Materials Science Practice Exam 1.0
1/28
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
29 Terms
Boron electronic structure 1s² 2s² 2p¹. Which are NOT valence? (Q1)
1s²
You are given the electronegativities of two atoms A and B. Predict the most likely type of bonding expected for compound AB. (Q2)
Bonding types: A=2.2, B=2.2
Covalent
You are given the electronegativities of two atoms A and B. Predict the most likely type of bonding expected for compound AB. (Q2)
Bonding types: A=0.8, B=1.0
Metallic
You are given the electronegativities of two atoms A and B. Predict the most likely type of bonding expected for compound AB. (Q2)
Bonding types: A=0.9, B=3.3
Ionic
You are given the electronegativities of two atoms A and B. Predict the most likely type of bonding expected for compound AB. (Q2)
Bonding types: A=2.1, B=2.7
Polar Covalent
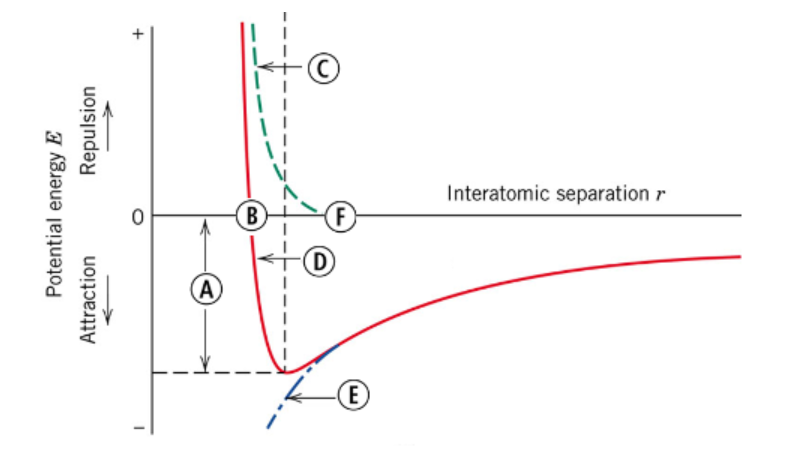
You are given the following potential energy vs interatomic separation bonding curve with critical points labeled A-F. Which lettered feature would represent the equilibrium bond energy? (Q3)
E (WRONG)
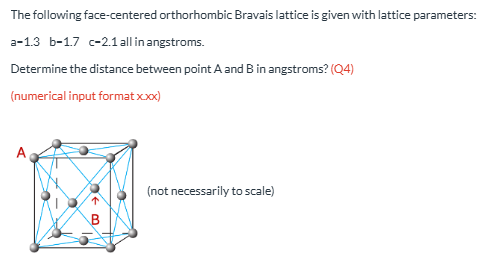
Distance A to B in angstroms. (a=1.3, b=1.7, c=2.1) (Q4)
1.61 WRONG
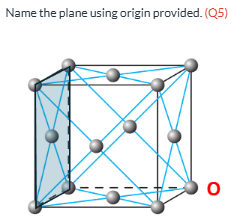
Name the plane (Q5)
(0 1 0)
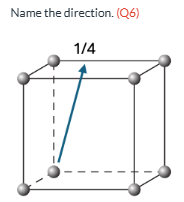
Name the direction (Q6)
[0 4 1] WRONG
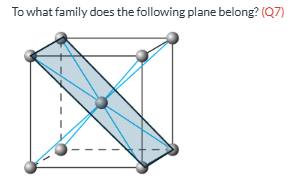
Family of the plane (Q7)
{110}
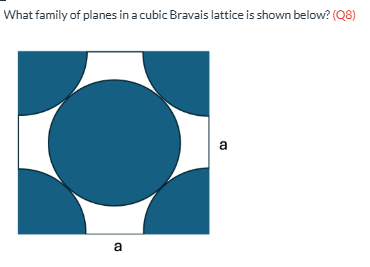
What family of planes in a cubic Bravais lattice is shown below? (Q8)
FCC {111} WRONG
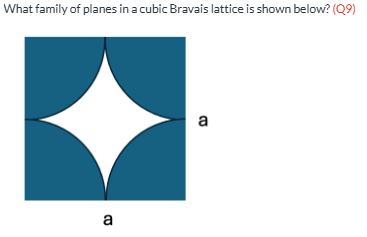
What family of planes in a cubic Bravais lattice is shown below? (Q9)
BCC {110} WRONG

This is the unit cell for the base-centered orthorhombic Bravais lattice. The lattice parameters a,b and c are all different but all the angles in this cell are 90oC. Assuming a basis of 1 atom per lattice point each centered at a lattice point (i.e. 0,0,0), how many atoms are there per unit cell? (Q10)
2
Determine the LPF (linear packing factor) of the [111] direction of a BCC unit cell assuming that the central atom is missing and is replaced by a vacancy. You may assume that the lattice does not distort due to the vacancy. Please put answer in decimal form. (Q11)
0.50
The close-packed planes in the SC metallic crystal structure are? (Q12)
there are no close-packed planes in this structure
Identify the defect(s) that could possibly occur if a few aluminum (Al) atoms were introduced into a copper (Cu) crystal structure? (Q13) MULTIPLE ANSWER
OPTIONS:
Vacancies
Substitutional impurities
Interstitial impurities
Twin planes
Dislocations
Pores or voids
Grain boundaries
Substitutional impurities
For a steel alloy it has been determined that a carburizing heat treatment of 12.9 h duration will raise the carbon concentration to 0.35 wt% at a point 2.3 mm from the surface. Estimate the time (in h) necessary to achieve the same concentration at a 6.8-mm position for an identical steel and at the same carburizing temperature. (Q14)
112.8
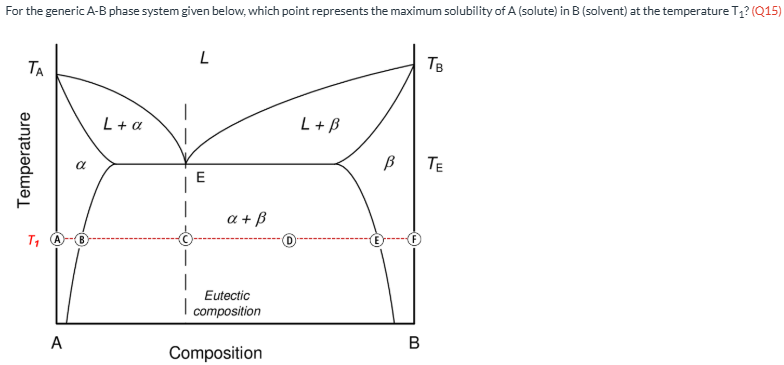
For the generic A-B phase system given below, which point represents the maximum solubility of A (solute) in B (solvent) at the temperature T1? (Q15)
E
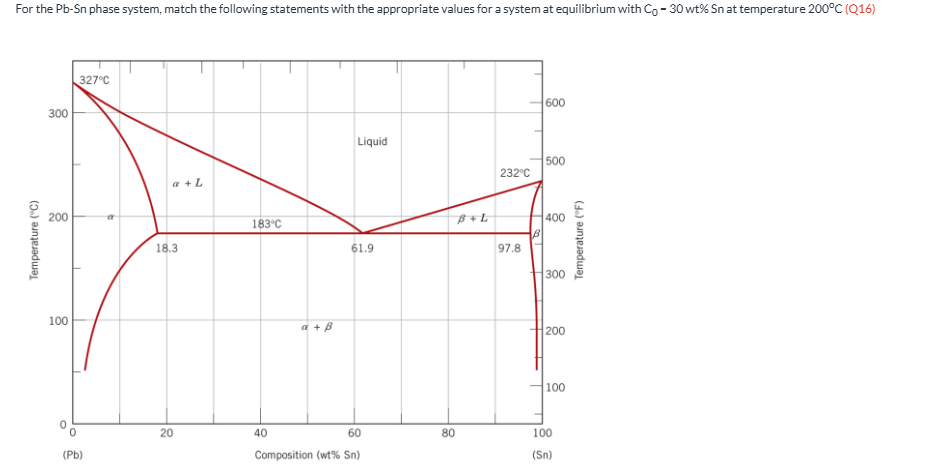
For the Pb-Sn phase system, match the following statements with the appropriate values for a system at equilibrium with C0 = 30 wt% Sn at temperature 200oC (Q16) The alpha phase composition is approximately ____ wt% Sn.
17
For the Pb-Sn phase system, match the following statements with the appropriate values for a system at equilibrium with C0 = 30 wt% Sn at temperature 200oC (Q16) The liquid phase composition is approximately ____ wt% Sn.
56
For the Pb-Sn phase system, match the following statements with the appropriate values for a system at equilibrium with C0 = 30 wt% Sn at temperature 200oC (Q16) The fractional amount of alpha phase is approximately _____%
67%
For the Pb-Sn phase system, match the following statements with the appropriate values for a system at equilibrium with C0 = 30 wt% Sn at temperature 200oC (Q16) The fractional amount liquid phase is approximately _____ %
33%
For the Pb-Sn phase system, match the following statements with the appropriate values for a system at equilibrium with C0 = 30 wt% Sn at temperature 200oC (Q16) The beta phase composition is approximately _____ wt% Sn.
N/A
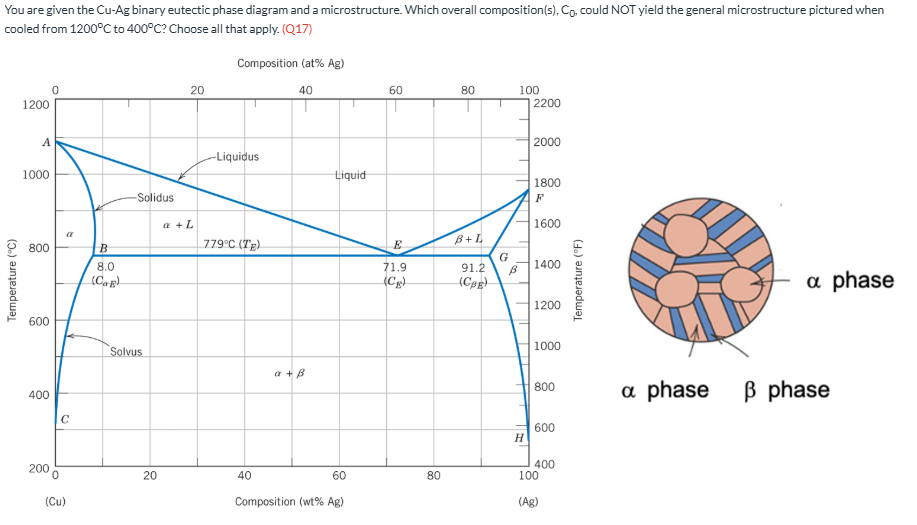
You are given the Cu-Ag binary eutectic phase diagram and a microstructure. Which overall composition(s), C0, could NOT yield the general microstructure pictured when cooled from 1200oC to 400oC? Choose all that apply. (Q17)
0 wt% Ag, 5 wt% Ag, 95 wt% Ag
A cylindrical metal specimen, with diameter of 5.1 mm is placed under a tensile load of 237,456 N. The modulus of the material is 107 GPa. Calculate the strain. (Q18)
0.108
A cylinder 1 m long and 15 mm in diameter is pulled in tension along its axial direction. If the strain in the axial direction is 0.005 and the Poisson's ration for this material is 0.36, what is the change in diameter (in mm). (Q19)
0.027 WRONG
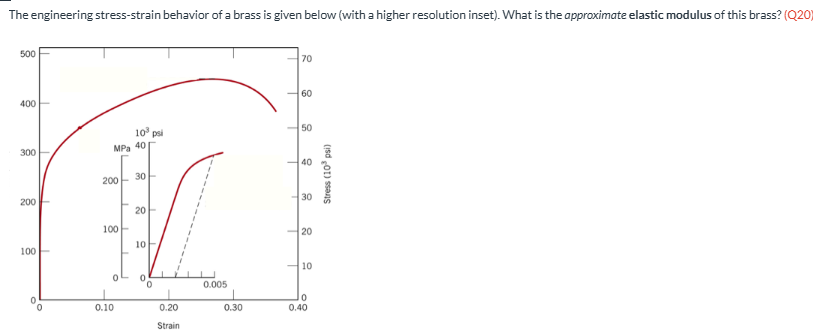
The engineering stress-strain behavior of a brass is given below (with a higher resolution inset). What is the approximate elastic modulus of this brass? (Q20)
100 GPa
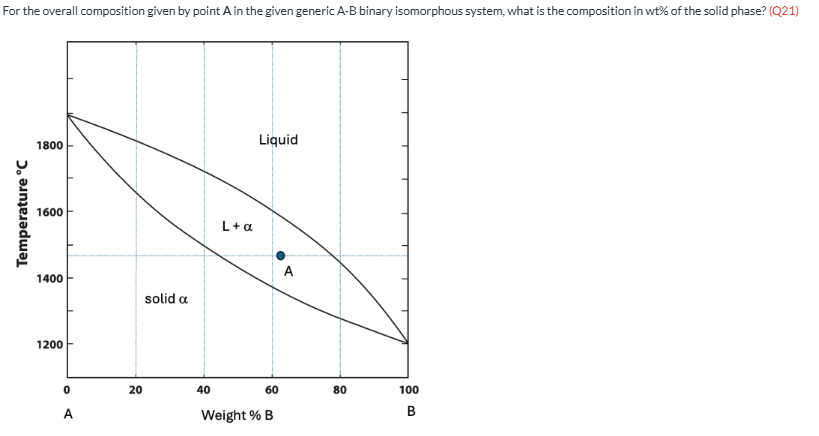
For the overall composition given by point A in the given generic A-B binary isomorphous system, what is the composition in wt% of the solid phase? (Q21)
45 wt%
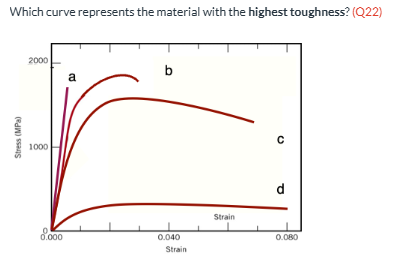
Which curve represents the material with the highest toughness? (Q22) A, B, C, or D
C