Week 3: Analytical techniques and instrumentation/toxicology
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Clinical chemistry - 4 basic disciplines
spectrometry
luminescence
electroanalytic methods
chromatography
Spectrometry measurement
measure either absorption or emission of radiant energy to determines the concentration of atoms or molecules
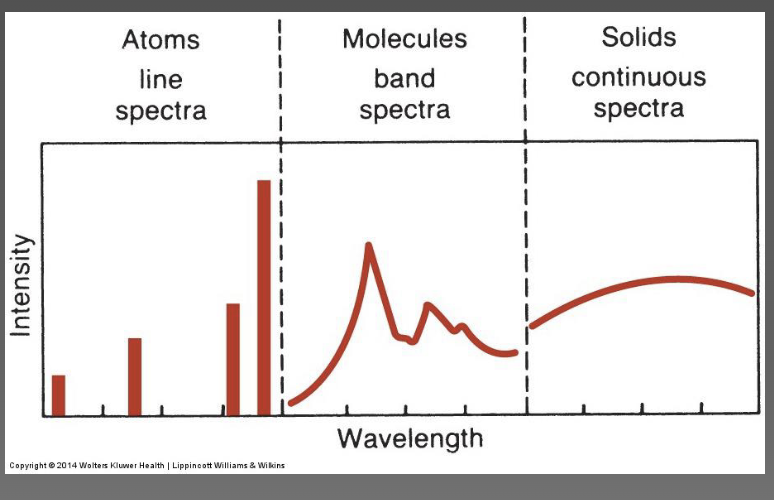
characteristics of absorption/emission spectra-line (atoms), band (molecules), and continuous (solids)
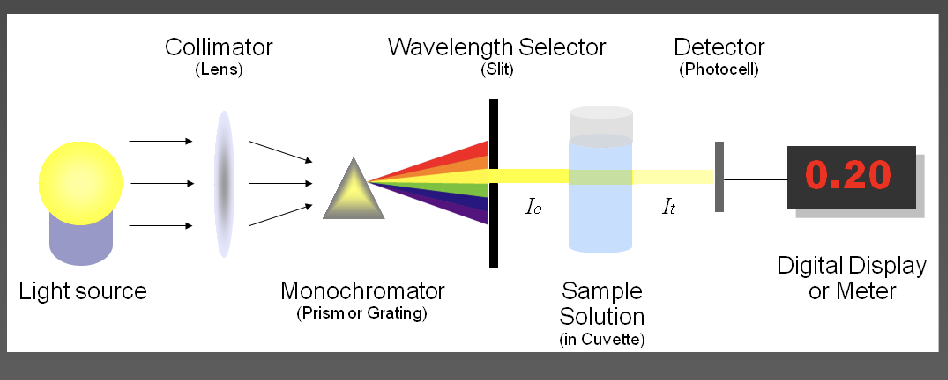
Spectrophotometer
used to measure light transmitted by a solution to determine concentration of the light-absorbing substance in solution
Luminescence
process of emitting light from a substance
bioluminescence is the emission of light by living organisms
chemiluminescence is a result of a chemical rxn
ex. luminol test gives blue glow in detecting blood
Fluorescence
fluorescence in several wavelengths can be detected by an array detector to detect compounds using HPLC
TLC (thin layer chromatography) plates can be visualized under UV if the compounds or a coloring reagent is fluorescent
Electroanalytic methods
pH electrode is universally used in clinical labs
ion-selective electrodes measure the hydrogen-ion concentration to indicate its acidity or alkalinity
acidosis and alkalosis describe the abnormal conditions that result from an imbalance in the pH of blood
excess acid or alkali in blood is typically caused by an underlying condition or disease
Electroanalytic methods - pH
any disease or condition that affects the lungs, kidneys, metabolism or breathing has the potential to cause acidosis or alkalosis
acidosis: blood pH <7.35 (diabetes)
alkalosis: blood pH >7.45 (severe vomitting)
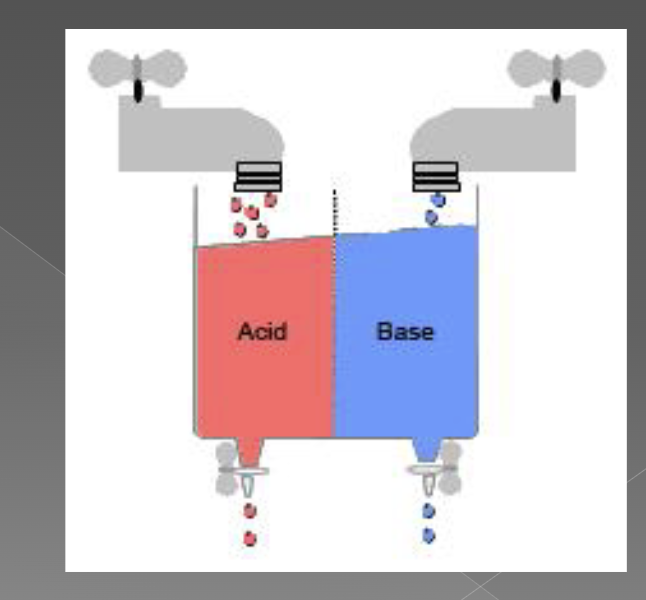
body balances between incoming acids and bases (faucet on) and elimination of acids and bases (drain on)
Electroanalytic methods - Urinalysis pH
benefits:
indicates metabolic acidosis or alkalosis
precipitation of crystals
treatment of UTIs
ex. formation of various types of kidney stones strongly influenced by urinary pH
by modifying urine pH through diet or meds, formation of crystals can be reduced or eliminated
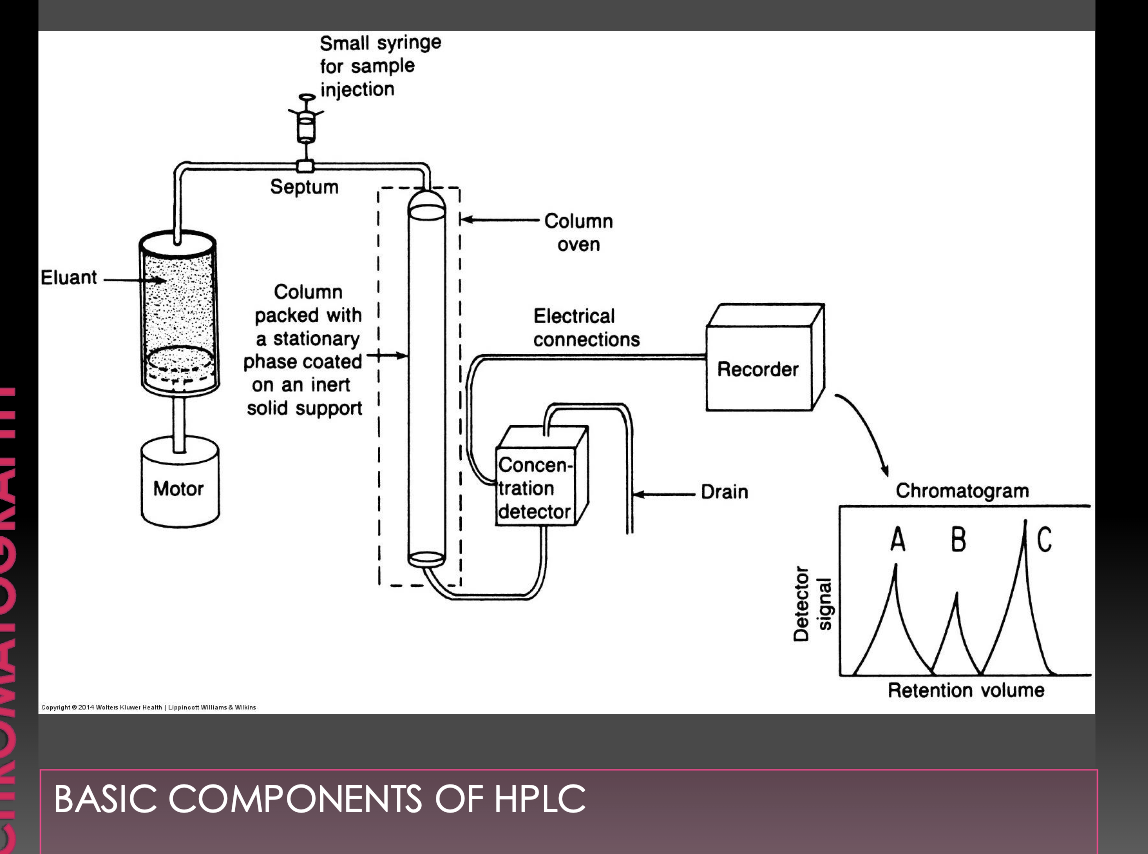
Chromatography
involves a group of techniques used to separate complex mixtures on the basis of different physical interactions between the individual compounds and the stationary phase of system
Mobile phase: gas or liquid
Stationary phase: solid or liquid; column holds stationary phase
Complex mixture: sample.
Separated components: eluate
Thin layer chromatography (TLC)
variant of column chromatography
thin layer of sorbent like silica gel is coated on glass
sample applied on the spot and the mobile phase (solvent) separates the sample into molecules
retention factor (Rf) is compared to standard known Rf

High performance liquid chromatography
separation and analysis of mixed compounds
mobile phase is liquid
thermally unstable compounds can be analyzed
qualitative and quantitative analysis can be performed
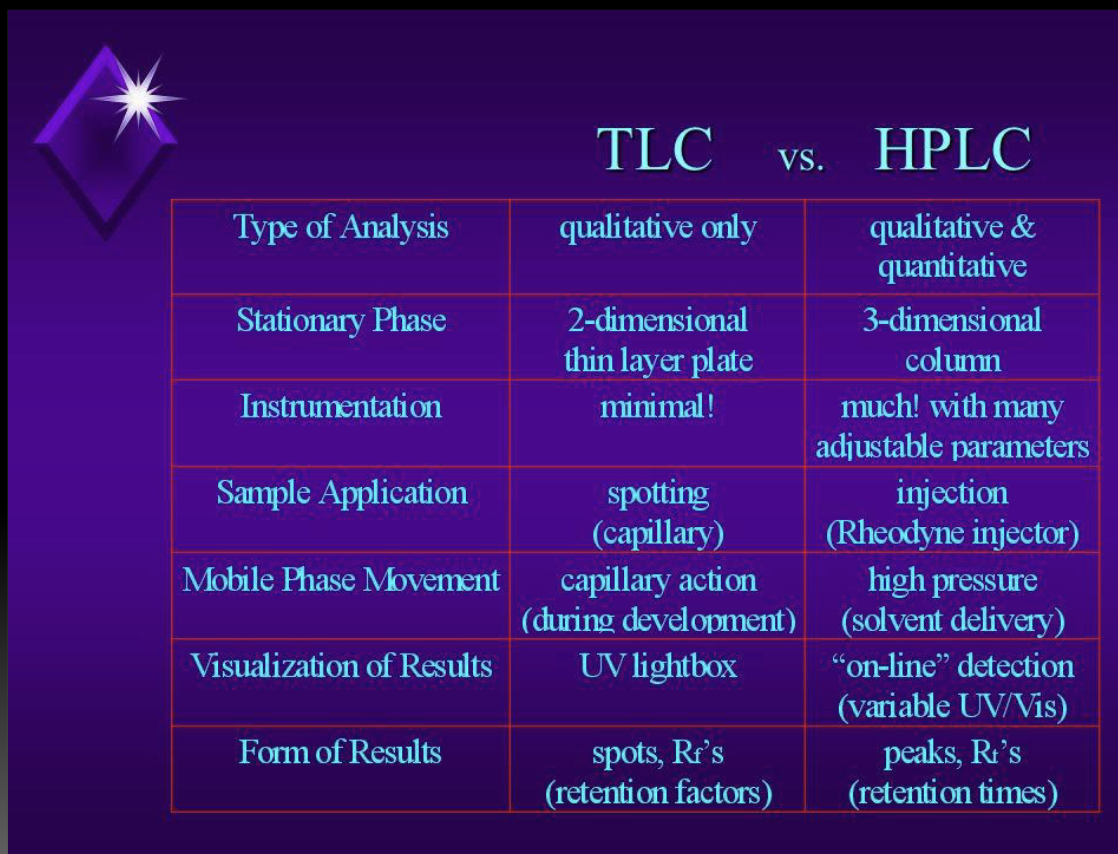
TLC vs. HPLC
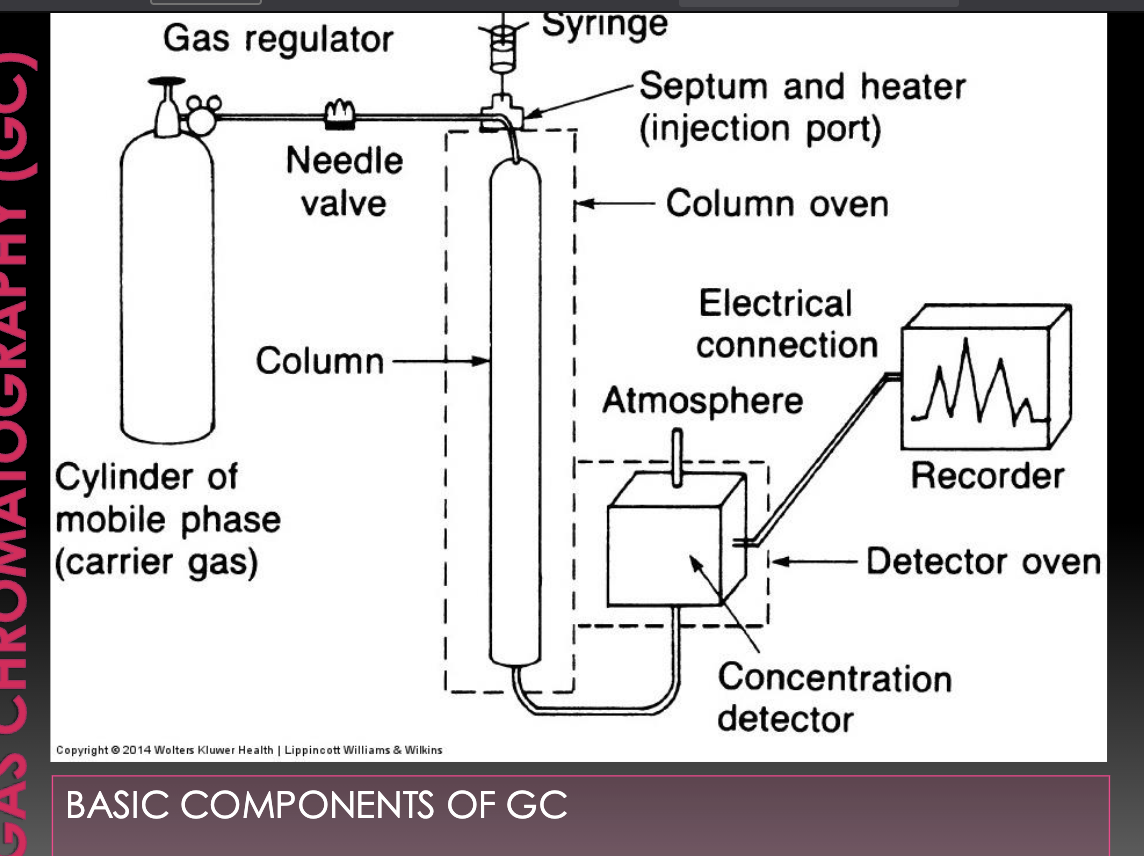
Gas chromatography mass spectrometry (GCMS)
gas mobile phase
complex samples separated in column
each component enters mass analyzer where it’s fragmented into unique and repeatable pattern
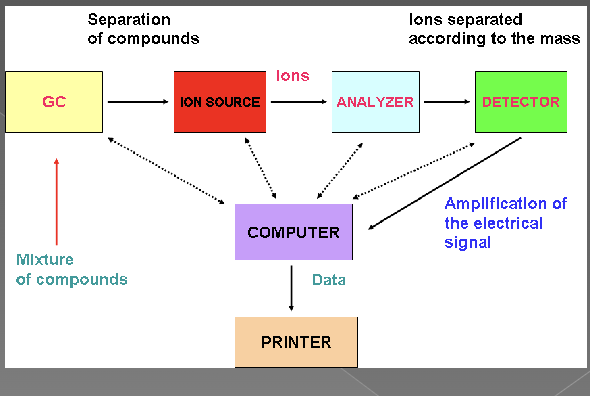
Mass spectrometry
analytical technique that sorts ions according to their mass to charge ratio
ionization source - small sample is ionized, usually to cations by loss of an electron
mass analyzer - ions are sorted and separated according to their mass and charge
detector - separated ions measured and results displayed on a chart as a spectrum (Fragmentation pattern)
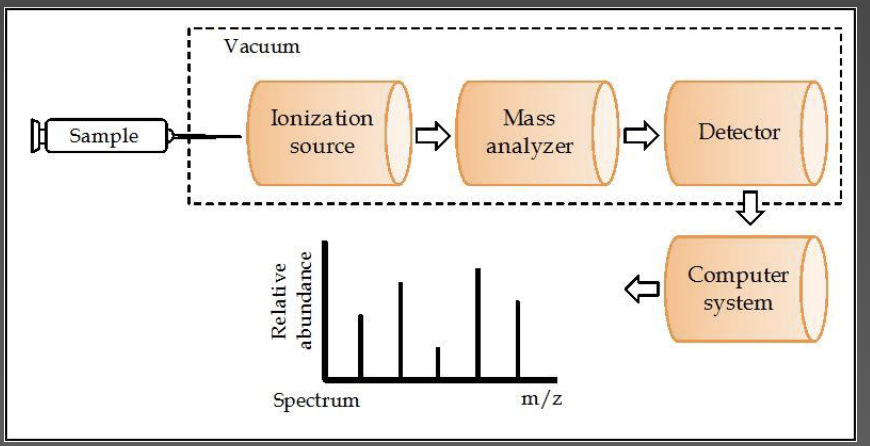
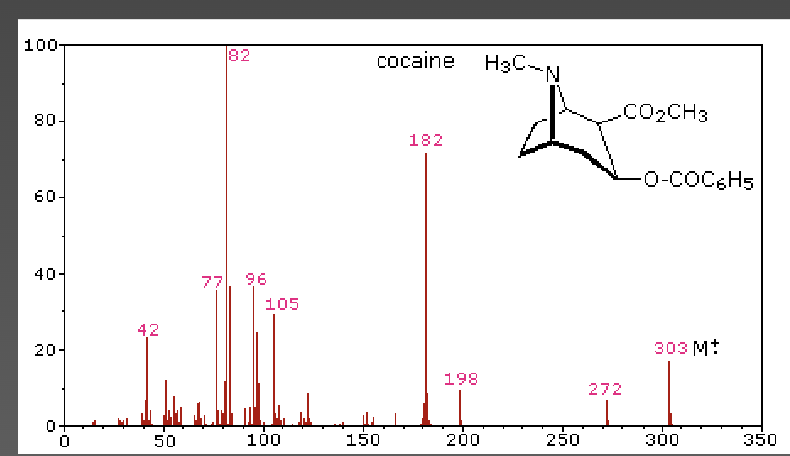
Mass spectrometry - cocaine
fragment pattern is considered to be the fingerprint of the compound
the more abundant ions (82, 182, 303) can be rationalized by certain cleavages and eliminations occurring to the structure of cocaine
Presumptive (screening)
test indicates the sample is probably the substance
Kastle-Meyer test will show that a sample is not blood or that the sample is probably blood
chemical indicator phenolphthalein is used to detect the possible presence of hgb (+ = pink)
Marquis reagent used as a simple spot-test to presumptively ID alkaloids (drugs/poisons) as well as other compounds
confirmatory testing
verify the substance’s identity, or to measure the % purity or other quantitative analysis
high specificity
high sensitivity
quantitative and qualitative
legally defensible results
avoids false pos/negs
Marijuana analysis
hemp plant - Cannabis sativa
plant contains mind-altering chemical delta-9-tetrahydrocannabinol (THC)
analysis of cannabis via color test, urinalysis, hair analysis, saliva test, comprises of presumptive testing and necessary confirmatory tests are required for ID
THC’s major metabolite is 11-nor-9-carboxy-delta-9-tetrahydrocannabinol (THC-COOH). not psychoactive
Marijuana analysis - screening
urinalysis: provide info to past exposure, but tests don’t necessarily differentiate between exposure to second hand smoke and actually smoking marijuana
false pos are common
Duquenois-Levine color test
Marijuana becomes purple with addition of Duquenois reagent and hydrochloric acid
upon addition of organic solvent, purple color transfers to organic layer, indicating that cannabinoids may be present
color gives 2 layers: purple (top) and purple/violet (bottom)
Marijuana analysis - confirm
TLC
Marijuana is a mixture of compounds
components spread out in TLC plate as solvent travels
chromatography occurs and a visualizing agent along with THC standard is used for confirmation
Rf calculated
GC
peaks are ID’d by their retention times (time of elution)
Chemical components of marijuana
Metabolite of Marijuana
metabolism of THC results in glucuronide
common soluble conjugates formed as a step towards metabolism and excretion

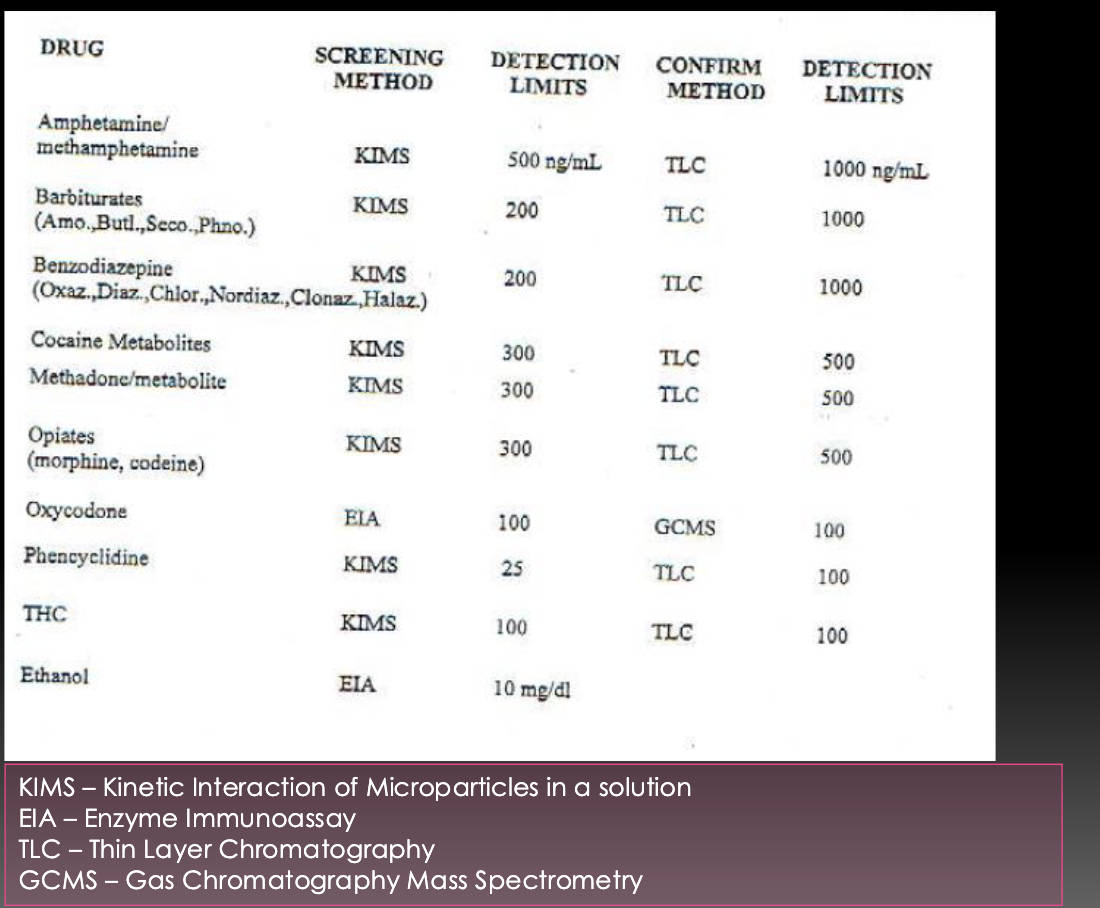
Drugs of abuse in urine
Clinical toxicology labs

Forensic toxicology labs
Xenobiotics
xenos - greek for ‘stranger’
xenobiotic compound foreign to the body
artificial components such as chemicals, pesticides and drugs
they are not normally found or produced in body
exogenous agents that may have adverse effects on a living organism
Toxicology
study of the adverse effects of xenobiotics in humans
scope is very broad
3 major disciplines
Mechanistic
descriptive
regulatory
Mechanistic toxicology
elucidates the cellular, molecular, and biochemical effects of xenobiotics
Descriptive toxicology
uses results from animal experiments to predict what level of exposure will cause harm in humans
regulatory toxicology
interpretation of the combined data from mechanistic and descriptive studies is used to establish standards that define the level of exposure that will not pose a risk to public health
ex. FDA, US environmental Agency, OSHA
Risk Assessment
RISK - the harm a hazard can do
HAZARD - object that can potentially hurt someone
EXPOSURE - takes into account the amount, frequency, route, and duration
RISK = HAZARD x EXPOSURE
Forensic toxicology
primarily concerned with medicolegal consequences of exposure to chemicals or drugs
focuses on establishing and validating the analytic performance of methods used to generate evidence in legal situations
Clinical toxicology
study of interrelationships between xenobiotics and disease states
emphasizes diagnostic testing and therapeutic interventinos
Environmental toxicology
evaluation of environmental chemical pollutants and their impact on human health
sources include organic and inorganic pollutants, pesticides and biological agents, all of which can have harmful effects on living organisms
Clinical poisoning facts
intentional suicide - 50% of poisoning cases
suicide - highest mortality rate
therapeutic or illicit drug accidental overdose common in adolescents and adults
accidental exposure - 30% of cases (most frequent in children)
remaining cases - homicide and occupational exposure
Routes of exposure
ingestion, inhalation, transdermal absorption most common
most often observed - ingestion
systemic effect occurs when the toxin is absorbed into circulation
Ingestion
toxins enter through pharmacokinetic processes
in most cases, passive diffusion occurs where toxic substances that are hydrophobic cross cell barriers
toxins not absorbed from GI tract don’t produce systemic effects but local effects
ex. diarrhea, bleeding and malabsorption of nutrients
local effects can cause systemic effect secondary to toxin exposure
Factors affecting absorption
pH
rate of dissolution
gastric motility (movement)
resistance to degradation in GI tract
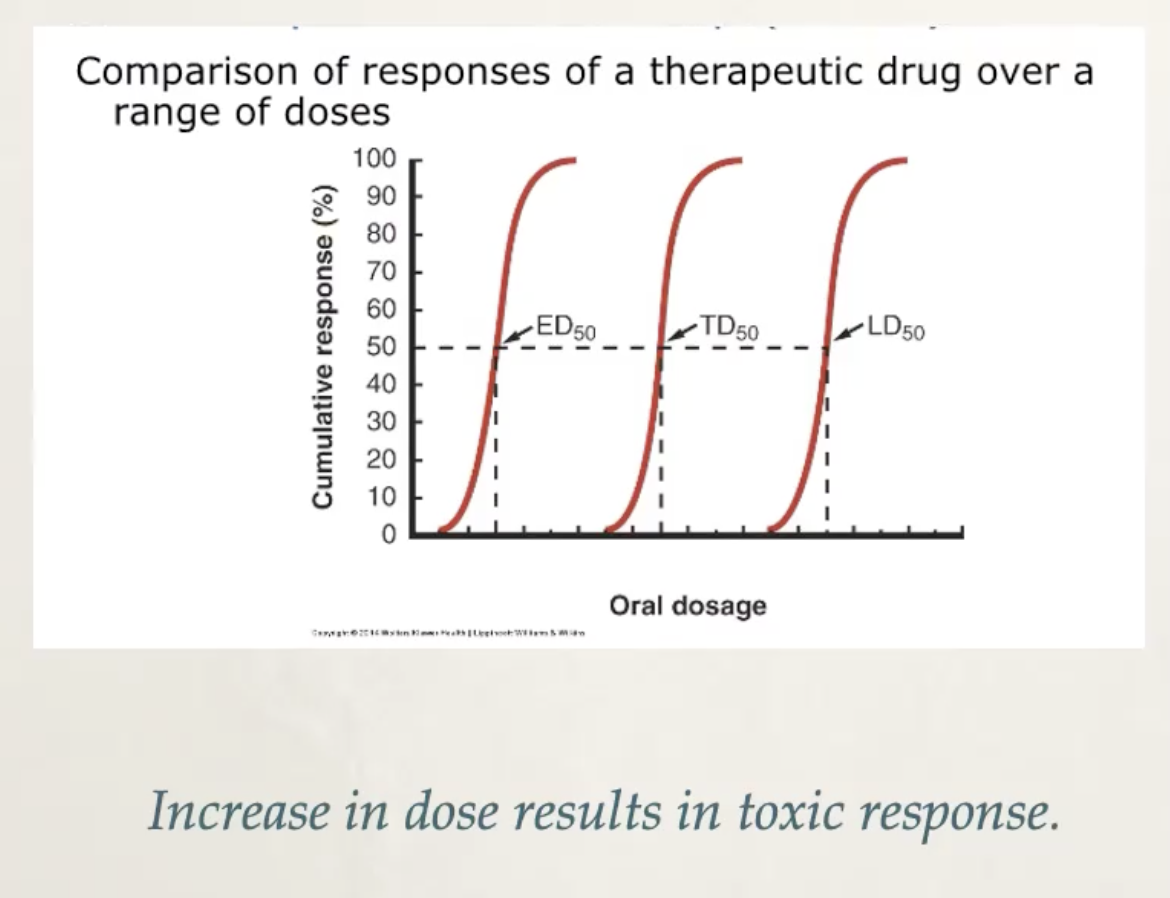
Dose-response relationship
dose-response relationship correlates the dose of a xenobiotic that will result in a harmful response
ED50 is the effective dose that produces a therapeutic effect in 50% of population
TD50 is the toxic in 50% of population. Associated with an early pathologic effect at lower than lethal doses
LD50 is the lethal dose in 50% of population
Individual dose-response
changing health effects based on change in xenobiotic exposure levels
Quantal dose-response
change in health effects of a defined population based on changes in exposure to xenobiotics
Acute toxicity
single, short term exposure to a substance, the dose of which is sufficient to cause immediate toxic effects
Chronic toxicity
repeated frequent exposure for extended periods for greater than 3 months and possibly years, at doses that are insufficient to cause an immediate acute response