Collision Theory, Energy & Reactions Test
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What are the 3 conditions needed for a reaction to occur? (collision theory)
1) Reactants must collide
2) The collisions must have sufficient energy
3) The reactants must be in the proper orientation during the collisions
What are the 4 signs of a chemical reaction?
1) Color change 2) Gas production 3) Temperature change 4) Precipitate formation
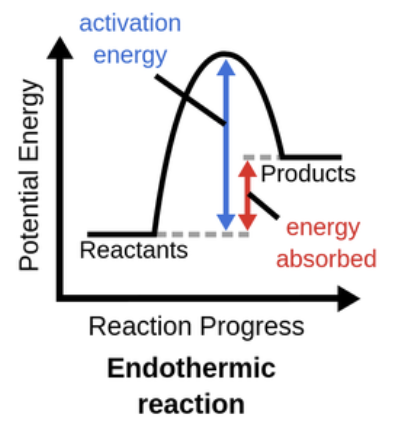
Practice drawing and labeling an endothermic graph.
Practice drawing and labeling an exothermic graph.
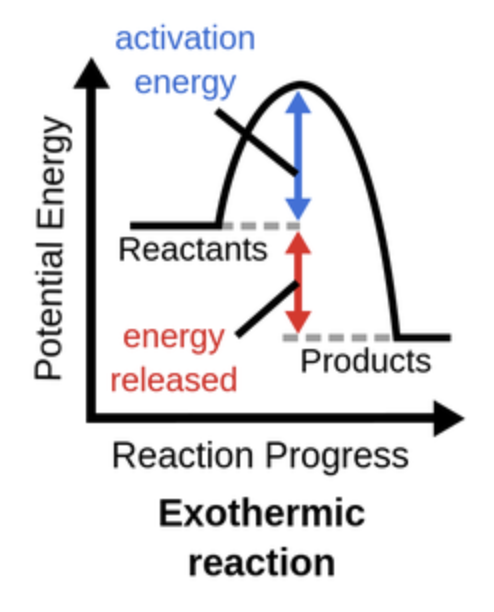
What is activation energy?
The minimum energy required for a chemical reaction to occur, allowing reactants to transform into products.
What is a catalyst?
A substance that increases the rate of a chemical reaction by lowering the activation energy without being consumed in the process.
What is an endothermic reaction?
Any process which requires/absorbs energy from its surroundings, usually in the form of heat (creating lower surrounding temp.) (e.g photosynthesis)
What is an exothermic reaction?
A chemical reaction that releases energy into it’s surroundings, usually in the form of heat or light.
What factor could increase the energy in collision theory and why?
Increasing the temperature could increase the energy of the particles, leading to more frequent and energetic collisions.
What factor could increase the correct orientations in collision theory and why?
Agitating the reaction mixture can help particles collide with the correct orientation, facilitating successful reactions.
What factor could increase collisions in collision theory?
Decreasing the volume of the container or increasing the concentration of particles could increase collisions.
What factor increases all three conditions of collision theory?
Agitating the particles
What are the rules for naming acids? (binary and oxy)
Binary Acids (No Oxygen): Use the format hydro- [root] -ic acid.
Example: 𝐻𝐶𝑙 = Hydrochloric acid.
Oxyacids (Contain Oxygen): Based on the polyatomic ion ("-ate" or "-ite").
If the anion ends in -ate, replace it with -ic acid 𝑆𝑂2−4 → Sulfuric acid).
If the anion ends in -ite, replace it with -ous acid (𝑆𝑂2−3 → Sulfurous acid).
What does a synthesis equation look like?
A + X —> AX
Ex: H2 (g) + O2 (g) —> H2O
A reaction where two or more reactants combine to form a single product.
What does a decomposition reaction look like?
AX —> A + X
Ex: HgO(s) —> Hg(l) + O2 (g)
A reaction in which a single reactant breaks down into two or more simpler substances.
What happens in a decomposition reaction with carbonate ions?
Carbonates break into metal oxide and carbon dioxide
Ex: ZnCO3(s) —> ZnO(l) + CO2 (g)
What happens in a decomposition reaction with chlorate ions?
Chlorates break into metal chloride and oxygen
Ex: Ni(ClO3)2(s) —> NiCl2(l) + O2 (g)
What happens in a decomposition reaction with hydroxide ions?
Hydroxides break into metal oxide and water
Ex: Fe(OH)3(s) —> Fe2O3(l) + H2O (g)
What does a combustion reaction look like?
CxHy + O2 —> CO2 + H2O
A substance combines with oxygen, releasing a large amount of energy in the form of heat and light. Products are water and carbon dioxide.
What does a single displacement reaction look like?
One element replaces a similarly charged element in a compound.
A + BX —> AX + B
Element + compound —> Element + compound
What does a double displacement reaction look like?
Similar ions of two compounds exchange places in an aqueous solution.
AX + BY —> AY + BX
Compound + compound —> Compound + compound
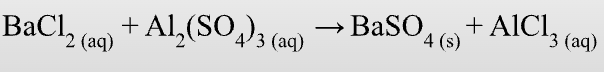
What does a neutralization/Acid-Base reaction look like?
A hydrogen ion is donated to a hyrdoxide to create water.
Acid + base —> H2O + salt
How do you predict if a reaction will occur based on an activity series?
Reaction predictions are made by comparing the reactivity of the elements involved; if a more reactive element displaces a less reactive one, the reaction will occur.
Remember more, then less.