MSE 2001 - Atomic Building Blocks (Memorization type terms)
1/25
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
What is an “Atom”?
Small bits of matter compiled of electrons, neutrons, and protons.
What is in the center of every atom?
The nucleus
What is inside every “Nucleus”?
Protons and Neutrons
What is the charge of a “Neutron”?
0
What is the charge of a “Proton”?
Position (+)
What is the charge of an “Electron”?
Negative (-)
What is the same about both Neutrons and Protons?
Same mass (1 amu)
What is the nucleus surrounded by?
Shells/orbitals of negatively charged electrons (electron cloud)
How does one find atomic number?
By looking at the number of protons
By looking at the number of electrons (IN NEUTRAL ATOMS ONLY)
What does the top number in the image represent?
Atomic Number
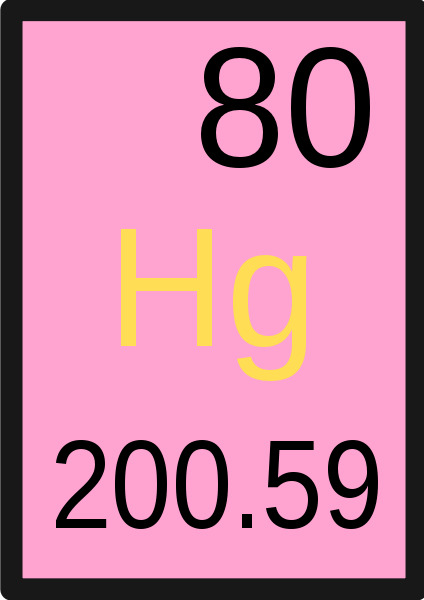
What does the letters in the center represent?
Symbol for element
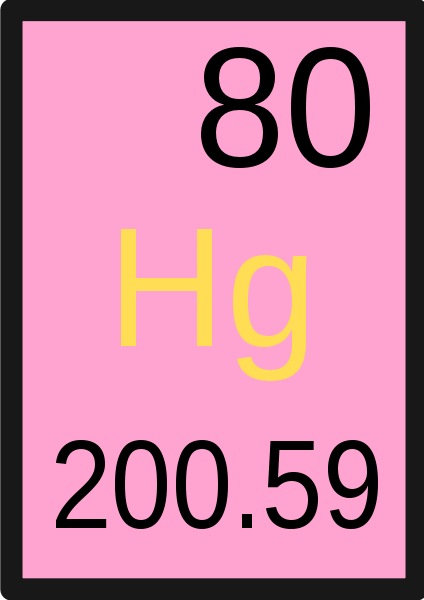
What does the number in the bottom represent?
A
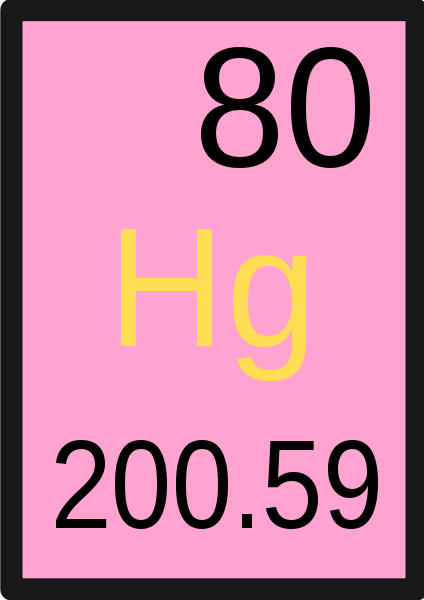
What does “amu” mean?
Atomic Mass Unit
How do you find the number of neutrons in an atom?
Subtract the atomic number from the amu
Bohr Model
One of the 1st accepted atomic models
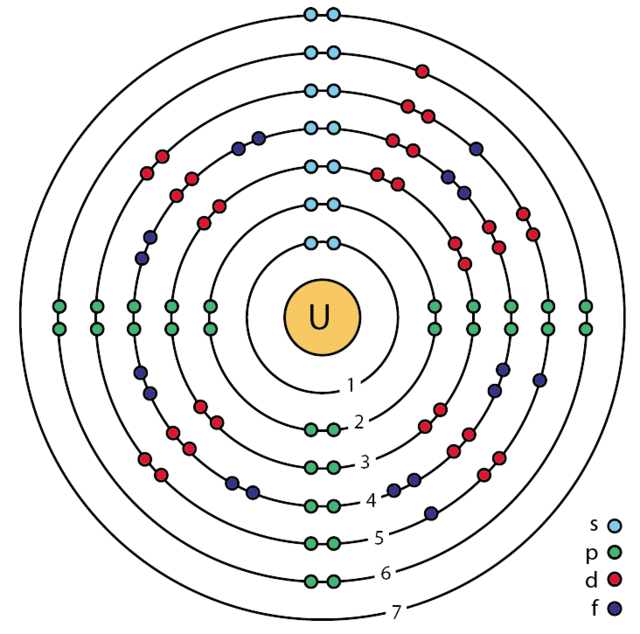
How was the Bohr model updated?
By using the wave mechanical model (quantum mechanics)
What does the wave mechanical model describe?
describes the energy levels of electrons in an atom
Includes orbital shape of electrons
What is the “Schrodinger’s Equation”?
Describes the properties of electrons in terms of wave motion. The solutions of the equation are identified by a set of integer values known as Quantum Numbers
What is “n”?
Principal Quantum Number
What is “l”? [L]
Angular momentum quantum number
What does the angular momentum number depend on?
Principal Quantum Number
What is “m”?
Magnetic Quantum Number
What is “ms”?
Electronic Spin
Pauli Exclusion Principle
No two interacting electrons may have the same four quantum numbers.
What is “Electron Configuration”?
Describe the distribution of all electrons within an atom
How do electrons fill subshells?
Electrons fill subshells starting with the lowest energy levels