2.1.2 k-o PROTEINS
1/20
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
21 Terms
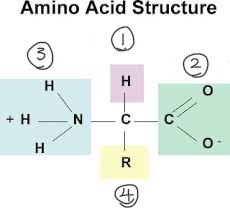
k) Draw and label a diagram of an amino acid.
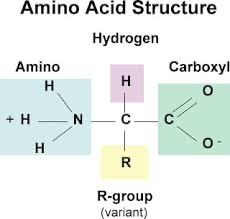
k) Identify the part of an amino acid which is variable.
R-group
k) State how many amino acids occur in life.
20
k) Describe the different types of amino acid in life.
nonpolar hydrophobic
polar hydrophilic
polar hydrophilic basic (positively charged)
polar hydrophilic acidic (negatively charged)
l) Draw a labelled diagram demonstrating the condensation and hydrolysis of peptide bonds.
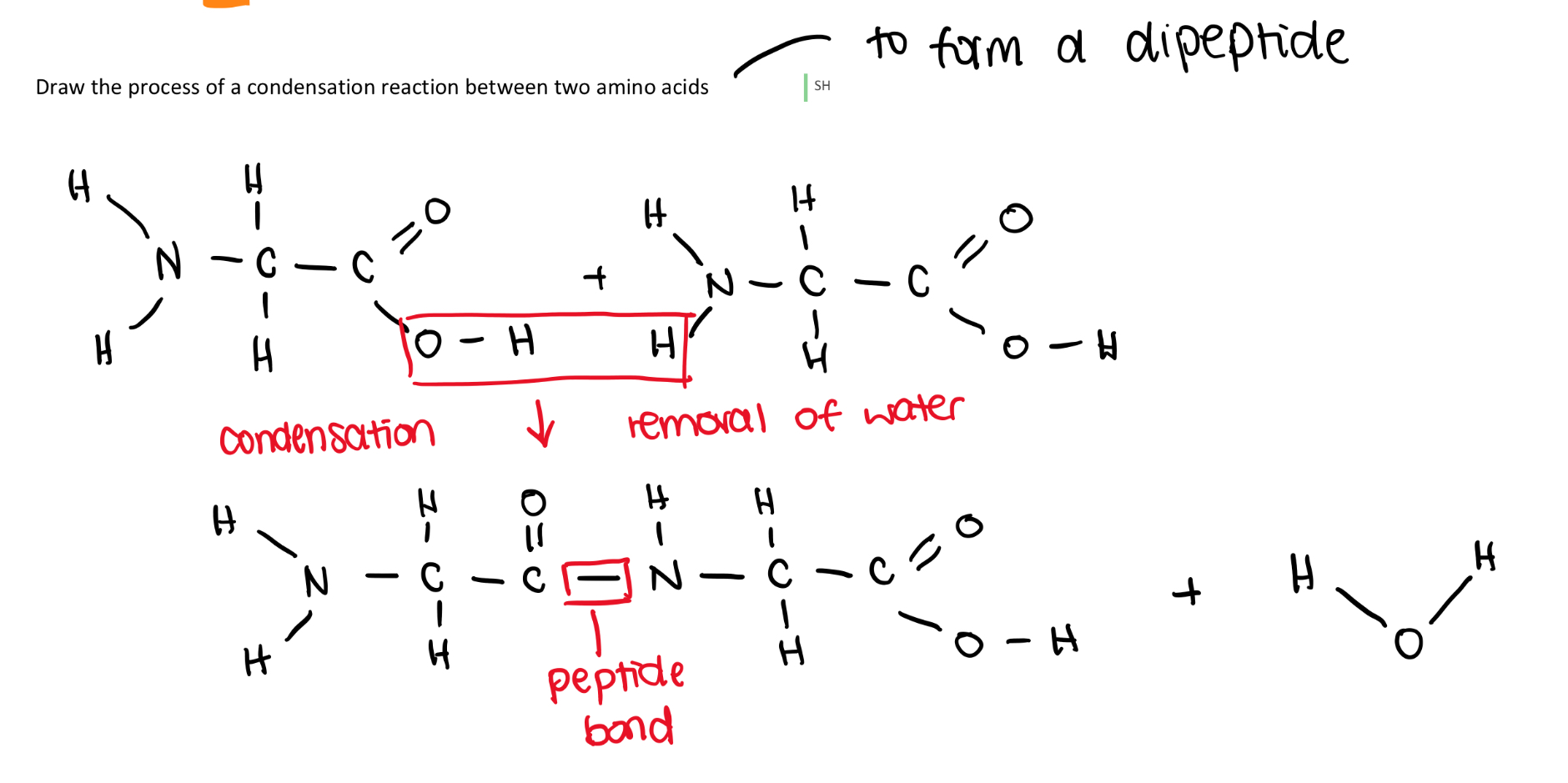
l) Explain how the variety of amino acids leads to a wide range of dipeptides and very quickly to an incredible variety of polypeptide chains.
20 amino acids therefore 400 dipeptides as the order does change the molecules.
There are 20³ tripeptides and so on giving rise to incredible variation in polypeptides.
l) Define the term ‘polypeptide chain’
chains of three or more amino acids
l) Define the term ‘protein’.
one or more polypeptide arranged as a macromolecule
l) Describe how one end of a polypeptide chain differs from the other end.
One end is the C terminal which is the carboxyl end and the other is the N terminal, the amine group.
m) Define the term ‘primary structure’ of a protein and describe how it is held together.
The number and sequence of amino acids in a polypeptide is its primary structure. Each amino acid is bonded to the next by peptide bonds.
m) Define the term ‘secondary structure’ of a protein and describe how it is held in place.
All or part may fold into a secondary structure most commonly and alpha-helix or a beta-pleated sheet.
To produce one of these there needs to be a regular repeating pattern of amino acids.
The structures are held together by h-bonds between N-H groups and C=Os.
m) Define the term ‘tertiary structure’ of a protein and describe how it is held in place.
The compact 3D shape adopted by most polypeptides. These molecules are called globular (near-spherical) proteins.
The R groups form the bonds that hold the structure in place.
H-bonds form between slightly charged R groups
Ionic bonds form between R groups carrying opposite charges
Disulphide bonds or bridges from between the sulphur containing R groups of 2 cysteine amino acids - strong covalent bonds
Hydrophobic interactions - protein folds so that hydrophobic R groups lie in the centre of the molecule away from the aqueous environment.
m) Define the term ‘quaternary structure’ of a protein and describe how it is held in place.
A protein with quaternary structure contains 2 or more polypeptide chains bonded together which have no function on their own and only form a functional molecule when bonded.
Bonds that hold tertiary structure also hold quaternary; ionic bonds, disulphide bridges and hydrophobic interactions
n) Define the term ‘globular protein’.
near spherical, water soluble proteins
n) Define the term ‘fibrous protein’.
long, insoluble structural proteins
n) Define the term ‘prosthetic group’.
non-protein component of a conjugated protein
n) Define the term ‘conjugated protein’.
a protein that is joined to a non-protein group (prosthetic group)
n) Draw a table to show an example of a conjugated protein, an enzyme and a peptide hormone.
*identify key structural components, properties and functions.
haemoglobin - each of the 4 polypeptide chains is bonded to a haem group. It carries oxygen.
Catalase - quaternary protein with 4 haem groups. The presence of iron II ions allows it to catalyse break down of hydrogen peroxide which is a harmful waste product of metabolism
Antidiuretic hormone - regulates the body’s retention of water by increasing water absorption of kidney’s collection duct
o) Give 3 examples of fibrous proteins.
keratin
elastin
collagen
o) Compare the structure, properties and functions of collagen, keratin and elastin.
collagen - connective tissue found in skin, tendons, ligaments and the nervous system - 4 polypeptides wound round each other to form a rope like structure
keratin - hair, skin and nails, many sulphur containing amino acids means many disulphide bridges which make it very strong - e.g. nails more disulphide bridges, making them less flexible
elastin - found in elastic fibres in blood vessels and alveoli allowing them to expand and return to normal as needed. Made by linking many soluble tropoelastin protein molecules which act as little springs together to form an insoluble macromolecule.
o) Compare the structure, properties and functions of globular and fibrous proteins.
Globular - spherical and water soluble due to folding so that hydrophobic regions are inside and hydrophilic regions are on the outside.
Fibrous - long, strong, insoluble molecules, tend to contain a limited range of amino acids with small R groups and arranged in a pretty regular pattern containing many hydrophobic amino acids.