Chemistry - energy changes
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
What are exothermic reactions?
Exothermic reactions are reactions that transfer energy to their surroundings (usually as heat or sometimes light)
Why do the reactants have more energy than the products in an exothermic reaction?
more energy is released forming new bonds in the products than is absorbed to break the original reactant bonds
What happens to the temperature of the surroundings in an exothermic reaction?
it increases
What happens when bonds break?
energy is absorbed (endothermic)
What happens when bonds form?
energy is released (exothermic)
Examples of exothermic reactions
- Combustion
- Neutralisation
- respiration
- forming new bonds
Uses of exothermic reactions
Self-heating cans, hand warmers, burning fuels
What is the energy profile diagram for an exothermic reaction
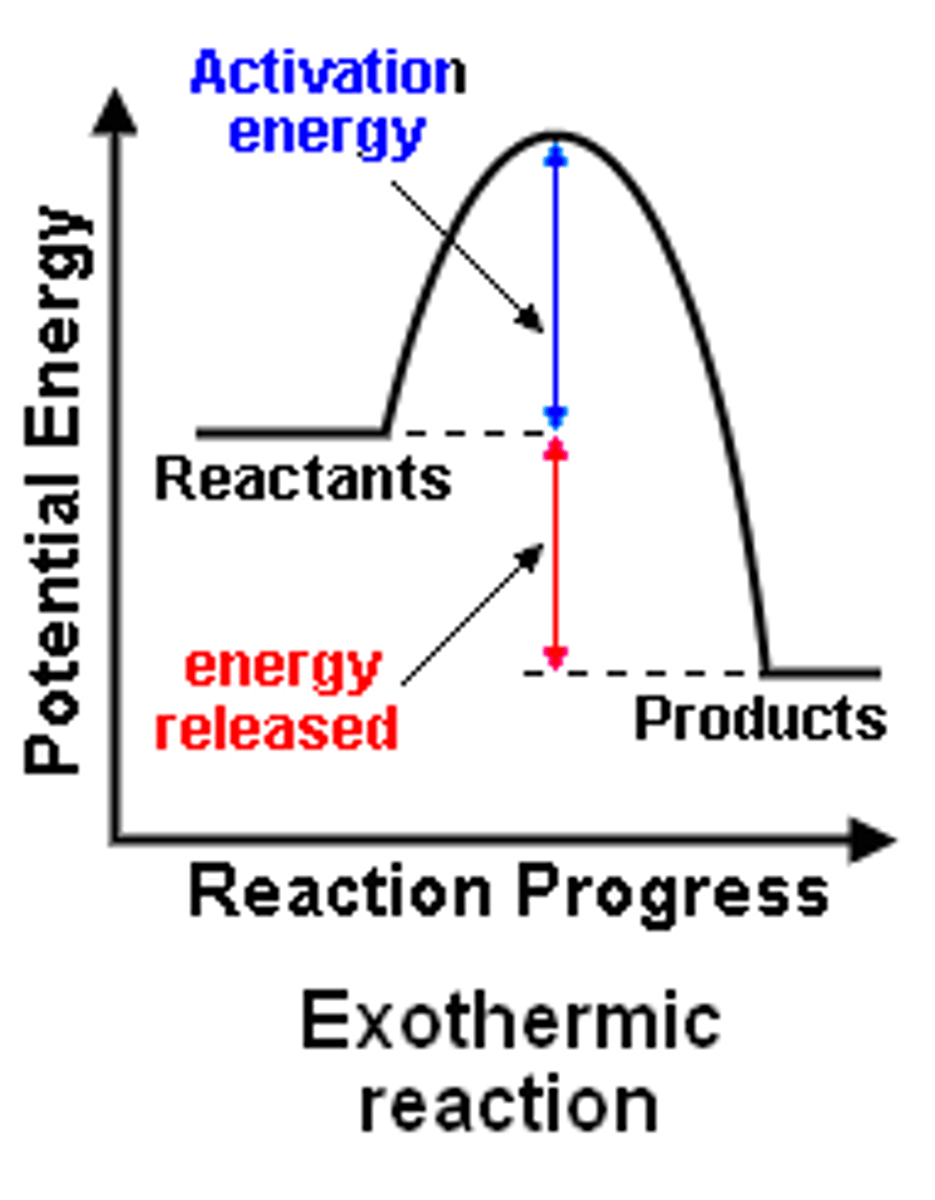
What is the energy transfer in exothermic reactions?
The chemical energy store decreases, and the thermal energy of the surroundings increases
What are endothermic reactions?
Endothermic reactions are reactions that take in energy from their surroundings
What is activation energy?
energy needed to start a reaction
What happens when reactants have less energy than the activation energy?
They will just bounce off eachother
Why do the products have more energy than the reactants in an endothermic reaction?
more energy is absorbed breaking old bonds in the reactants than is released to make new bonds
Why do endothermic reactions absorb energy?
the energy required to break the bonds in the reactants is greater than the energy released when new bonds form in the products
Why do exothermic reactions release energy?
because the total chemical energy stored in the bonds of the products is less than the energy stored in the bonds of the reactants