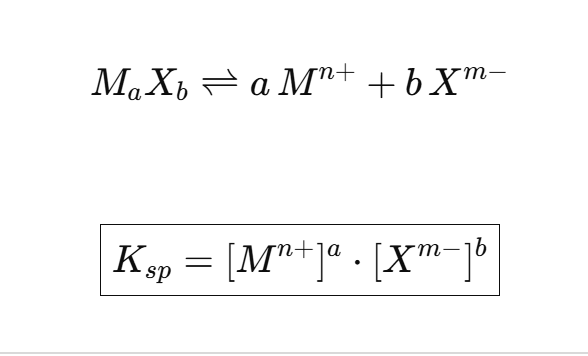Chem Exam 2
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
I.C.E Tables
Initial
Change in concentration
End/Equilibrium.
Kb
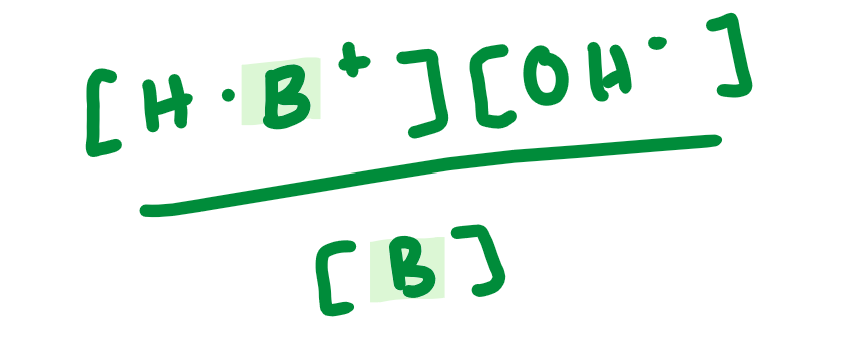
Ka
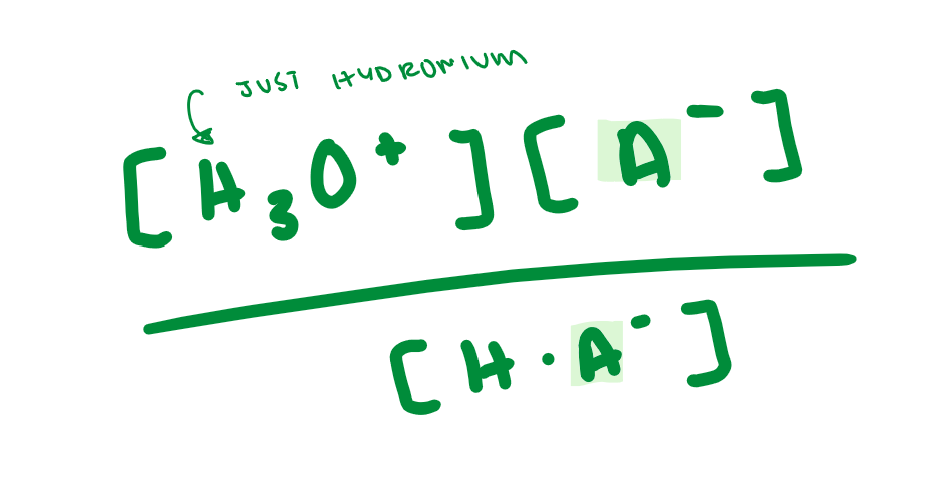
H+ = …
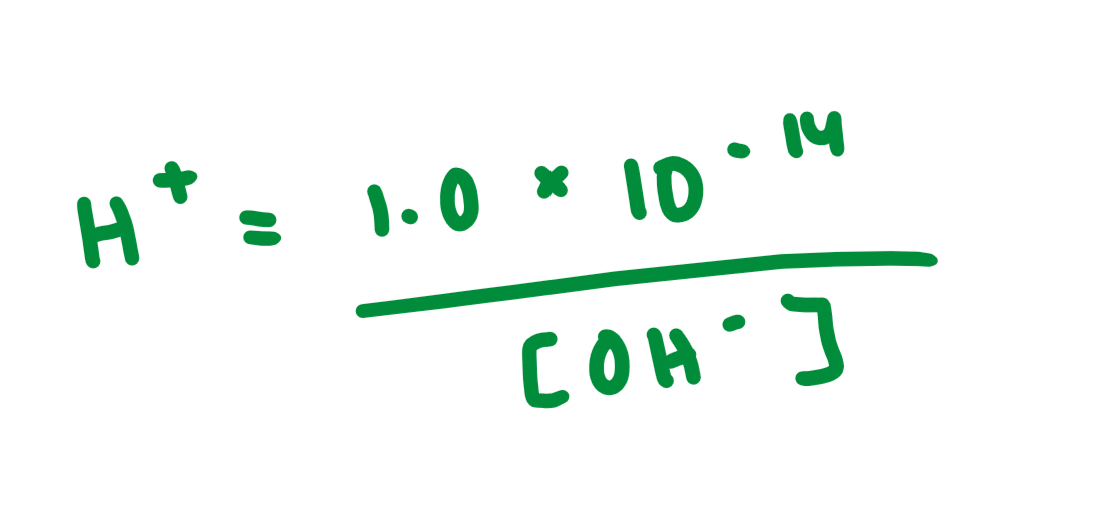
Kw
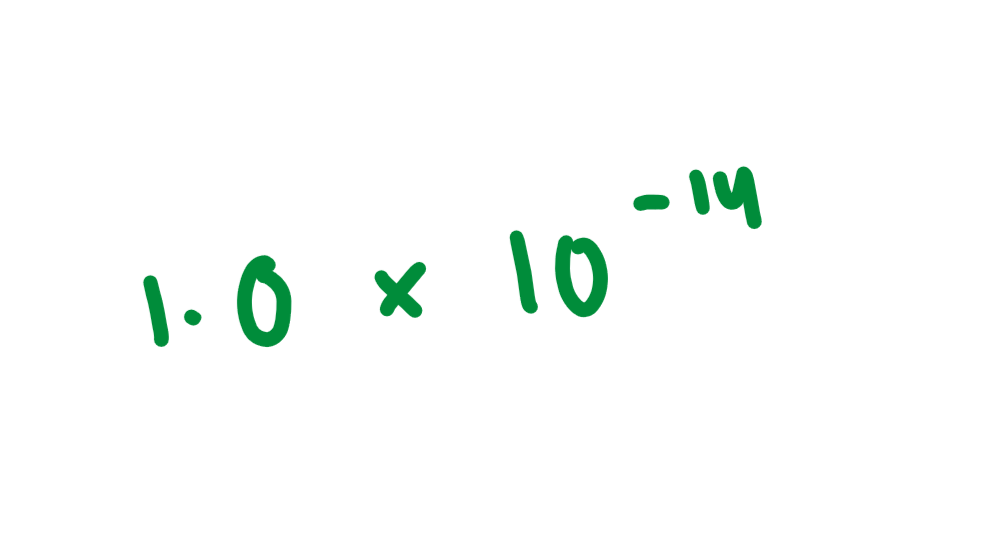
Steps to Acid-Base EQ
Dissociate completely.
Conduct main reaction w/ I.C.E table.
Find Kb
Plug in ICE table values equal to Kb
Solve for X
Conduct second reaction, determine if negligible.
Find final concentrations
Find H+
Kc
Equilibrium constant in terms of concentration. Brackets are concentrations and exponents on coefficients from balanced equation.
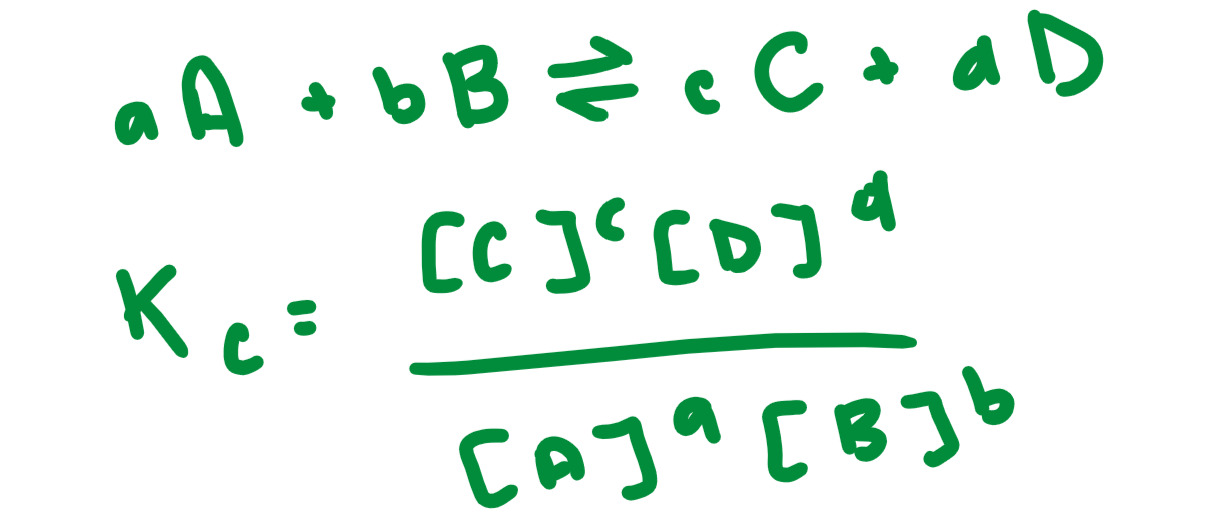
Concentration
moles per liter (mol/L)
Quadratic Formula
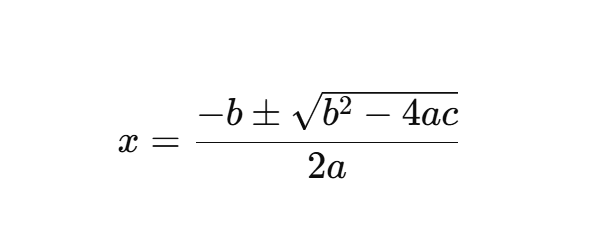
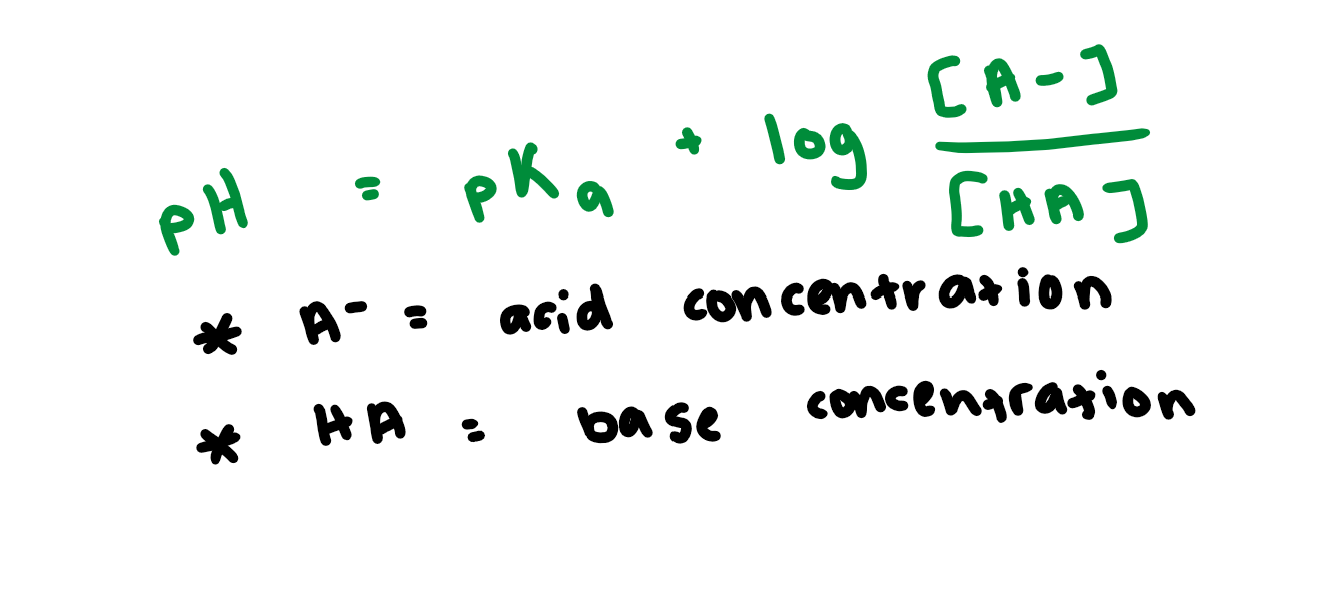
Henderson-Hasselbach EQ
Strong Acids (everything else weak)
HCl (hydrochloric acid)
HBr (hydrobromic acid)
HI (hydroiodic acid)
HNO₃ (nitric acid)
HClO₄ (perchloric acid)
H₂SO₄ (sulfuric acid — strong for first H⁺)
Strong bases (everything else weak)
(LiNaKoCaSrBa)
LiOH
NaOH
KOH
Ca(OH)₂
Sr(OH)₂
Ba(OH)₂
pH
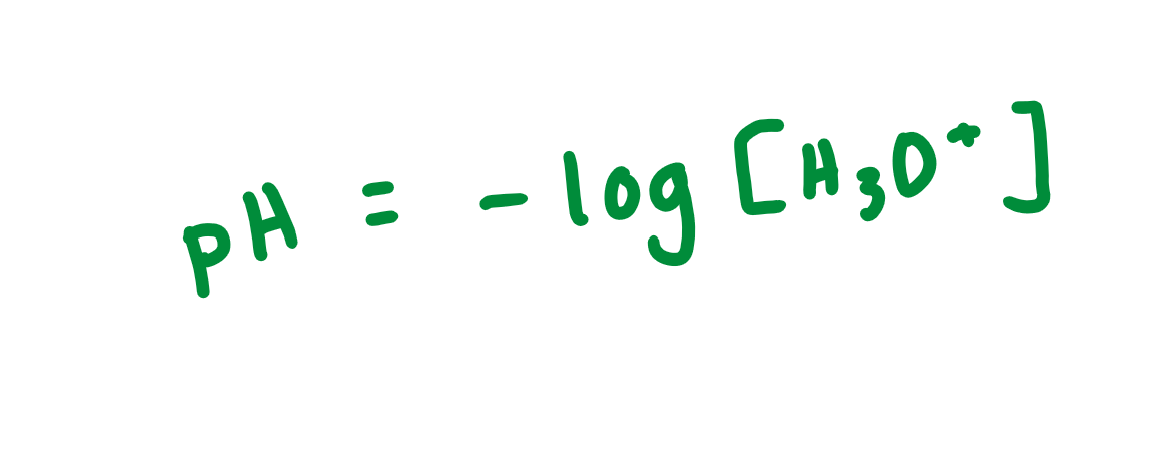
pOH
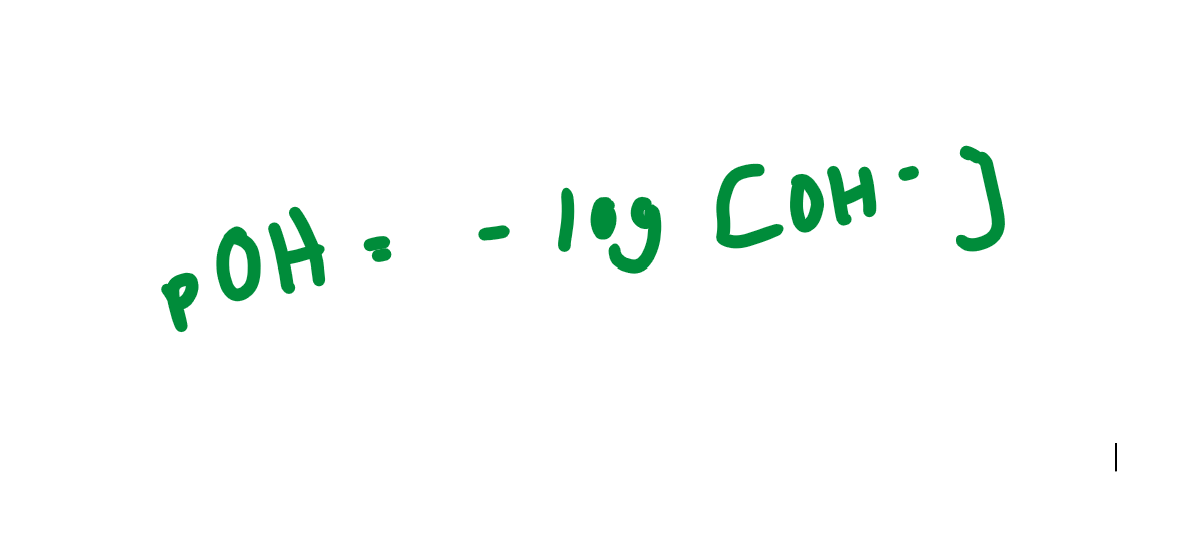
Brønsted-Lowry acid
Proton donor and creates conjugate base
Brønsted-Lowry base
Proton acceptor and creates conjugate acid
Conjugate base of weak acid: more acidic solution =
More soluble
Conjugate base of strong acid: more basic solution =
Less soluble
Increase solubility by…
Removing ions (Or add H)
Decrease solubility by…
Adding a common ion (or add OH)
Weak acid conjugate base
Acid increases solubility
Strong acid conjugate base
No effect
If X is basic from weak acid, solubility…
Increases
If X is neutral from strong acid, solubility…
Doesn’t change.
What determines if acid affects solubility
Whether anion reacts with H+
Larger Ka of conjugate acid means weaker what and less what effect?
Weaker base and less pH effect.
Higher Ksp
More soluble
Cation
Positive (left side of table)
Anion
Negative (right side of table)
Ksp
Ksp=∏(ion concentration)coefficient
Multiply all ions
Raise to power of their coefficient
Ignore the solid