Soil Science Test 3
1/92
Earn XP
Description and Tags
Once againnnnnnn
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
93 Terms
potential, free
water potential is a measure of the - or - energy of water, measured in units of pressure
higher, lower
water always moves from - potential to - potential
mass flow
water moves via __ , or movement in response to a pressure or tension gradient
positive, megapascals
turgor pressure is - in living cells, at about 0.1-1 MPA (short for -)
phloem
the movement of fluid in the - is driven by positive pressure
tension, negative
the movement of water in the non living cells of the xylem is driven by - or - pressure
permeable, water
non-woody, fine roots are the most - portion of the root system and have the greatest ability to absorb -
root hairs
fine roots can be covered by —, which significantly increase the absorptive surface area
apoplastic, xylem, outside
water can move across the root by the - pathway, which includes interconnected cell walls, intercellular spaces, - tracheids and vessels, and the areas - the plasma membrane/the non living ones
symplastic, inside, plasmodesmata
water can move across the root by the - pathway, which is everything - the plasma membrane, and the movement of water via - between cells connected by the plasma membrane. Also called the cell to cell pathway
suberin, hydrophobic
at the endodermis, the apoplastic pathway is blocked by a band of -, which is a Highly - (waterproof) substance that seals off the route of water in the apoplast
symplastic, apoplastic
the purple line represents the - pathway, and the green line is the - pathway
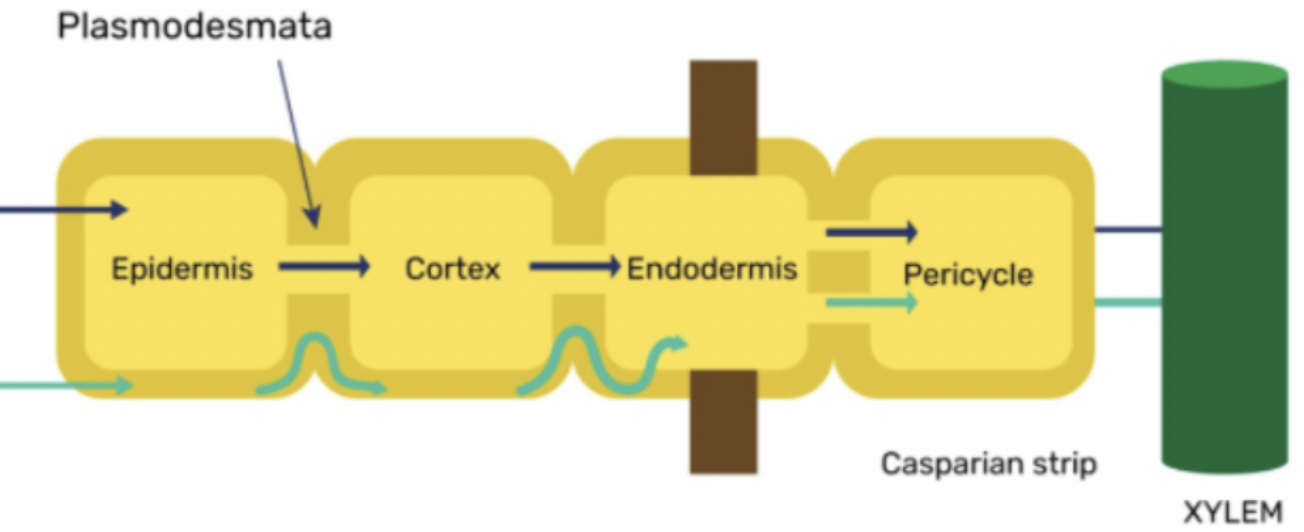
casparian strip
the suberin layer is the —, and once it is reached, the water must move to the symplastic pathway
checkpoint
the endodermis acts as a selective - by not having channels or transporters for certain harmful molecules like heavy metals
root pressure, meters
Root cells actively pump ions into the xylem and the root endodermis holds the ions there. As ions accumulate in the xylem, water enters the xylem from the roots by osmosis, pushing the water/ion solution upward by positive pressure. This force is called —. This force can only pump the water/ion mixture a few - up
capillary, cohesion, adhesion
Water eventually moves into the xylem, made up of narrow tubes (tracheids and vessels). When water is in a narrow tube, it gets - action, and water moves up by - from collective water molecules as well as - from the tube walls. But, water can only go so far with this alone
transpiration, stomates, gas
_ is the loss of water from aerial parts of plant, especially via - on leaves, and is also the Movement of water molecules in - phase from sub-stomatal cavity to outside air
negative, transpiration
The bulk of water absorbed and transported through plants is moved by - pressure generated by the evaporation of water from the leaves, or -
transpiration pull
— is the suction force caused by transpiration, and The main factor that causes water to move up the xylem
mineral, cooling, photosynthesis, turgor
Transpiration draws water and - salts from the roots to the stems and leaves, Evaporation of water from the cells in the leaves causes evaporative -. Water transported to the leaves is used for -, and Water transported to the leaves used to maintain - pressure of the leaf cells, especially guard cells!
temperature, humidity, intensity, air
factors that affect transpiration include the - of the air, air -, light -, and wind/- movement
evaporation, transpiration, 86
Higher temperatures increase the rate of -. The higher the temperature, the greater the rate of - Until stomates close in response to temperatures being too high, at about - degrees F
secondary, negative
Xylem vessels and tracheids have strong - cell walls, which allows them to withstand - pressure force
mass flow
examples of — include soil water flow, xylem flow, and phloem flow
diffusion, substomatal
_ is water movement due primarily to differences in concentration of water molecules, an example being the movement of water from the - cavity to outside air
osmosis
_ is the movement of water across a semi-permeable membrane
solute
Osmotic potential also known as the - potential, or (ΨS)
osmotic, solute, pressure
water potential inside of a plant is the sum of - or - potential and - potential
water
the formula for - potential inside the plant is ψW = ψS + ψ
direction
the difference in water potential across the membrane, or Δ ψW, determines the - water moves across the membrane
less, more
Water moves from state of higher free energy (- negative) a to lower free energy (- negative)
into
When there is More solutes inside cell vs. outside: water moves - the
cell
equl
When the Number solutes inside the cell _ the number outside, there is no net movement of water
out
When there is Less solutes inside cell vs. outside, water moves - of cell
plasmodesmata, gradient
Guard cells are not connected by - and are Independent/isolated from other cells. This isolation allows water potential - to be set up, which could not happen if they were connected by (first blank)
molecules, compounds
_ are two or more atoms bonded together, while _ are two or more different elements bonded together. all compounds are molecules, but not all molecules are compounds
ions
atoms or molecules carrying some sort of charge are called -
absorb, adsorb
Plant roots - nutrient ions, soil particles - them
surface
to adsorb is to attract a thin layer of molecules to a -, where they stick
growth, nutrition, supplying
an element is essential when A lack of the element stops a plant from completing - or reproduction, The element is directly involved in plant -, and a shortage of the element can be corrected only by - that element
air, water
Plants get C, H, and O from the - and - they take in
soil, roots
mineral nutrients, or those other than C, H, and O; are Primarily absorbed from the - via the -
nitrogen, phosphorous, potassium
the primary macronutrients are the mineral nutrients used in largest amounts: -, -, and -
endodermis, xylem, used
For nutrients to get from the soil to the xylem, they must cross two membranes: at or before the - and into the -. If the nutrients are going to be -, they must pass a third membrane
transport proteins
nutrients cross the plasma membrane via —
channel, carrier
there are two types of transport proteins: - and -
tunnel, active
channel proteins are a pore or - things move through quickly, and are always - in transport
protein, conformation, usually
in carrier proteins, Solute binds to - on one side of membrane, protein changes “-”, solute is released on the other side of the membrane. transport is - active
response, spontaneous
in passive transport, no energy is required, the solute moves in - to concentration gradient across the membrane, and Movement is -
simple, facilitated
there are two types of passive transport: - diffusion and - diffusion
transport, spontaneously, small
in simple diffusion, there is no - protein involved, Solute moves - right through the lipid bilayer in response to its concentration gradient, and Only -, non-charged solutes can pass through a membrane
transport, spontaneous, charged
in facilitated diffusion, a - protein is involved (usually channel but can be carrier). Movement still - and still goes with the concentration gradient, but there are special considerations for - solutes
electrochemical, negative, positive
if dealing with charged solutes in facilitated diffusion, you must consider the - gradient. Plasma membranes have a slight -charge on their “inside” side, which affects the movement of ions. For cations, the concentration gradient and electrical gradient is usually favorable, since it is - and attracted to the slight (second blank)
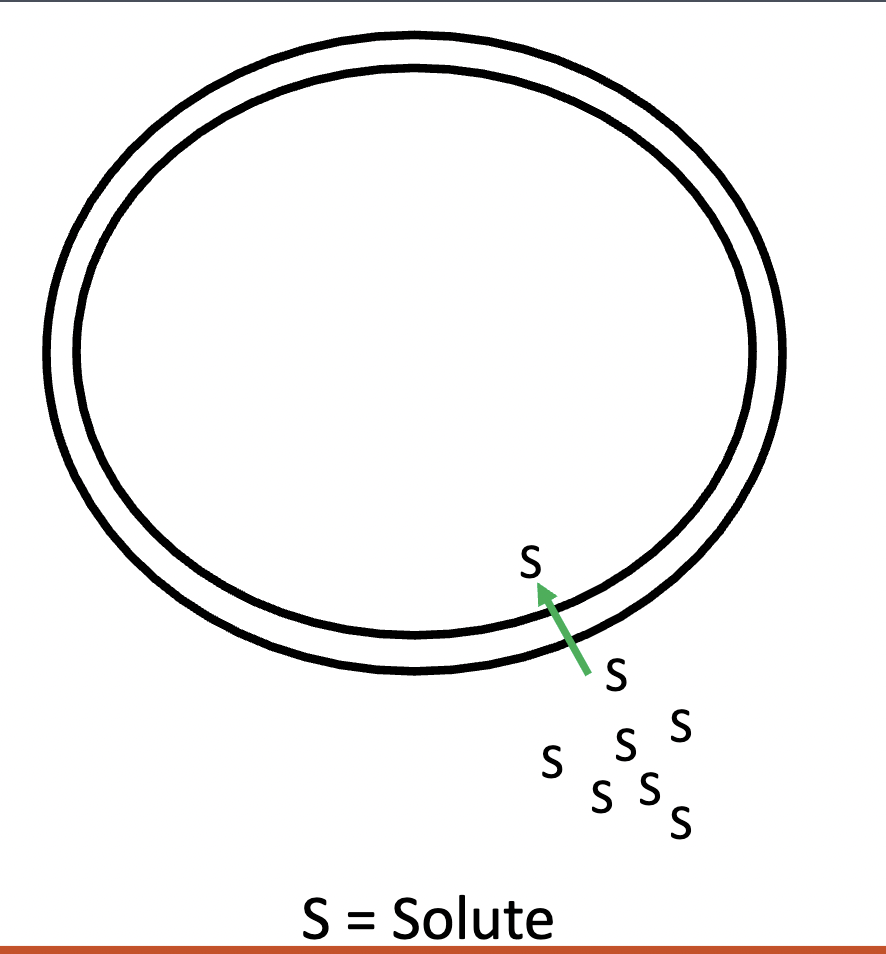
simple diffusion
this graph represents —, a passive transport type
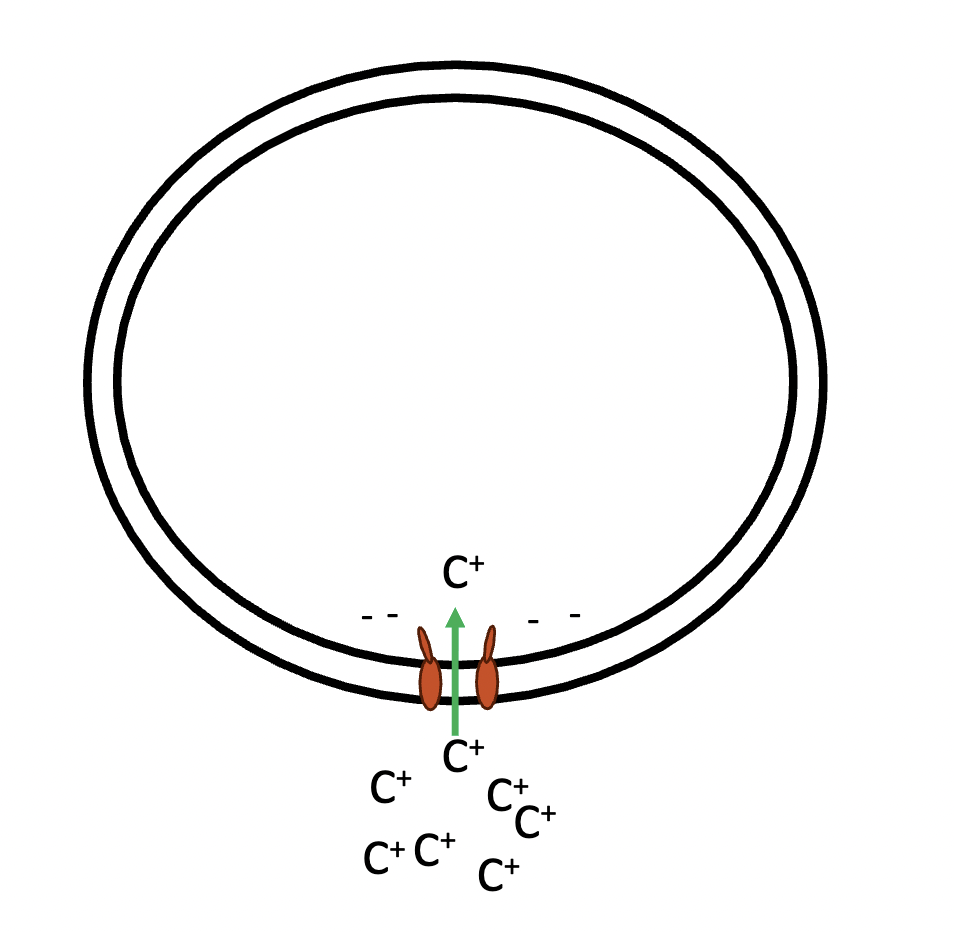
facilitated diffusion
this graph represents —, a passive transport type
active transport, energy
in —, Movement is against the electrochemical gradient, thus it requires an input of -, and is always mediated by carrier proteins.
primary active, symport, antiport
there are 3 types of active transport: — transport, secondary active -, and secondary active -
directly, non spontaneous
Primary active transport - utilizes energy to move something across a membrane, against their electrochemical gradient. Transport is mediated by carrier protein, and the Process is __
phosphate, electrochemical
an example of primary active transport is the cleaving of ATP into ADP + Pi (inorganic -) + energy. The resulting energy is used to drive hydrogen ions (H+) against their - gradient
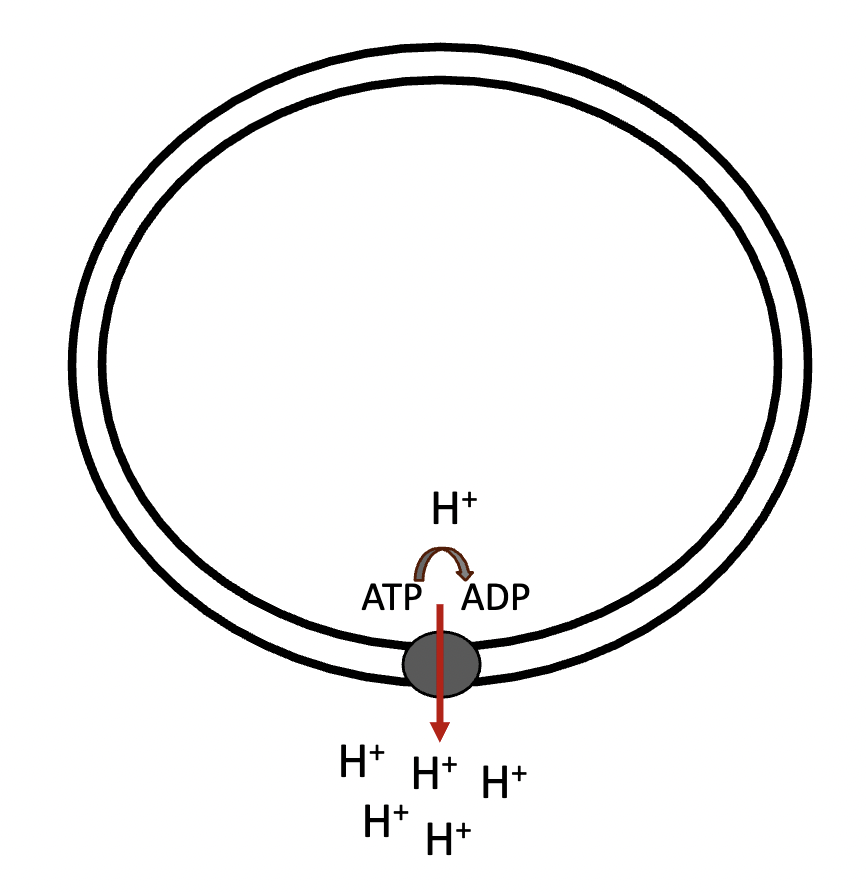
against, with
secondary active transport pairs the movement of a solute - its gradient with the movement of a solute - its gradient (usually hydrogen). It moves things via symport or antiport.
same, releases
in secondary active symport, there is movement of hydrogen ions and solute in the - direction, and the hydrogen’s movement with the gradient - energy that powers the movement of solute against its gradient
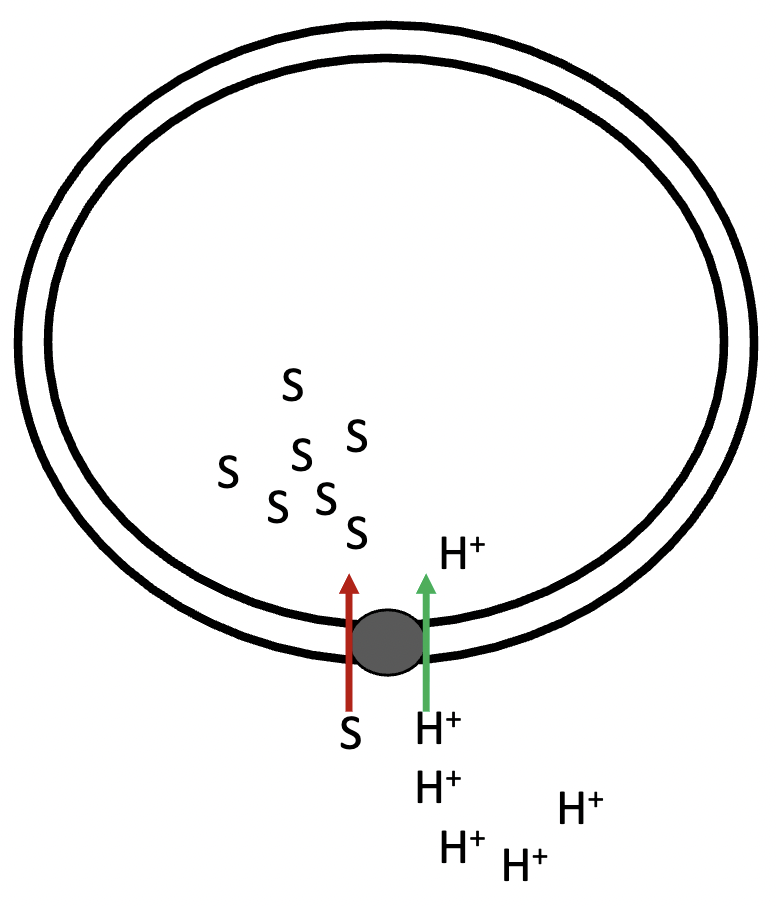
opposite, cation
in secondary active antiport, the hydrogen ions and solute move in - directions. A - is transported against its electrochemical gradient by pairing it with the movement of the hydrogen ion with its gradient
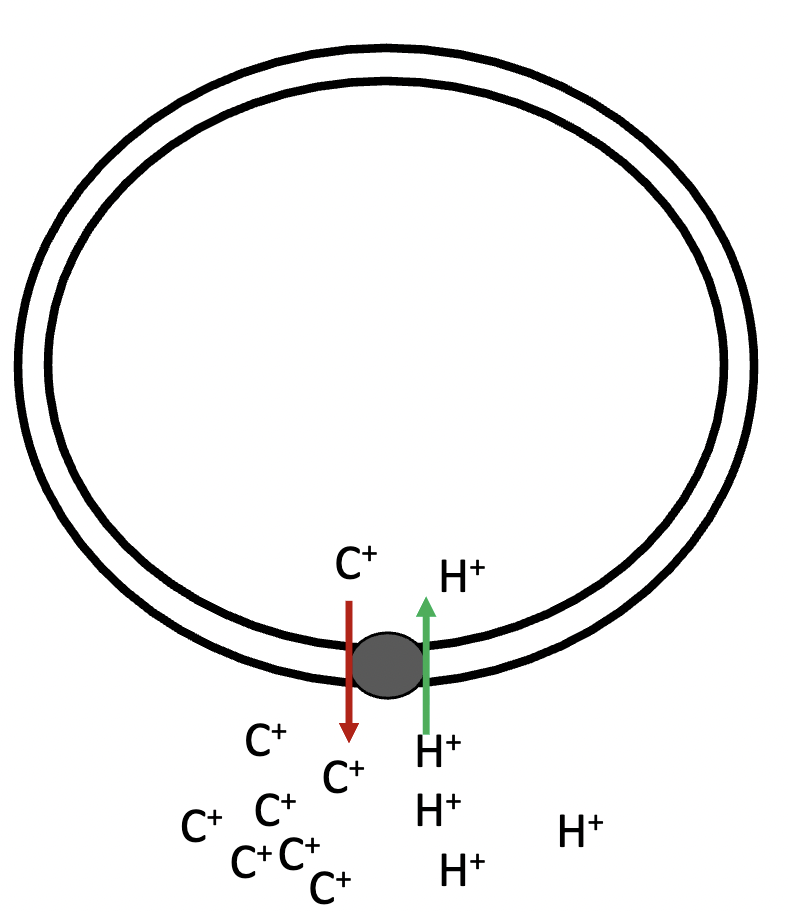
with, against
in both secondary active symport and antiport, the hydrogen ion is moving - its gradient, and this releases energy. this energy powers the transport of the solute - its gradient
ionic
The nutrients plants use are the charged, - forms of the elements
nitrate, ammounium
Some ions have one or more different charges and chemical forms, like nitrogen, which can be - (NO3-) or - (NH4+)
colloids
soil _ are tiny clay and humus particles, both inorganic and organic, that carry a slight electrical charge
adsorb, exchange
cation exchange capacity is the degree to which a soil can - and - ions, or a soil’s capacity to hold cations
cation exchange
Mineral nutrients adsorbed by the surface of soil particles can be replaced by other cations in the — process
leached, nutrient
Mineral cations adsorbed on the surface of soil particles Are not easily lost when the soil is - by water, and Provide a - reserve available to plant roots
larger, higher
A soil with higher CEC generally has a - reserve of mineral nutrients; has - negative charge, and can hold more cations
soil, surface
CEC is highly dependent on the - type and - area
repelled, soil solution
Mineral anions like nitrate (NO3-) and chloride (Cl–) tend to be - by the negative charge on surface of soil particles, thus remain dissolved in the — and are more succeptible to leaching
clay, humus, higher
Colloids adsorb pesticides as well as nutrients, and - and - tend to tie up many chemicals. You Often have to apply - chemical rates to high-CEC, clayey soils than to low-CEC, sandy soils
more, leaching, less
Smaller amounts of fertilizer, applied - often, are needed in low-CEC soils to prevent - losses. Larger amounts may be applied - frequently in high-CEC soils
hydrogen
The main thing that gets exchanged with nutrients at CE sites is -
pH
soil - is a measure of the acidity or alkalinity of the soil, or can be defined as hydrogen ion concentration
acids, water
When - dissolve in -, they release hydrogen ions. Strongly acidic solutions contain a lot of hydrogen ions.
combines, neutralizes, hydroxide
A base is a substance that - with hydrogen ions and - an acid. More - ions means a more basic solution
salt, charged
A product of the reaction of a base and an acid is a -. If it is dissolved in water, the individual components may become - again.
vigorous, larger
Nitrogen speeds growth, and plants receiving adequate amounts have - growth and - leaves
dark, pale
Plants make large amounts of chlorophyll, and on Nitrogen well-fed plants, leaves are - green. On poorly fed ones, they are - green
weaker, wind
Easily injured growth is encouraged when excess nitrogen is used, and plant stems are -, easily topple in rain, break in -
hardening off
Overly rapid growth from nitrogen also delays the _-_ process that protects many plants from cold, exposing them to winter damage
ammonium, nitrate
the only 2 forms of nitrogen that plants can absorb and utilize are - and - (NH4+ and NO3-)
nitrogen
_ is the most commonly deficient element in plant growing systems
chemicals, immobilized
Nutrients taken from soil by plants cannot be used by other plants, nor can - in the bodies of living microorganisms, animals, or fresh organic matter. The nutrients in living bodies or fresh OM are said to be -
immobilization
The removal of free nutrients from the soil by soil life is said to be -
ionic, mineralization
Plants need nutrients in simple, inorganic, - forms. Thus, plants cannot use immobilized nutrients until they have been changed to simple, inorganic forms by microbial decomposers. This process is called -
nitrogen fixation
— is the Conversion of atmospheric N2 into ammonia (NH3) by some types of bacteria. Ammonia is then quickly converted to
ammonium (NH4+)
Haber Bosch
To convert atmospheric nitrogen (N2) into ammonia (NH3), very high temperatures and pressure are necessary. This was industrially realized by the — process in 1913
Carl, Fritz
The Haber Bosch process was realized by the German chemists - Bosch and - Haber
anion, freely
nitrate is an - and requires active transport. It is not attracted to CE sites and moves -in the soil. Can be easily leached from the soil by precipitation or overirrigation