Combustion and oxidation of alcohols
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
What are some uses of alcohols
Fuels
Solvent
Disinfectants
Feedstock
What is the equation for the combustions of an alcohol
Using ethanol as an example:
C2H6O + 3O2 ⟶ 2CO2 + 3H2O
What can the OH groups within alcohols be oxidised by (oxidising agent)
It can be oxidised by strong oxidising agents, mainly use acidified potassium dichromate (VI)
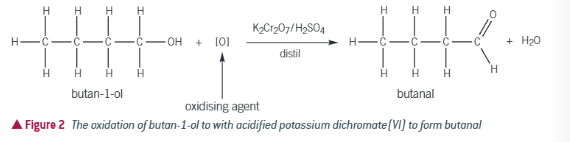
What forms when you partially oxidise a primary alcohol
An aldehyde will form (HO)
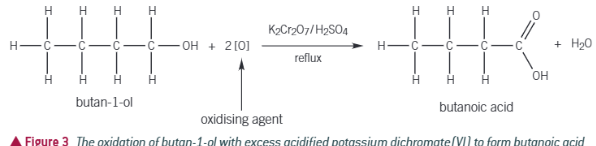
What forms when you fully oxidise a primary alcohol
A carboxylic acid will form ( COOH)
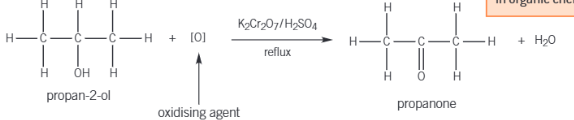
What forms when you oxidise a secondary alcohol
A ketone will form (double bond O to carbon)
Why can’t a tertiary alcohol be oxidised
Carbon in which the -OH group is attached to has no hydrogen which is required to form a carbonyl group
What are the conditions needed to form carboxylic acid in primary alcohol, and why are they needed
Needs to be heated under reflux and excess oxidising agent. As reflux prevents loss of reactants as once it is vapour it will condense and go back into product. Without it, product will evaporate fully. You use excess oxidising agent, as when aldehyde condenses again it will undergo further oxidisation to form the carboxylic acid.
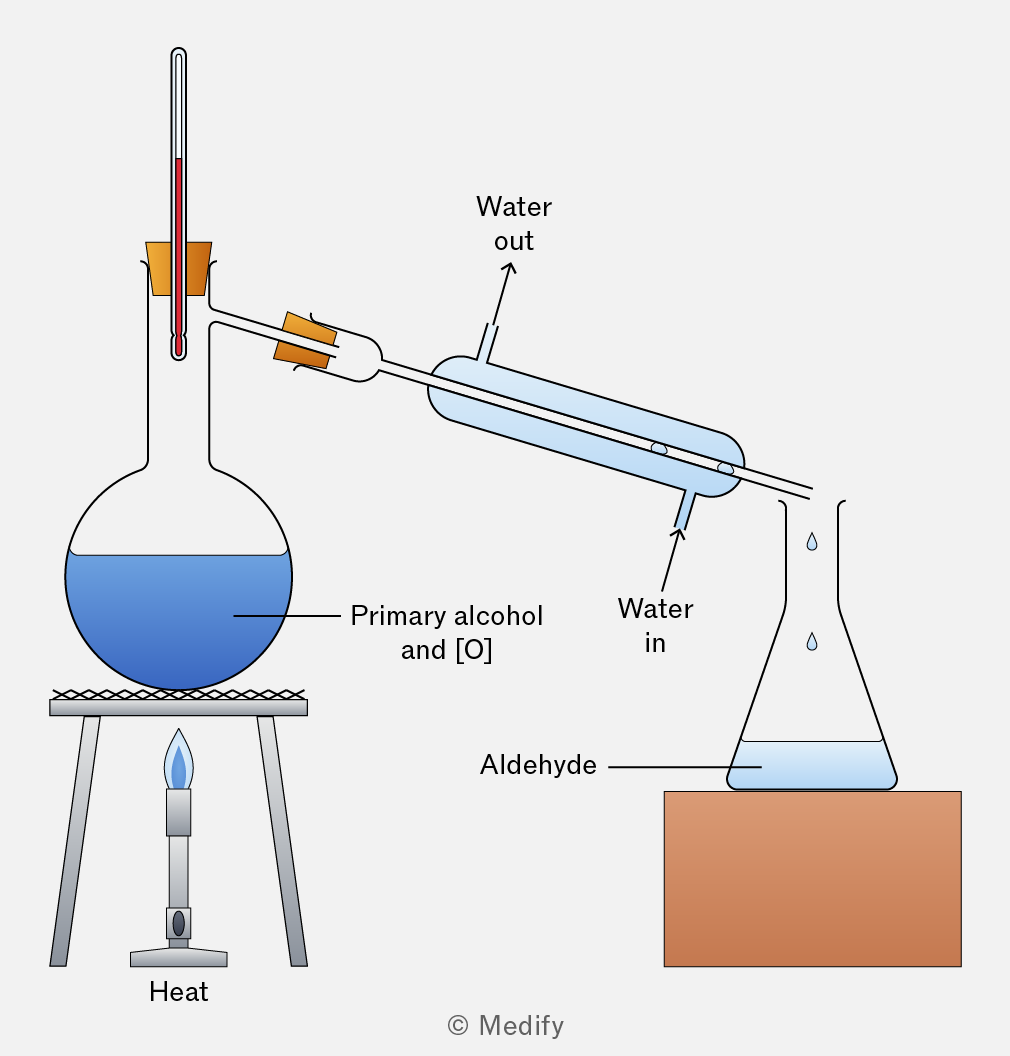
What are the conditions needed to form aldehyde in a primary alcohol and why
Alcohol oxidised with limited oxidising agent and then distilled immediately. Aldehyde will vaporise and is collected, preventing further oxidisation to a carboxylic acid.
What is the colour change when dilute sulfuric acid is added to potassium dichromate
Cr2,O72-(orange) → Cr3+(green)
Reflux diagram
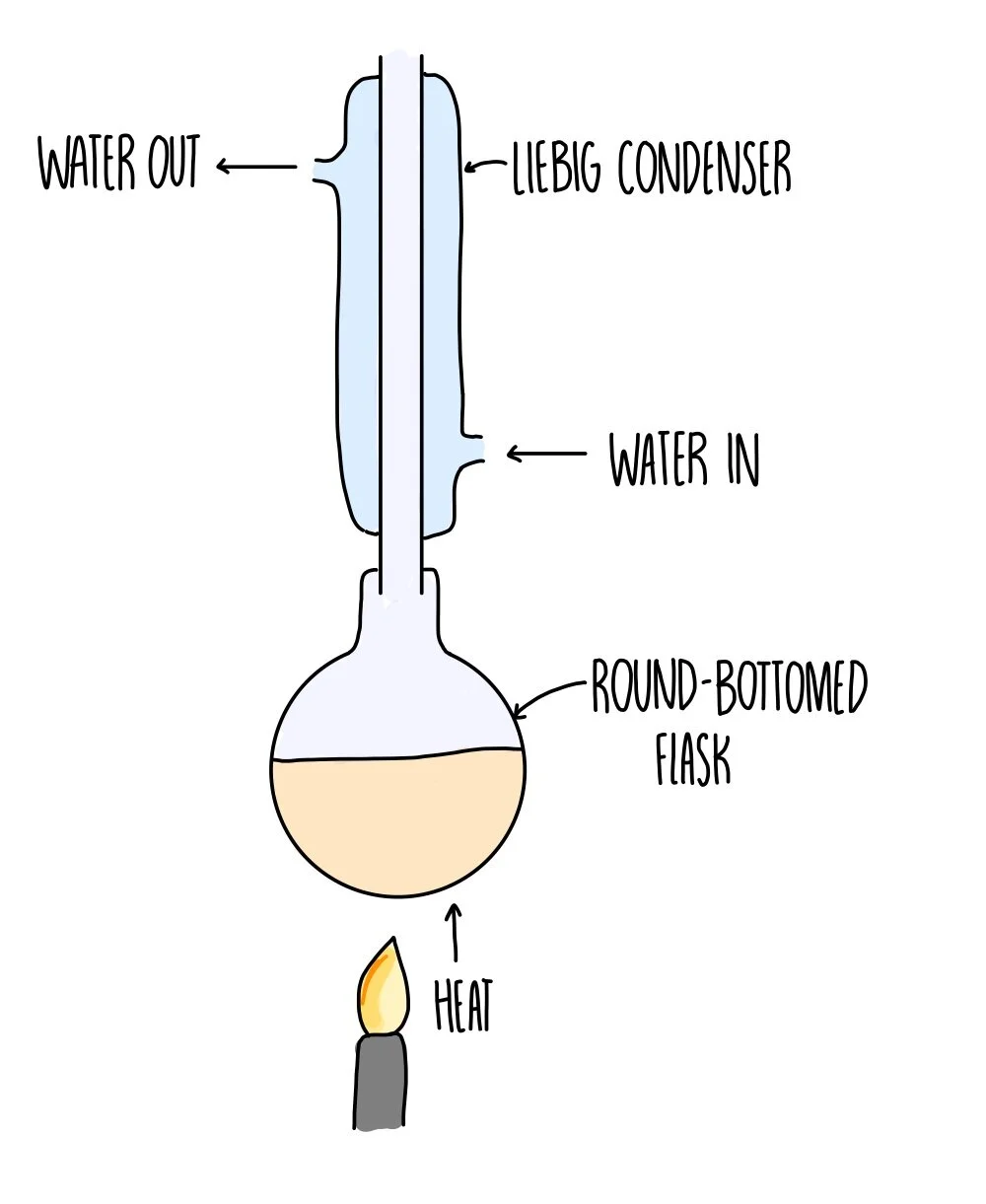
Distil diagram
Bent bit called still head