3. Atomic Structure
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
proton relative charge
+1
neutron relative charge
0
electron relative charge
-1
relative mass of proton
1
relative mass of neutron
1
relative mass of electron
1/1840
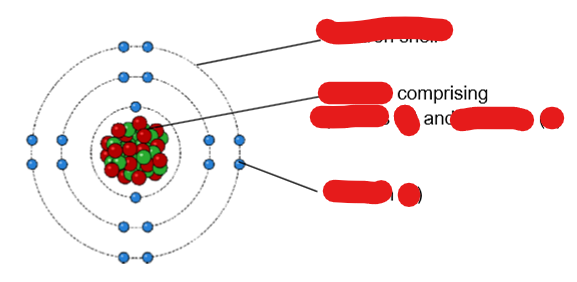
fill in blanks
electron shell nucleus protons (p) neutrons (n) electron (e)
for first _ elements, valence shell can only hold _ electrons. After that, valence shell can hold ___ electrons
20 8 18
atoms are _________ _________ (no. of ________ = no. of __________)
electrically neutral protons electrons
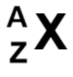
what is A? What is Z?
nucleon no., proton no.
what are isotopes? (isotopes are _____ of the _____ element with the _______ number of protons, but ___________ number of neutrons.)
isotopes are atoms of the same element with the same number of protons, but different number of neutrons
isotopes have _______ __________ properties (due to ______ number of __________)
same chemical same electrons
isotopes have _______ __________ ___________ properties (due to ______ number of __________)
slightly different physical different neutrons
ions' no. of protons and no. of electrons are ________
different
what makes an ion positive (cation)? (______ protons than electrons)
more protons than electrons
what makes an ion negative (anion)? (______ protons than electrons)
less protons than electrons
defenition of relative atomic mass Ar (the _________ mass of ____ _____ of an element compared to /_ the mass of an _____ of carbon-12)
average one atom 1/12 atom
Ar is to ____ decimal place
one
For elements to have similar chemical properties, they are in the same _______ and have the _____ __________ of _______ ______
group same number valence electrons
electronic structure of atom: ________ of atom and ________ of _________
identity number electrons
isoelectronic: have the _____ ______ ________ of _______
same total number electrons