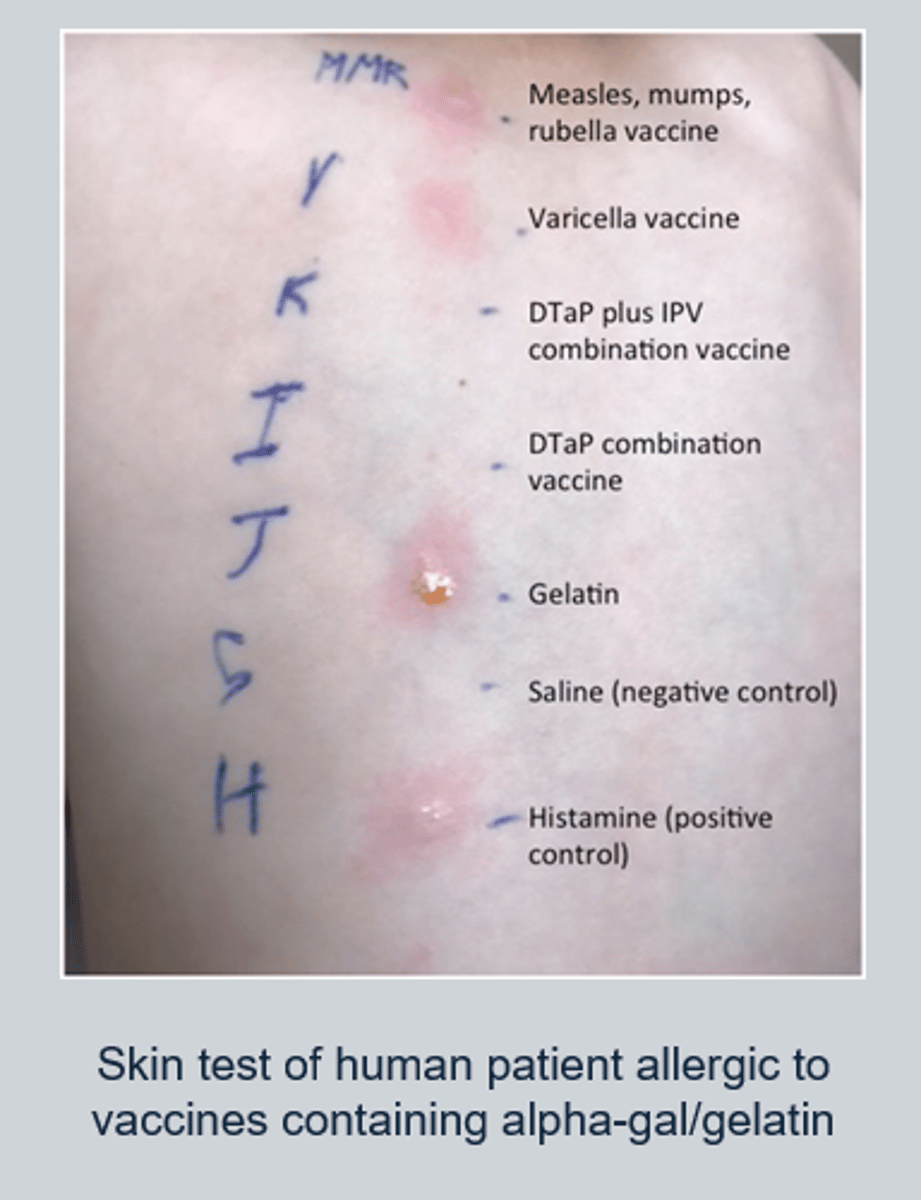
What are type 1 hypersensitivity reactions to vaccines
Ig-E mediated allergic response when antigen molecule encountered--> degranulation of mast cells and release of histamine = anaphylactic shock within a few minutes to an hour
When a person is first exposed to a component of the vaccine (e.g., a protein, preservative, or other substances), their immune system may mistakenly recognize it as harmful.
The immune system responds by producing IgE antibodies against that specific component (this is called sensitization).
IgE antibodies bind to mast cells and basophils, which are immune cells involved in allergic responses.
On subsequent exposure to the same vaccine (or components of it), the IgE antibodies on mast cells and basophils recognize the allergen (vaccine component) and become activated.
This activation triggers the release of inflammatory mediators, including histamine, prostaglandins, and leukotrienes, from the mast cells.
Type II hypersensitive reaction
A Type II hypersensitivity reaction (also called cytotoxic hypersensitivity- but not cytotoxic t cell involvement just refers to death of cells via IgM or IgG cytotoxicity)
occurs when the immune system mistakenly targets and destroys the body's own cells by wrongly recognising self-antigens as foreign antigens.
This happens when antibodies (usually IgG or IgM) bind to self-antigens on the surface of cells, leading to complement activation (which can use cell lysis by forming a MAC complex to lyse the cell and/or promoting phagocytosis by immune cells like macrophages and neutrophils (opsonisation).
Or they can promote phagocytosis by the antibodies directly opsonising the pathogens themselves without complement involvement.
Occur within hours to days.
What are alloantibodies?
Alloantibodies are antibodies produced by an individual in response to alloantigens, which are antigens from another individual of the same species (same species but genetically different individuals). The mother (dam) produces antibodies against foreign antigens (such as pathogens or other antigens) and then passes these antibodies to her offspring.
Since the antibodies are produced by the dam and are directed against the calf’s own antigens (such as the calf's blood group antigens), these antibodies are considered alloantibodies in the calf.
Autoantibodies
These are antibodies produced by an individual against their own self-antigens. Essentially, they are antibodies that mistakenly target and attack the individual's own tissues or cells.
Give two examples of type II hypersensitivities that can occur from vaccines
Bovine neonatal pancytopenia (BNP) - BVDV inactivated vaccine given to cows. Problem occurs if given to pregnant cows (severe reduction in newborn calf's blood cells due to destruction of bone marrow cells) when the dam produces maternal antibodies to the vaccine and the calf inherits these alloantibodies which mistakenly recognise calf's blood cells as foreign and attack them.
Renal autoantibodies - Contaminants lead to autoantibody production and cause interstitial nephritis. Making annual vaccinations a risk factor for CKD in cats (best to vaccinate more cats but less often)
Type III hypersensitivity
Immune complex-mediated: Antibodies (usually IgG or IgM) bind to soluble antigens (from pathogens, drugs, or other substances).
These complexes can get deposited in tissues (such as kidneys, joints, or blood vessels), where they activate the complement system.
This leads to inflammation and tissue damage, caused by neutrophil attraction, which release damaging enzymes.
Occur within 1-3 weeks
Give examples of type III hypersensitivities to vaccines
Rabies vaccine can cause cutaneous vasculitis:
- Typically, a mild reaction with some hair loss
- Breed predisposition (e.g. poodle)
Type IV sensitvity
Type IV hypersensitivity is a cell-mediated immune reaction, also called delayed-type hypersensitivity (DTH),
where T cells (not antibodies) mediate the response by encountering the foreign antigen and recruiting macrophages and neutrophils as part of a cell-mediated inflammatory response that causes harm to the body's own healthy cells as a side effect, which which takes time to occur (delayed response):
- T helper cells (CD4+ Th1 mostly) recognize antigens presented by antigen-presenting cells (APCs) MHC II.
- Th1 release cytokines, which recruit more innate immune cells like macrophages (primarily) and CD8+ cytotoxic T cells.
-persistent stimulation of immune response
Occurs within days to weeks (but typically within 48-72 hours).
How do you get type IV hypersensitivity reactions to vaccines?
Type IV hypersensitivity (also known as delayed-type hypersensitivity, DTH) to vaccines occurs when the immune system’s T cells (particularly CD4+ T helper cells and CD8+ cytotoxic T cells) are activated by components of the vaccine, leading to an inflammatory response. The inflammation and tissue damage caused by the recruitment of immune cells like macrophages and neutrophils takes time to develop, typically peaking between 48-72 hours after exposure, which is why it is called delayed-type hypersensitivity.
Granuloma - fibrous stroma and an infiltration of macrophages, giant cells, and lymphocytes around the persistent antigen
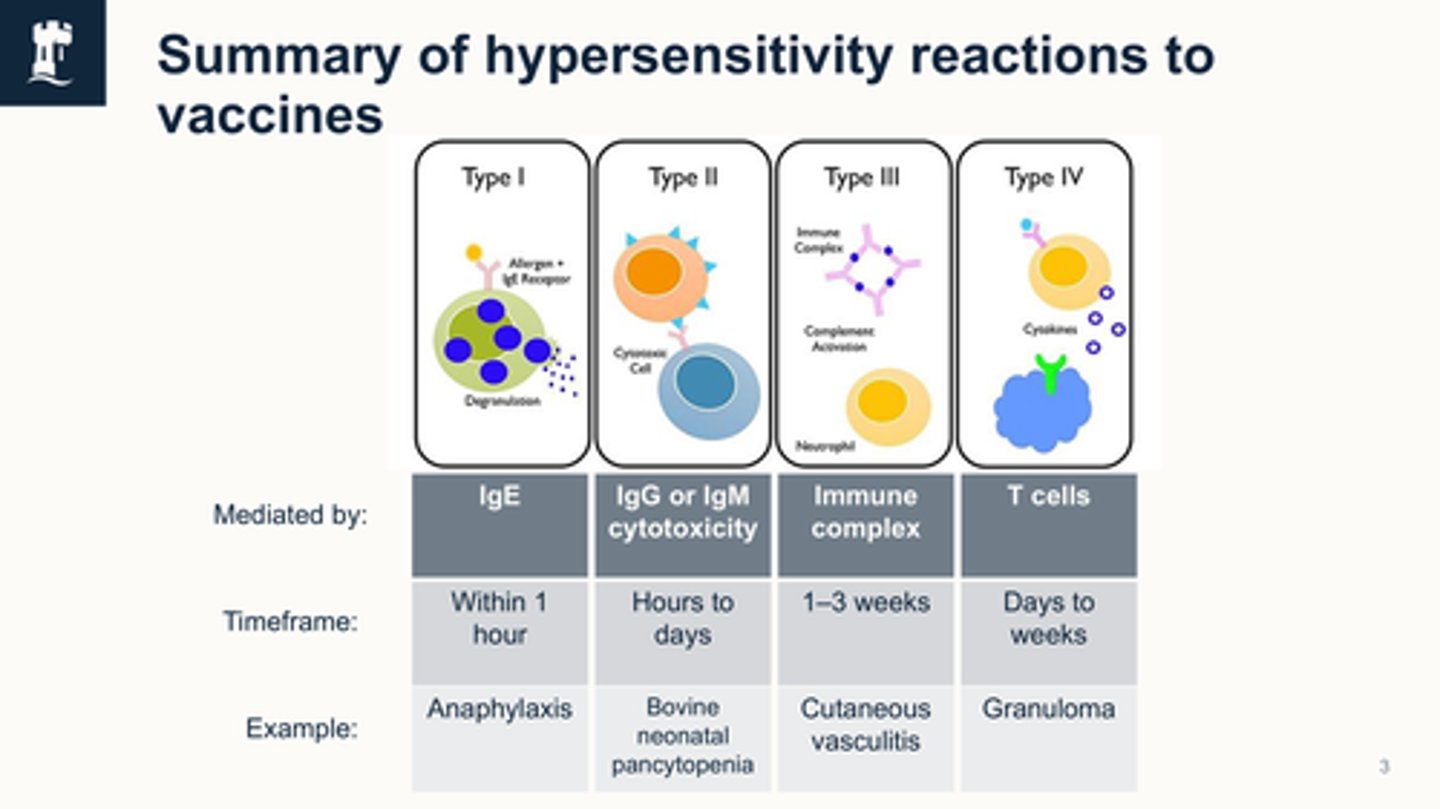
summarise hypersensitivy reactions to vaccines
Who do you report adverse effects to?
VMD in 15 calendar days or immediately if you think that a whole batch is effected
Veterinary Medicines Directorate Pharmacovigilance Team
Date reaction / lack of efficacy observed