CELL & MOLECULAR AGEING
1/75
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
76 Terms
define ageing
ageing: complex biological process in which changes at the molecular, cellular and organ levels results in a progressive inevitable and inescapable decrease in the body’s ability to respond appropriately to internal and/ or external stressors
outline lifespan
different from ageing
life expectancy is increasing
increased % elderly in population
increase in age-related diseases
ageing VS age-related disease
ageing is not a disease - occurs in every multi-cellular animal
has universal molecular aetiology
ageing occurs in the absence of disease
who came up with the traditional theories of ageing
Galen
Roger Bacon
Charles Darwin
outline Galen’s traditional theory of ageing
Galen (AD129-199)
ageing is not a disease
death is inevitable as ‘the body deteriorates of itself’ but life could be prolonged
importance of a healthy youth as the basis for a robust old age incl. diet, walking, wine
outline Roger Bacon’s traditional theory of ageing
Roger Bacon (1220-1292)
wear and tear theory
result of abuses and insults to the body - good hygiene may slow process
outline Charles Darwin’s traditional theory of ageing
Charles Darwin (1809-1892)
programmed mechanism of ageing - biological evolutionary process
loss of irritability in nervous and muscular tissue
what are the two main categories of modern biological theories of ageing
programmed theories
ageing follows a biological timetable
damage or error theories
environmental assaults to living organisms that induce cumulative damage at various levels
what theories are within the programmed theories of ageing
programmed longevity
endocrine theory
immunological theory
programmed theories of ageing: programmed longevity
switching on and off of certain genes
changing expression results in genetic instability
programmed theories of ageing: endocrine theory
biological clocks (e.g. circadian rhythms) act through hormones
programmed theories of ageing: immunological theory
immune system is programmed to decline » increased vulnerability to infectious disease
this leads to ageing and death
what theories are within the damage/ error theories of ageing
wear and tear theory
rate of living theory
cross-linking theory
free radicals theory
somatic DNA damage theory
damage/ error theories of ageing: wear and tear theory
cells and tissues have vital parts that wear out resulting in ageing
damage/ error theories of ageing: rate of living theory
the greater an organism’s rate of oxygen basal metabolism, the shorter its lifespan
damage/ error theories of ageing: cross-linking theory
accumulation of cross-linked proteins damages cells and tissues
damage/ error theories of ageing: free radicals theory
superoxide and other free radicals generated cause damage to the macromolecular components of cell (proteins, nucleic acids, lipids, carbohydrates)
outline superoxides
superoxides are naturally produced by the body and needed
however they damage DNA and mitochondria if left in the body for too long
in health, cell has mechanisms to remove superoxides but as you age these mechanisms become less efficient and effective
damage/ error theories of ageing: somatic DNA damage theory
accumulation of DNA damages results in eventual ageing
as you age you also accumulate DNA damage
summary of biological theories of ageing
multiple theories proposed
however no consensus because many interact with each other in complex ways
what determines the ‘ageing phenotype’
cell and molecular events that contribute to the ageing process and together determine the ‘ageing phenotype’
what does ‘hallmark’ mean in terms of ageing
cellular and molecular mechanisms that cause ageing
what criteria should each hallmark fulfil
should manifest during normal ageing
experimental aggravation should accelerate ageing
experimental amelioration (improvements) should retard the normal ageing process and hence increase healthy lifespan
what are the cell and molecular hallmarks of ageing (9)
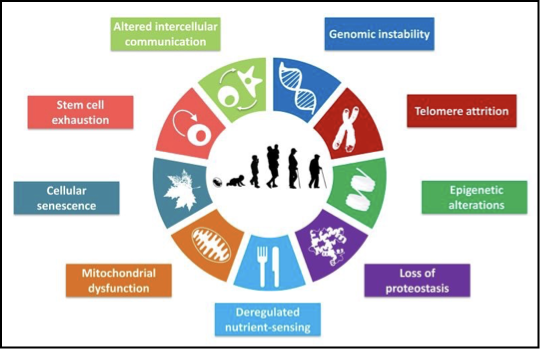
outline genomic instability
integrity and stability of DNA is continuously challenged by endogenous e.g. ROS and exogenous agents e.g. UV
there will be a natural accumulation of genetic damage throughout life - nuclear and mitochondrial DNA
how do DNA alterations result in dysfunctional cells
DNA alterations may affect essential ageing genes and transcriptional pathways
this results in dysfunctional cells
tissue and organismal homeostasis and tissue renewal may be jeopardised
genetically controlled longevity
heritable component in human longevity - esp. at extreme ages
large number of genes identified, modification of which affects longevity
but phenotypic differences in ageing between monozygotic twins
20-30% of ageing is controlled by genetics, the rest is environmental factors
what is an ageing syndrome
Hutchinson-Gilford Progeria (HGPS)
outline HGPS
rare genetic, fatal disorder with striking features resembling premature ageing
mutation in LMNA gene
associated with atherosclerosis, high BP, strokes, angina, heart failure
usually die by 14.5 years old
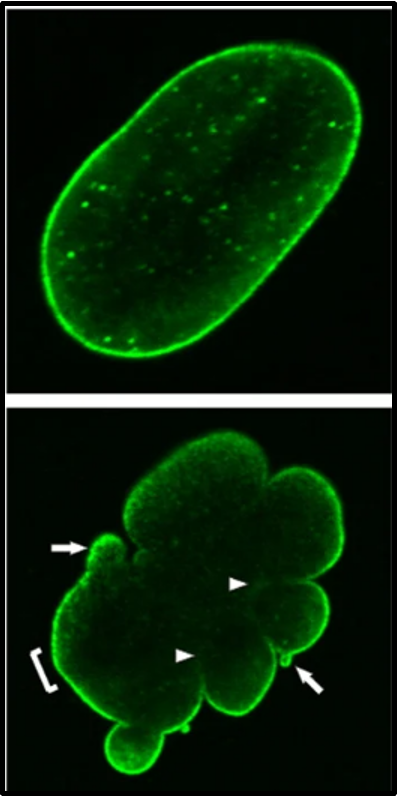
outline LMNA gene (mutation) in HGPS
LMNA encodes for lamin A (encoding nuclear envelope protein)
defective lamin A protein (truncated) makes the nucleus unstable
cellular instability appears to lead to the process of premature ageing in progeria
what is an oral implication of progeria
overcrowding of teeth in mandible
what are telomeres
telomere: repetitive DNA sequences found at either ends of chromosomes that protect the ends of chromosomes
DNA needs protecting because the ends can fray - telomeres prevent this
outline the change in length of telomeres
telomeres progressively shorten with each cell division
telomerase maintains the length of telomeres
most mammalian somatic cells do not express telomerase
what kind of enzyme is telomerase
specialised DNA polymerase able to replicate terminal ends of linear DNA
what if telomeres become dysfunctional
pathological telomere dysfunction accelerates ageing in mice and humans
experimental stimulation of telomerase can delay ageing in mice
across species, do shorter telomeres predict shorter lifespans
no, across species, short telomeres do not necessarily predict short lifespan - humans have shorter telomeres than rodents but have longer lifespans
telomere attrition II
outline epigenetic changes
chemical changes DNA that does not affect underlying DNA
epigenetic changes: age-associated epigenetic markers
increase - histone H4K16 acetylation, H4K20 trimethylation or H3K4 trimethylation
decrease - H3K9 methylation or H3K27 trimethylation
what are the different mechanisms of epigenetic changes
alterations in DNA methyltransferases
histone acetylases, deacetylases, methylases and demethylases
protein complexes implicated in chromatin remodelling
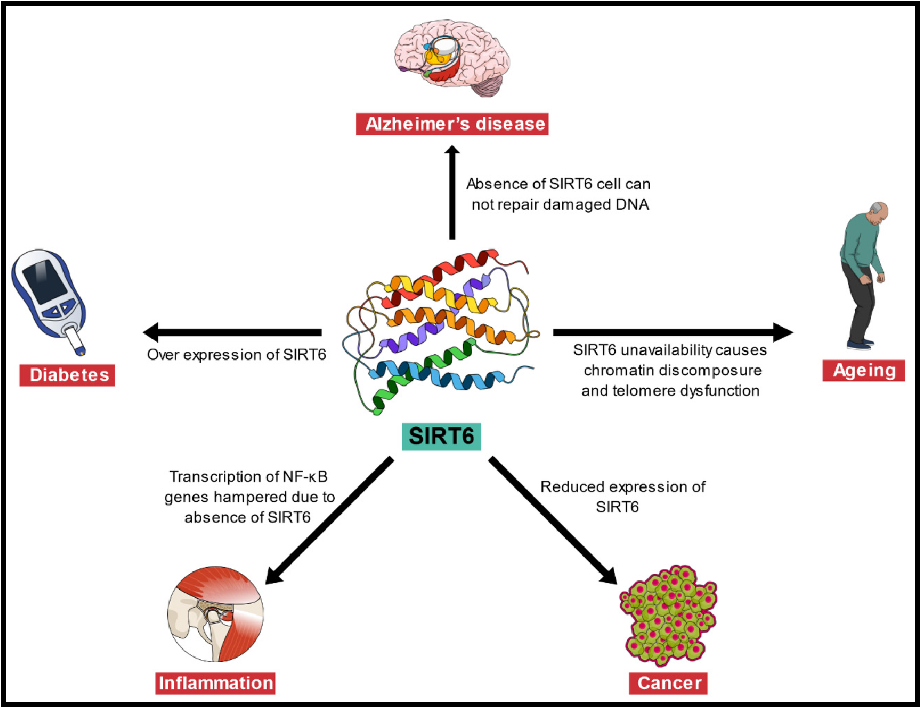
outline sirtuins
family of enzymes that has important roles in DNA repair, telomere maintenance and age-related pathological conditions
e.g. loss of SIRT6 gene in mice leads to ageing
gain of function extends longevity
class III histone deacetylases
SIRT6 and links to pathological conditions
what is proteostasis
proteostasis: the process of protein homeostasis - balance of synthesis, folding, degradation
what do Alzheimer’s, Parkinson’s and cataracts have in common
chronic expression of unfolded, misfolded proteins will begin to aggregate which contributes to the development of some age-related pathologies:
Alzheimer’s
Parkinson’s
cataracts
name the two principal proteolytic systems associated with protein quality control decline with ageing
autophagy-lysosomal
ubiquitin-proteasome
what is an experimental example of proteostasis being associated with age-related pathologies
mutant mice deficient in protein from the heat-shock family exhibit accelerated ageing phenotypes
loss of proteostasis depends on the ______
loss of proteostasis depends on the tissue (so some organs may begin to fail and age before others)
outline deregulated nutrient sensing
dietary restriction increases lifespan or healthspan in eukaryote species - seen in unicellular and multicellular organisms
the insulin and IGF-1 signalling (IIS) pathway is the most conserved ageing controlling pathway in evolution
decreased nutrient signalling extends longevity
what does Rapamycin do in mice
rapamycin can extend longevity in mice
restricting diet to restrict ____ can improve longevity
restricting diet to restrict mTOR (protein) can improve longevity
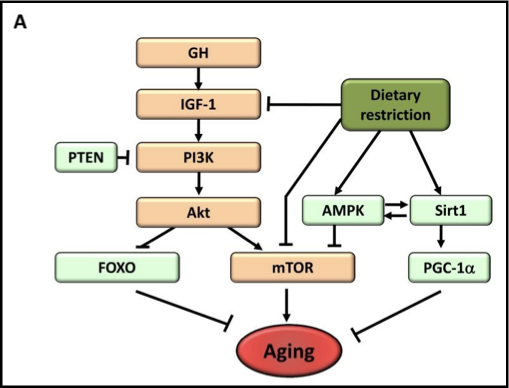
light green = improves longevity
define calorie restriction
calorie restriction: the sustained restriction of dietary energy intake compared with the energy requirement for weight maintenance
what does responsible prescription of calorie restriction require
adequate intake of carbohydrate, fat, protein and micronutrients to ensure satisfaction of recommended daily allowance
are there a lot of randomised controlled trials on the effects of calorie restriction on biological ageing in humans
there are very few
expensive
time-consuming
lots of difficult to control factors
controlling your diet for _ years extends your life for _ year
controlling your diet for 2 years extends your life for 1 year

outline mitochondrial dysfunction
multiple converging mechanisms:
accumulation of mutations and deletions in mtDNA (mitochondrial DNA)
oxidation of mitochondrial proteins by ROS produced in electron transport chain
destabilisation of macromolecular organisation of respiratory chain (super)complexes
» destabilisation of mitochondria and breakdown of electron transport chain
why does the efficacy of the respiratory chain decrease with age
efficacy of respiratory chain decreases with age due to:
electron leakage
reduced ATP production
mitochondrial dysfunction: outline the conventional and current theories of the role of ROS in ageing
conventional theory: increased ROS production causes progressive mitochondrial deterioration and global cellular damage
current theory: as age increases, ROS increases to maintain survival until the levels become too high and aggravate age-associated damage
what do ROS trigger
proliferative and survival signals in response to physiological signals and stress conditions
(this is how the current theory of ROS was arrived at - because it can respond to signals it also helps maintain survival to a certain point as we age)
describe the pathway from mitochondrial damage to neurodegeneration
mitochondrial damage » pathologic inflammation » synaptic degeneration » neurodegeneration
mitochondrial damage initiates ROS production
this stimulates pro-inflammatory signals and cytokine production which leads to inflammation
but there is nothing to attack because the damage is intracellular so it damages synapses instead
this begins a cascade that results in global neurodegeneration
what conditions are the previous pathway observed in
Alzheimer’s and other neurological disorders
outline the relationship between melatonin and ROS
melatonin is a neuroprotective hormone that has a largely antioxidant effect
exclusively synthesised in the mitochondrial matrix
it soaks up ROS within synapses of neurones
therefore maintaining melatonin levels via pharmaceuticals or by circadian regulation is an area of therapeutic interest
outline cellular senescence
irreversible cell-cycle arrest mechanism
when cells are still living but not proliferating, permanently left the cell cycle therefore protected from dysregulated growth
this results in large phenotypic changes
why does cellular senescence result in large phenotypic changes
cell is no longer in the cell cycle and therefore no longer well regulated
no protein level checks, lots of cytokine and growth factors
DNA coiling is also dysregulated
who was cellular senescence first described by
Hayflick and Moorhead (1961)
DNA becomes damaged because of telomere attrition
therefore cells leave cell cycle permanently to try and protect themselves (cell division is arrested)
this is replicative senescence
protective mechanism for unrestricted growth of damaged cells
why is there an accumulation of senescent cells with ageing
there is an accumulation of senescent cells with ageing due to:
increase in rate of generation of senescent cells
and/ or
decrease in their rate of clearance
state the 5 distinct paracrine mechanisms by which senescent cells could promote tissue dysfunction
induction of senescence in neighbouring cells (paracrine senescence)
perturbation of the stem cell niche (causing stem cell dysfunction)
disruption of extracellular matrix
induction of aberrant cell differentiation
stimulation of tissue inflammation (senescence-associated secretory phenotype)
outline stem cell exhaustion
decrease in cell cycle activity of stem cells with ageing
consequence of accumulation of DNA damage, overexpression of cell cycle-inhibitory proteins (senescence), telomere shortening
what process declines with age
haematopoiesis declines with age
» increased incidence of anaemia and myeloid malignancies
outline altered intercellular communication
inflammaging: inflammation increases with age
accumulation of pro-inflammatory tissue damage
senescent cells secrete pro-inflammatory cytokines
immunosenescence (failure of immune system to clear infectious agents)
—
» impact on cell communication
what type of signalling tends to be deregulated in ageing and why
neurohormonal signalling tends to be deregulated in ageing as inflammatory reactions increase
outline inter-organ coordination in altered intercellular communication
age related changes in one tissue can lead to ageing-specific deterioration of other tissues
e.g. impaired kidney function can increase the risk of heart disease in humans
however this means that lifespan-extending therapies targeting one tissue can retard the ageing process in other tissues
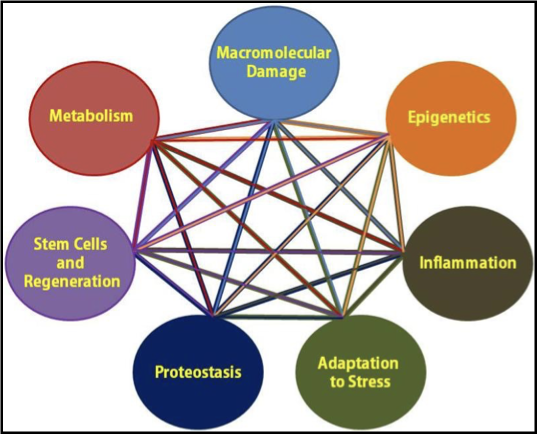
what is an alternative model to the cell and molecular hallmarks of ageing
the pillars of ageing (2013)
what are the pillars of ageing
intertwined processes that promote ageing
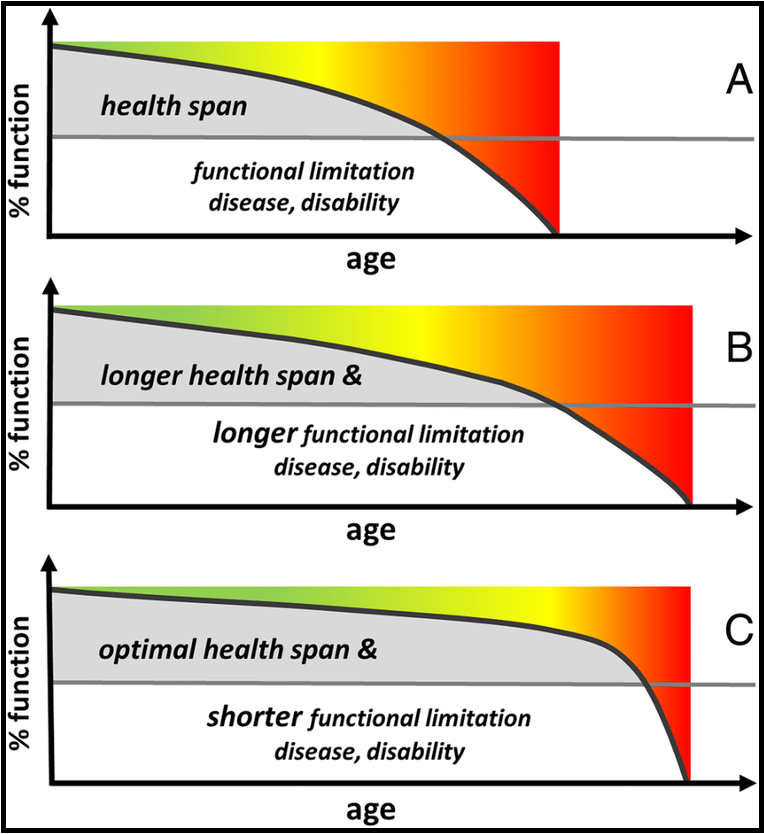
what is a challenge of increasing lifespan/ life expectancy
while life expectancy continues to rise, healthspan is not keeping pace
current disease treatment often decreases mortality but does not reverse the decline in overall health
elders are sick for longer, often coping with multiple chronic diseases simultaneously
is there a consensus on biological ageing
no, there are many different interlinked theories