CHEM403L: Isolation & Hydrolysis of Casein from Milk
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Amino Acids
These are organic molecules that contain an amino group (--NH2) and a carboxyl group (--COOH).
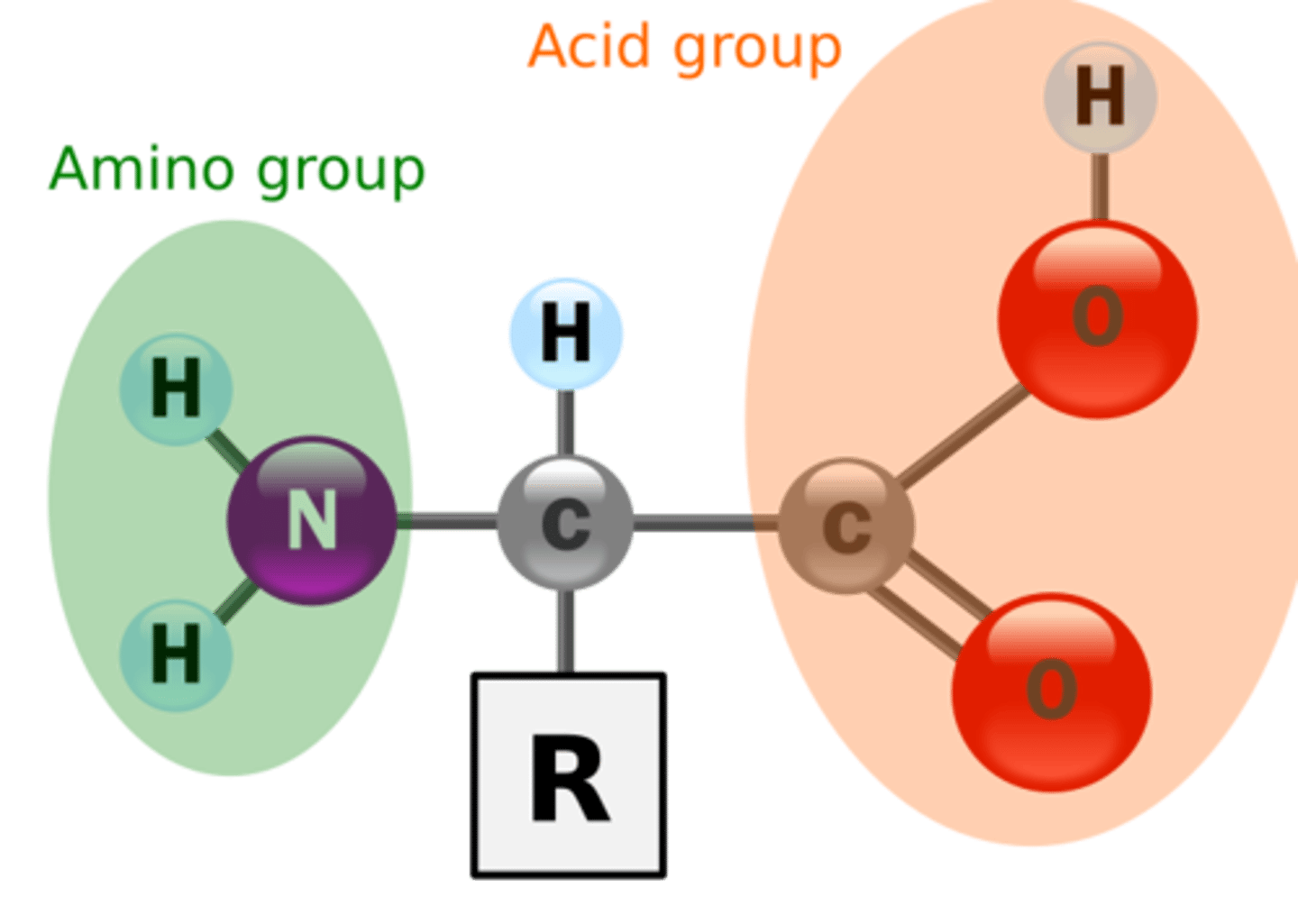
b. carboxyl, a. amino
The [a. amino; b. carboxyl] group is the acidic portion of an amino acid, while the [a. amino; b. carboxyl] group is identified to be basic.
Polar
Nonpolar
Acidic
Basic
Aromatic
The 20 essential amino acids are classified into 5 categories according to their chemical properties. Enumerate these 5 categories.
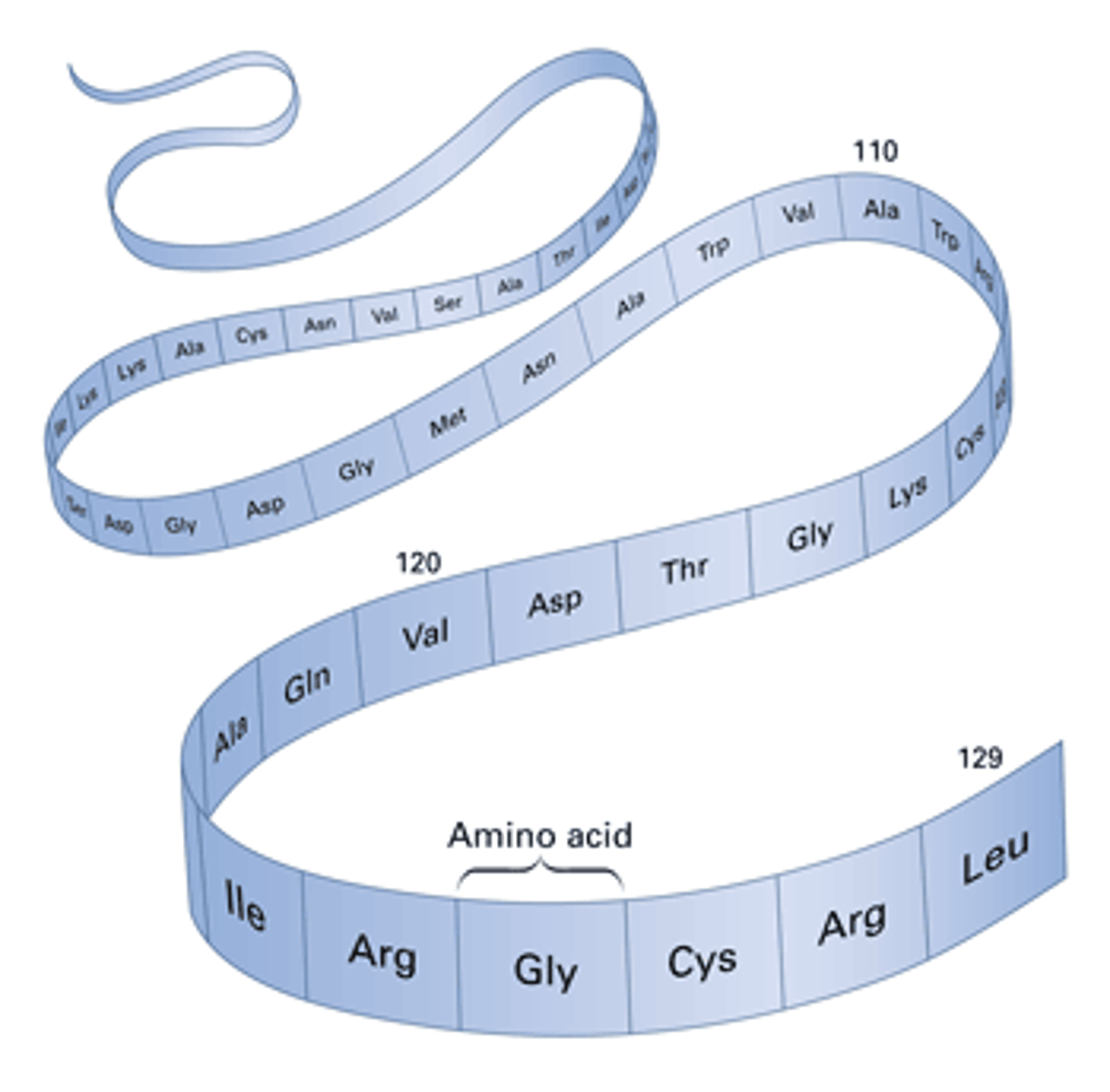
Primary Structure
This type of protein structure is the specific linear sequence of amino acids that make up a polypeptide chain.
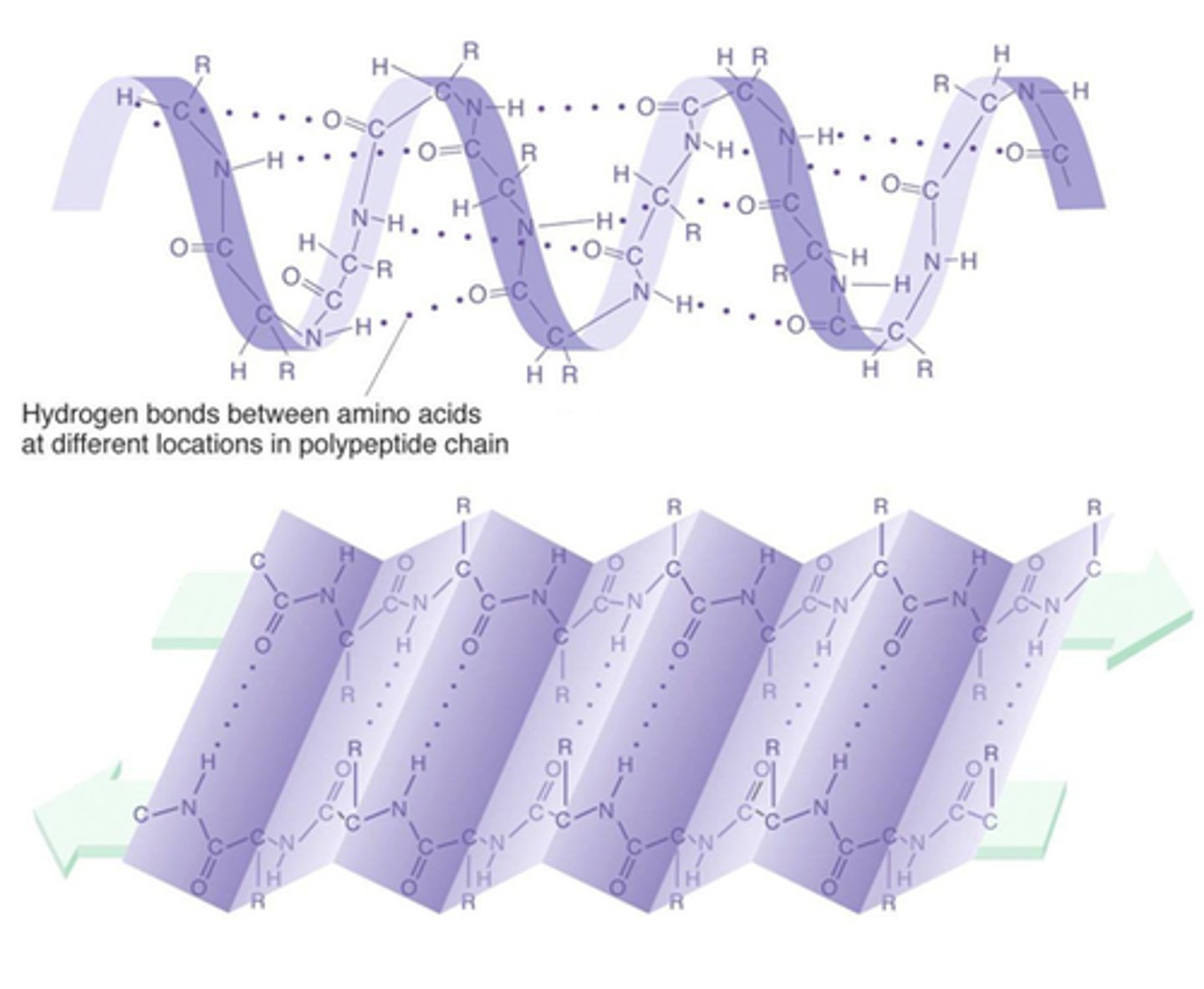
Secondary Structure
This type of protein structure occurs when the sequence of amino acids are linked by hydrogen bonds.
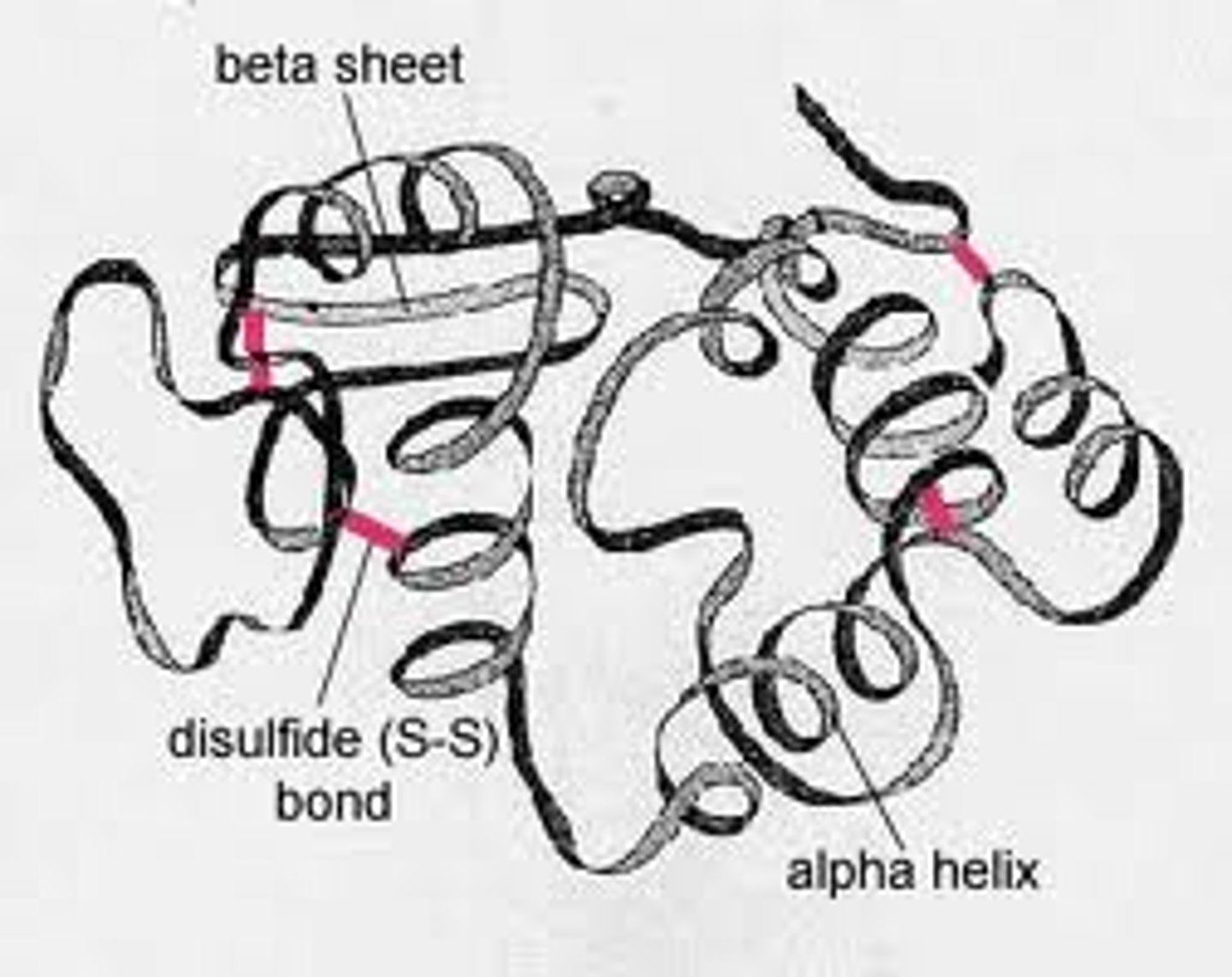
Tertiary Structure
This type of protein structure occurs when certain attractions are present between alpha helices and pleated sheets.
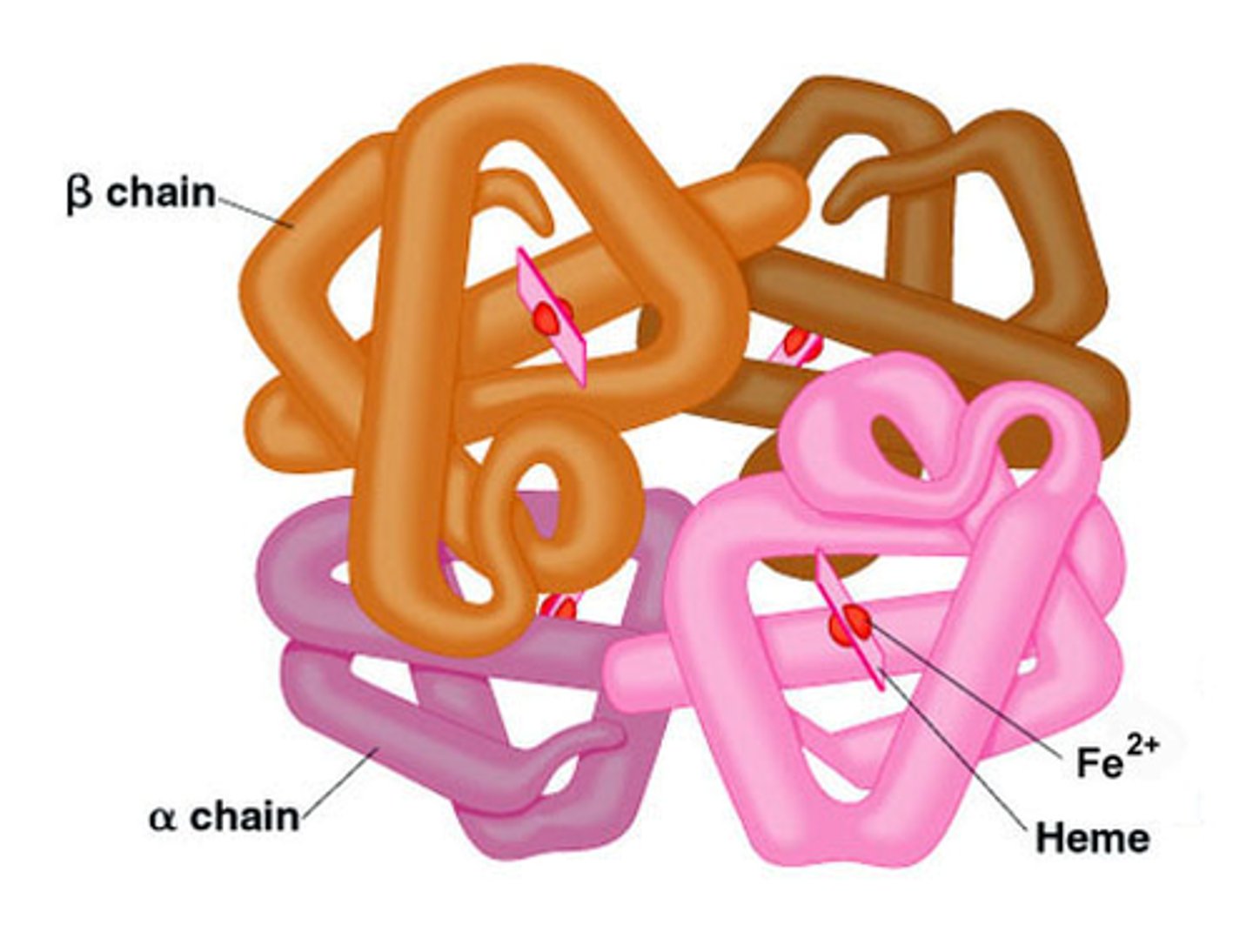
Quaternary Structure
This type of protein structure refers to the arrangement of subunits in a multi-subunit protein. Weak interactions between the subunits help stabilize the overall structure.
Essential Amino Acids
These amino acids cannot be synthesized by the body and must be obtained through diet.
Nonessential Amino Acids
These amino acids can be synthesized by the body
TRUE
TRUE OR FALSE: Alanine and Aspartic Acid are two examples of nonessential amino acids.
Casein
What is the primary protein found in mammalian milk?
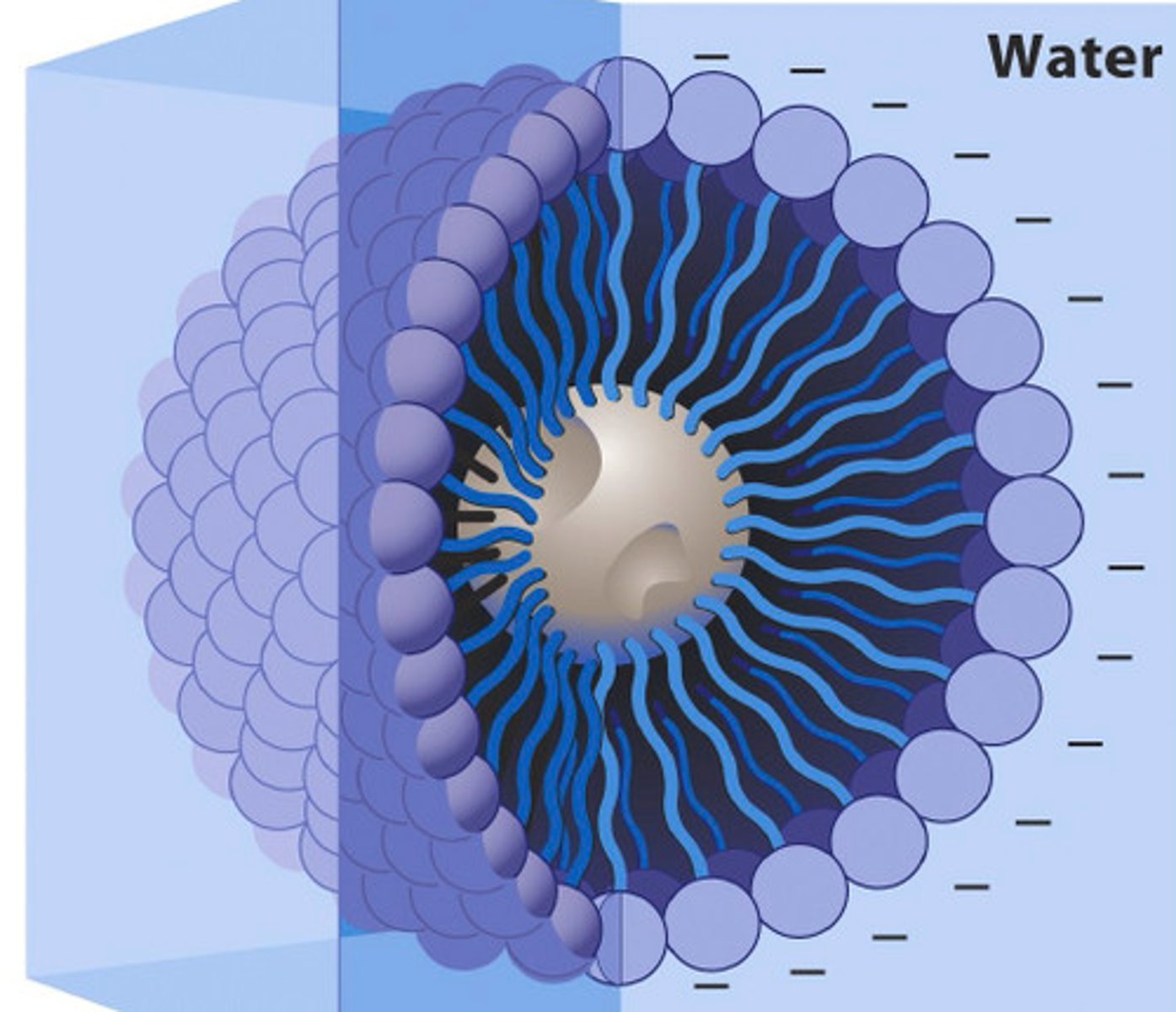
Micelle
This term refers to a small, loosely bound aggregation of surfactant molecules that form a colloidal suspension in a liquid.
Colloid
This term refers to a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance.

a. outside
The hydrophilic region of a micelle is located [a. outside; b. inside] the molecule.
b. inside
The hydrophobic region of a micelle is located [a. outside; b. inside] the molecule
b. polar, a. nonpolar
In a micelle, the hydrophilic region is known to be [a. nonpolar; b. polar] while the hydrophobic region is known to be [a. nonpolar; b. polar].
TRUE
TRUE OR FALSE: Water molecules will be attracted to the hydrophilic region of a micelle.
Zwitterion
This term refers to a molecule that contains both positively and negatively charged functional groups, resulting in an overall net charge of 0.
Whey Protein
This refers to a mixture of proteins derived from a liquid byproduct of cheese production. It is commonly used as a dietary supplement to support muscle growth and recovery.
Isoelectric Point
The ______ is the pH at which a particular molecule (like a protein or amino acid) carries no net electrical charge.
Isoelectric Precipitation
The principle behind this concept includes that at isoelectric point, the protein's net charge becomes neutral, causing it to aggregate and precipitate out of solution because of reduced solubility.
Protein Hydrolysis
This term refers to the process of breaking down proteins into smaller peptides or individual amino acids by adding water. This process is usually catalyzed by enzymes (proteases) or by chemical agents such as acids or bases.
FALSE (water as a base)
TRUE OR FALSE: In acidic hydrolysis, water acts as an acid, accepting hydrogen ions to form hydronium ions (H3O+), which is essential in the breakdown process.
a. strong
In the hydrolytic process, the usage of [a. strong; b. weak] acids and bases are required to effectively cause the breakdown of peptide bonds in proteins into amino acids.
55°C
In the isolation of casein from milk, the solution was heated to a temperature of _____ °C, which was chosen to facilitate the precipitation of casein without causing denaturation of the proteins.
b. H2SO4
Which of the following choices is the strong acid utilized in the Acid Hydrolysis of casein?
a. HNO3
b. H2SO4
c. HBr
d. CH3COOH
c. Ba(OH)2
Which of the following choices is the strong base utilized in the Base Hydrolysis of casein?
a. NaOH
b. KOH
c. Ba(OH)2
d. Ba(OH)
Coagulate
Using milk with fat can interfere with the isolation of casein because fat globules in milk can interact and _______ with casein micelles, contaminating the experimental sample.
Neutralization
This chemical process involves the reaction between an acid and a base to form salt and water, effectively eliminating excess hydrogen or hydroxide ions from the solution. Doing this in solutions can help stabilize amino acids by preventing degradation.
black, orange
After autoclaving, the acid hydrolyzate solution turned color ____ while the base hydrolyzate turned color ______.
b. tryptophan
In Acid Hydrolysis, which amino acid is primarily degraded, leading to the formation of a black color in the hydrolyzate?
a. proline
b. tryptophan
c. phenylalanine
d. tyrosine
Humin
What is the term referring to the black color of the acid hydrolyzate?
d. none of the above
When subjecting the Base Hydrolyzate to colorimetric tests, the following amino acids are absent EXCEPT:
a. cysteine
b. arginine
c. threonine
d. none of the above
15 psi, 120°C, 5 hours
What are the parameters for autoclaving the protein samples?
_____ psi
_____ °C
_____ hours
a. and d.
Which of the following choices describe the purpose of autoclaving the protein samples?
a. eliminate unwanted microbes in the sample
b. enhance protein solubility by denaturing proteins
c. remove volatile compounds that interfere with analysis
d. speed up the process of hydrolysis
a. complete
Both acid and base hydrolysis are considered to be [a. complete; b. partial hydrolysis.
Partial Hydrolysis
This type of hydrolysis involves the incomplete breaking of chemical bonds using water, resulting in smaller fragments rather than fully breaking down the molecule into its simplest components.
Complete Hydrolysis
This type of hydrolysis refers to the complete breakdown of a molecule into its simplest components using water.
TRUE
TRUE OR FALSE: After acid and base hydrolysis, you will expect to obtain free amino acids in the experimental sample.
b. 4.6
At which pH level does the protein casein form a zwitterion?
a. 3.4
b. 4.6
c. 5.1
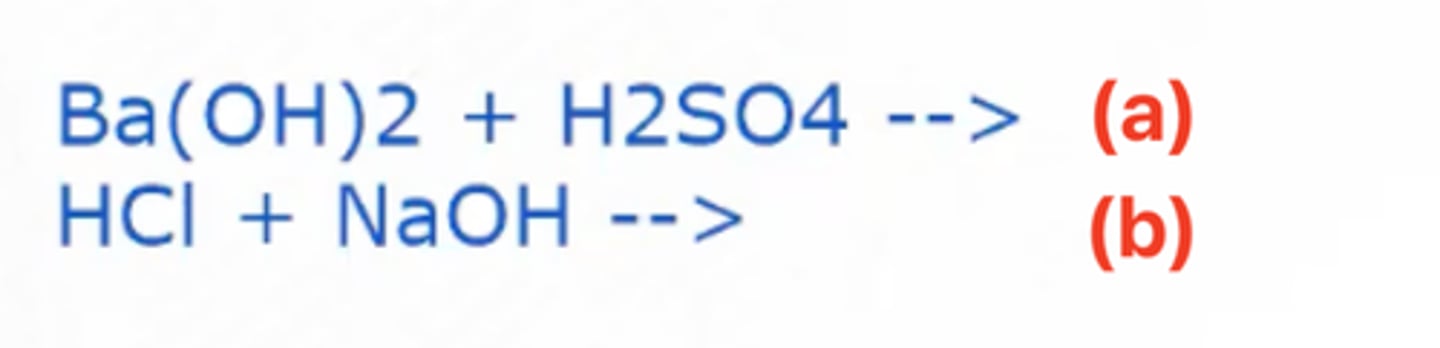
BaSO4 (Barium Sulfate) + H2O (Water)
Given the chemical reaction in the image, what are the products formed in (a)?
NaCl (Sodium Chloride) + H2O (Water)
Given the chemical reaction in the image, what are the products formed in (b)?
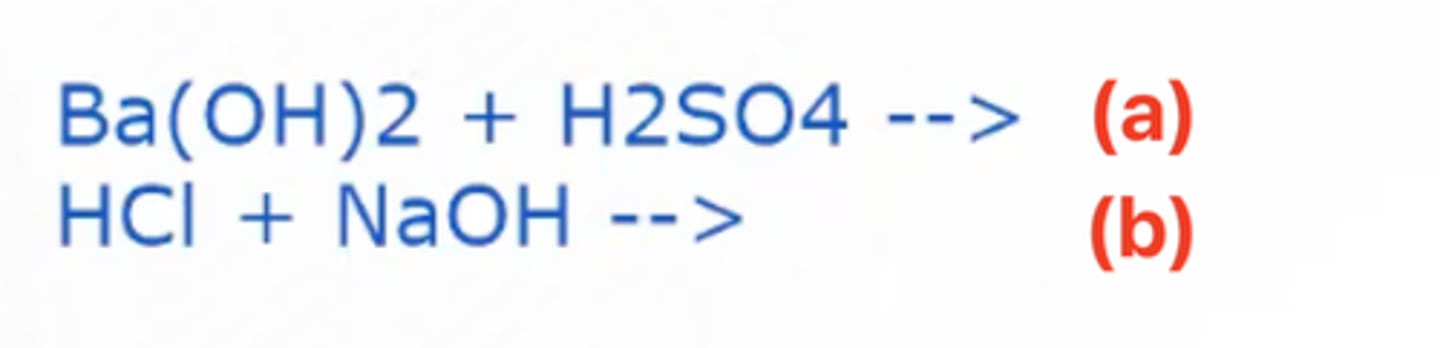
b. HCl and NaOH forms a soluble salt in water after the neutralization reaction
Why is the usage of Ba(OH)2 and H2SO4 much more preferred in the experiment rather than other strong acids and bases such as HCl and NaOH?
a. Ba(OH)2 provides a higher pH range for protein stability
b. HCl and NaOH forms a soluble salt in water after the neutralization reaction
c. Ba(OH)2 has stronger buffering capacity compared to NaOH
d. Sulfuric acid (H2SO4) is less corrosive than hydrochloric acid (HCl)
a. carboxyl group, amine group, alcohol group
Which 3 functional groups are mainly formed after a hydrolysis reaction?
a. carboxyl group, amine group, alcohol group
b. amide group, carboxyl group, alcohol group
c. carboxyl group, amine group, aldehyde group
d. amine group, ketone group, alcohol group