CHEM 3438 Exam 3
1/106
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
107 Terms
The heme in hemoglobin is held in the enzyme (in part) by a ligand interaction between the heme iron and
a. a leucine in the protein
b. a disulfide bond
c. an aspartate in the protein
d. an alanine in the protein
e. a histidine in the protein
e. a histidine in the protein
If oxygen leaves hemoglobin and takes an electron with it, oxidizing the iron(II) to iron(III),
a. the oxygen becomes superoxide
b. the oxygen becomes water
c. the oxygen becomes hydrogen peroxide
d. this can't happen, oxygen can't gain or lose electrons
e. this can't happen, the oxidation state of iron in hemoglobin isn't iron(II)
a. the oxygen becomes superoxide
Myoglobin's primary role in the body is to
a. transport oxygen from the lungs to respiring tissues
b. bind superoxide to prevent its toxicity
c. bind toxins like carbon monoxide and cyanide
d. prevent hemoglobin from aggregating
e. hold a reserve of oxygen for muscle that is employed when oxygen levels become dangerously low
e. hold a reserve of oxygen for muscle that is employed when oxygen levels become dangerously low
Fetal hemoglobin
a. does not bind oxygen
b. is severely affected by sickle cell disease
c. is monomeric
d. has a higher oxygen affinity than adult hemoglobin
e. is completely unnecessary, since the fetus does not inhale air
d. has a higher oxygen affinity than adult hemoglobin
A sample of blood proteins is separated on a 2D-gel. If the patient is young enough to express significant amounts of both fetal and adult hemoglobin, and fetal hemoglobin has less positive charges than adult hemoglobin but the sizes are nearly identical, where would they be found on the gel relative to one another?
a. At the same position in the isoelectric focusing dimension, but fetal hemoglobin will be found higher on the gel in the SDS-PAGE dimension
b. At the same position in the isoelectric focusing dimension, but fetal hemoglobin will be found lower on the gel in the SDS-PAGE dimension
c. At the same position in the SDS-PAGE dimension, but fetal hemoglobin will be found at lower pH in the isolectric focusing dimension
d. At the same position in the SDS-PAGE dimension, but fetal hemoglobin will be found at higher pH in the isolectric focusing dimension
e. The will be in the exact same spot
c. At the same position in the SDS-PAGE dimension, but fetal hemoglobin will be found at lower pH in the isolectric focusing dimension
The T-state of hemoglobin binds oxygen more tightly than the R-state.
a. True
b. False
b. False
2,3-BPG modulates the oxygen affinity of the hemoglobin tetramer (shown) by binding
a. in the center of the tetramer, where it can contact all of the subunits
b. on the proximal side of the heme, near the oxygen binding site
c. on the distal side of the heme, near the histidine that binds to the heme iron
d. BPG does not bind to hemoglobin, it modulates oxygen affinity only indirectly
a. in the center of the tetramer, where it can contact all of the subunits
A major benefit of the sigmoid oxygen binding curve (oxygen saturation versus partial pressure of oxgyen) for hemoglboin is that
a. it increases the amount of oxygen transferred from lung to oxygen deprived tissue
b. the hemoglobin is never saturated an any oxygen pressure
c. it decreases the amount of oxygen picked up in the lung
d. it decreases the amount of oxygen delivered to oxygen deprived tissue
e. it increases the amount of carbon dioxide picked up in the lung
a. it increases the amount of oxygen transferred from lung to oxygen deprived tissue
On the structure of a hemoglobin tetramer, mark the site where 2,3-BPG binds. If there is more than one major site (i.e. four related by the tetramer's symmetry) then pick any one of the major sites.
Top Left Coordinates(116, 151)Bottom Right Coordinates(231, 277)
According to the figure above, which of the following statements are true? (Pick the single best answer.)
a. Hemoglobin is normally purple, which is a mix of blue R-state and red T-state.
b. The T-state binds oxygen more strongly than the R-state.
c. Hemoglobin binds oxygen more tightly than either pure T-state or pure R-state would.
d. At low oxygen concentrations, hemoglobin starts out mostly in the T-state but as oxygen increases, the hemoglobin becomes mostly like the R-state.
e. A low oxygen, hemoglobin in mostly in the R-state but as oxygen increases, the hemoglobin mostly becomes the T-state.
d. At low oxygen concentrations, hemoglobin starts out mostly in the T-state but as oxygen increases, the hemoglobin becomes mostly like the R-state.
In the MWC model of cooperativity as applied to hemoglobin, at very low oxygen pressure
a. the alpha subunits are in the T state but the beta subunits are in the R state
b. the alpha subunits are in the R state but the beta subunits are in the T state
c. hemoglobin sickles
d. the T state is strongly favored
e. the R state is strongly favored
d. the T state is strongly favored
The binding of 2,3-bisphosphoglycerate (2,3-BPG) to hemoglobin
a. lowers the affinity for oxygen
b. competes with oxygen as a ligand for the heme iron
c. changes the pKa of the proximal histidine
d. causes sickling of the hemoglobin
e. raises the affinity for oxygen
a. lowers the affinity for oxygen
One of the benefits of allosterism in hemoglobin, which results in a sigmoid oxygen binding curve,is that
a. there is no advantage, it is an artifact of evolution
b. there is maximal change in oxygen binding between the lung oxygen pressure and the typical(or active) tissue oxygen pressure
c. it allows hemoglobin to function as a monomer, saving the energy cost of maintaining andexpressing multiple genes
d. hemoglobin is able to bind to oxygen more tightly than myoglobin at tiss ue oxgyenconcentrations, saving oxygen for other tissues
e. oxygen binds more strongly to hemoglobin at all oxygen pressures than if it wasn't allosteric
b. there is maximal change in oxygen binding between the lung oxygen pressure and the typical(or active) tissue oxygen pressure
Carbon dioxide binds to hemoglobin as a
a. cationic group
b. heme ligand
c. carbamate
d. bicarbonate
e. alcohol
c. carbamate
Which one of the following will cause hemoglobin to release oxygen?
a. increase in O2 concentration
b. increase in pH
c. decrease in CO2 concentration
d. decrease in temperature
e. decrease in pH
e. decrease in pH
At low pH, the affinity of hemoglobin for oxygen
a. is not different than at high pH
b. increases
c. decreases
c. decreases
This graph of hemoglobin oxygen saturation clearly shows that
a. hemoglobin has a lower affinity for oxygen when the pH is low or the carbon dioxide concentration is high
b. hemoglobin has a lower affinity for oxygen when the pH is high or the carbon dioxide concentration is high
c. hemoglobin has a lower affinity for oxygen when the pH is low or the carbon dioxide concentration is low
d. hemoglobin has a lower affinity for oxygen when the pH is high or the carbon dioxide concentration is low
a. hemoglobin has a lower affinity for oxygen when the pH is low or the carbon dioxide concentration is high
Sickled red blood cells take that shape because of long rods of polymerized hemoglobin.
a. True
b. False
a. True
Hemoglobin mainly polymerizes from the oxygenated form.
a. True
b. False
b. False
During a sickle cell disease 'crisis'
a. hemoglobin dissociates into monomers
b. hemoglobin polymerizes into fibers
c. hemoglobin saturates with oxygen
b. hemoglobin polymerizes into fibers
In thalassemia, faulty hemoglobin results in the destruction of red blood cells.
a. True
b. False
a. True
Sickle cell disease is prevalent in some parts of the world because it
a. provides some protection against malaria
b. only becomes an issue when a person overheats
c. thrives better in tropical climates
d. is caused by a pathogen that thrives in salt water
e. it is transmitted to cannibals that eat the brains of their ancestors
a. provides some protection against malaria
When a cofactor is a small organic molecule, it is known as a(n)
a. vitamin.
b. isoenzyme.
c. regulator.
d. zymogen.
e. coenzyme.
e. coenzyme
Minerals are inorganic ________________________.
a. cofactors
b. proteins
c. vitamins
d. macronutrients
a. cofactors
Metal ions such as Zn 2+ and Fe 3+ are often needed by enzymes as
a. inhibitors.
b. isozymes.
c. substrates.
d. cofactors.
e. allosteres.
d. cofactors
Coenzymes such as water-soluble vitamins are needed in only small amounts because
a. only small amounts of enzymes are present in each cell.
b. each vitamin molecule can be reused many times as a cofactor.
c. only small amounts of substrates are available at any one time.
d. they can be eliminated in the urine.
e. they can be stored in the liver for future use.
b. each vitamin molecule can be reused many times as a cofactor.
Of the following, __________ will lower the activation energy for a reaction.
a. raising the temperature of the reaction
b. increasing the concentrations of reactants
c. adding a catalyst for the reaction
d. removing products as the reaction proceeds
e. increasing the pressure
c. adding a catalyst for the reaction
The most general description of how enzymes catalyze chemical reactions is that they
a. change the free energy change for the reaction
b. bind substrates tightly
c. bind products tightly
d. change equilibrium constants
e. lower activation energy
e. lower activation energy
The active site of an enzyme
a. is remote from the site of substrate attachment.
b. catalyzes the reaction.
c. includes the entire enzyme.
d. increases the energy of reaction.
e. is converted to a product.
b. catalyzes the reaction.
A catalyst can increase the rate of a reaction __________.
a. by providing an alternative pathway with a lower activation energy
b. by lowering the overall activation energy (Ea) of the reaction
c. by lowering the activation energy of the reverse reaction
d. by changing the value of the frequency factor (A)
e. All of these are ways that a catalyst might act to increase the rate of reaction.
a. by providing an alternative pathway with a lower activation energy
The rate of a reaction depends on __________.
a. collision frequency
b. collision orientation
c. collision energy
d. all of the above
e. none of the above
d. all of the above
The most important result of the Third Law of Thermodynamics (at least for our purposes in this class) is
a. a definition of the heat capacity
a. a definition of the heat capacity
Which of the following is one of the 'laws' of thermodynamics?
a. The total energy of an isolated system is conserved
a. The total energy of an isolated system is conserved
In the derivation of the Michaelis-Menten Equation for a noncompetitive inhibitor, the mass balance equation for substrate (the full version of the equation before making any simplifying assumptions) is
a. St = [E] + [ES] + [ESI]
b. St = [S] + [ES] + [ESI] + [P]
c. St = [ES] + [E]
d. St = [S] + [E]
e. St = [S] + [I] + [ESI]
b. St = [S] + [ES] + [ESI] + [P]
When deriving the Michaelis-Menten equation, we make an assumption that at time zero (t=0), the amount of product is zero ([P]=0).
a. True
b. False
a. True
In the derivation of the Michaelis-Menten Equation, the mass balance equation for enzyme is
a. Et = [E] + [ES]
b. Et = [S] + [ES] +[P]
c. Et = 2[ES] + [E]
d. Et = [S] + [E]
e. Et = 0
a. Et = [E] + [ES]
The steady-state assumption is that [ES] = 0.
a. True
b. False
b. False
In the derivation of the Michaelis-Menten Equation, we make an assumption of 'steady state'. This assumption is
a. d[S]/dt=0 ([S] doesn't change)
b. [P] <<< [S] ([P] is much smaller than [S])
c. [ES] = all of the enzyme we put into the reaction
d. that no product is formed
e. d[ES]/dt = 0 ([ES] doesn't change)
e. d[ES]/dt = 0 ([ES] doesn't change)
In Michaelis-Menten kinetics, if [S] = K M, then the rate of reaction v =
a. 2 * Vmax
b. 0
c. 0.5 * Vmax
d. k1 k2 [ES]
e. Vmax
c. 0.5 * Vmax
The HIV protease inhibitor Indinavir inhibits the enzyme because it assembles the normal peptide substrate of HIV protease. Indinavir is a
a. competitive inhibitor
b. noncompetitive inhibitor
c. suicide inhibitor
d. uncompetitive inhibitor
e. group specific agent/inhibitor
a. competitive inhibitor
A fatty acid hydroxylase enzyme has a Km for palmitic acid of 5.81 micromolar and a turnover rate of 1,385.8 micromole substrate per minute per micromole enzyme. If 75.5 nanomolar enzyme is mixed with an unknown concentration of substrate and the reaction rate was 48.5 micromolar substrate per minute, how much substrate was in the reaction to start with? Report your answer in micromolar to the nearest 0.1 uM.
5
You are studying a new enzyme and want to determine Km and Vmax. You collect your initial rate data at multiple substrate concentrations, take the reciprocal of v and s, and do a Lineweaver-Burke plot. If the slope of the resulting line is 0.0245 micromolar minutes and the y-intercept is 0.0025 per min, what is the Km of this enzyme for its substrate? Report your answer in micromolar to the nearest 0.1 uM.
9.8
Vmax is
a. the rate of an enzyme reaction at extremely high substrate concentrations
b. the rate of an enzyme reaction at extremely low substrate concentrations
c. dependent on the rate of substrate binding to the enzyme, k1
d. the maximal amount of substrate that can be bound to one molecule of enzyme
e. independent of the enzyme concentration
a. the rate of an enzyme reaction at extremely high substrate concentrations
Enzyme A obeys simple Michaelis-Menten kinetics. If the rate of reaction under a given set of conditions is 9 uM/min and the concentration of substrate is doubled, the new initial rate would be
a. 27 uM/min
b. There is not enough information to determine the answer
c. 3 uM/min
d. 18 uM/min
e. 81 uM/min
b. There is not enough information to determine the answer
Which of the following inhibitors can be overcome by increasing substrate concentration?
a. competitive
b. noncompetitive
c. uncompetitive
d. none of the above
a. competitive
Enzymes speed up chemical reactions by
a. lowering the equilibrium constant
b. raising the equilibrium constant
c. lowering the activation energy
d. raising the activation energy
e. magic
c. lowering the activation energy
A compound that binds to the surface of an enzyme, and changes its shape so that a substrate cannot enter the active site, is called a(n)
a. proenzyme.
b. noncompetitive inhibitor.
c. cofactor.
d. irreversible inhibitor.
e. competitive inhibitor.
b. noncompetitive inhibitor.
"For an uncompetitive inhibitor, the same (effective) Vmax is achieved no matter what inhibitor concentration is used (including 0)."
a. True
b. False
b. False
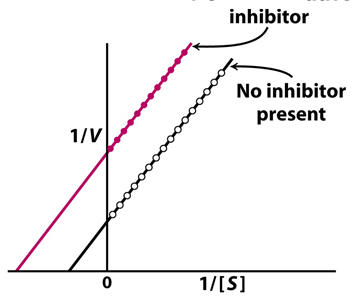
The graph above clearly indicates that the inhibitor used is a
a. competitive inhibitor
b. pure noncompetitive inhibitor
c. mixed noncompetitive inhibitor
d. Trojan Horse Inhibitor
e. uncompetitive inhibitor
e. uncompetitive inhibitor
You are developing a medical test for tissue damage using the presence of the enzyme pyruvate dehydrogenase in the bloodstream as a marker for that damage. The amount of the enzyme activity present in blood is proportional to the amount of damaged tissue. If the activity is measured with a saturating concentration of substrate, what is the ratio of pyruvate dehydrogenase enzyme in a patient versus a normal (control) patient with no damage if the measured rates of reaction are 38.74 uM/min and 3.42 uM/min, respectively? Report the ratio, which is unitless, to the nearest 0.1.
11.3
A fatty acid hydroxylase enzyme has a Km for palmitic acid of 8.29 micromolar and a turnover rate of 1,231.2 micromole substrate per minute per micromole enzyme. If 80.9 nanomolar enzyme is mixed with an unknown concentration of substrate and the reaction rate was 52.33 micromolar substrate per minute, how much substrate was in the reaction to start with? Report your answer in micromolar to the nearest 0.1 uM.
9.2
Consider an enzymatic reaction in which the initial concentration of substrate is low. If the amount of enzyme is held constant, but the amount of substrate is increased, the rate of an enzyme catalyzed reaction will
a. increase in an exponential fashion.
b. decrease at first, then increase in a linear fashion.
c. be inhibited by the higher concentrations of substrate.
d. increase at first in a linear fashion, then remain at a constant high rate.
e. stay the same.
d. increase at first in a linear fashion, then remain at a constant high rate.
A negative Gibb's free energy change for a reaction indicates that it
a. will release heat to the surroundings
b. will absorb heat from the surroundings
c. will be spontaneous in the reverse of the direction indicated
d. is at equilibrium
e. is spontaneous in the direction indicated
e. is spontaneous in the direction indicated
In the derivation of the Michaelis-Menten Equation for a noncompetitive inhbitor, the mass balance equation for enzyme is
a. Et = [E] + [ES] + [EI] + [ESI]
b. Et = [S] + [ES] +[EI] + [P]
c. Et = [E] + [S]
d. Et = [E] + [I] + [EI]
e. Et = [E] * [I]
a. Et = [E] + [ES] + [EI] + [ESI]
In Michaelis-Menten kinetics, Vmax is
a. the rate of reaction at an infinitely low enzyme concentration7)3
b. the rate of reaction at an infinitely high substrate concentration
c. the rate of reaction when [E] >>> [S]
d. the rate of reaction at an infinitely low substrate concentration
e. the rate of reaction at an infinitely high enzyme concentration
b. the rate of reaction at an infinitely high substrate concentration
You are developing a medical test for tissue damage using the presence of the enzyme pyruvate dehydrogenase in the bloodstream as a marker for that damage. The amount of the enzyme activity present in blood is proportional to the amount of damaged tissue. If the activity is measured with a saturating concentration of substrate, what is the ratio of pyruvate dehydrogenase enzyme in a patient versus a normal (control) patient with no damage if the measured rates of reaction are 45.12 uM/min and 4.33 uM/min, respectively? Report the ratio, which is unitless, to the nearest 0.1.
10.4
A fatty acid hydroxylase enzyme has a Km for palmitic acid of 28.91 micromolar and a turnover rate of 1,131.8 micromole substrate per minute per micromole enzyme. If 53.8 nanomolar enzyme is mixed with an unknown concentration of substrate and the reaction rate was 47 micromolar substrate per minute, how much substrate was in the reaction to start with? Report your answer in micromolar to the nearest 0.1 uM.
97.8
Enzyme A obeys simple Michaelis-Menten kinetics. If the rate of reaction under a given set of conditions is 9 uM/min and the concentration of enzyme is doubled, the new initial rate would be
a. 27 uM/min
b. There is not enough information to determine the answer
c. 3 uM/min
d. 18 uM/min
e. 81 uM/min
d. 18 uM/min
Penicillin forms an acyl-enzyme intermediate with its target enzyme.
a. True
b. False
a. True
Transition-state analogs are
a. extremely effective competitive inhibitors
b. not very effective as enzyme inhibitors
c. chemically unstable
d. suicide inhibitors
e. usually uncompetitive inhibitors
a. extremely effective competitive inhibitors
Irreversible inhibition can be eliminated by adding more substrate to a reaction.
a. True
b. False
b. False
Injecting a rabbit with a transition-state analog results in
the production of monolonal antibodies
a. suicide inhibition
b. product inhibition
c. formation of antibodies that are enzymes (abzymes)
d. the production of super-intelligent rabbits that want to take over the world even more than lab mice do
c. formation of antibodies that are enzymes (abzymes)
Which of the following inhibitors is not a reversible inhibitor?
a. competitive
b. suicide
c. noncompetitive
d. uncompetitive
b. suicide
Fetal hemoglobin has a lower affinity for oxygen than adult hemoglobin.
a. True
b. False
b. False
The 2,3-bisphosphoglycerate (2,3-BPG) binding site in hemoglobin
a. is in the center of the tetramer, contacting all four domains
b. is the same as the oxygen binding site
c. only exists in fetal hemoglobin
d. is surrounded by negatively charged residues
e. is in the alpha domain
a. is in the center of the tetramer, contacting all four domains
We always assume that E + S == ES is at equilibrium when working through enzyme kinetics derivations.
a. True
b. False
b. False
An irreversible inhibitor is one that
a. reacts covalently with a cofactor.
b. binds to the enzyme with hydrophobic interactions.
c. binds covalently to the enzyme active site.
d. forms hydrogen bonds with the substrate.
e. reacts covalently with the substrate.
c. binds covalently to the enzyme active site.
Deprenyl is a monoamine oxidase inhibitor (MAOI) that contains no reactive groups except an amine and an alkyne. The compound selectively alkylates MAO when the enzyme tries to oxidize the inhibitor. When The mode of inhibition is
a. uncompetitive
b. suicide
c. competitive
d. noncompetitive
b. suicide
Thalassemia is most prevalent in North America.
a. True
b. False
b. False
When carbon dioxide hydrates and then deprotonates, the product is bicarbonate.
a. True
b. False
a. True
2,3-Bisphosphoglycerate binds to
a. the positively charged hole at the center of the hemoglobin tetramer
b. adult hemoglobin, but only in the oxygenated state
c. the oxygen binding site in each monomer of hemoglobin, directly competing with oxygen for this site
d. myoglobin
e. fetal hemoglobin
a. the positively charged hole at the center of the hemoglobin tetramer
Unlike the oxygen binding curve (oxygen saturation versus partial pressure of oxygen) for hemoglobin, the curve for myoglobin
a. is completely linear for all oxygen concentrations (the form is a line)
b. is significantly sigmoidal
c. is not sigmoid at all
d. is inverted, with higher saturation at lower oxygen concentrations
e. This question is nonsense; myoglobin doesn't bind oxygen
c. is not sigmoid at all
You are measuring the rate of an enzyme called leukotriene B4 hydroxylase that is important in regulating the immune system. Assuming the enzyme follows simple Michaelis-Menten Kinetics, if the Km of this enzyme for its substrate (leukotriene B4) is 5.42 micromolar and the kcat is 7.31 micromole substrate per minute per micromole enzyme, what initial rate of reaction would be observed if you add enzyme and substrate to a cuvette resulting in concentrations (after dilution) of 116.1 nanomolar and 19.61 micromolar, respectively? Report your answer in the unit nanomolar substrate per minute to the nearest 1 nM/min. (Hint: Check your answer - what should the rate roughly by if the substrate concentration is above Km?)
665
In the absence of oxygen, hemoglobin heavily favors the
a. T-state
b. R-state
c. dimeric quaternary structure
d. monomeric quaternary structure
e. trimeric quaternary structure
a. T-state
The shift of hemoglobin from the low affinity state to the high affinity state is induced by
a. carbamate formation
b. high [CO2]
c. oxygen binding causing the iron to move into the plane of the heme
d. low pH
e. deoxygenation
c. oxygen binding causing the iron to move into the plane of the heme
Which has the higher affinity for oxygen?
a. adult hemoglobin
b. fetal hemoglobin
b. fetal hemoglobin
You are developing a medical test for tissue damage using the presence of the enzyme pyruvate dehydrogenase in the bloodstream as a marker for that damage. The amount of the enzyme activity present in blood is proportional to the amount of damaged tissue. If the activity is measured with a saturating concentration of substrate, what is the ratio of pyruvate dehydrogenase enzyme in a patient versus a normal (control) patient with no damage if the measured rates of reaction are 32.36 uM/min and 6.44 uM/min, respectively? Report the ratio, which is unitless, to the nearest 0.1.
5
You are studying a new enzyme and want to determine Km and Vmax. You collect your initial rate data at multiple substrate concentrations, take the reciprocal of v and s, and do a Lineweaver-Burke plot. If the slope of the resulting line is 0.0179 micromolar minutes and the y-intercept is 0.007 per min, what is the Km of this enzyme for its substrate? Report your answer in micromolar to the nearest 0.1 uM.
2.6
Penicillin is an example of a _________________ inhibitor.
a. suicide
b. competitive
c. uncompetitive
d. noncompetitive
a. suicide
Because of the steady state assumption, we can assume that the rate of product formation is exactly equal to the rate of product synthesis.
a. True
b. False
b. False
One difference between hemoglobin and myoglobin is
a. myoglobin binds oxygen, hemoglobin does not
b. hemoglobin utilizes a heme cofactor, myoglobin does not
c. myoglobin is monomeric, whereas hemoglobin exists as a tetramer
d. myoglobin utilizes a heme cofactor, hemoglobin does not
e. hemoglobin binds oxygen, myoglobin does not
c. myoglobin is monomeric, whereas hemoglobin exists as a tetramer
"Hemoglobin is a tetramer, but myoglobin is a dimer."
a. True
b. False
b. False
A shift of pH of just 0.2 (from 7.4 to 7.2 for example) can change amount of oxygen delivered from lungs to the acidic tissue by over 10%.
a. True
b. False
a. True
Fetal hemoglobin needs a much higher oxygen affinity than adult hemoglobin. This is accomplished by expressing a slightly different gene in the fetus. This fetal gene has a higher oxygen affinity because
a. it will not bind 2,3-BPG
b. it has more heme cofactors per chain
c. it has dimeric quaternary structure instead of dimeric
d. the hemoglobin become purely monomeric, and has a similar oxygen binding curve to myoglobin
a. it will not bind 2,3-BPG
A fatty acid hydroxylase enzyme has a Km for palmitic acid of 7.74 micromolar and a turnover rate of 1,335.1 micromole substrate per minute per micromole enzyme. If 55.5 nanomolar enzyme is mixed with an unknown concentration of substrate and the reaction rate was 46.18 micromolar substrate per minute, how much substrate was in the reaction to start with? Report your answer in micromolar to the nearest 0.1 uM.
12.8
You are studying a new enzyme and want to determine Km and Vmax. You collect your initial rate data at multiple substrate concentrations, take the reciprocal of v and s, and do a Lineweaver-Burke plot. If the slope of the resulting line is 0.0587 micromolar minutes and the y-intercept is 0.0067 per min, what is the Km of this enzyme for its substrate? Report your answer in micromolar to the nearest 0.1 uM.
8.8
A fatty acid hydroxylase enzyme has a Km for palmitic acid of 14.12 micromolar and a turnover rate of 1,498.1 micromole substrate per minute per micromole enzyme. If 83.8 nanomolar enzyme is mixed with an unknown concentration of substrate and the reaction rate was 48.3 micromolar substrate per minute, how much substrate was in the reaction to start with? Report your answer in micromolar to the nearest 0.1 uM.
8.8
Penicillin inhibits bacterial cell wall biosynthesis.
a. True
b. False
a. True
There are two types of thalassemia, alpha and omega.
a. True
b. False
b. False
Substrates are bound to enzymes by
a. multiple weak interactions.
b. covalent bonds.
c. disulfide bonds.
d. the hydrophobic effect.
a. multiple weak interactions.
The first step of an enzyme catalyzed reaction is
a. catalytic turnover
b. the binding of substrate by the enzyme
c. the release of product
d. dissociation of the ES complex
e. ES -> E + P
b. the binding of substrate by the enzyme
Sickle cell patients have episodes, or periodic crises, where multiple organs can be damaged at once.
a. True
b. False
a. True
You are measuring the rate of an enzyme called leukotriene B4 hydroxylase that is important in regulating the immune system. Assuming the enzyme follows simple Michaelis-Menten Kinetics, if the Km of this enzyme for its substrate (leukotriene B4) is 4.41 micromolar and the kcat is 17.86 micromole substrate per minute per micromole enzyme, what initial rate of reaction would be observed if you add enzyme and substrate to a cuvette resulting in concentrations (after dilution) of 62.2 nanomolar and 18.57 micromolar, respectively? Report your answer in the unit nanomolar substrate per minute to the nearest 1 nM/min. (Hint: Check your answer - what should the rate roughly by if the substrate concentration is above Km?)
898
Compared to the same reaction when not catalyzed by an enzyme, the equilibrium constant for an enzyme catalyzed reaction is
a. the same
b. larger
c. smaller
d. there is no equilibrium constant for an enzyme-catalyzed reaction
a. the same
You are measuring the rate of an enzyme called leukotriene B4 hydroxylase that is important in regulating the immune system. Assuming the enzyme follows simple Michaelis-Menten Kinetics, if the Km of this enzyme for its substrate (leukotriene B4) is 3.3 micromolar and the kcat is 6.61 micromole substrate per minute per micromole enzyme, what initial rate of reaction would be observed if you add enzyme and substrate to a cuvette resulting in concentrations (after dilution) of 223.9 nanomolar and 25.95 micromolar, respectively? Report your answer in the unit nanomolar substrate per minute to the nearest 1 nM/min. (Hint: Check your answer - what should the rate roughly by if the substrate concentration is above Km?)
1313
A fatty acid hydroxylase enzyme has a Km for palmitic acid of 22.08 micromolar and a turnover rate of 1,487.2 micromole substrate per minute per micromole enzyme. If 56 nanomolar enzyme is mixed with an unknown concentration of substrate and the reaction rate was 47.53 micromolar substrate per minute, how much substrate was in the reaction to start with? Report your answer in micromolar to the nearest 0.1 uM.
29.4
Hemoglobin is a(n)
a. αβ dimer
b. monomer
c. β2 dimer
d. α2 dimer
e. α2β2 tetramer
e. α2β2 tetramer
You are measuring the rate of an enzyme called leukotriene B4 hydroxylase that is important in regulating the immune system. Assuming the enzyme follows simple Michaelis-Menten Kinetics, if the Km of this enzyme for its substrate (leukotriene B4) is 4.94 micromolar and the kcat is 18.93 micromole substrate per minute per micromole enzyme, what initial rate of reaction would be observed if you add enzyme and substrate to a cuvette resulting in concentrations (after dilution) of 184 nanomolar and 18.59 micromolar, respectively? Report your answer in the unit nanomolar substrate per minute to the nearest 1 nM/min. (Hint: Check your answer - what should the rate roughly by if the substrate concentration is above Km?)
2752
You are developing a medical test for tissue damage using the presence of the enzyme pyruvate dehydrogenase in the bloodstream as a marker for that damage. The amount of the enzyme activity present in blood is proportional to the amount of damaged tissue. If the activity is measured with a saturating concentration of substrate, what is the ratio of pyruvate dehydrogenase enzyme in a patient versus a normal (control) patient with no damage if the measured rates of reaction are 29.85 uM/min and 5.42 uM/min, respectively? Report the ratio, which is unitless, to the nearest 0.1.
5.5
Which of the following do you know if a reaction is known to be sponataneous as written?
a. It has a negative enthalpy change
b. It has a postivie entropy change
c. It has a positive enthalpy change
d. It has a negative change in Gibb's free energy
e. It has a negative entropy change
d. It has a negative change in Gibb's free energy