Unit 3:Aircraft Structural Materials
1/222
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
223 Terms
Section A: Metals
Section A: Metals
What are commonly used aerospace materials? (List seven of them for structural)
Wood
Steel
Aluminum Alloys
Titanium Alloys
Magnesium Alloys
Nickel Alloys
Fiber-Reinforced Composites
What are three factors to think about when picking materials for aircraft parts? (List all three and their details)
Function
– What is the component use for?Material Properties
– Strength-to-weight ratio (strong but not heavy)
– Stiffness (how much it bends or holds shape)
– Toughness (can it take a hit without breaking?)
– Resistance to corrosion (won’t rust or weaken easily)
– Fatigue and effects of environmental heating (can it handle repeated stress or heat?)Production
– Machinability (easy to cut, shape, or work with)
– Availability and consistency of material (easy to get and reliable quality)
Three common aluminum metals.
2024-T3
6061-T6
7076-T6
Cyclic Stresses
Average commercial aircraft
30 year life cycle
60,000 Hours
2,500 Days – 357 weeks – 6.85 Years
20,000 Flights
667 flights per year
100,000 miles of taxiing
4 times around the Earth’s circumference
Total average maintenance and service cost are double the original purchase price
Flight Stress – Pressure differential fuselage to outside
0 kPa to 60 kPa (8.6 psi)
The cabin is pressurized to keep passengers comfortable, but outside air at cruising altitude has very low pressure.
This means the pressure pushes outward from inside the aircraft toward the outside.
The fuselage is stressed every flight because it has to constantly handle this pressure difference.
Flight Stress – Temperature differential ground to cruise
Ground temp to -56°C (-69°F)
Aircraft experience huge temperature swings from the hot ground to freezing altitudes.
The material must handle extreme cold and shrinking without becoming brittle or cracking.
Flight Stress – Impact load of landing
Landing gear now supports aircraft
Wings flex from upward lift force to downward force of their own weight
Tires accelerate from 0 kph to 400 kmph (this creates a puff of smoke)
On landing, the landing gear takes the full hit, supporting the whole plane.
Wings stop lifting and suddenly carry their own weight, flexing downward.
Tires go from standing still to spinning fast in an instant, causing stress and heat (and that puff of smoke).
What are the three main flight stresses? (List all three)
Pressure differential (fuselage to outside)
Temperature differential (ground to cruise)
Impact load of landing
What are the five basic types of structural stress in aircraft? (List all five)
Tension (pulling apart)
Compression (pushing together)
Shear (sliding forces)
Bending (combination of tension & compression)
Torsion (twisting)
What happens when you reduce material density in aircraft construction? (Keep in mind)
Reduces airframe weight and improves performance
What benefits come from a lighter aircraft structure? (Keep in mind)
Better fuel efficiency, climb rate, G-force loading
What is a downside of making aluminum stronger? (Keep in mind)
Stronger aluminum = More brittle
Property of Metal – Strength
Determined by the percentage of a parent metal and other elements used to make an alloy
(How tough the metal is overall)
Property of Metal – Tensile Strength
Ability to resist tension or pulling forces (often represented as KSI)
(How much pulling it can take before snapping)
Tension
Property of Metal – Yield Strength
Ability to resist deformation
(How much force it can take before it bends permanently)
Property of Metal – Shear Strength
Ability to resist opposing forces
(Resists sliding forces that try to cut or split the metal)
Property of Metal – Bearing Strength
Ability of a joint to withstand crushing/compression i.e. buckling/bending
(How well it handles pressure without getting crushed)
Property of Metal – Hardness
Ability to resist penetration, cutting, or abrasion
(How scratch-resistant and tough the surface is)
Annealing
Property of Metal – Malleability
Ability to bend, form, or shape
(Can be hammered or rolled into shape without cracking—Opposite of Hardness)
Property of Metal – Ductility
Ability to be drawn into wire stock or stretched
(Can be stretched without breaking)
Property of Metal – Brittleness
Ability to resist breaking or shattering when exposed to stresses
(Breaks easily without bending—Opposite of Toughness)
Property of Metal – Conductivity
Ability for a metal to conduct heat or electricity
(How well heat or electricity flows through it)
Thermal Conductivity
Electrical Conductivity
Property of Metal – Thermal Expansion
Ability for material to expand and contract when exposed to varying temperatures
(Material changes size when temperatures change)
Coefficient of Expansion
Property of Metal – Elasticity
Ability to return to the original shape
(Bounces back after being stretched or bent)
Elastic Limits
Property of Metal – Toughness
Ability to resist tearing or breaking when bent or stretched
(Handles impacts and force without cracking—Opposite of Brittleness)
Property of Metal – Fusibility
Ability to be joined/fused by heating or melting
(Can be welded or melted together easily)
Property of Metal – Density
Material's mass per unit volume
(How heavy it is for its size)
Tension
A pulling force that tries to stretch a material.
(Example: Pulling on a rope puts it under tension.)
Annealing
A heat treatment process that softens metal to decrease internal stress, making it easier to work with and less brittle.
(Used after metal has been hardened from working or welding.)
Thermally Conductive
A material’s ability to transfer heat.
(Example: Aluminum is thermally conductive—it heats up quickly.)
Electrical Conductivity
A material’s ability to allow electricity to flow through it.
(Example: Copper wires are used because copper has high electrical conductivity.)
Coefficient of Expansion
A number that tells how much a material expands or shrinks when the temperature changes.
(The higher the number, the more it expands with heat.)
Elastic Limit
The maximum amount a material can be stretched or bent before it won’t return to its original shape.
(Example: A rubber band stretches and returns to shape—but if you stretch it too far, it stays deformed. That’s past the elastic limit.)
Wrought VS Cast
Wrought:
Metal that has been worked into shape by rolling, hammering, or other mechanical means (not cast).
(Example: Wrought aluminum sheets used in aircraft skins.)
Cast:
Metal that has been melted and poured into a mold to solidify into a shape.
(Example: Engine blocks are often cast because of their complex shapes.)
Metalworking Processes – Hot Working
Forming metal at an elevated temperature
(Example: Forging a steel crankshaft while it’s glowing red hot)
Rolling
Forging
Metalworking Processes – Pressing
Using a press to make parts
(Example: Stamping car body panels out of sheet metal)
Metalworking Processes – Drop Forging
Hammering process where a hot piece is placed between two dies and hammered
(Example: Making wrenches or other strong hand tools)
Metalworking Processes – Hammering
Used on small parts and parts are hammered into shape (think blacksmith)
(Example: Shaping horseshoes or custom metal brackets by hand)
Smith Forging
Metalworking Processes – Cold Working / Strain Hardening
Bending metal back and forth below its critical temperature range... will decrease ductility
(Example: Bending aluminum tubing for an aircraft fuel line, which hardens it)
Cold-Rolling
Cold-Drawing
Metalworking Processes – Cold Drawing
Squeezes tubing to smaller sizes through use of successive smaller dies
(Example: Making hypodermic needles or precision tubes)
Cold drawing is a cold working process where metal is pulled through a die to reduce its diameter, improve surface finish, and increase strength and hardness. It’s commonly used on:
Steel bar stock
Wire
Rod
Tubing
Metalworking Processes – Extrusion
Similar to cold drawing, but using dies to make shape (i.e. angle bar, z-channel, t-sections). Aluminum is good for this.
(Example: Process of forcing metal through a die, hot or cold.)
Wrought
Metal that has been worked into shape by rolling, hammering, or other mechanical means (not cast).
(Example: Wrought aluminum sheets used in aircraft skins.)
Casting (What is it, and a potential problem?)
Metal that has been melted and poured into a mold to solidify into a desired shape.
Usually lower in strength and more brittle.
(Example: Engine blocks are often cast because of their complex shapes.)
Rolling
A process where metal is passed through rollers to reduce its thickness and make it uniform
(Example: Making flat aluminum sheets for fuselages.)
Extruding
Process of forcing metal through a die, forcing the metal to take a particular shape
Most extruding is done while metal is hot, but softer metals like lead and tin can be done cold
Forging
Shaping metal by applying compressive force, usually by hammering or pressing.
(Example: Forged aircraft landing gear components.)
Smith Forging
A manual forging process where metal is heated and shaped by hammering, typically done by a blacksmith.
(Example: Custom metal brackets or artistic metalwork.)
Cold Rolling
Rolling metal at or near room temperature to improve surface finish and strength.
(Example: Used for making precision structural components like aircraft skin panels.)
Cold Working (What is it, and a potential problem?)
Done at temps below the critical range, and results in straining and hardening of the metal
Because of this, the metal may have to be reheated to allow it to be worked with
What are three facts about aluminum alloy? (List three)
Cutting-edge (1920s–60s)
Most abundant metal in the earth’s crust
Pure aluminum is relatively soft
Alloyed
When a metal (like aluminum) is mixed with other compatible metals to improve its properties.
(Example: Aluminum alloy is stronger than pure aluminum because it’s alloyed with elements like copper or zinc.)
Note: Strength increases considerably when it is alloyed.
Aerospace Material – Aluminum Alloy (List five benefits)
Currently most widely used material
Readily formed (Easy to shape or manufacture into parts)
Moderate cost
Excellent resistance to chemical corrosion
Excellent strength-to-weight ratio
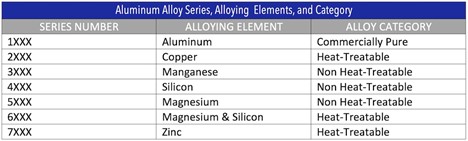
Aluminum Alloying Agents and their 1st Numbers (No need to memorize chart)
Most common alloy is 2024 (24ST)
– 93.5% aluminum, 4.4% copper, 1.5% manganese, and 0.6% magnesium
Need to know… The First Numbers of Each…
Examples from earlier lessons (A-F coding style):
A (1100) – Pure aluminum
AD (2117) – Durable aluminum (copper alloyed)
B (5056) – Magnesium alloy
D (2017) – Copper alloy
DD (2024) – Copper alloy (most common)
E (7050) – Zinc alloy
What is the most common aluminum and it’s make up?
DD 2024 Aluminum with Copper
93.5% aluminum, 4.4% copper, 1.5% manganese, and 0.6% magnesium
Nonferrous Metals
Pure aluminum lacks sufficient strength to be used for aircraft construction, however, when it is alloyed (mixed with other compatible metals) its strength is increased considerably.
1XXX – Aluminum
2XXX – Copper
3XXX – Manganese
4XXX – Silicon
5XXX – Magnesium
6XXX – Magnesium and Silicon
7XXX – Zinc
8XXX – Other elements
Clad Aluminum Alloys (Alclad Aluminum)
Alclad is a corrosion-resistant aluminum sheet
Formed by thin, pure aluminum surface layers
Metallurgically bonded (rolled onto) a high-strength aluminum alloy core material
The pure aluminum coating is approximately 5% of the total thickness on each side, which is usually only a few thousandths thick
It has a melting point of about 500°C (932°F
What is Heat Treating
Process of heating and cooling metal to achieve desired result (either harder, stronger, more resistant to impact, softer, more ductile).
What is aging in heat treating?
Aging is a heat treatment process where a metal is held at an elevated temperature for a period of time to increase strength and hardness.
Some alloys, such as 2017 and 2024, develop their full properties after solution heat treatment followed by about four days of cooling (aging) at room temperature. (Due to the copper)
What are the three types of heat treating aluminum? (List all three)
Solution Heat Treatment
Precipitation Heat Treatment
Annealing
Heat Treating Aluminum – Solution Heat Treatment
PowerPoint Definition:
Heated and mixed with alloys then quenched
Additional Key Info:
Quenching (rapid cooling) is critical—it locks the alloying elements in place.
This prepares the aluminum for aging to reach full strength.
Common with heat-treatable aluminum alloys like 2024 and 7075.
Heat Treating Aluminum – Precipitation Heat Treatment
PowerPoint Definition:
Heated for a certain time and allowed to cool, then reheated again and cooled… resulting in a harder and more corrosion-resistant metal.
Additional Key Info:
This process is also called artificial aging.
Some aluminum alloys like 2024 and 2017 develop full strength through natural aging—just sitting at room temperature.
Especially effective for copper-based alloys.
Increases strength, hardness, and corrosion resistance.
Natural Aging: When a heat-treated aluminum alloy is left at room temperature for a period of time (usually several days) to gain full strength without needing additional heat.
(Example: Alloys like 2024 and 2017 naturally age due to their copper content.)
Artificial Age-hardening, Precipitation-Hardening or Precipitation Heat Treatment: A process where a heat-treated aluminum alloy is reheated to a lower temperature and held for several hours, then cooled, to increase strength and hardness.
(This is a controlled aging process that speeds up what natural aging does, often used for alloys like 7075.)
Heat Treating Aluminum – Annealing
PowerPoint Definition:
Process that softens metal by heating to high temp, then cooled to 500°F at 50 degrees per hour rate
Additional Key Info:
Used to soften aluminum, relieve stress, and restore ductility.
Common for non-heat-treatable alloys like 3003.
Makes the metal easier to shape or repair.
(Not on PowerPoint) Non-Heat Treatable Alloys
Aluminum alloys that can’t be heat treated because they don’t contain the right alloying elements—like magnesium, which can’t handle heat and could explode if treated that way.
3003 – Almost pure aluminum with a little magnesium. Lightweight and corrosion resistant, but not strong.
5052 – Most important non-heat treatable alloy. About 2.5% magnesium and a small amount of chromium.
Used for fuel tanks, oil tanks, and fluid lines.
Gets stronger through cold working, not heat.
How Are Metals Identified After Heat Treatment?
After heat treatment, aluminum alloys are marked with the letter “T” followed by a number.
*T = Solution heat treated other than F, O, or H (memorize this one)
T2 = Annealed
T3 = Solution heat treated
T4 = Solution heat treated then natural aging at room temp
T5 = Artificially aged after being rapidly cooled during fabrication
T6 = Solution heat treated, then artificially aged
T7 = Solution heat treated, then stabilized to control its growth and distortion
Note:
T2 through T7 = Do NOT memorize — not on the test
Hardness Identification
*T = Indicates the basic temper of the metal
This means the aluminum was heat treated to improve its strength.
Example: 2024-T4 – used in aircraft parts; strong because it’s heat treated and aged.
(Think of it like baking the metal to make it tougher.)
*F = As Fabricated
No heat treatment or cold working has been done—used just how it was made.
Example: 3003-F – used in siding, trim, or decorative panels.
(Think of it as “factory fresh,” no changes.)
*O = Annealed
Metal was softened by heat, so it’s easier to bend or form.
Example: 1100-O – used in foil or ducting where flexibility matters.
(Like softening butter so you can spread it.)
*H = Strain hardened / cold worked
Metal was made stronger by bending, rolling, or stretching—without using heat.
Example: 5052-H32 – used for fluid tanks and riveted parts.
(Like squishing clay to make it firm without baking it.)
Other Designations (Do NOT memorize):
H1 = Strain hardened only
H2 = Strain hardened and partially annealed
H3 = Strain hardened and stabilized
NOTE:
Only study T, F, O, and H.
H1, H2, H3 are not on the test.
What does the letter T mean in aluminum heat treatment and hardness identification?
T means the aluminum has been heat treated (heated and cooled in a controlled process) to improve strength and hardness.
What does the letter F mean in aluminum hardness identification?
F means the metal is as fabricated—it’s been formed into shape, but no heat treatment or cold working was done.
(Important: This is not the same as forging or heat treating—no internal hardening happens.)
What does the letter O mean in aluminum heat treatment and hardness identification?
O means the aluminum is annealed, which means it’s been softened using heat so it can be easily bent or formed.
What does the letter H mean in aluminum hardness identification?
H means the aluminum has been strain hardened (cold worked) to increase strength—no heat is used in this process.
What’s the difference between forging and heat treatment?
Forging is used to shape metal by hammering or pressing it—usually while it’s hot.
Heat treatment is used to change the metal’s properties (like hardness or strength) by heating and cooling it in a controlled way.
Forging = shaping
Heat treatment = hardening or softening
(Not on PowerPoint) Reheat Treatment
Heat-treatable metals (like 2024 rivets) are reheated to soften them for use.
Clad metals = max 3 times.
Common Aluminum – (2024-T3)
High-strength aluminum alloy
Considered aircraft quality
Excellent fatigue resistance
Lower corrosion resistance than 6061
Used for:
Fuselage
Wing skins
Aircraft structure
Cowls
Also ideal for:
Repairs and restoration
Has a shiny finish
Common Aluminum – (6061-T6)
Common Aluminum – (6061-T6)
Known for high corrosion resistance and good surface finish
Easy to weld
Strength is close to mild steel
Used for:
Wings and fuselage (non-critical parts)
Seat rails
Handles
Popular in homebuilt aircraft
Common Aluminum – (7075-T6)
Used to strengthen aircraft structures
Alloy mix includes:
Copper
Magnesium
Zinc
Very strong, but
Hard to weld (due to copper)
Not impossible to weld, just difficult
Hardness Testing— Rockwell Tester
Measures resistance to penetration
(Means how well the metal resists something pressing into it)
Also measures depth and hardness of the material
(How deep the tool sinks in and how firm the material is throughout)
Uses a penetrator forced into the metal
Verifies consistent hardness through the part
Hardness Testing— Brinell Tester
Uses a hardened spherical ball pressed into the surface
Measures resistance to penetration only
Uses hydraulic pressure
- 10 sec for ferrous metals
- 30 sec for nonferrous metals
Hardness Testing— Barcol Tester
Handheld tool for soft materials like aluminum, brass, or copper
Mainly used on installed parts
Works by hand in any position
Magnesium and its alloys
Magnesium is very light and suitable for aircraft use (about 2/3 the weight of aluminum)
Obtained primarily from electrolysis of sea water or brine from deep wells
Lacks strength in its pure form… so alloys are added (can be heat-treated)
Highly susceptible to corrosion and tends to crack (corrosion protection: chemically treated to form oxide film)
Readily burns in dust or small particle form (use caution when grinding or machining)
Beech Bonanza have magnesium-skinned ruddervators and are prone to corrosion… new parts hard to get and replacing with another material is prohibitive due to flight control balancing issues
Titanium and its Alloys
Lightweight and high strength (about 50% lighter than stainless steel)
Excellent corrosion resistance (but must be protected from nitrogen and oxygen at high temperatures)
Typically seen in landing gear systems and airframe systems like:
• Engine mounts
• High bypass fan of engines
• Stringers
• Hydraulic piping
• Engine exhaust
Nickel and its alloys
Two main types of nickel in aircraft use – Monel and Inconel
Monel is 68% nickel and 29% copper, and can be welded or machined
Monel is used in areas where great toughness is needed
Inconel is 80% nickel and 14% chromium
Inconel has frequent use in turbine engines and exhaust systems due to its ability to withstand corrosion and high heat areas
What is Monel?
68% nickel and 29% copper,
What is Inconel?
80% nickel and 14% chromium
Copper and its alloys
Very little use structurally
Great electrical and thermal conductivity (electrical wiring)
Brass
Brass is a copper alloy that contains zinc
Brass are often used in bushings, bearings, valve seats, and fuel metering
Bronze
Bronze is a copper alloy that contains tin
Just like Brass, bronze are often used in bushings, bearings, valve seats, and fuel metering
Ferrous metals
Any alloy containing iron as its chief ingredient is called ferrous metal and the most common is steel
Iron – fairly soft, fairly heavy, and low strength to weight ratio
Used in engine parts such as valve guides/piston rings or where its porous and wear characteristics allow it to hold lubricant film (helps retain oil through tiny surface pores.)
Iron is produced by mixing iron ore with coke (a grey, hard and porous coal-based fuel with a high carbon content) and limestone and submitting it to hot air
What is Steel (SAE and its numbers)
To make steel, pig iron (the product of smelting iron ore) is re-melted. Pure oxygen is forced into the molten metal where it combines with carbon and burns.
SAE (Society of Automotive Engineers) classifies steel alloys using a four-digit numerical index system, similar to aluminum.
SAE 1030:
1st digit = principal alloy element
2nd digit = percent of alloying element
Last two digits = percentage of carbon in the steel
How do you read SAE 1030?
SAE 1030:
1st digit = principal alloy element
2nd digit = percent of alloying element
Last two digits = percentage of carbon in the steel
Steel Alloys – Carbon
Carbon is the most common alloy found in steel. The amount of carbon affects hardness and strength.
More carbon = stronger steel
Steel Alloys – Low Carbon Steel
Used in safety wire, cable, bushings, rods.
Low carbon means it’s softer and easier to form, but not as strong. Good for flexibility, not high strength.
Steel Alloys – Medium Carbon Steel
Good for forging or machining when surface hardness is needed.
Stronger than low carbon steel, but still workable.
Steel Alloys – High Carbon Steel
Very hard, used in springs, files, cutting tools.
High carbon = very strong and hard, but also more brittle.
Steel Alloys – Sulfur
Is also considered an impurity.
Causes steel to be brittle when rolled or forged.
Must be removed during refining.
✅ When unwanted (impurity):
Makes steel brittle
Causes hot shortness (cracks when hot worked)
Bad for rolling, bending, forging
✅ When added on purpose:
Improves machinability
Helps chips break easier when cutting
Used in free-machining steels
Reduces ductility & toughness
Steel Alloys – Silicon
Acts as a hardener. Improves ductility (ability to stretch without breaking).
Steel Alloys – Phosphorous (List two things)
Raises yield strength and corrosion resistance. Makes steel stronger and longer lasting.
Steel Alloys – Nickel
Adds strength and hardness.
Common in bolts, nuts, pins, etc.
Steel Alloys – Chromium
Increases strength, hardness, wear resistance, and corrosion resistance.
Found in bearing balls and rollers.
Heat Treatment of Steel – Hardening (Explain the Process)
Consists of heating steel to a temp just above upper critical point for a specified time period and then cooled rapidly by plunging it in oil, water, or brine. At 200°F the part should be removed from the solution and allowed to cool until cold (tempering).
Heat Treatment of Steel – Tempering
Reduces brittleness caused by hardening and softens. It is always done at temperatures below the critical point. (Tempering involves reheating the hardened steel to a lower temperature, then cooling it, which relieves internal stress and adjusts hardness.)