IB Bio year 2 Unit 7: Plants and water potential
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Adaptations for gas exchange in leaves
What processes do leaves carry out?
Respiration and photosynthesis
List why leaves need special adaptations
Adapted to exchange oxygen and carbon dioxide
Adapted to reduce water loss
Diagram of a leaf (Look at notes)
Adaptations of Leaf Structure
Waxy cuticle: The waxy cuticle covers the epidermis cells and reduces the evaporation of water from the leaf.
Epidermis: The epidermis provides protection for the mesophyll cells within the leaf. Epidermal cells are transparent, allowing light to reach the mesophyll cells where photosynthesis is carried out.
Spongy mesophyll: The irregular shape of spongy mesophyll cells increases the surface area for gas exchange. The spongy mesophyll cells are surrounded by air spaces.
Air spaces: Air spaces around spongy mesophyll cells facilitate the diffusion of gases between the surrounding atmosphere and the mesophyll cells.
Stomata: Stomata are pores which allow gases to enter and exit the leaf. Stomata are usually more common on the lower epidermis of the leaf. The stomata are open and closed by guard cells.
Veins/Vascular bundles: Veins provide support for the leaf. They contain xylem and phloem tissue. Xylem transports water and minerals from the roots. Phloem transports nutrients up and down the plant.
Transpiration
is the movement of water through a plant, and its evaporation through the stomata of the leaves.
is an inevitable consequence of gas exchange, as water in mesophyll cells evaporates, and diffuses through the open stomata.
Factors Affecting Transpiration
*Water diffuses from high concentration of water particles to low concentration of water
Light intensity: As light intensity increases, more stomata open. If there are more open stomata, then more water can diffuse out of the leaf, increasing the rate of transpiration.
Temperature: As the temperature increases, the water particles gain kinetic energy, and move faster. Faster moving water particles diffuse through the stomata of the leaf at a faster rate. Additionally, higher temperatures increase the rate of evaporation, which also increases the rate of transpiration.
Humidity: As humidity increases, the concentration of water outside the leaf increases. This decreases the concentration gradient between the inside and outside of the leaf. Water particles will diffuse slower, resulting in a slower rate of transpiration.
Air flow (wind): As air flows past the leaf, it moves water vapour away from the leaf, reducing the concentration of water outside the stomata of a leaf. This increases the concentration gradient, resulting in an increase in the rate of transpiration.
Stomatal Density
is the number of stomata per unit area of a leaf.
*Can use microscope to determine stomatal density
*A microscope with a graticule is required, so the the area of the leaf viewed under the microscope can be calculated.

Reliability of Quantitative Data
Quantitative data is any data which involves numbers.
Carrying out more replicates provides the scientist with greater confidence regarding the variation within data collected.
Water is the Medium for life
The medium for metabolic reactions (water is a solvent)
A transport medium
Involved in many chemical reactions
Ex) Hydrolysis reactions and photosynthesis
Water is a Polar Covalent molecule
Water is a polar covalent molecule because the oxygen atom forms two Covalent bonds with hydrogens
*Two hydrogen atoms form covalent bonds with an oxygen atom.
-The electrons are shared unequally between the oxygen and hydrogen atoms in water molecules.
-The oxygen atom has a more positive nucleus than the hydrogen atoms, and has a greater attraction for (the negatively charged) electrons.
Oxygen has a slightly negative charge (δ-)
Hydrogen has a slightly positive charge (δ+)
NOTE: If asked to draw two water molecules and a hydrogen bond, the hydrogen bond should be a dashed line. The covalent bonds between the oxygen and hydrogens in a water molecule should be solid lines.
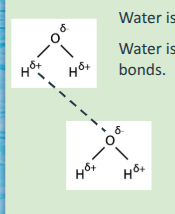
Cohesion and Adhesion
-Water molecules are cohesive and adhesive due to their polarity.
Cohesion
is attraction between the same kind of molecules, whereas adhesion is attraction between different kinds of molecules.
Water molecules are attracted to other water molecules. The negatively charged oxygen of one water molecule is attracted to the positively charged hydrogens of another water molecule, forming a hydrogen bond.
Is caused by hydrogen bonding!
Surface tension
is the property of a substance to resist an external force.
A relatively strong surface tension forms at the surface of water due to the cohesive nature of water molecules.
Surface tension allows animals like water striders to walk on water, and the surface of a lake becomes their habitat.
Cohesion and Transport in the Xylem
Xylem: vessels are thin narrow tubes which transport water (and dissolved minerals) from the roots to the leaves of plants.
The cohesive nature of polar water molecules allows water to form a continuous column of water molecules moving up the xylem/stem of plants.
Adhesive
The polar water molecules are attracted to other polar molecules, forming hydrogen bonds.
Adhesion - Capillary Action in Soil
Capillary action describes the ability of water to flow against gravity in a narrow space.
Water adheres to the polar soil particles and moves up the soil towards the roots of plants.-
Adhesion - Capillary Action in the Cell Wall
Cellulose in the cell walls of plants is polar.
Water can move from cell to cell by capillary action, as the polar water adheres to the polar cellulose.
Solvation
Solution: is a homogeneous mixture of a solvent and a solute.
Solute: dissolves in a solvent to form a solution. The solute is present in a smaller quantity than the solvent.
Solvent: is a substance in a which a solute is dissolved to form a solution.
Solvation: describes the interaction between a solvent and a solute. The solute particles interact with the solvent through various intermolecular forces such as hydrogen bonding.
What happens when a solute dissolves in water?
When a solute dissolves in water, the solute particles interact with water particles through hydrogen bonding.
What happens when Polar molecules dissolves in water?
When polar molecules dissolve in water, hydrogen bonds form between water molecules and the other polar molecules.
What happens when ionic compounds dissolve in water?
When ionic compounds dissolve in water, water molecules surround the ions of the compound.
The partially negatively charged oxygen atoms of water are attracted to positive ions (cations). The partially negative hydrogen atoms of water are attracted to negative ions (anions).
The attraction between water molecules and ions helps separate and disperse ions within a solution.
Water as the Universal Solvent
Water is often called the universal solvent, as it allows a wide variety of hydrophilic substances to dissolve in it, because of its polarity.
Hydrophilic substances are charged substances that mix and dissolve with water
*Most metabolic reactions will only occur when the reactants are dissolved in water.
Water is a Transport Medium in Plants
Ionic minerals are dissolved in water as it is transported up the xylem of a plant.
Organic molecules such as glucose and amino acids dissolve in water, and are transported through the phloem of plants.
Water is a Transport Medium in Animals
Nutrients
Oxygen
Carbon dioxide
Hormones
Waste products of metabolism
Antibodies
Heat
Water is the Medium for Metabolism
Metabolism is the complex network of interdependent and interacting chemical reactions occurring in living organisms.
Enzymes are biological catalysts which speed up the rate of chemical reactions. Enzymes are involved in controlling metabolism.
Metabolic reactions occur in aqueous solutions, with the reactants and enzymes dissolved in water.
Water Does Not Dissolve Hydrophobic Substances
Hydrophobic/nonpolar substances are not charged and do not readily mix with water.
Some molecules found in living organisms are hydrophobic, and do not dissolve in water. The functions of these substances depend on them being hydrophobic.
Examples:
Lipids are hydrophobic, allowing them to be used as an energy store without significantly changing the water potential of cells.
Physical Properties of Water: Buoyancy
Buoyancy is the upward force exerted by a fluid (liquid or gas) on an object immersed in the fluid
Liquid water is a denser fluid than air, providing greater buoyancy for aquatic animals and allowing them to float or swim more easily.
This helps many aquatic animals conserve energy and allows them to stay afloat without expending a lot of effort.
Ex) The ringed seal has a layer of blubber that provides buoyancy and helps it float on the surface of the water.
Physical Properties of Water: Viscosity
Viscosity is the measure of a fluid's resistance to flow. The higher the viscosity, the more difficult it is for animals to move through the fluid.
Water has a higher viscosity than air
Ex) Black-throated loon, have a streamlined body shape which allows them to smoothly move through water.
Physical Properties of Water: Thermal Conductivity
Thermal conductivity is a measure of a material's ability to conduct heat.
Water has a much higher thermal conductivity than air. Aquatic animals are more likely to lose heat to the environment, and must be adapted to reduce heat loss.
Ex)
Ringed seals have a layer of insulating blubber to prevent heat loss. Seals will huddle together out of the water (on land), decreasing overall exposed surface area and reducing heat loss.
Black-throated loons are insulated by feathers which are coated with a hydrophobic oil to keep the feathers dry, which reduces heat loss.
Physical Properties of Water: Specific Heat Capacity
Specific heat capacity is the energy required to raise the temperature of 1g of a substance by 1 K (or 1°C).
Water has a very high specific heat capacity (4.186 J/g°C), as energy is required to break the hydrogen bonds between water molecules. The specific heat capacity of air is much lower than water.
The temperature of large bodies of water is much more stable than air temperature, as the water can absorb or release much more heat energy without experiencing significant temperature change.
Animals cells (which are primarily composed of water) are also resistant to temperature change, which helps maintain constant body temperature in endotherms such as birds and mammals.
Physical Properties of Water - Ringed Seal
Buoyancy: The ringed seal has a layer of blubber which allows it to remain buoyant while in the sea, reducing the energy required to swim in the sea.
Viscosity: The streamlined body shape of the seals allow them to efficiently move through water. The flippers use drag to facilitate movement.
Thermal conductivity: The seal’s layer of blubber insulates the animal when in water. Seals tend to huddle, reducing exposed surface area and heat loss when on land.
Specific heat capacity: Ringed seals are endotherms and are adapted to maintain a constant body temperature. The high specific heat capacity of the water in their bodies helps maintain a stable body temperature.
Physical Properties of Water - Black-Throated Loon
Buoyancy: The black-throated loon is able to adjust its density by changing the volume of air in its air sacs. This allows the bird to control its depth when swimming in water.
The black-throated loon is able to increase its buoyancy in air by increasing its surface area by spreading its wings. The bones of birds are hollow, further reducing their density and increasing the buoyancy of the black-throated loon, allowing it to fly.
Viscosity: The streamlined shape of black-throated loons allow them to efficiently move through air and water. The webbed feet help the birds to move through water.
Thermal conductivity: The black-throated loon is an endotherm that maintains a constant body temperature. The bird’s feathers trap air and provide insulation. The feathers are covered in a hydrophobic oil which keeps the feathers dry.
Specific heat capacity: the high specific heat capacity helps the black-throated loon to regulate its body temperature, which is especially important when it is diving into cold water.
Why was water not present when planet Earth formed?
Temperatures were too high for water to exist as a solid(ice). Gaseous water would have moved away by the solar winds
How has Earth gained oceans/water?
In the outer solar system, temperatures were low enough de to distance from the sun so that ice formed objects such as comets and asteroids. Earth’s water probably arrived from asteroids, which have similar ratios of heavy hydrogen atoms as Earth’s water. This likely happened about 4 billion years ago.
Why does Earth not lose its water?
The large Earth creates enough gravity to retain water. The Earth is in a habitable zone with a temperature that allows for liquid water and for the water cycle to occur.
Relationship between the search for extraterrestrial life and the presence of water
Scientists believe that liquid water is required for life to exist on planets.
Scientists are currently searching for exoplanets with liquid water which may support extraterrestrial life.
It must be in the Goldilocks zone, where the temperature is neither too hot nor too cold for liquid water. “Just right”
Sexual Reproduction in Flowering Plants
Flowers contain the reproductive organs of flowering plants.
Many flowers contain both male stamens and female carpels.