Quiz 1: Cell Membrane
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
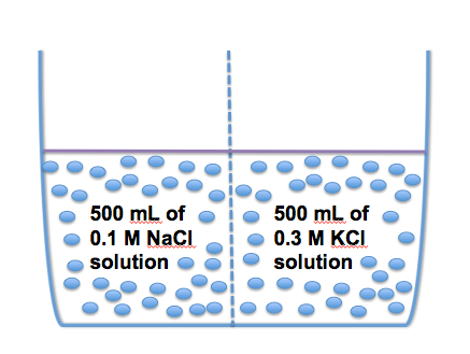
For Questions #1 & 2, consider the image above. A tank is partitioned into equal volumetric halves (500 mL each) by a membrane that is permeable only to water. Initially, the right-hand compartment contains a 0.3 molar (M) aqueous solution of potassium chloride (KCl) and the left-hand compartment contains a 0.1 molar (M) aqueous solution of sodium chloride (NaCl).
Initially:
the KCl solution is hypotonic compared to the NaCl solution
the KCl solution is isotonic compared to the NaCl solution.
the KCl solution is hypertonic compared to the NaCl solution.
the KCl solution is hypertonic compared to the NaCl solution.
At dynamic equilibrium:
The solution on the right has a greater osmotic pressure than the solution on the left.
The sodium concentration is equal on both sides of the partition.
The potassium concentration is equal on both sides of the partition.
The solution on the right has a greater total volume than the solution on the left.
The solution on the right has a greater total volume than the solution on the left.
If the size of a gradient decreases, then:
the resistance decreases.
flux decreases.
flux increases.
the resistance increases.
flux decreases.
Sodium channels and aquaporins are similar in that:
they both move water across the cell membrane.
they are both uniporters.
they are both passive transporters.
they are both active transporters.
they are both passive transporters.
The Na/K-ATPase:
maintains a high K+ concentration outside of our cells.
works without any energy expenditure.
moves K+ ions downhill and Na+ ions uphill.
maintains a high Na+ concentration outside of our cells.
maintains a high Na+ concentration outside of our cells.
In the epithelial cells of our gut, the Na/K-ATPase, Na/glucose-cotransporter, and glucose-uniporter work together to:
move Na+ ions into the gut lumen.
move glucose molecules from the gut lumen to the circulation (blood).
move K+ ions into the circulation (blood).
move glucose molecules from the circulation (blood) to the gut lumen.
move glucose molecules from the gut lumen to the circulation (blood).
Blood potassium levels are typically in the range of 3.5 to 5.2 mM (millimolar). If a person's blood potassium level is 6.2 mM, then they:
are hyperkalemic, and the resting membrane potential in their cells is probably too low (hyperpolarized).
are hyperkalemic, and the resting membrane potential in their cells is probably too high (depolarized).
are hypokalemic, and the resting membrane potential in their cells is probably too high (depolarized).
are hypokalemic, and the resting membrane potential in their cells is probably too low (hyperpolarized).
are hyperkalemic, and the resting membrane potential in their cells is probably too high (depolarized).
For Questions #8-10, consider a typical mammalian cell that has 4 mM Cl- ions inside the cell and 120 mM Cl- ions outside the cell. Assume the cell is at a resting membrane potential (VR) of -70 mV.
Which value is closest to the Nernst potential for chloride (ECl)?
-90 mV
0 mV
+65 mV
+130 mV
-90 mV
For Questions #8-10, consider a typical mammalian cell that has 4 mM Cl- ions inside the cell and 120 mM Cl- ions outside the cell. Assume the cell is at a resting membrane potential (VR) of -70 mV.
If Cl- channels in the cell membrane open, in which net direction would Cl- ions move?
out of the cell
into the cell
into the cell
For Questions #8-10, consider a typical mammalian cell that has 4 mM Cl- ions inside the cell and 120 mM Cl- ions outside the cell. Assume the cell is at a resting membrane potential (VR) of -70 mV.
If the chloride concentration outside the cell decreases to 110 mM:
the chemical driving force acting on Cl- increases.
the membrane potential would not change.
the chemical driving force acting on Cl- decreases.
the Nernst potential for chloride would not change
the chemical driving force acting on Cl- decreases.