7A AOL
5.0(1)
Card Sorting
1/118
Earn XP
Last updated 12:53 AM on 12/7/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
119 Terms
1
New cards
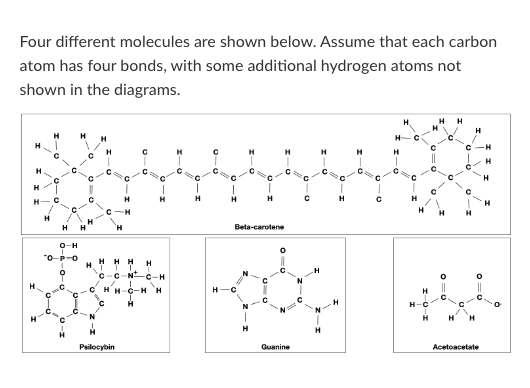
T/F: Beta-carotene would be more likely to cross a pure lipid bilayer than Acetoacetate.
True
2
New cards
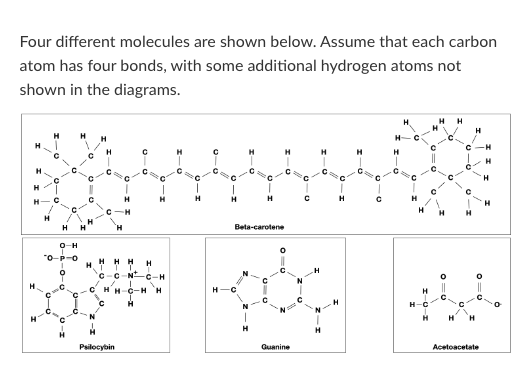
T/F: Guanine could form a hydrogen bond with water.
True
3
New cards
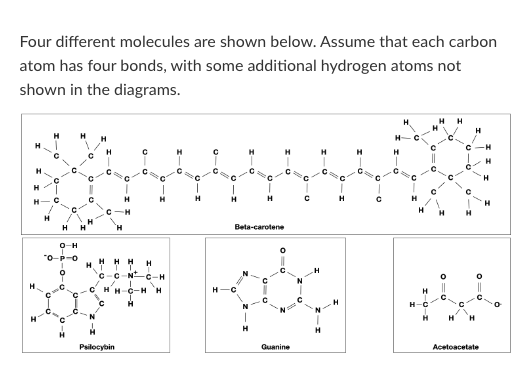
T/F: Guanine could form a hydrogen bond with an Acetoacetate molecule.
True
4
New cards
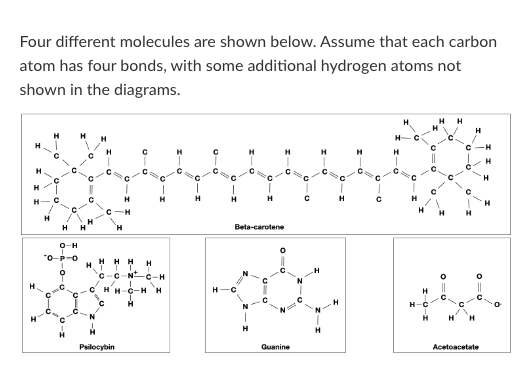
Which of the following words best describes beta-carotene?
A) Polar
B)Non polar
C)Amphiathic (has both polar and nonpolar regions)
A) Polar
B)Non polar
C)Amphiathic (has both polar and nonpolar regions)
Nonpolar
5
New cards
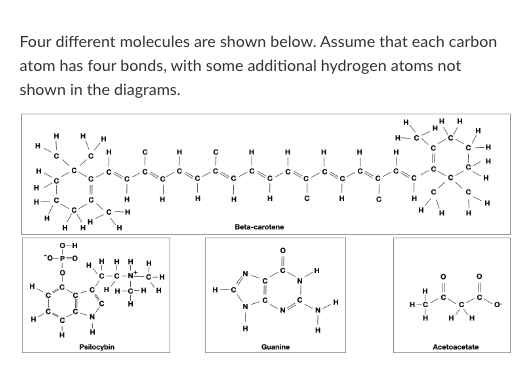
Psilocybin is hydrophobic.
False
6
New cards
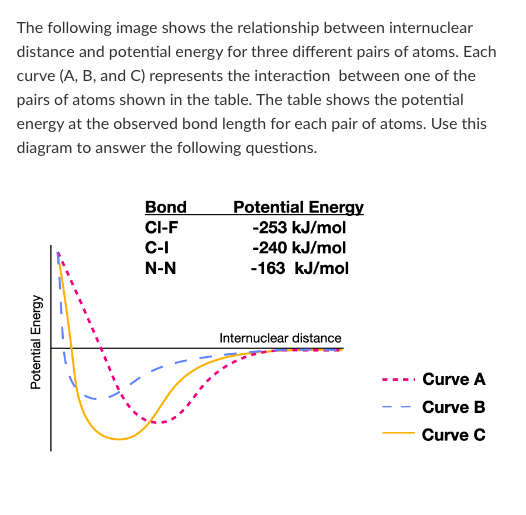
Which curve represents the interaction between a Cl atom and an I atom?
Curve A
Curve B
Curve C
Curve A
Curve B
Curve C
Curve A
7
New cards
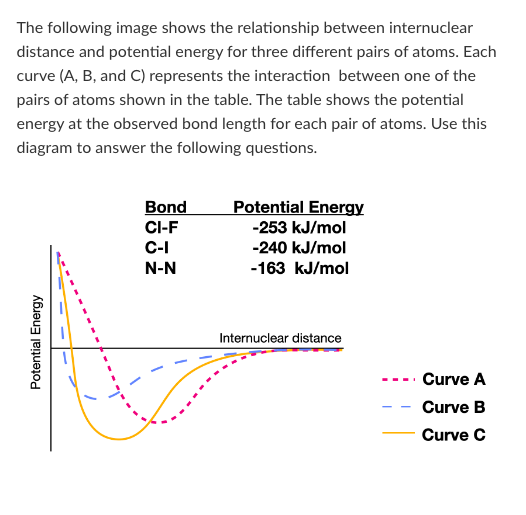
Which atom pair forms a bond with the longest internuclear distance?
CI-F
Cl-l
N-N
CI-F
Cl-l
N-N
C-I
8
New cards
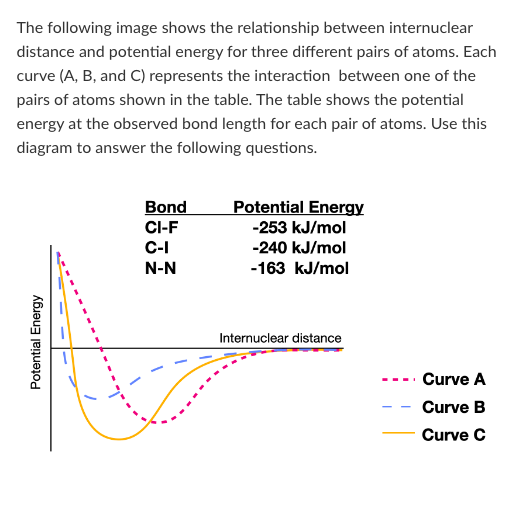
Which atom pair forms the weakest bond?
CI-F
Cl-l
N-N
CI-F
Cl-l
N-N
N-N
9
New cards
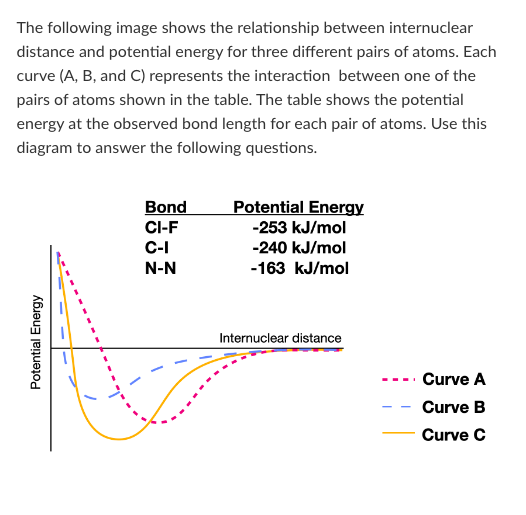
T/F: Breaking a bond between two N atoms would release 163 kJ/mol of energy.
False
10
New cards
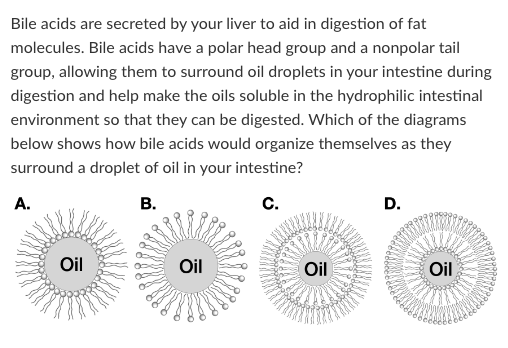
Diagram B
11
New cards
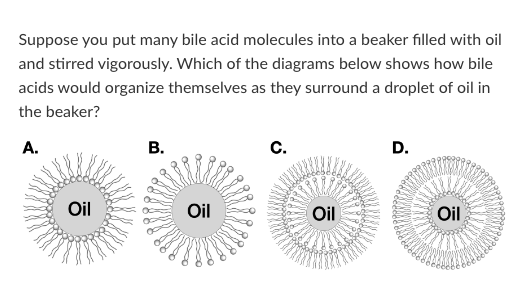
Diagram C
12
New cards
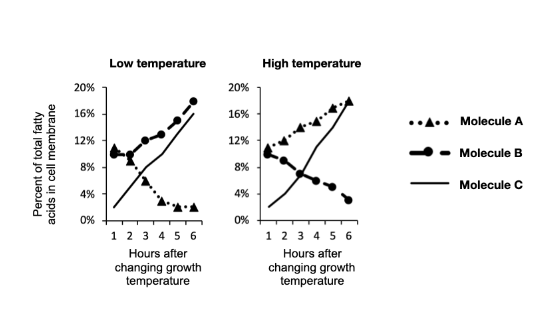
Which molecule is represented by "Molecule A" in the graphs?
Lauric Acid
Stearic Acid
Hopene
More than one of these is possible
Lauric Acid
Stearic Acid
Hopene
More than one of these is possible
Stearic Acid
13
New cards
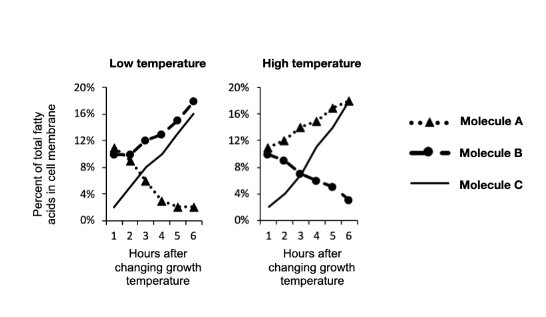
Which molecule is represented by "Molecule B" in the graphs?
Lauric Acid
Stearic Acid
Hopene
More than one of these is possible
Lauric Acid
Stearic Acid
Hopene
More than one of these is possible
Lauric Acid
14
New cards
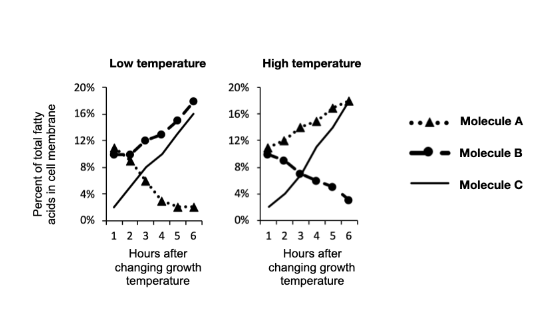
You anticipate that as global climate change increases the temperature of the Earth over time, these bacteria will adapt to the increasing temperature by changing their membrane composition. Which molecule(s) do you expect to decrease in abundance over time as bacteria adapt?
Lauric Acid
Stearic Acid
Hopene
More than one of these is possible
Lauric Acid
Stearic Acid
Hopene
More than one of these is possible
Lauric Acid
15
New cards
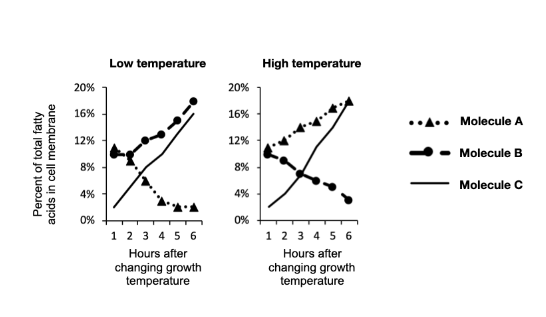
Imagine that you were to repeat this experiment, but this time you wanted to compare Eicosapentaenoic acid, a polyunsaturated fatty acid with many double bonds, and Docasanoic acid, a saturated fatty acid with no double bonds. Both of these fatty acids are 20 carbons long. Under which conditions would you expect to see a decrease in Docasanoic acid?
Low Temeprature
High Temperature
Both
Neither
Low Temeprature
High Temperature
Both
Neither
Low Temperature
16
New cards
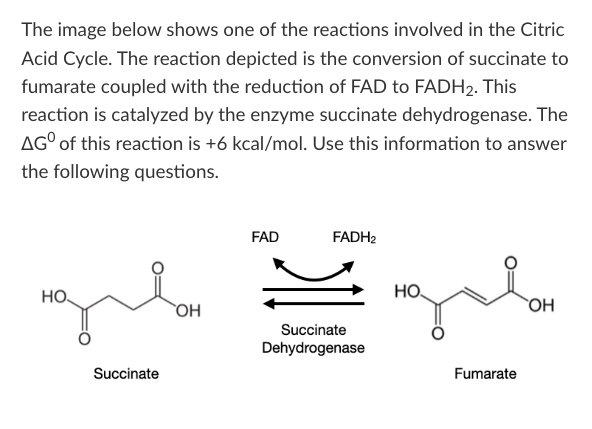
T/F: Because the ∆G0 of this reaction is positive, this reaction will only occur in the cell if coupled to an exergonic reaction.
False
17
New cards
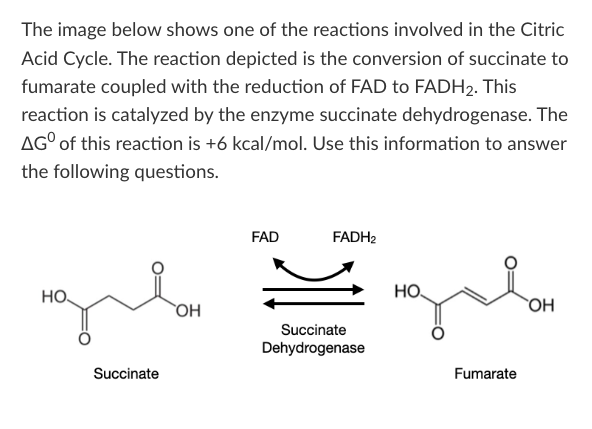
T/F: Succinate dehydrogenase will be used up during this reaction, so it must be replenished by the cell if the reaction is to continue occurring over time.
False
18
New cards
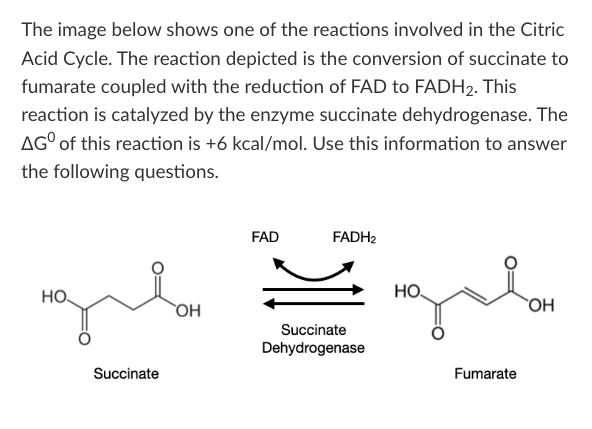
Which of the following statements accurately describes the effect of succinate dehydrogenase on this reaction?
A) Succinate dehydrogenase lowers the overall ∆G of this reaction, making it more favorable.
B) Succinate dehydrogenase provides the activation energy for this reaction to proceed.
C) Succinate dehydrogenase will make both the forward and reverse reactions occur at a faster rate.
D) Succinate dehydrogenase will speed up the forward reaction and slow down the reverse reaction
E) More than one of these is true.
F) None of these is true.
A) Succinate dehydrogenase lowers the overall ∆G of this reaction, making it more favorable.
B) Succinate dehydrogenase provides the activation energy for this reaction to proceed.
C) Succinate dehydrogenase will make both the forward and reverse reactions occur at a faster rate.
D) Succinate dehydrogenase will speed up the forward reaction and slow down the reverse reaction
E) More than one of these is true.
F) None of these is true.
Succinate dehydrogenase will make both the forward and reverse reactions occur at a faster rate.
19
New cards
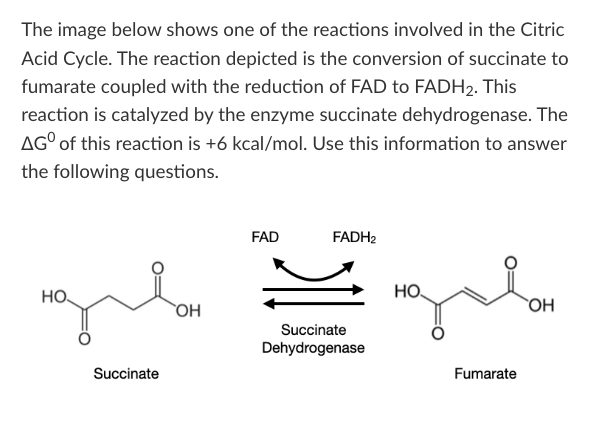
T/F: When this reaction is at equilibrium, the rate of the forward and reverse reactions will be equal to each other.
True
20
New cards
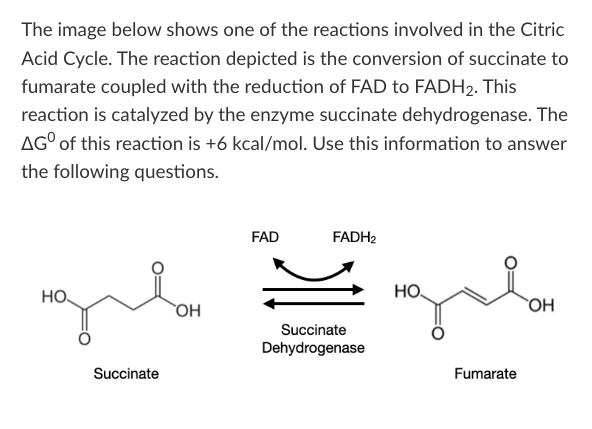
T/F: If standard enthalpy (∆H0) increases over the course of this reaction, then standard entropy (∆S0) could decrease.
True
21
New cards
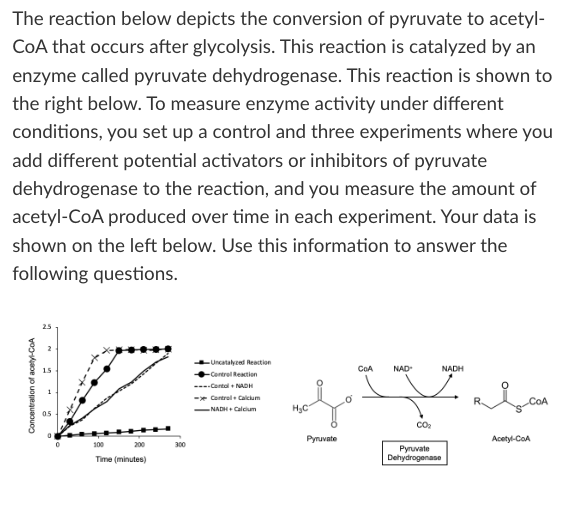
What is the effect of calcium on pyruvate dehydrogenase activity?
Calcium is an activator
Calcium is an inhibitor
Not enough information
Calcium is an activator
Calcium is an inhibitor
Not enough information
Calcium is an activator
22
New cards
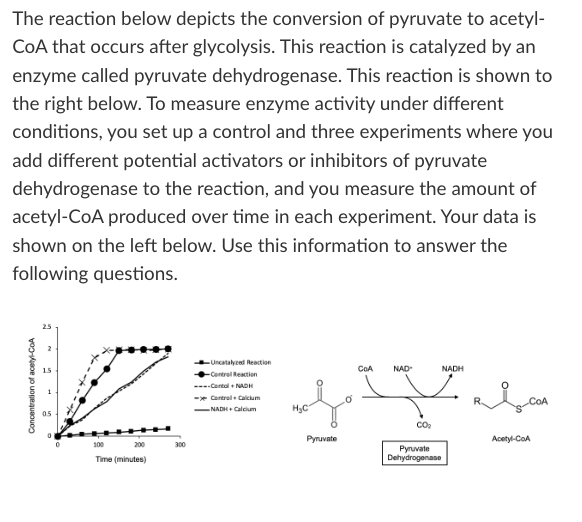
T/F: The addition of calcium and NADH together decreases pyruvate dehydrogenase activity.
True
23
New cards
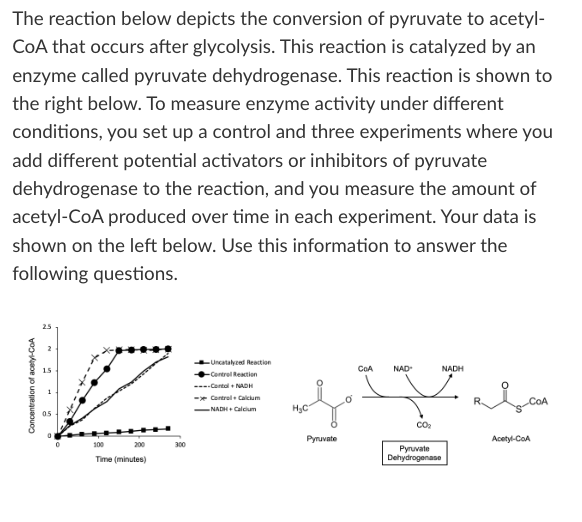
T/F: The addition of calcium to this reaction would result in less NADH production over the course of 100 minutes compared to the control reaction.
False
24
New cards
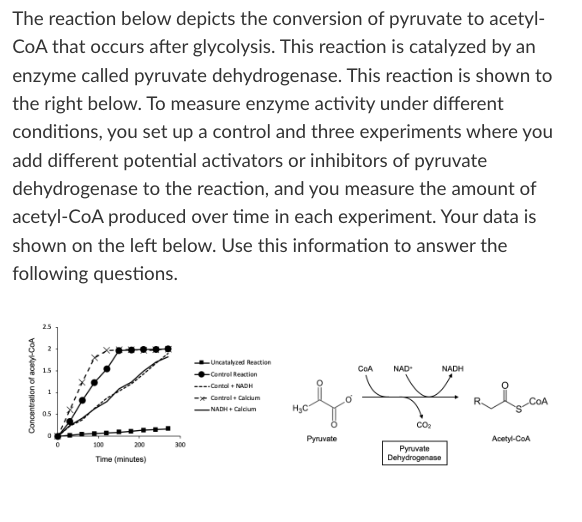
If you are doing an experiment where you want to produce the greatest amount of CO2 possible within 50 minutes, which of the following would you add to your control reaction?
NADH
Calcium
Both of these
Either of these
Neither of these
NADH
Calcium
Both of these
Either of these
Neither of these
Calcium
25
New cards
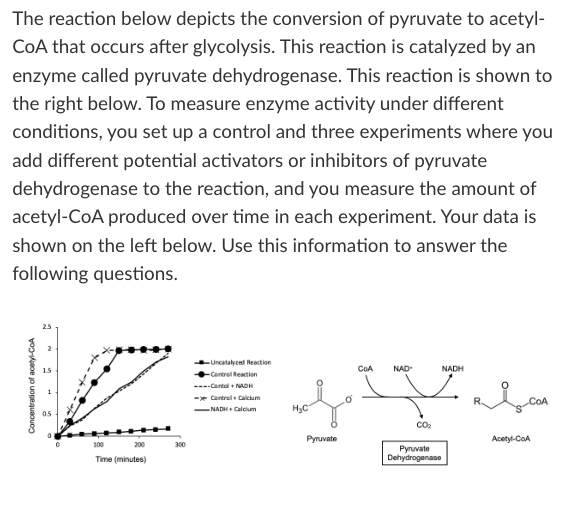
Pyruvate is produced during glycolysis in the cytosol of a cell. For the citric acid cycle to occur, pyruvate needs to be transported into the mitochondrion. Based on what you know about the structure of pyruvate, would you expect that pyruvate would diffuse through the mitochondrial membrane, or would you expect a dedicated pyruvate membrane transport protein to exist?
Pyruvate should be able to diffuse through the mitochondrial membrane
A dedicated pyruvate membrane transport protein must exist.
Pyruvate should be able to diffuse through the mitochondrial membrane
A dedicated pyruvate membrane transport protein must exist.
A dedicated pyruvate membrane transport protein must exist.
26
New cards

Which free energy graph best represents the "Control + Calcium" experiment?
Graph A
Graph B
Graph C
Graph D
None of the above
Graph A
Graph B
Graph C
Graph D
None of the above
Graph C
27
New cards

Which free energy graph best represents the "Uncatalyzed reaction" experiment?
Graph A
Graph B
Graph C
Graph D
None of the above
Graph A
Graph B
Graph C
Graph D
None of the above
Graph D
28
New cards

Which free energy graph best represents the effect of increasing the concentration of substrate by 10x in the control experiment?
Graph A
Graph B
Graph C
Graph D
None of the above
Graph A
Graph B
Graph C
Graph D
None of the above
Graph B
29
New cards
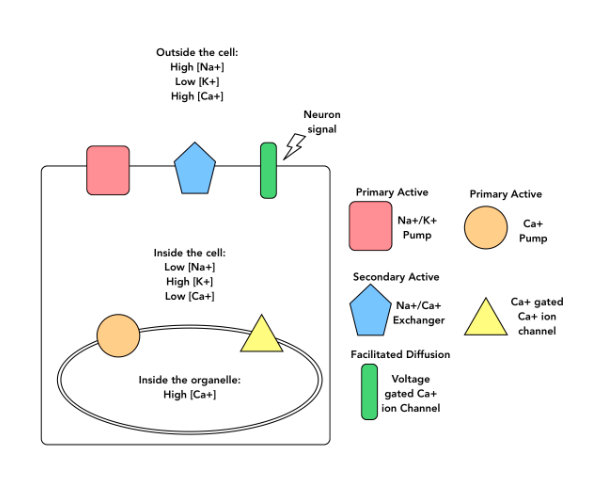
Based on the diagram and your knowledge of how membrane transport proteins work, which direction do you expect the Na+/Ca2+ to move its substrates through the cell membrane?
Both Na+ and Ca2+ into the cytosol
Both Na+ and Ca2+ outside of the cell
Na+ into the cytosol, and Ca2+ outside of the cell
Na+ outside of the cell, and Ca2+ into the cytosol
Both Na+ and Ca2+ into the cytosol
Both Na+ and Ca2+ outside of the cell
Na+ into the cytosol, and Ca2+ outside of the cell
Na+ outside of the cell, and Ca2+ into the cytosol
Na+ into the cytosol, and Ca2+ outside of the cell
30
New cards
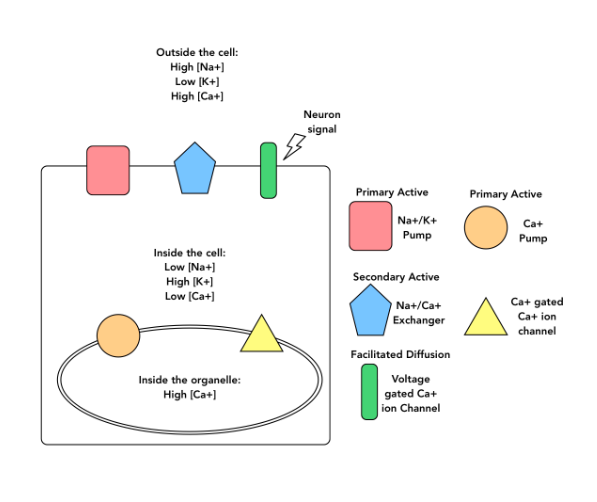
T/F: Based on the information provided, the Ca2+ gated Ca2+ ion channel is a passive transport mechanism.
True
31
New cards
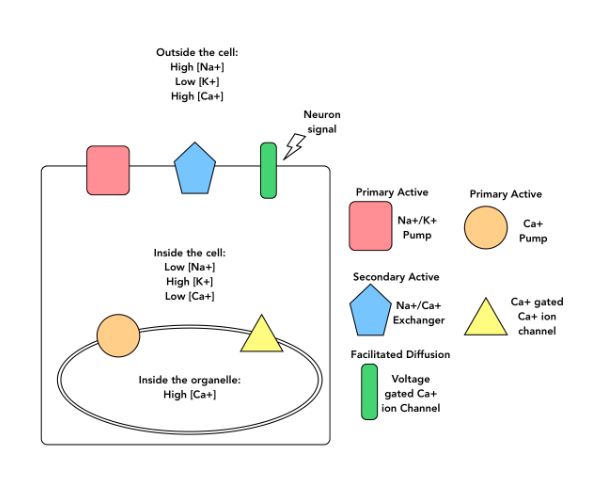
If the Na+/K+ pump stopped working, which of the following would you expect to observe?
Na+ concentration would increase outside of the cell.
Ca2+ concentration would increase inside the cytosol of the cell
The rate of Ca2+ movement through the voltage-gated Ca2+ ion channel (in response to a neuron signal) would decrease.
More than one of these would occur.
Na+ concentration would increase outside of the cell.
Ca2+ concentration would increase inside the cytosol of the cell
The rate of Ca2+ movement through the voltage-gated Ca2+ ion channel (in response to a neuron signal) would decrease.
More than one of these would occur.
More than one
32
New cards
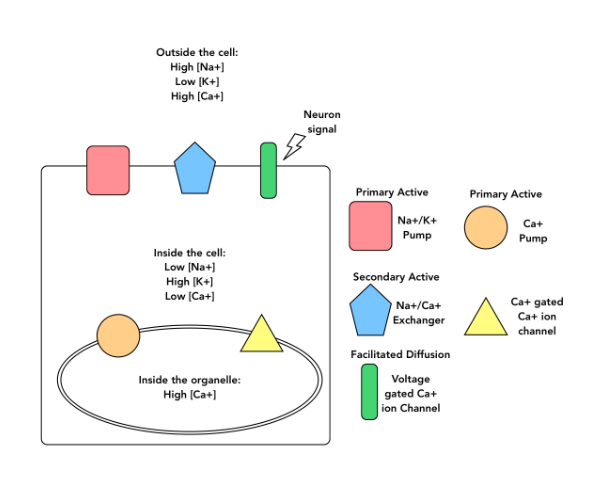
Based on the diagram and your knowledge of how membrane transport proteins work, which direction do you expect the Ca2+ pump to move Ca2+ through the organelle membrane?
From the cytosol into the sarcoplasmic reticulum
From the sarcoplasmic reticulum into the cytosol
It depends on the concentration of Ca2+ in the cytosol and the sarcoplasmic reticulum.
From the cytosol into the sarcoplasmic reticulum
From the sarcoplasmic reticulum into the cytosol
It depends on the concentration of Ca2+ in the cytosol and the sarcoplasmic reticulum.
From the cytosol into the sarcoplasmic reticulum
33
New cards
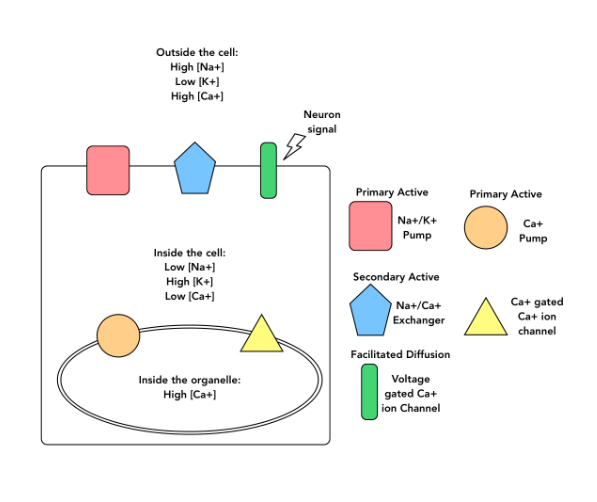
Verapimil is a medication used to treat patients who are experiencing abnormally fast heartbeats. This drug works by preventing the voltage-gated Ca+ ion channel from opening in response to neuron signaling. When a patient is treated with Verapimil, what would be the effect on the movement of ions through the Ca2+ gated Ca2+ channel?
More Ca2+ ions would move through this channel.
Fewer Ca2+ ions would move through this channel
It depends
More Ca2+ ions would move through this channel.
Fewer Ca2+ ions would move through this channel
It depends
Fewer Ca2+ ions would move through this channel
34
New cards

T/F: The observations above suggest that the liquid in cup A could have been a very highly concentrated salt and sugar solution.
True
35
New cards

The observations above suggest that immediately after using the liquid in cup B to water your plant, the liquid in the soil of your potted plant was ________ compared to the cytosol of your plant cells.
Hypertonic
Hypotonic
Isotonic
Hypertonic
Hypotonic
Isotonic
Hypotonic
36
New cards

You have another plant that is a little bit different. You happen to know that its cell membranes are permeable to water and solutes. On Day 1, this plant looked similar to your original plant on Day 1. If you were to water this plant with the solution in cup A, this plant would be _________ compared to the plant shown above.
More shriveled
Less shriveled
Similarly shriveled
More shriveled
Less shriveled
Similarly shriveled
Less Shriveled
37
New cards

T/F: On day 3, there is no longer any water moving in or out of your plant’s cells.
False
38
New cards

T/F: Observation 4 is consistent with hypothesis 1.
True
39
New cards

T/F: Observation 1 is consistent with hypothesis 1 and inconsistent with hypothesis 2.
True
40
New cards

T/F: Observation 3 is inconsistent with hypothesis 1.
False
41
New cards
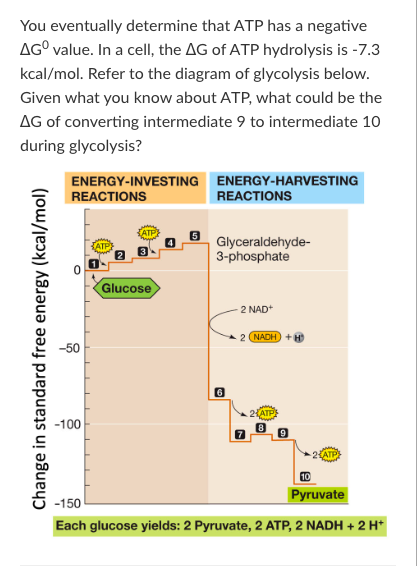
A) +16.0 kcal/mol
B) +14.0 kcal/mol
C)-1640 kcal/mol
D)-16.0 kcal/mol
B) +14.0 kcal/mol
C)-1640 kcal/mol
D)-16.0 kcal/mol
-16.0 kcal/mol
42
New cards
In a cell, the ∆G of ATP hydrolysis is -7.3 kcal/mol. This indicates that:
A) The products of ATP hydrolysis are more stable than the reactants.
B) The products of ATP hydrolysis are less stable than the reactants.
C) The products of ATP hydrolysis and the reactants have approximately equal stability.
A) The products of ATP hydrolysis are more stable than the reactants.
B) The products of ATP hydrolysis are less stable than the reactants.
C) The products of ATP hydrolysis and the reactants have approximately equal stability.
The products of ATP hydrolysis are more stable than the reactants.
43
New cards
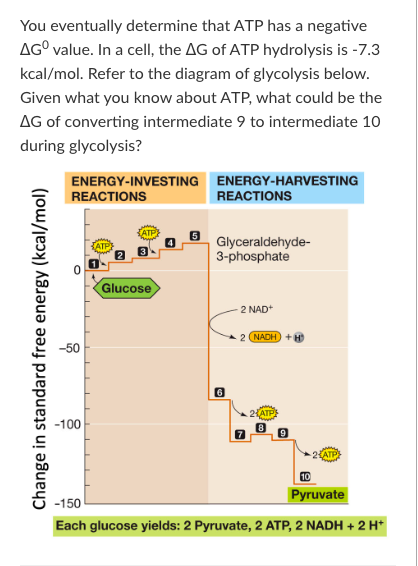
T/F: Glycolysis is an example of a catabolic pathway. Therefore, over the course of glycolysis, entropy decreases.
False
44
New cards
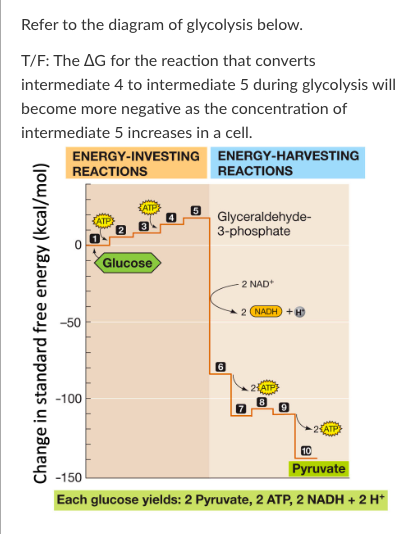
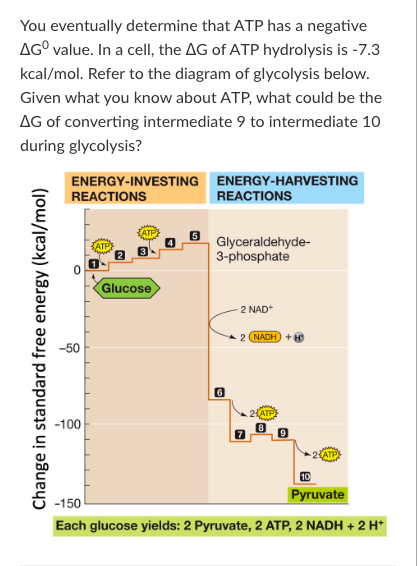
T/F: The ∆G for the reaction that converts intermediate 4 to intermediate 5 during glycolysis will become more negative as the concentration of intermediate 5 increases in a cell.
False
45
New cards
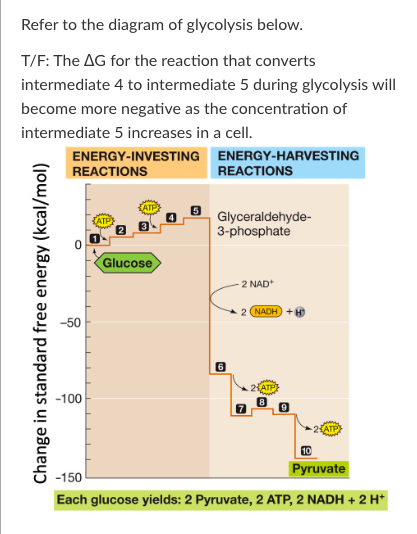
T/F: The conversion of intermediate 7 to intermediate 8 is not coupled to ATP hydrolysis. Therefore, the ∆G of this reaction is most likely negative under cellular conditions.
True
46
New cards
Which organelle would you expect to see a lot of in human stomach cells that secrete a lot of enzymes?
Lysosome
Golgi Apparatus
Vacuole
Chloroplasts
Mitochondria
Lysosome
Golgi Apparatus
Vacuole
Chloroplasts
Mitochondria
Golgi Apparatus
47
New cards
Which organelle would you expect to see a lot of in neurons that must continuously use the Na+/K+ pump to maintain an electrochemical gradient?
Lysosome
Golgi Apparatus
Vacuole
Chloroplasts
Mitochondria
Lysosome
Golgi Apparatus
Vacuole
Chloroplasts
Mitochondria
Mitochondria
48
New cards
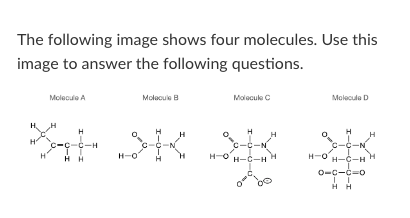
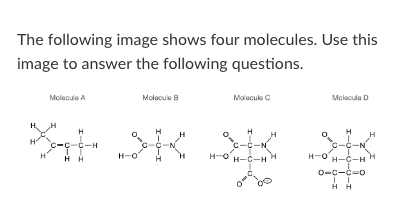
Which of the four molecules depicted above could not exist in nature?
Molecule A
Molecule B
Molecule C
Molecule D
All of these could exist
Molecule A
Molecule B
Molecule C
Molecule D
All of these could exist
Molecule D
49
New cards
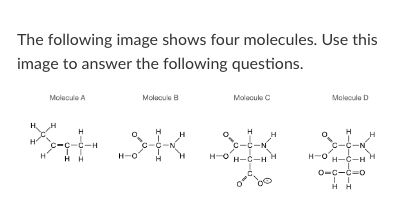
How many non-polar bonds exist in molecule B?
3
50
New cards
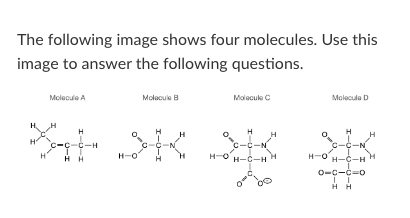
Which of molecules above could form Van der Waals interactions with another identical molecule?
Molecule A
Molecule B
Molecule C
Molecule D
All of these could exist
Molecule A
Molecule B
Molecule C
Molecule D
All of these could exist
More than one of the above
51
New cards
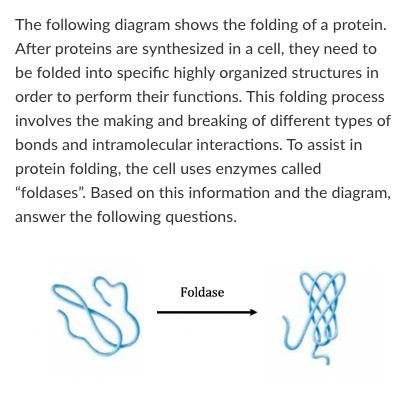
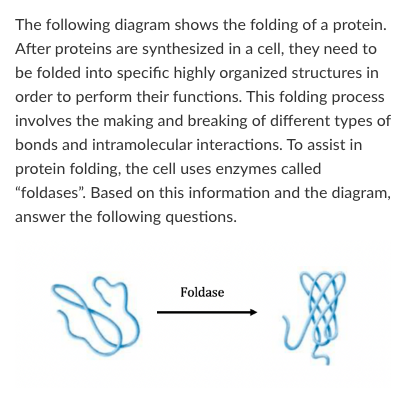
T/F: The entropy of the protein decreases over the course of this reaction
True
52
New cards
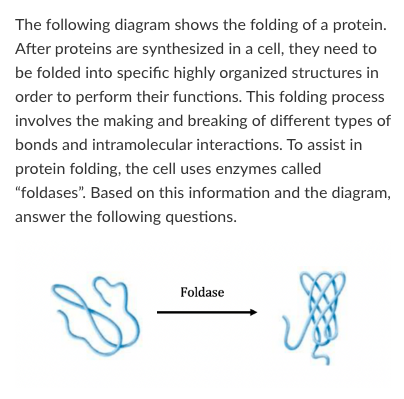
T/F: As this reaction proceeds, a small amount of energy will be released as heat.
True
53
New cards
T/F: Foldase provides the energy required to make this reaction proceed in the direction shown by the arrow.
False
54
New cards
T/F: Because this process uses an enzyme, we know that this process is endergonic without the enzyme.
False
55
New cards
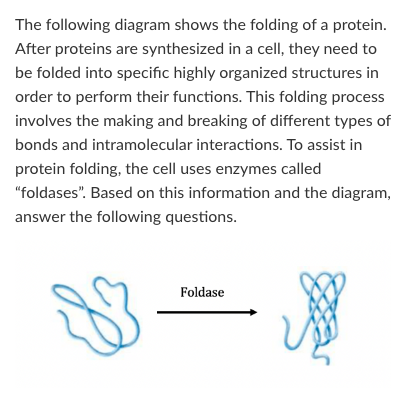
T/F: When foldase is present, the ∆G of this reaction becomes more negative.
False
56
New cards
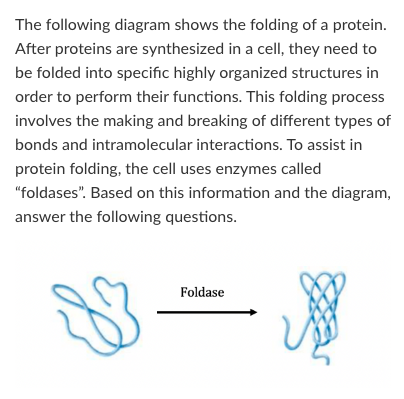
Because the protein folding reaction proceeds in the presence of a specific enzyme and no other components, we can conclude that:
A) The reaction in the presence of the enzyme will not proceed any faster if the temperature is raised a few degrees
B) The reverse reaction (protein unfolding) would not proceed in the presence of the enzyme
C) The reaction would proceed in the absence of the enzyme but at a slower rate
D) The reaction would not proceed in the absence of the enzyme
A) The reaction in the presence of the enzyme will not proceed any faster if the temperature is raised a few degrees
B) The reverse reaction (protein unfolding) would not proceed in the presence of the enzyme
C) The reaction would proceed in the absence of the enzyme but at a slower rate
D) The reaction would not proceed in the absence of the enzyme
The reaction would proceed in the absence of the enzyme but at a slower rate
57
New cards
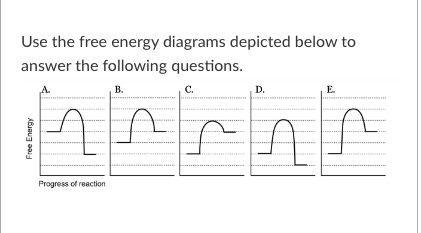
T/F: The reaction depicted in free energy diagram C could be coupled to the reaction depicted in free energy diagram D to produce an exergonic reaction.
False
58
New cards
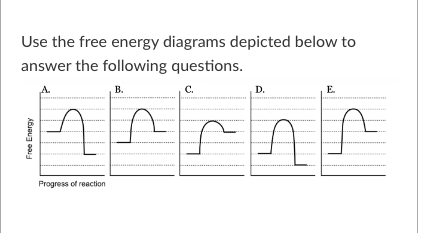
T/F: Because these diagrams depict changes in free energy over the course of a reaction, it is not possible to determine their relative reaction rates if no enzymes are involved.
False
59
New cards
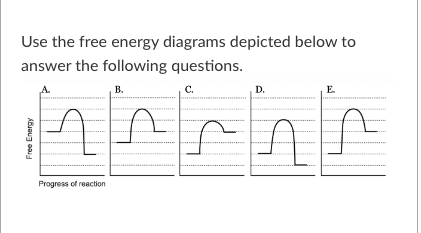
T/F: The reaction depicted in free energy diagram C could represent ATP hydrolysis.
False
60
New cards
Athletes eat large meals containing carbohydrate and fat when they are in training but their body weight (mass) remains nearly constant. Which of the following statements correctly describes what happens to most of the mass consumed?
It is converted into adenosine triphosphate (ATP).
It is released as carbon dioxide and water.
It is converted to energy
It is converted into adenosine triphosphate (ATP).
It is released as carbon dioxide and water.
It is converted to energy
It is released as CO2 and H2O
61
New cards
T/F: Mouse DNA must contain equal amounts of thymine and guanine.
False
62
New cards
T/F: Mouse DNA may contain a different number of purines and pyrimidines.
False
63
New cards
T/F: Mouse DNA must denature at the same temperature as human DNA.
False
64
New cards
Imagine you have discovered a new species of bacteria. To begin your investigation of this organism, you run an assay on the total nucleotide content of the bacterial DNA. The thymine content of DNA from the bacterial cells is 40%. You also run an assay on the total nucleotide content of E. coli, another species of bacterium. The thymine content of DNA from E. coli is 24%.
T/F: the DNA from your new bacterial species will denature at a lower temperature than the DNA from E. coli.
T/F: the DNA from your new bacterial species will denature at a lower temperature than the DNA from E. coli.
True
65
New cards
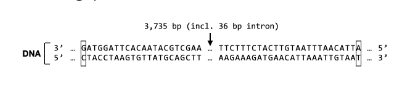
T/F: The sequence shown on your computer screen includes promoter regions for the S Protein gene and Gene X.
False
66
New cards
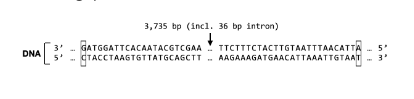
T/F: RNA polymerase will read the sequence for the S Protein gene from the 5’ end to the 3’ end of the template strand.
False
67
New cards
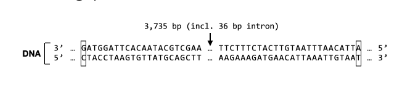
T/F: The primary structure of the proteins expressed from each of these genes will be the same.
False
68
New cards
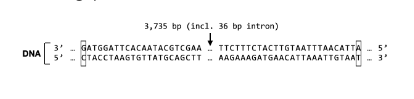
T/F: The tertiary structure of S Protein and the protein expressed from Gene X will most likely be the same.
False
69
New cards
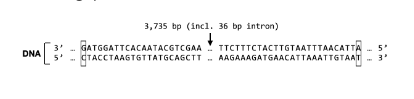
The amino acid sequence of Protein X begins Met-Gln-Leu. Which strand is the template strand for the Protein X gene?
Top strand
Bottom strand
Top strand
Bottom strand
The top strand shown in the diagram
70
New cards
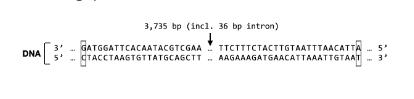
How many total amino acids would you find in S Protein?
1253
1254
1255
1233
1243
A different number not shown above
Not possible to say without more information
1241
1253
1254
1255
1233
1243
A different number not shown above
Not possible to say without more information
1241
1,243
71
New cards
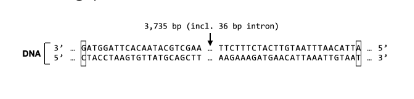
What is the second to last amino acid in S Protein?
Thr
Met
lle
Tyr
None
Thr
Met
lle
Tyr
None
None of these
72
New cards
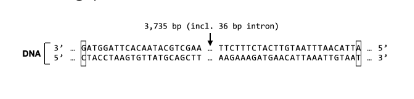
T/F: When transcribing Gene X, RNA polymerase will read the DNA sequence from the right side of your computer screen to the left side of your computer screen.
False
73
New cards
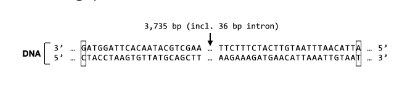
Which of the following changes to a codon would you expect to be most damaging to the resulting protein (all codons are written 5'->3')?
AGA->CGG
AAG->CGU
UCG->AGU
GAA->AAA
AGA->CGG
AAG->CGU
UCG->AGU
GAA->AAA
GAA->AAA
74
New cards
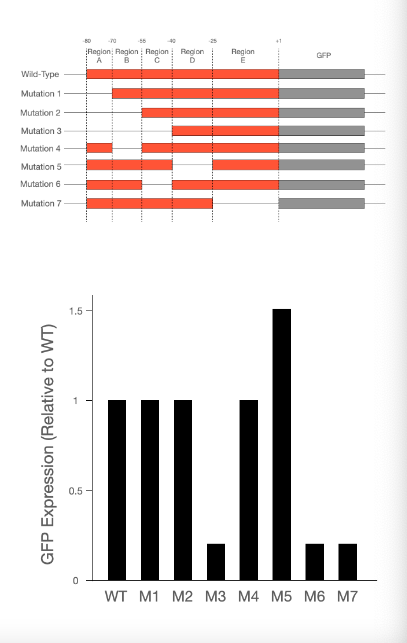
T/F: Region B could be a promoter sequence.
False
75
New cards
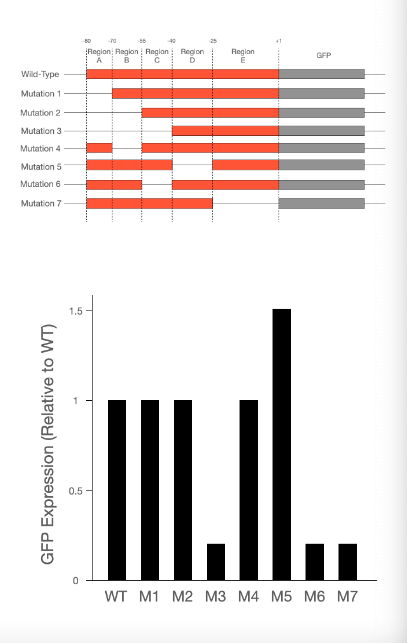
T/F: Region A does not appear to play a role in transcription.
True
76
New cards
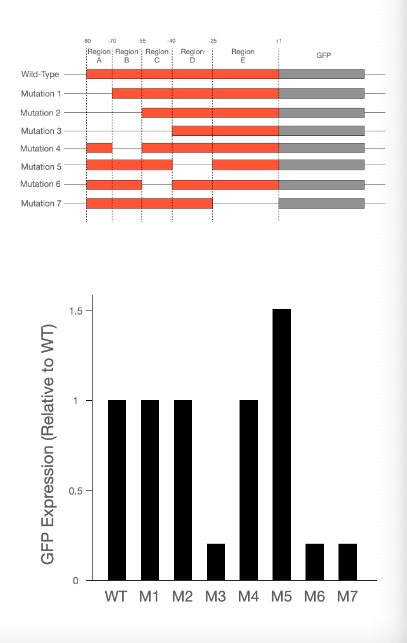
T/F: Region C could contain a transcription factor binding site.
True
77
New cards
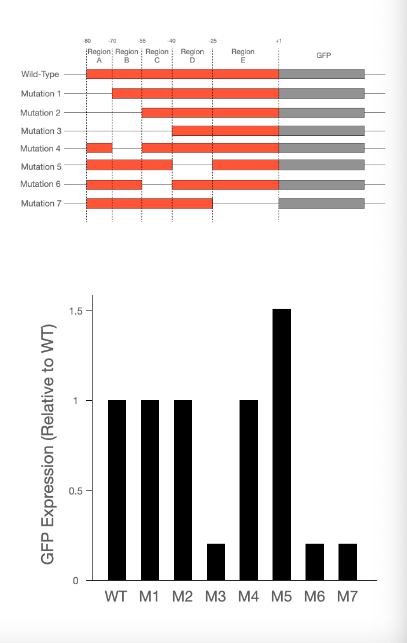
T/F: When region D is deleted, expression of GFP increases.
True
78
New cards
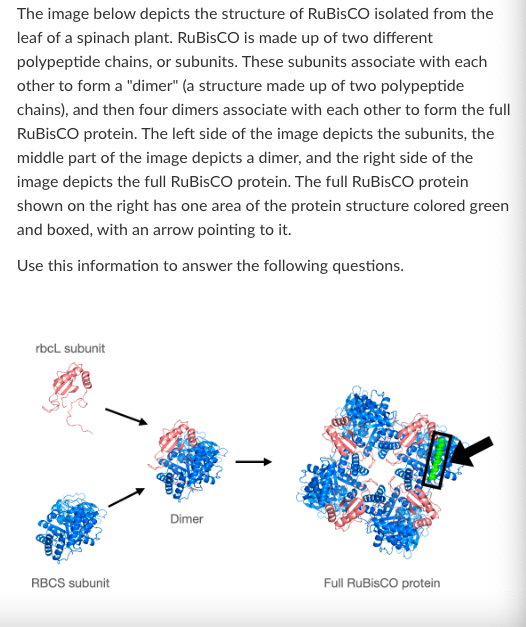
What is the highest level of protein structure shown in the 'RBCS subunit' image?
Primary
Secondary
Tertiary
Quartenary
Primary
Secondary
Tertiary
Quartenary
Tertiary
79
New cards
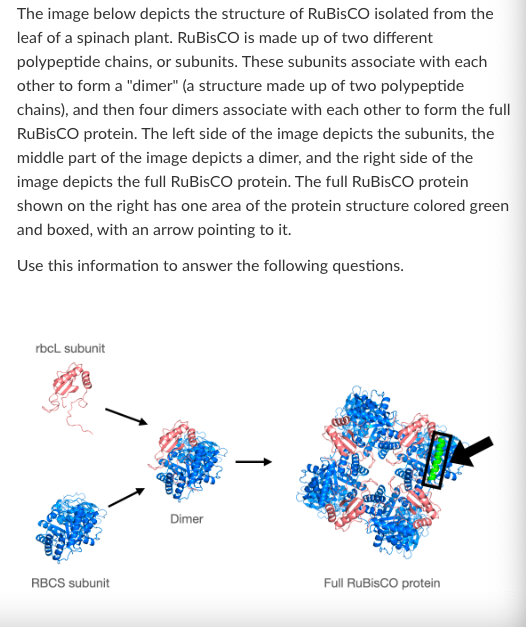
What is the highest level of protein structure shown in the 'Dimer' image?
Primary
Secondary
Tertiary
Quartenary
Primary
Secondary
Tertiary
Quartenary
Quarternary
80
New cards
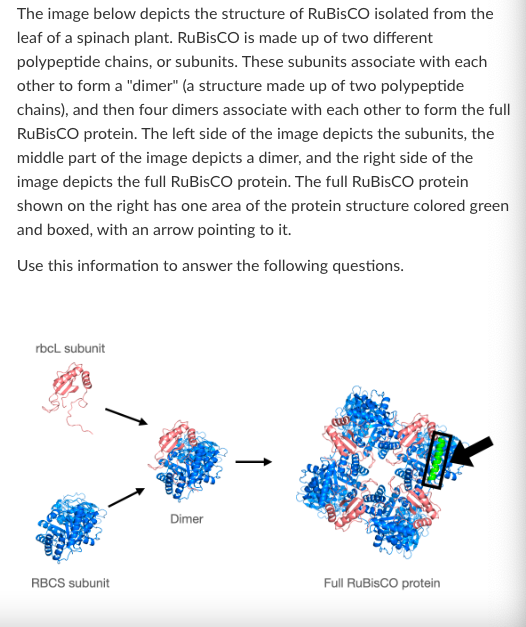
T/F: Disrupting the secondary structure of RuBisCO would most likely affect its primary structure.
False
81
New cards
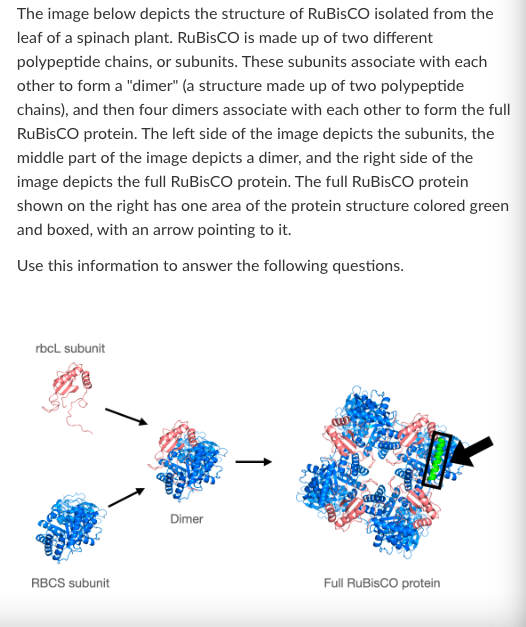
T/F: If heat were added to RuBisCO, the primary structure would be the first level of structure to be disrupted.
False
82
New cards
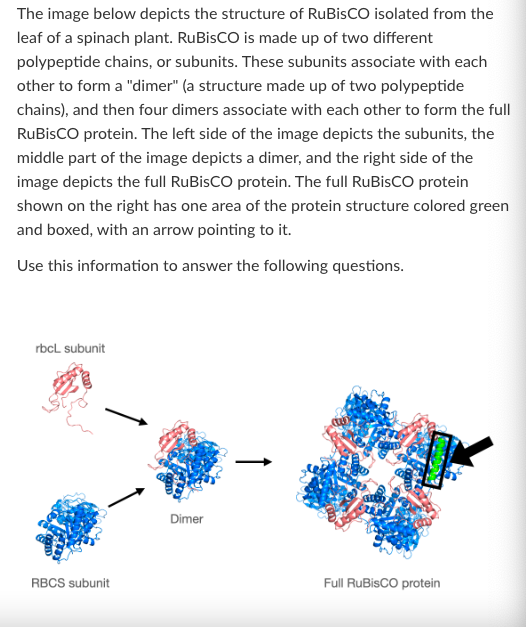
T/F: The tertiary structure of RuBisCO is formed primarily by covalent bonding between amino acids that are not necessarily adjacent to each other in the primary structure.
False
83
New cards
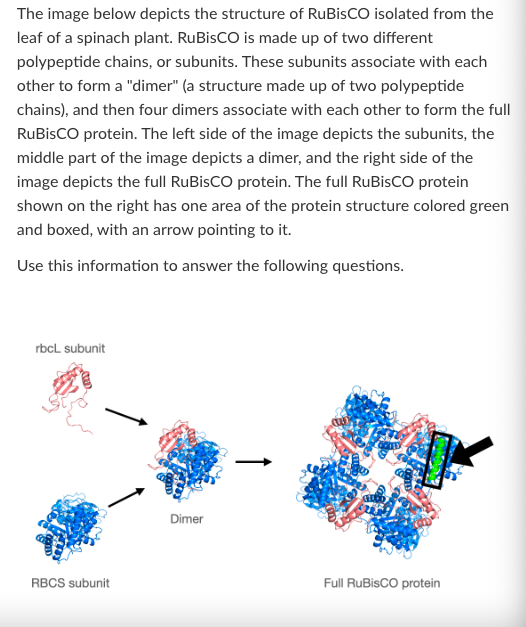
What level of protein structure is shown in the boxed green section of the full RuBisCO protein?
Primary
Secondary
Tertiary
Quartenary
Primary
Secondary
Tertiary
Quartenary
Secondary
84
New cards
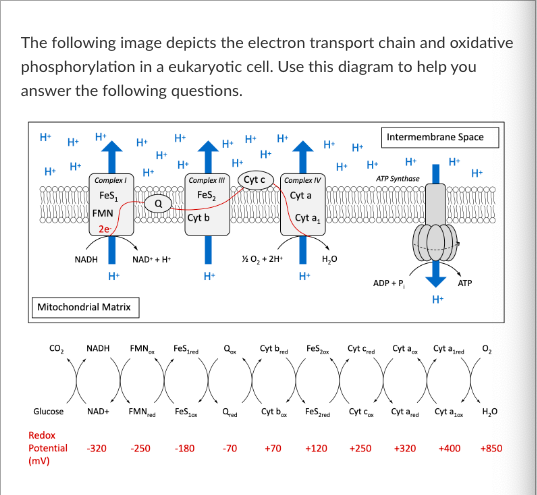
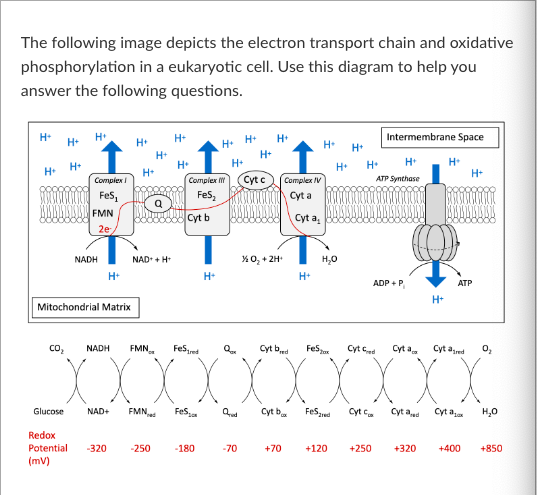
T/F: FMNox is an example of a reducing agent.
False
85
New cards
True
86
New cards
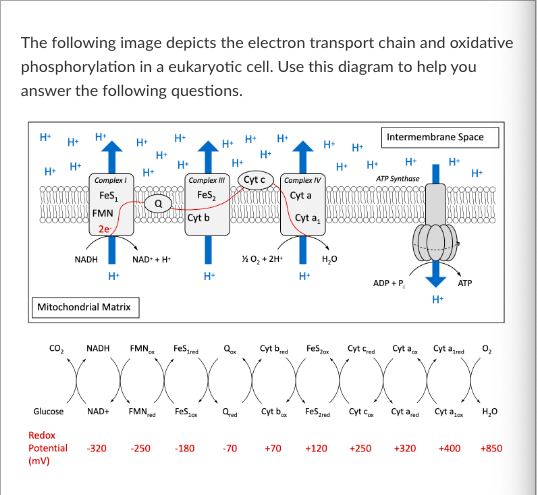
T/F: At the same time the total amount of NADH increases in a cell, the total amount of NAD+ in the cell will decrease.
True
87
New cards
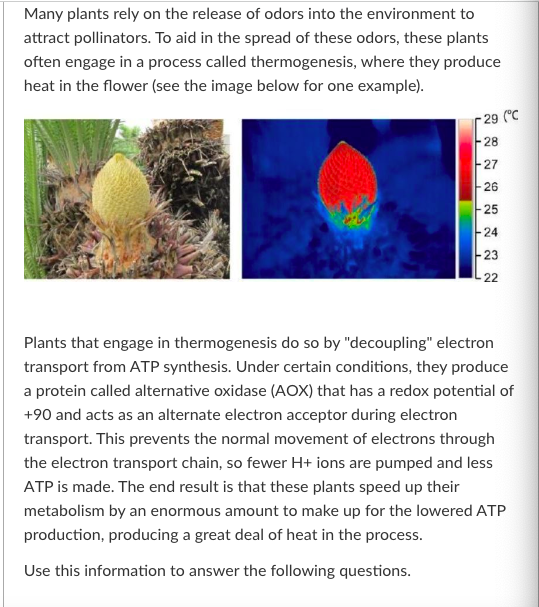
Given that you know that the redox potential of AOX is +90, which molecule in the electron transport chain could AOX be accepting electrons from?
Cyt a1
FeS1
FMN
FeS2
More than one of these
Cyt a1
FeS1
FMN
FeS2
More than one of these
More than one of these
88
New cards
If a plant cell begins producing AOX, the amount of Cyt cox will ______ compared to before AOX was present.
Increase
Stay the same
Decrease
Increase
Stay the same
Decrease
Increase
89
New cards
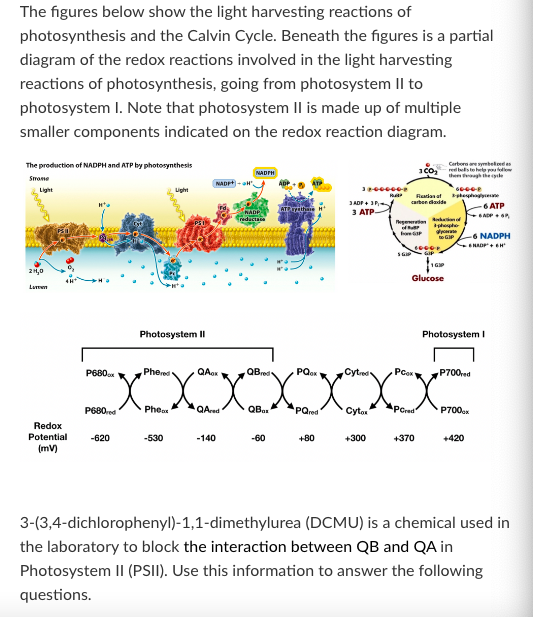
T/F: When a plant is treated with DCMU, NADPH accumulates.
False
90
New cards
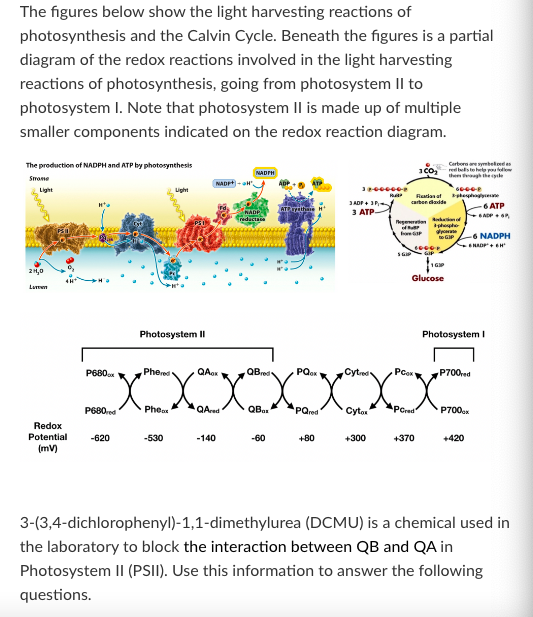
T/F: After a plant is treated with DCMU, light energy will still be absorbed by chlorophyll molecules in photosystem I.
True
91
New cards
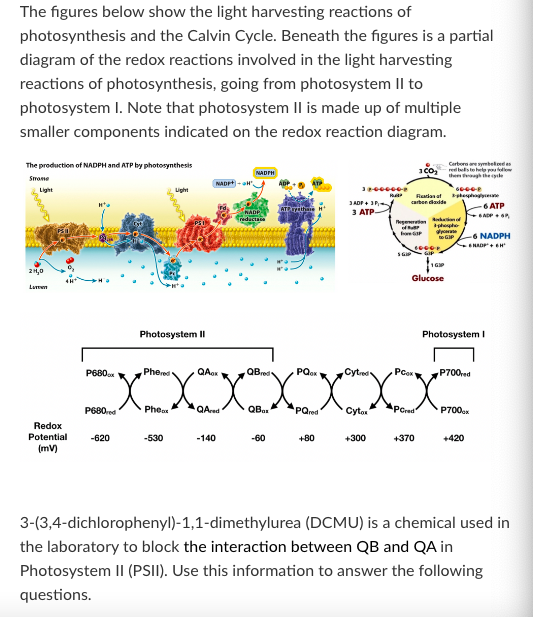
T/F: When a plant is treated with DCMU, Pc accumulates in its reduced form
False
92
New cards
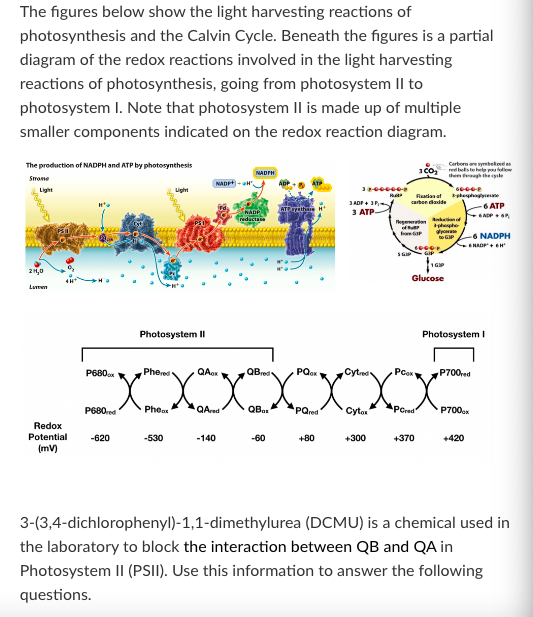
Treating a plant with DCMU will cause the plant's mass to ________ over time.
Increase
Decrease
Stay the same
Increase
Decrease
Stay the same
Decrease
93
New cards
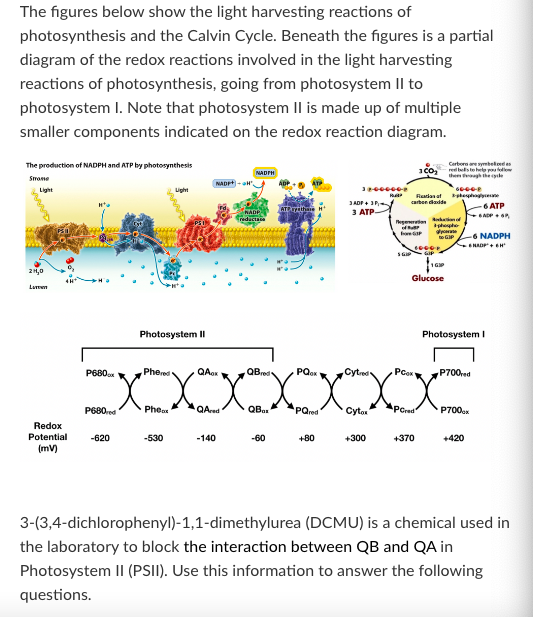
T/F: When a plant is treated with DCMU, G3P will accumulate in the chloroplast.
False
94
New cards
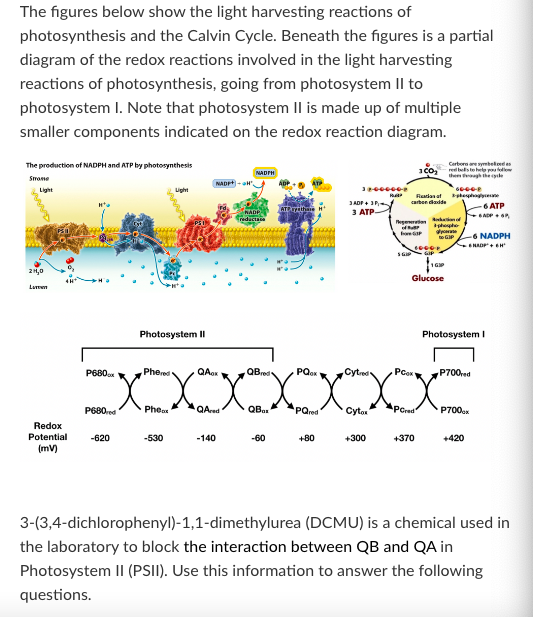
Adding a particular drug changes the redox potential of P700 from +420 to +310. Which of the following would you expect to observe in plant cells treated with this drug?
A) Phe would accumulate in its oxidized form.
B) ATP would continue to be produced in the chloroplast.
C) Pc would accumulate in its oxidized form.
D) Levels of 3-phosphoglycerate would decrease.
E) More than one of the above
F) None of the above
A) Phe would accumulate in its oxidized form.
B) ATP would continue to be produced in the chloroplast.
C) Pc would accumulate in its oxidized form.
D) Levels of 3-phosphoglycerate would decrease.
E) More than one of the above
F) None of the above
None of the above
95
New cards
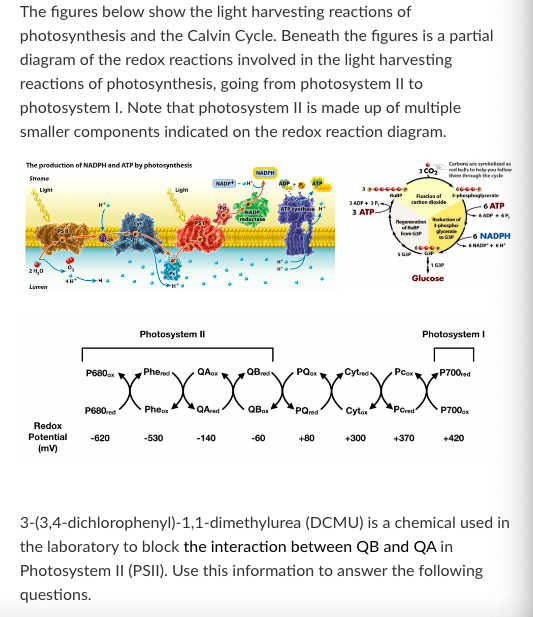
Prometryn is an herbicide that acts as a photosystem II inhibitor (it prevents photosystem II from splitting water). If a plant were treated with prometryn, which of the following effects would you expect to observe?
G3P production would decrease
3-phosphoglycerate would accumulate in the chloroplast
NADPH would accumulate in the chloroplast
Pc would accumulate in its oxidized form
ATP production would decrease in the chloroplast
More than one of the above
None of the above
G3P production would decrease
3-phosphoglycerate would accumulate in the chloroplast
NADPH would accumulate in the chloroplast
Pc would accumulate in its oxidized form
ATP production would decrease in the chloroplast
More than one of the above
None of the above
More than one of the above
96
New cards
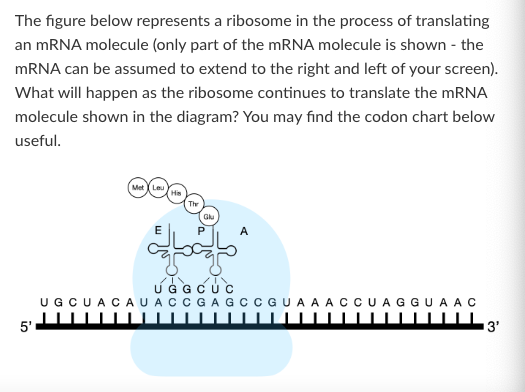
Which of the following describes where the ribosome began translating this mRNA molecule?
At the AUG nearest to the 5' cap
At the beginning of exon 1
At the +1 transcription start site
At the 5' cap
At the promoter
At the AUG nearest to the 5' cap
At the beginning of exon 1
At the +1 transcription start site
At the 5' cap
At the promoter
At the AUG nearest to the 5' cap
97
New cards
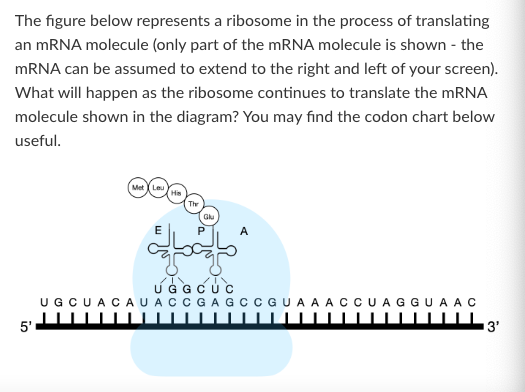
T/F: The next amino acid added to the polypeptide will form a peptide bond with the Met amino acid.
False
98
New cards
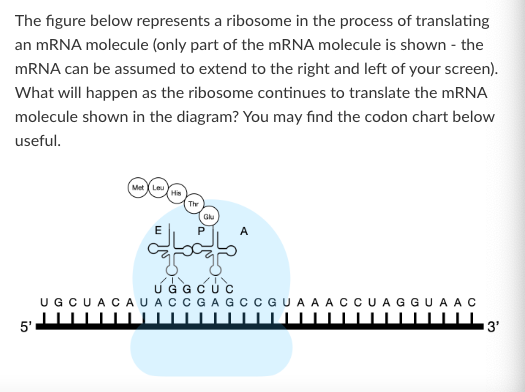
What is the anticodon of the tRNA that brought histidine into the growing polypeptide chain?
3'-AUC-5'
3'-GAU-5'
3'-GUA-5'
3'-CAU-5'
None of the above
3'-AUC-5'
3'-GAU-5'
3'-GUA-5'
3'-CAU-5'
None of the above
3'-GUA-5'
99
New cards
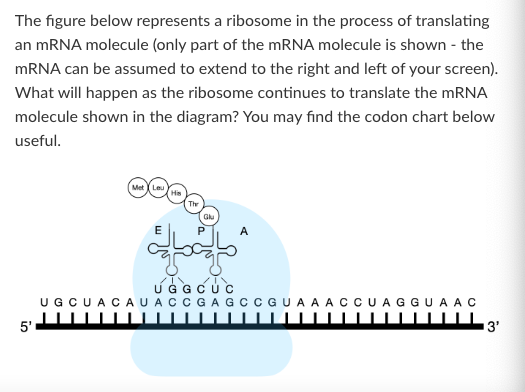
What amino acid will be attached to a tRNA with the anticodon sequence 3'-AUG-5'?
NONE OF THESE
100
New cards
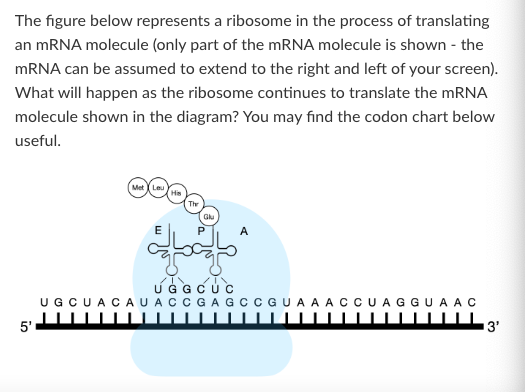
In a certain mutant strain of bacteria, 10% of the tRNAs with the anticodon 5’-ACG-3’ are modified so that they have an anticodon of 5’-AGC-3’. These bacteria will synthesize:
Proteins in which serine is inserted at some positions normally occupied by tyrosine
Proteins in which tyrosine is inserted at some positions normally occupied by serine.
Proteins in which arginine is inserted at some positions normally occupied by alanine
Proteins in which alanine is inserted at positions normally occupied by arginine
Shorter proteins on average than the wild-type bacterium
Longer proteins on average than the wild-type bacterium
Proteins in which serine is inserted at some positions normally occupied by tyrosine
Proteins in which tyrosine is inserted at some positions normally occupied by serine.
Proteins in which arginine is inserted at some positions normally occupied by alanine
Proteins in which alanine is inserted at positions normally occupied by arginine
Shorter proteins on average than the wild-type bacterium
Longer proteins on average than the wild-type bacterium
Proteins in which arginine is inserted at some positions normally occupied by alanine