13.2 Characteristic Organic Reactions
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
Saturated HCs have only __ bonds = m__ # of H atoms
Unsaturated HCs have d__ or t__ bonds
single, max, double, triple

Homolytic fission
> b__ c__ b__ in a way where each a__ takes an __ from bond to form 2 r__
breaking covalent bond, atom, e-, radicals
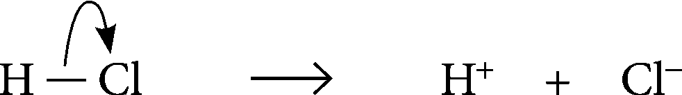
Heterolytic fission
> b__ c__ b__ in a way the m__ e-__ a__ takes b__ e- to form _ve ion, leaving a _ve ion
breaking covalent bond, more negative, atom, both, -, +
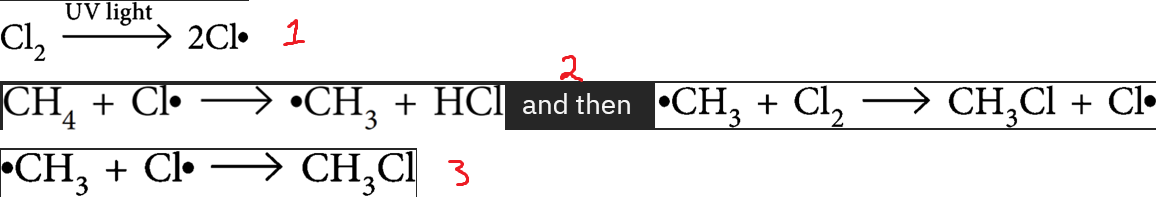
Free radical reactions
1. i__
> b__ c__ b__ using e__ from __ light → 2 free r__
2. p__
> free radicals a__ other mols to make m__ free radicals
> these radicals attack o__ mols = c__ reaction
3. t__
> 2 free r__ react & form p__ (no more free radicals)
initiation, break covalent bond, energy, UV, radicals, propagation, attack, more, other, chain, termination, radicals, product
Nucleophiles
Ø e- r__ species that can d__ a pair of e-
Ø “nucleo” + “phile” = loves n__ i.e. loves +ve charge itself is -ve
e.g. a__/s__ reactions
rich, donate, nucleus, addition, substitution
Electrophiles
Ø e- d__ species that can a__ a pair of e-
Ø “electro” + “phile” = loves e- i.e. loves -ve charge itself is +ve
e.g. _-bond breaking
deficient, accept, pi
Addition reaction
> where ≥_ mols c__ to make _ product
> e.g. hydration, h__
2, combine, 1, hydrogenation
Elimination
> small mol. (e.g. H2O/HCl) r__ from mol. & makes _/≡ bonds
removed, =
Substitution
> atom/g__ of atoms r__ w/ another atom/group of atoms
> needs __ light
group, replaced, UV
Hydrolysis
> b__ of mol. via __ (can be sped up w/ a__/alkali)
breakdown, H2O, acid
Condensation
> j__ 2 mols via r__ small mol (__ or HCl)
join, removing, H2O
Redox
Oxidation: O is __/ e- __ / incr. O n__ / _ removed ;; [__] can be used to show O from o__ agent
Reduction: O is __/ e- __ / __ O # / H __ ;; [H] can be used to show H from r__ agent
added, removed, number, H, O, oxidising, removed, added, decr, added, reducing
Electrophilic addition
> e- rich r__ in m__ is a__ by e__, then a__ occurs
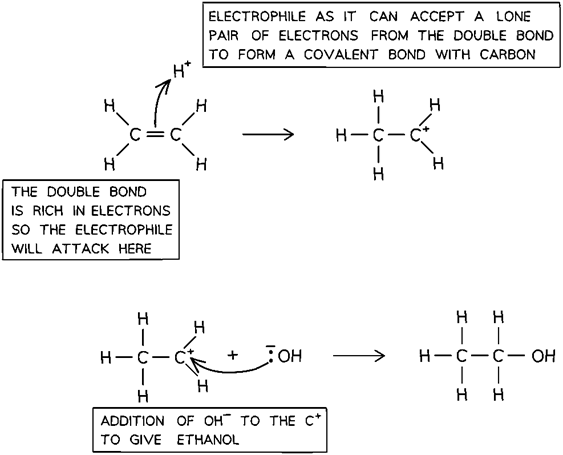
region, mol, attacked, electrophile, addition
Nucleophilic substitution
> e- rich nucleophile displaces h__ atom
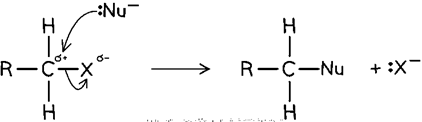
halogen
Nucleophilic addition
> e- d__ region in mol attacked by nucleophile, then a__ occurs

deficient, addition