Jurisprudence 3 - Immunization Administration, Epinepherine
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Immunizing pharmacists must complete an immunization administration certification program of no fewer than ______, approved by the FL BOP.
20 hours

Immunizing pharmacists must enter into a protocol under a ______ who is a physician licensed under chapter 458 or 459, FS.
supervisory practitioner

Immunizing pharmacists must maintain at least _______ of professional liability insurance.
$200,000

Once certified, an immunizing pharmacist must report immunizations to the state registry of immunization information. What is this registry in Florida?
Florida SHOTS
Immunizing pharmacy technicians must successfully complete an immunization administration certification program of no few than ______
6 hours

Immunizing pharmacy interns must successfully complete an immunization administration certification program of new few than ______
20 hours

True or False - Pharmacy intern immunization certification transfers when you become a pharmacist.
False - the certification does NOT transfer, the certification must be redone once a pharmacist license is obtained.
What types of vaccines are covered by the pharmacist-MD/DO protocol?
1. CDC adult immunization schedule vaccines
2. International travel vaccines recommended by CDC
3. Emergency use vaccines licensed by FDA: COVID
4. Vaccines authorized by the Florida Board during state emergencies
1 certified pharmacist may supervise up to _____ immunizing interns or technicians.
5
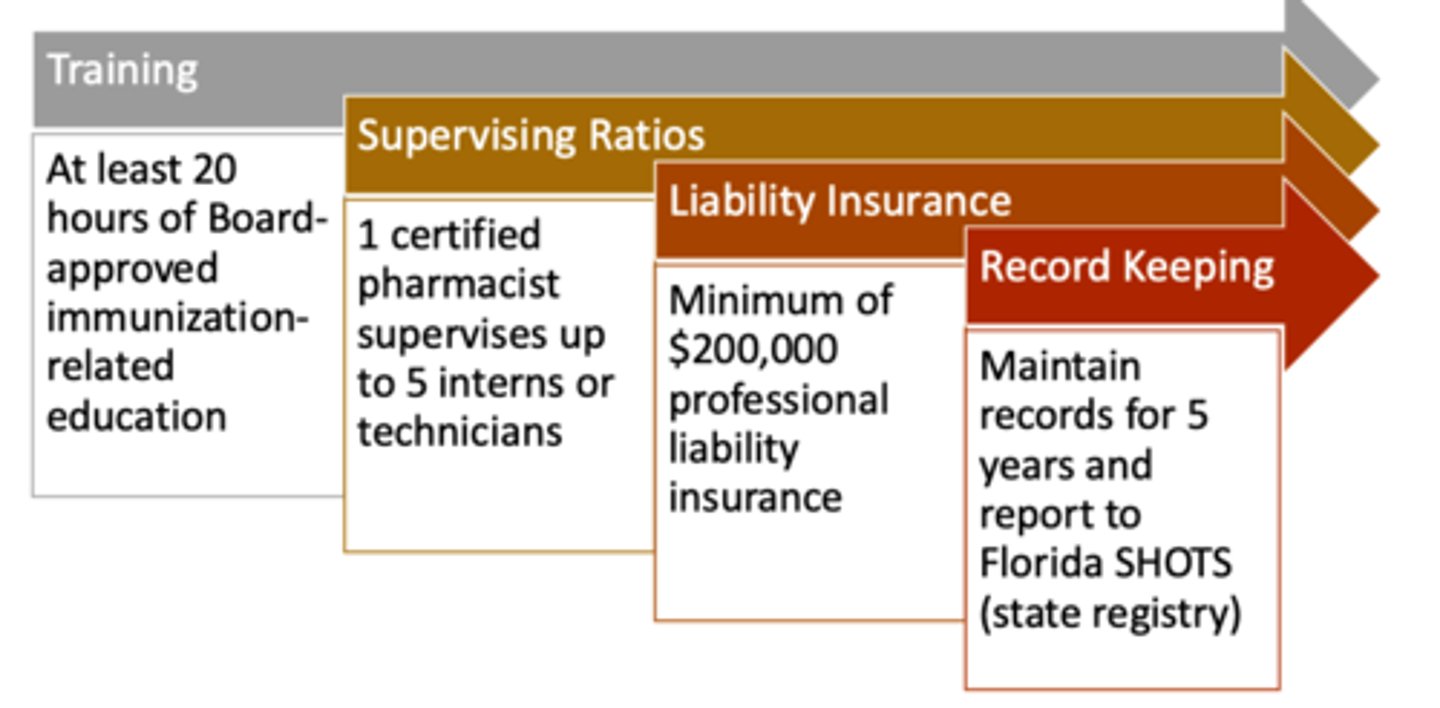
Immunization records must be maintained for _____ and reported to Florida SHOTS (state registry)
5 years
True or False: Epinephrine must be on hand for all immunization activities to address unforeseen allergic reactions
True; + Pharmacists, interns, or technicians may also use diphenhydramine as needed
True or False; there must be a separate epinephrine available for general availability
true

True or False: You are NOT permitted to administer vaccines without a current and active CPR card.
True
Pharmacists must report immunizations to Florida SHOTS within ______.
30 days + Notification of the patient's primary care provider (PCP) must also occur, if applicable
Adverse Drug Reactions (ADRs) due to vaccines must be reported to ______ and the patient's PCP when relevant.
Vaccine Adverse Event Reporting System (VAERS)
________ are required by federal law to be offered to the patient or caregiver PRIOR to the administration of 13 specific vaccines (before each dose of a multi-dose series, if applicable).
Vaccine Information Statements (VIS)
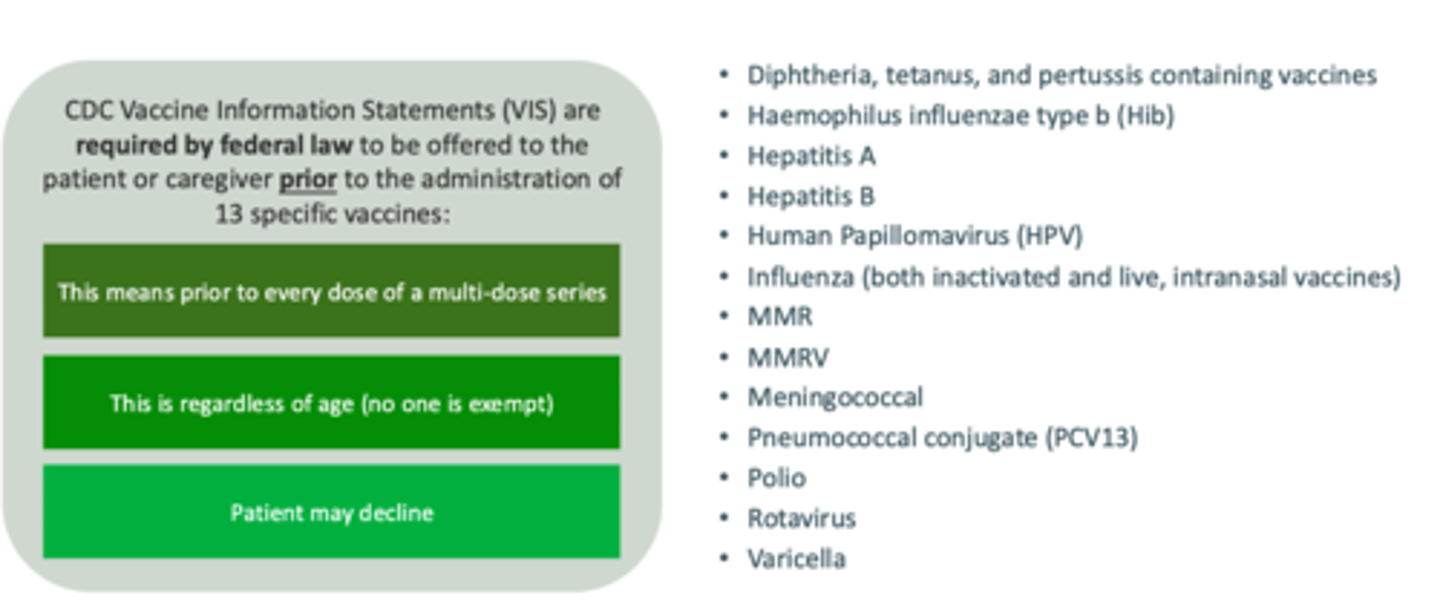
True or False - there are combination VIS statements for certain vaccines, like Tdap.
False - a separate VIS must be given for each of the components. There are no combination VIS's.
What vaccine information must be reported per federal law?
1. Edition date of the VIS
2. Date the VIS is provided
3. Vaccine manufacturer
4. Office address + name + title of person who administered the vaccine
5.Lot number

True or False - Vaccines cannot, under any circumstance, be stored in a "dormitory style" or bare style combined fridge/freezer unit.
True

What is the temperature range for "room"?
20 to 25C
68 to 77F
What is the temperature range for "fridge"?
2 to 8C
36 to 46F
What is the temperature range for "freezer"?
-10 to -25C
-13 to 14F
What is the temperature range for "ultra-cold freezer"?
-90 to -60C
-130 to -76F
Per the CDC, how frequent should the temperature logs be monitored for immunizations.
Once daily at minimum.
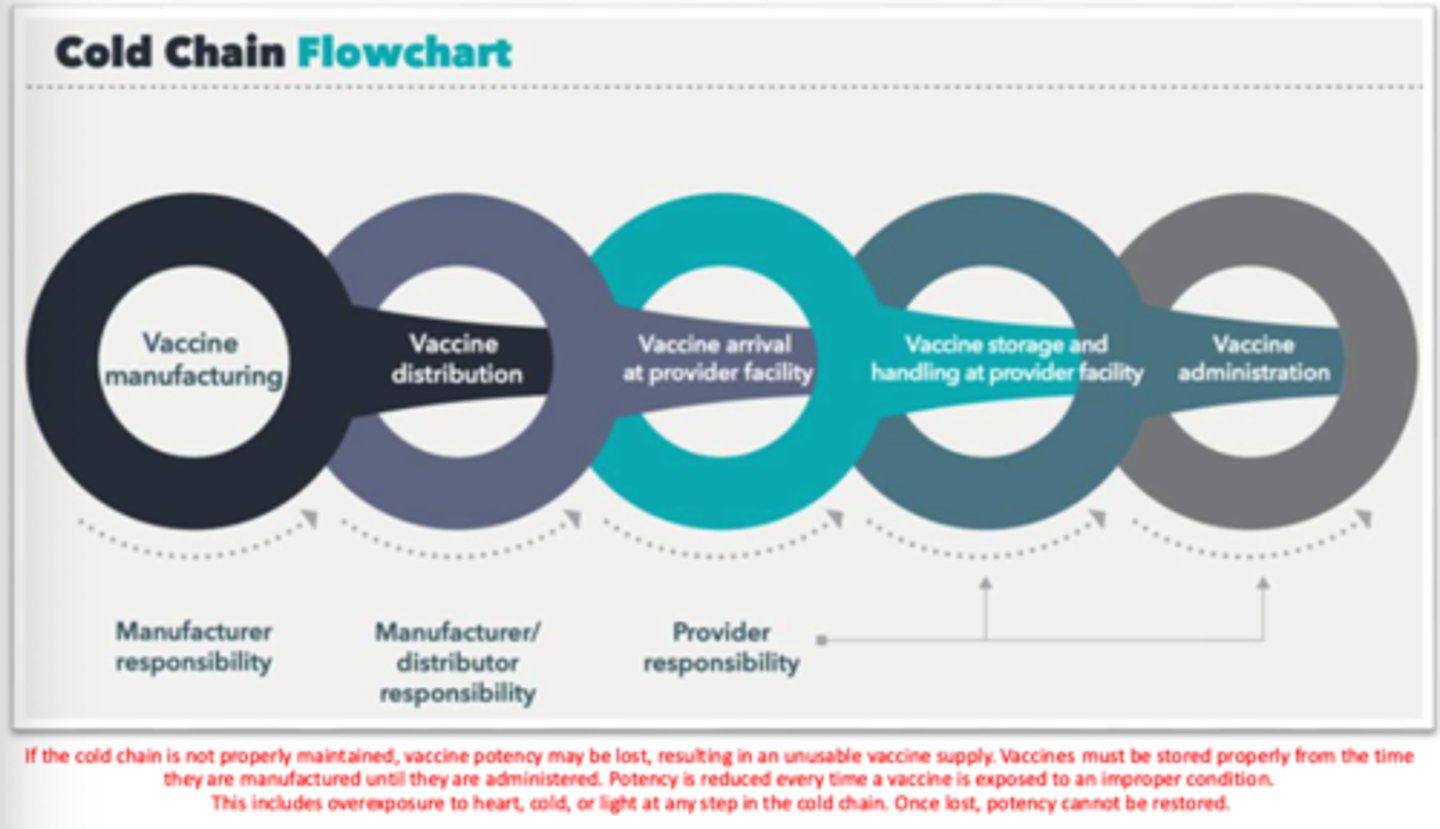
If the cold chain is not properly maintained, vaccine ____ may be lost, resulting in an _____ supply.
potency; unusable