RATE LAWS, INTEGRATED RATE LAWS, AND HALF LIVES
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
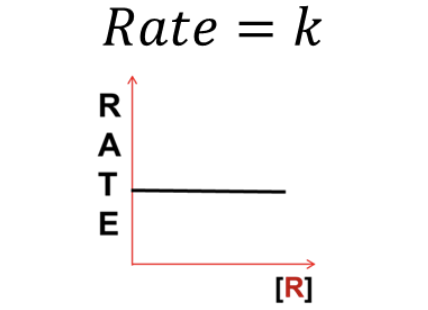
What RATE LAW does this graph represent?
Zeroth Order (Rate = k)
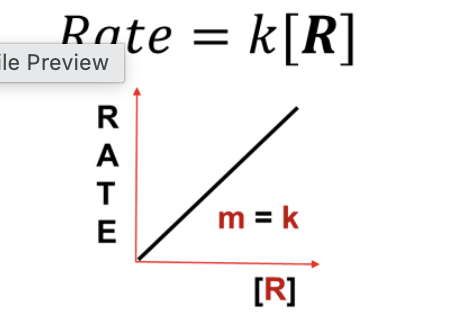
What RATE LAW does this graph represent?
First Order (Rate = k [R])
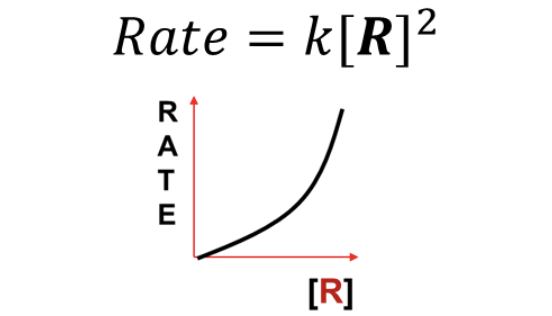
What RATE LAW does this graph represent?
Second Order (Rate = k [R]^2)
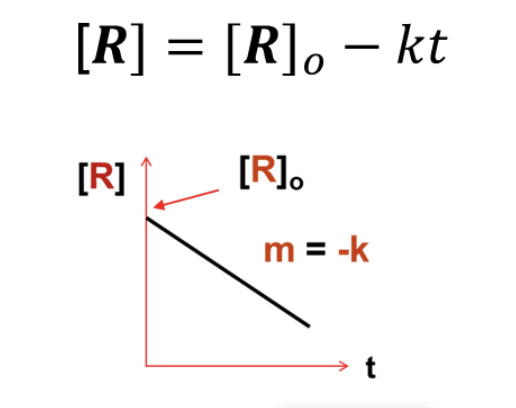
What INTEGRATED RATE LAW does this graph represent?
Zeroth Order ([R] = [R]o - kt)
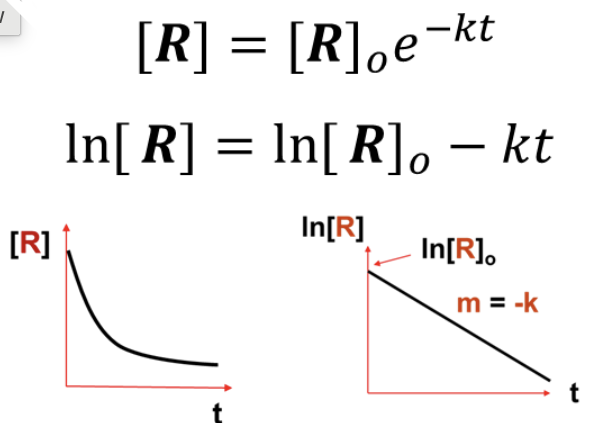
What INTEGRATED RATE LAW does this graph represent?
First Order ([R] = [R]o * e^-kt OR ln[R] = ln[R]o - kt)
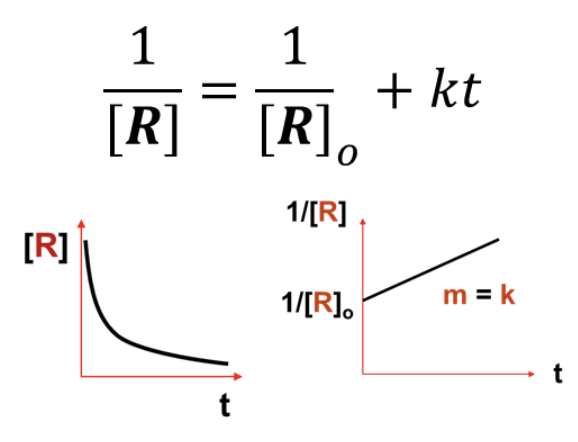
What INTEGRATED RATE LAW does this graph represent?
Second Order (1/[R] = 1/[R]o + kt)
What is the RATE LAW for a ZEROTH ORDER reaction?
Rate = k
What is the RATE LAW for a FIRST ORDER reaction?
Rate = k[R]
What is the RATE LAW for a SECOND ORDER reaction?
Rate = k[R]²
What is the INTEGRATED RATE LAW for a ZEROTH ORDER reaction?
[R] = [R]0 - kt
What is the INTEGRATED RATE LAW for a FIRST ORDER reaction?
[R] = [R]0 * e^-kt OR ln[R] = ln[R]0 - kt
What is the INTEGRATED RATE LAW for a SECOND ORDER reaction?
1/[R] = 1/[R]0 + kt
What is the HALF LIFE of a ZEROTH ORDER reaction?
t1/2 = [R]0/2k
What is the HALF LIFE of a FIRST ORDER reaction?
t1/2 = ln(2)/k
What is the HALF LIFE of a SECOND ORDER reaction?
t1/2 = 1/k[R]0
What TYPE of graph is ZEROTH ORDER?
Constant
What TYPE of graph is FIRST ORDER?
Exponential
What TYPE of graph is SECOND ORDER?
Quadratic (power of two)