Cell bio exam 3
1/70
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
71 Terms
CYTOSKELETON
Direct movement of things inside the cell
Muscle contraction
Cell division
Cytoplasmic streaming
Provide structure and shape to the cell
Allow the cell to move the membrane
THREE TYPES OF CYTOSKELETON
Intermediate filaments (cytoplasm and nucleus)
Microtubules (radiate from a primary location to reach many part of the cells)
Actin (microfilaments)(throughout the cells but concentrated along cell periphery and in processes)
Intermediate
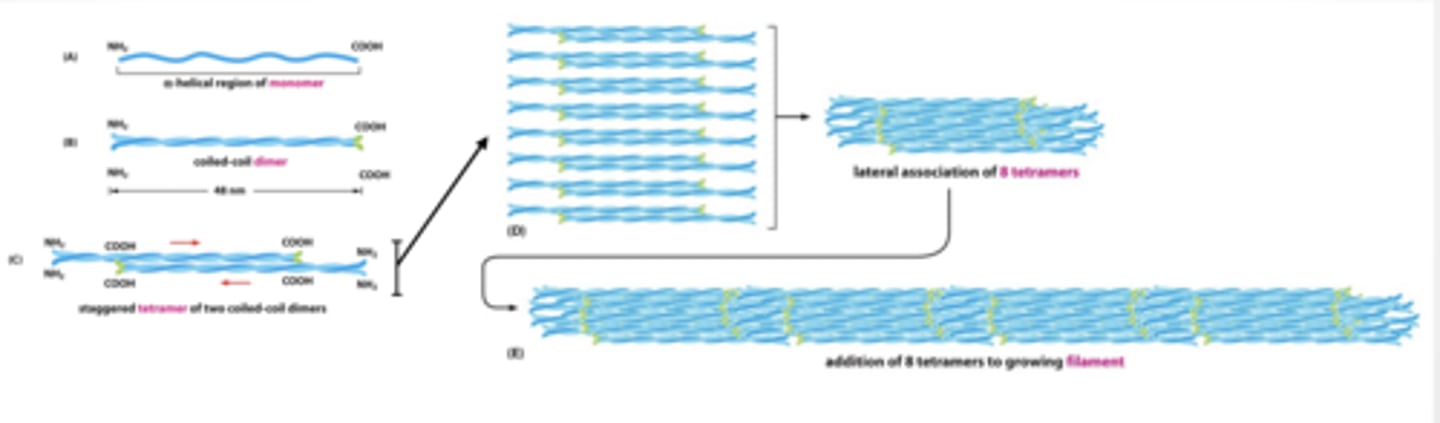
INTERMEDIATE FILAMENTS FORM LONG ROPE LIKE STRANDS OF STACKED TWISTED PROTEIN
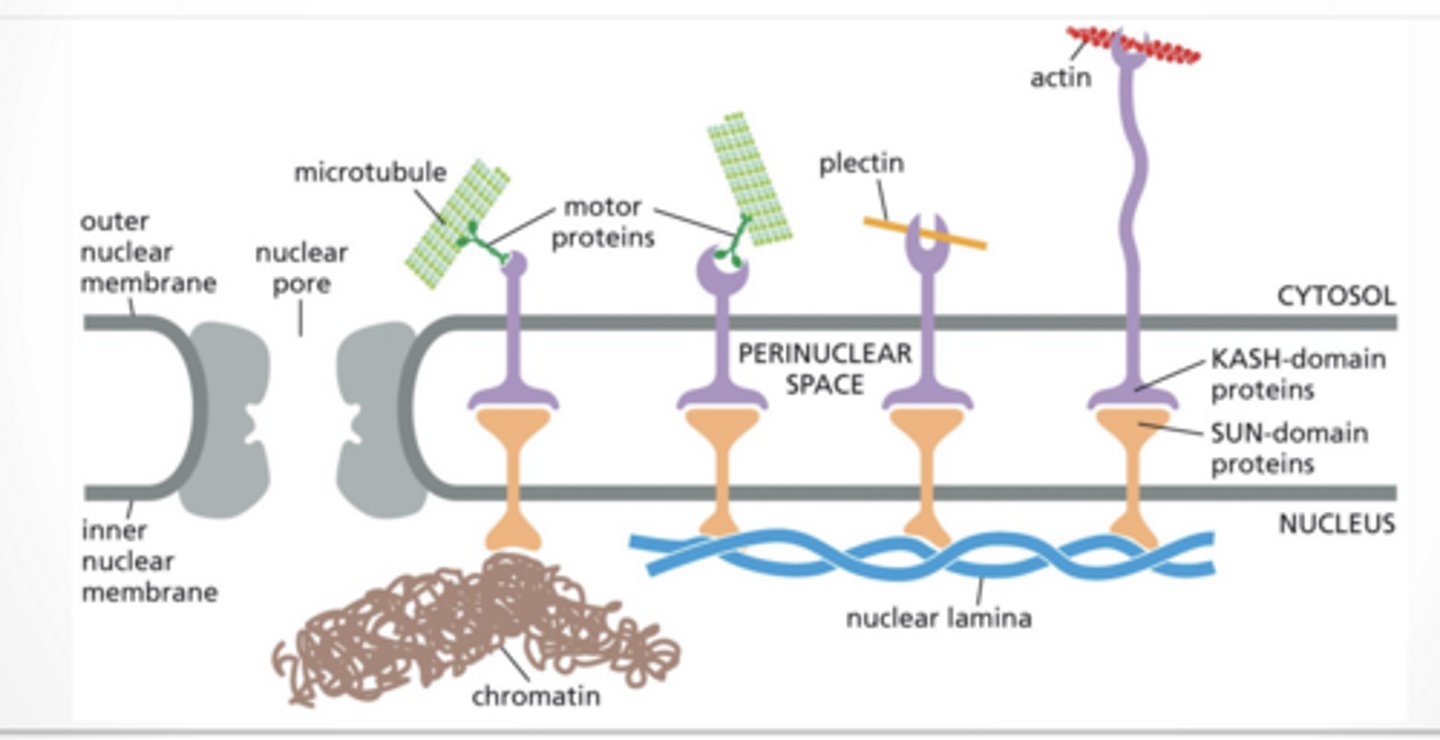
INTERMEDIATE FILAMENTS PROTECT FROM MECHANICAL STRESS Nucleus is surrounded and supported by the nuclear lamina
Intermediate filaments called lamin for lamina
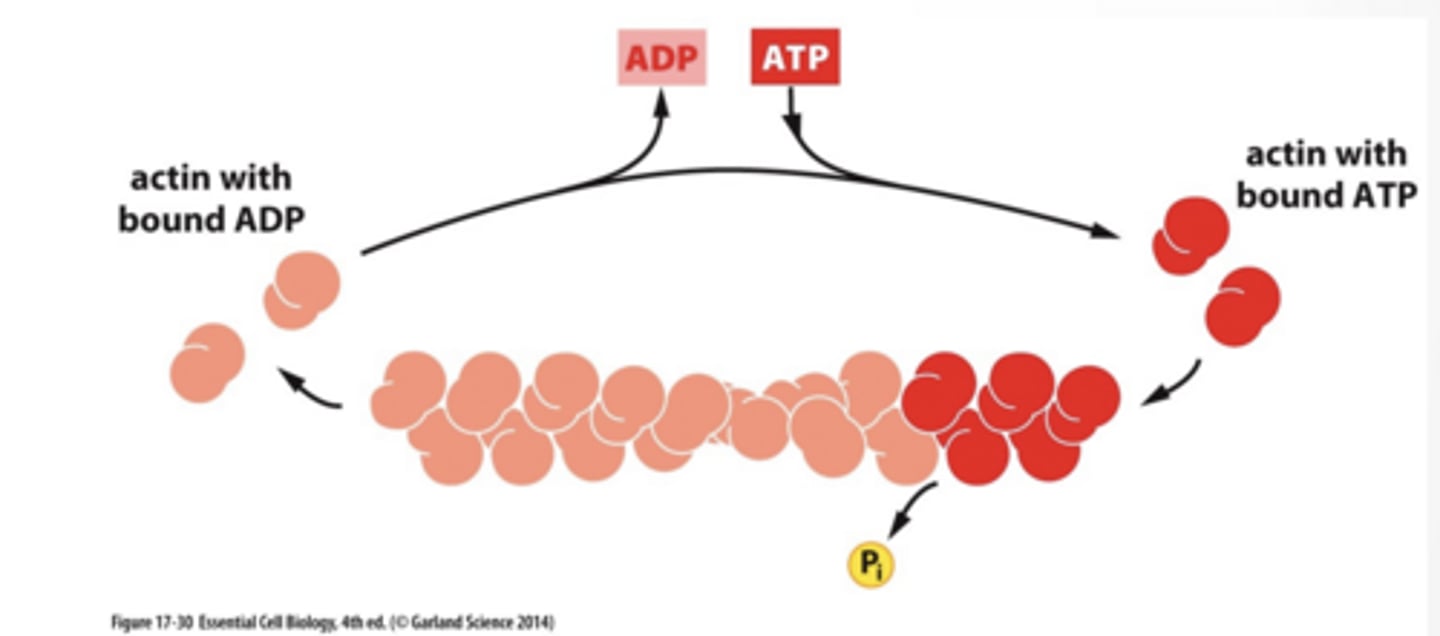
ACTIN ASSEMBLY IS DRIVEN BY TREAD MILLING
Monomers assemble into a strand
ATP bound actin binds to growing strand actin hydrolyzes ATP to ADP reducing binding affinity for other actin high monomer concentration drives assembly at both ends ADP bound actin disassembles
MANY CELL SHAPES ARE DRIVEN BY ACTIN
ACTIN IS USED TO SUPPORT CELL MOVEMENT
Actin assembly pushes the cell membrane into long protrusions
Lamellipodia
Actin filament formation drives the membrane forward by interaction proteins on the membrane
Actin filaments interact with adhesion molecules to attach membrane to substrate
MICROTUBULES
MICROTUBULES Have directionality plus and minus end
Tubulin isoforms assemble to form filament
alpha and beta dimers form filament
gamma forms stable base
MICROTUBULES ASSEMBLY IS GOVERNED BY DYNAMIC INSTABILITY
GTP bound alpha/beta dimers assemble onto growing + end of microtubules
alpha/beta dimers naturally hydrolyze GTP to GDP
GDP bound alpha/beta dimers interaction is not as strong as GTP bound dimers
Microtubules collapse is called catastrophe
CYTOSKELETAL ELEMENTS INTERACT WITH EACH OTHER
HOLDING THE NUCLEUS IN PLACE
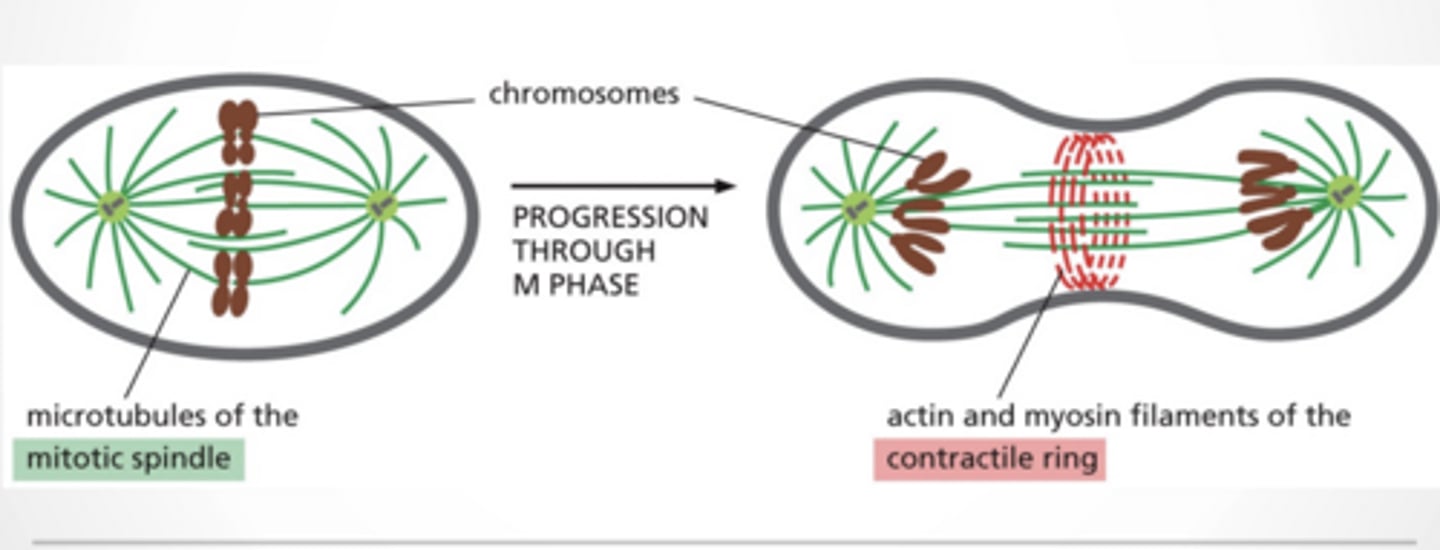
CELL DIVISION
Microtubules = mitotic spindle
Actin + myosin =
Physically split the cell
Form contractile ring
Tighten like a belt
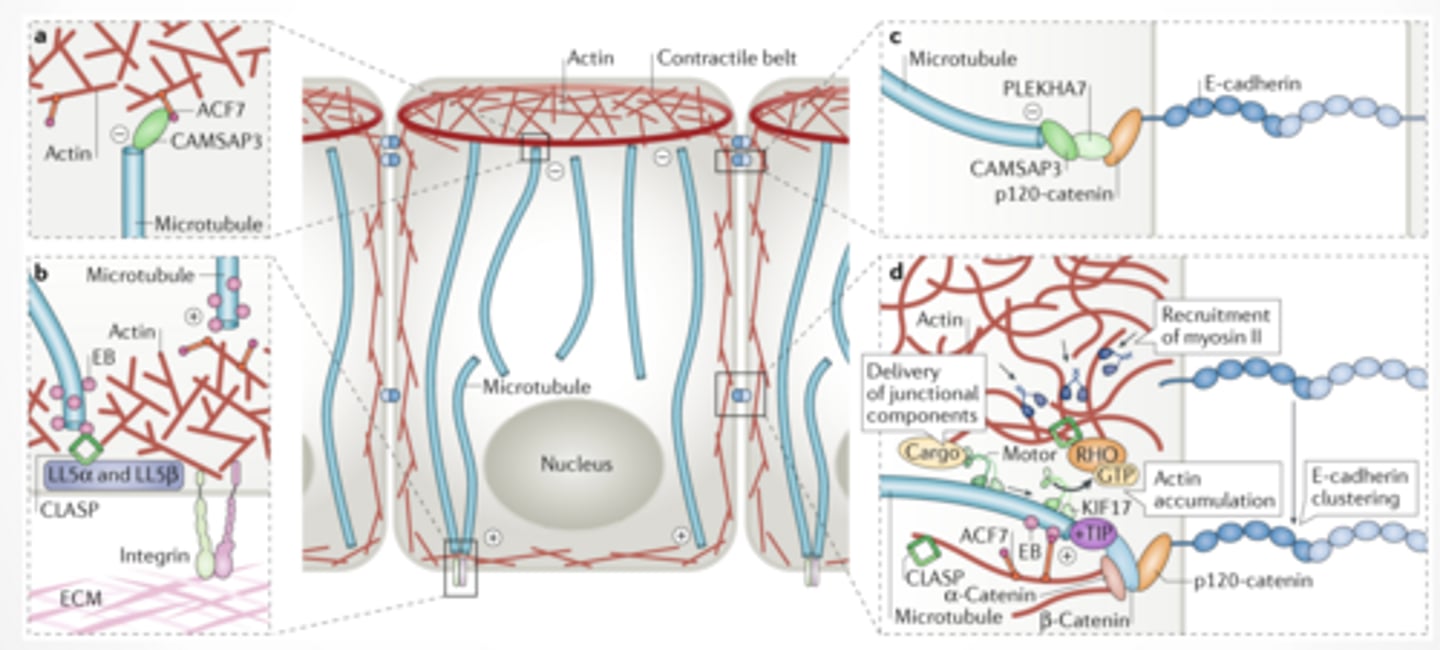
ACTIN + MICROTUBULES IN CELL POLARITY
Microtubules:
Deliver materials (vesicles)
Define direction
Actin:
Forms cortex (outer structure)
Controls shape + tension
Adhesion proteins:
Anchor cells to each other
Cell division feet
feet
ACTIN AND MICROTUBULES INTERACT IN CELL POLARITY feet
ACTIN AND MICROTUBULES INTERACT IN CELL POLARITY feeet
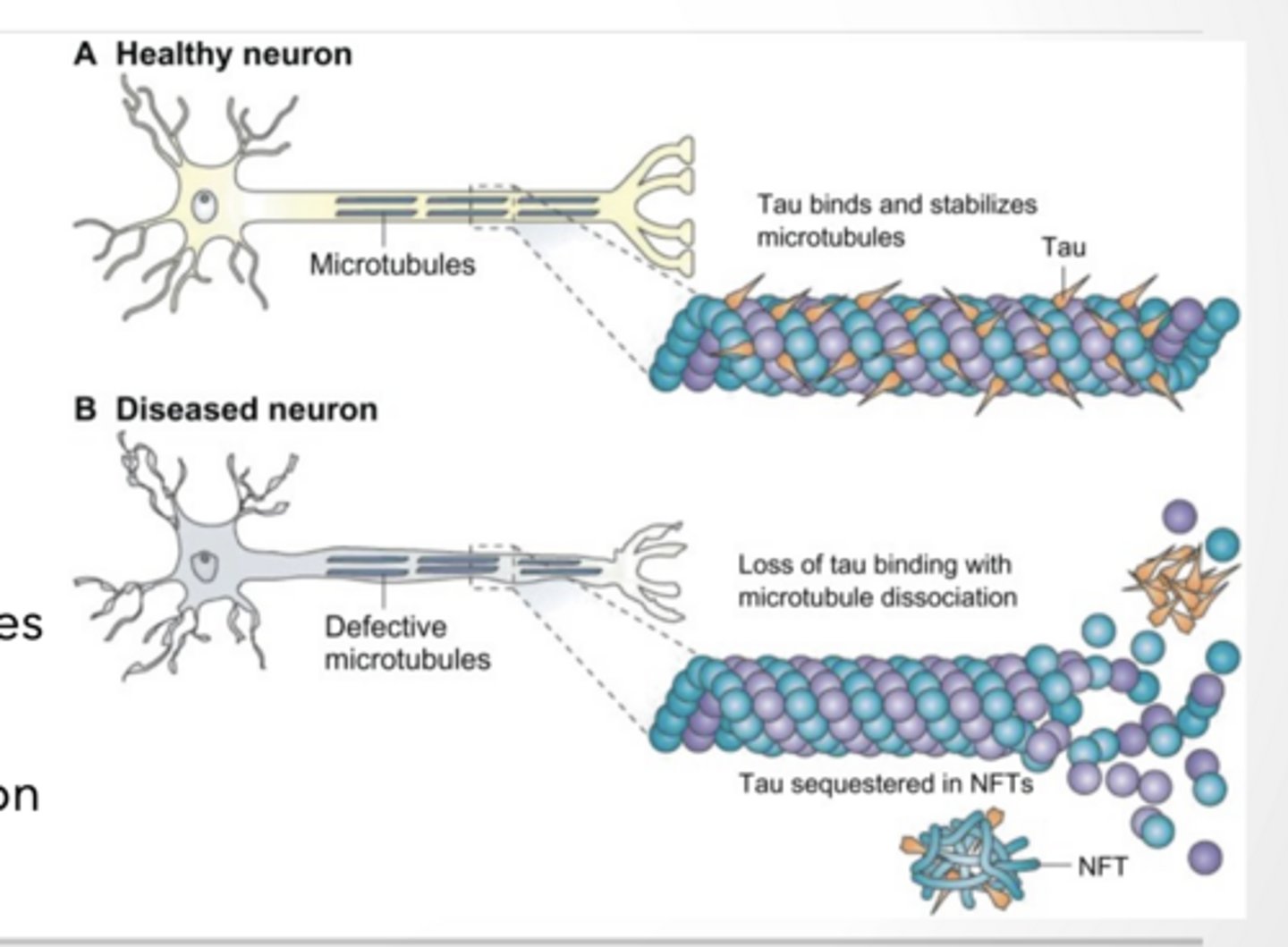
PROTEINS CAN STABILIZE MICROTUBULES TO PREVENT CATASTROPHE
And Actin FIbers
Stabile microtubules are important in transport and cell division
Microtubules are rigid, hollow protein rods, roughly 25 nm in diameter, that form a key component of the eukaryotic cytoskeleton
Tau stabilizes microtubules
over phosphorylation can lead to neurodegeneration
Other molecules will cleave microtubules
These interactions can be facilitated or inhibited by post translation modification of tubulin dimer
Tubulin is building block of Microtubules
ACTIN FIBERS CAN TAKE MANY SHAPES
Actin filaments are thin and flexible
Important in cell movement and muscle contraction Lots of proteins regulate actin structure and polymerization
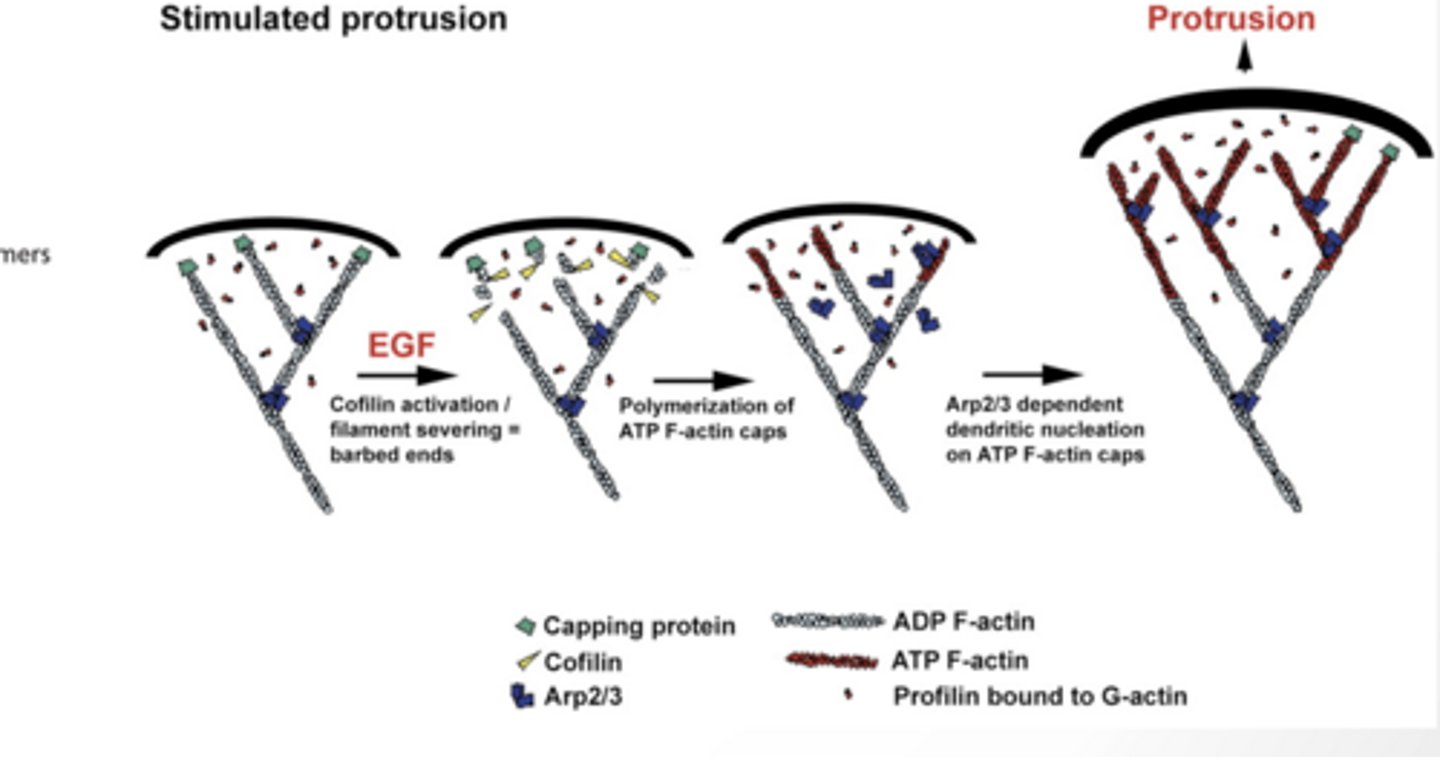
ROLE OF ACTIN MODIFYING PROTEINS IN CELL MOVEMENT
Cofilin cuts old filaments → creates new ends (“barbed ends”) + ends
ATP- F actin adds to those ends → filament grows
Arp2/3 makes branches → creates a web network
This pushes the membrane forward (protrusion)
Meanwhile, old filaments behind get broken down
CELL SIGNALING CAN ALTER ACTIN STRUCTURE
Structure is determined by interaction with regulatory proteins
What interactions occur are determined by cell signaling pathways
DRUGS AFFECT FILAMENTS
Binds and stabilizes microtubules
Binds tubulin dimers and prevents their polymerization
TABLE 17–2 DRUGS THAT AFFECT FILAMENTS
Actin-specific Drugs — Action
Binds and stabilizes filaments
Caps filament plus ends, preventing polymerization there
Binds actin monomers and prevents their polymerization
HOW DOES PACLITAXEL WORK TO TREAT CANCER?
Paclitaxel (Taxol) works by freezing microtubules in place
Normally: microtubules constantly grow and shrink
This is required for mitosis (cell division)
During anaphase, microtubules depolymerize, acting as the pulling force to move chromosomes to opposite poles.
Paclitaxel:
stabilizes microtubules (they can’t break down)
spindle fibers get stuck
Result: Cells get stuck in mitosis → can’t divide → die
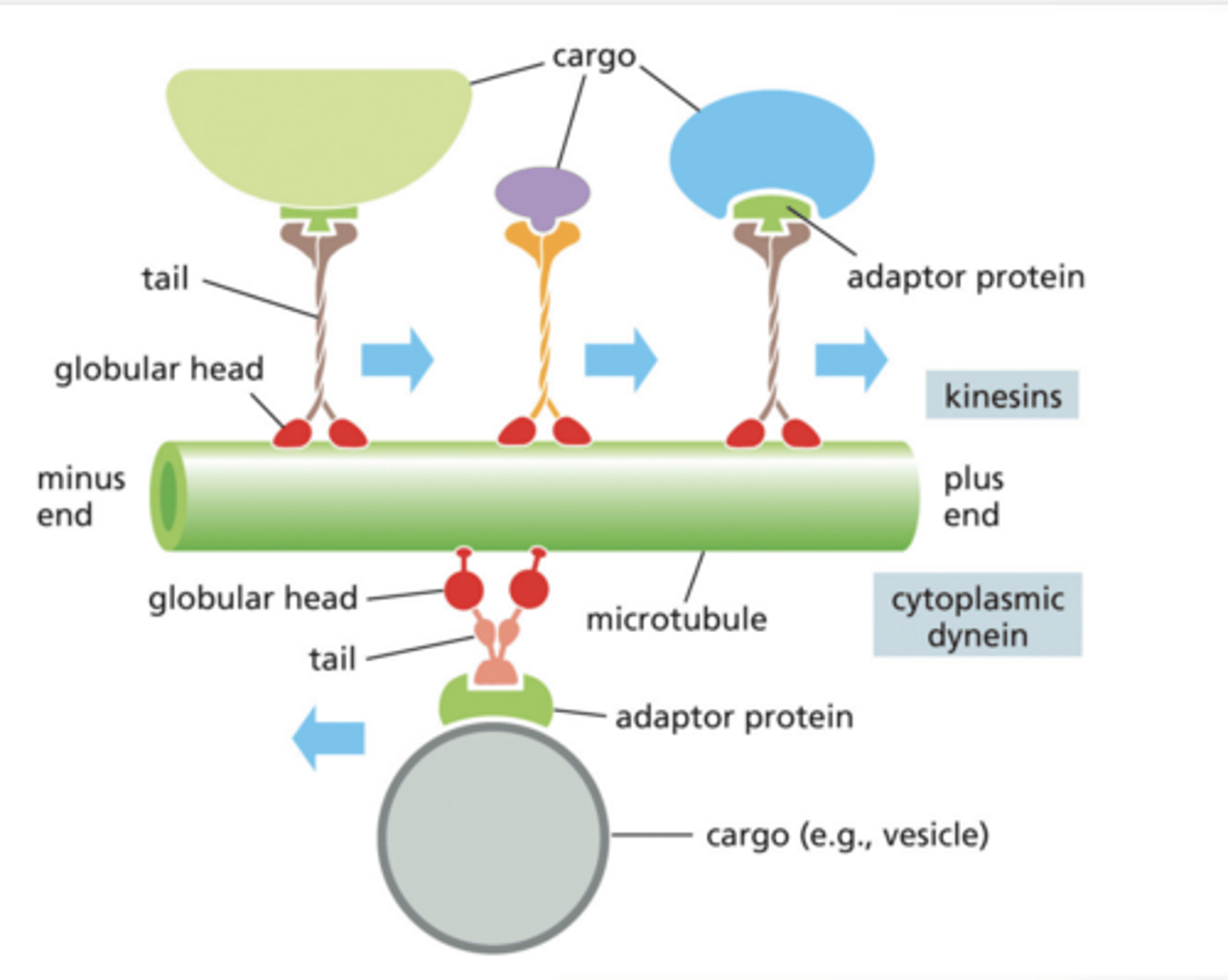
MOTOR PROTEINS CAN CARRY CARGO IN TWO DIRECTIONS ALONG MICROTUBULES
Microtubules are like train tracks
Motor proteins are like delivery trucks
Kinesin → goes to + end (usually toward cell edge)
Dynein → goes to – end (usually toward nucleus)
Globular Head = walks on microtubule (uses ATP)
Tail = holds cargo
Adaptor protein = connects cargo to motor protein
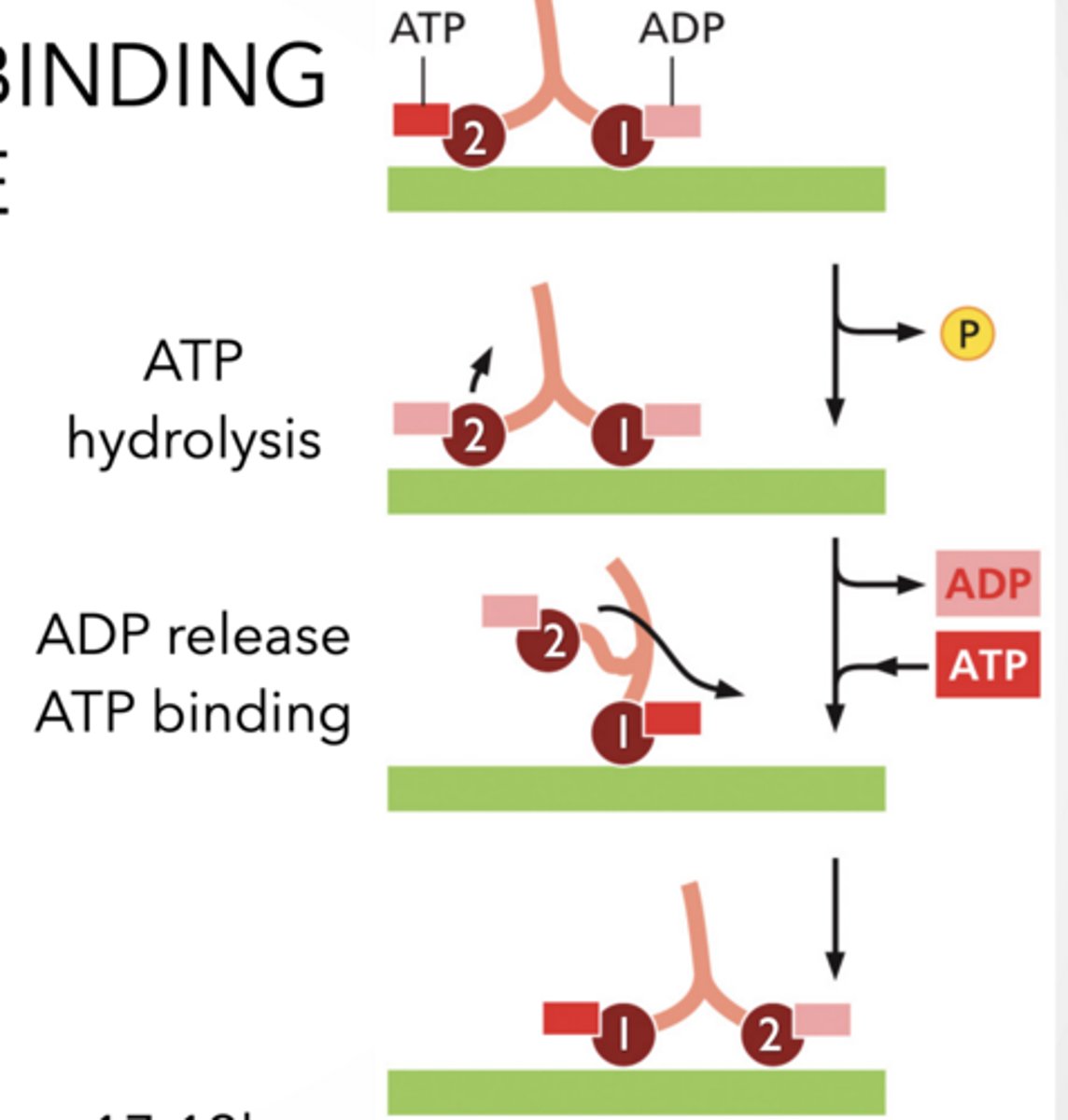
ATP HYDROLYSIS AND BINDING ALTERS PROTEIN SHAPE Hydrolyzing ATP provide energy to lift leg
P binds to other leg to lock it
Energy generated from ATP hydrolysis makes reversal energetically unlikely
Binding of ATP swings leg forward.
Compare to the BCR-ABL arm that swings in and out without ATP - low energy needed for shift
THIS IS HOW THE MOTOR PROTEINS " walk"
walking Feet
feet
MOTORS PROTEINS,
MICROTUBULES ARE USED TO TRANSPORT CARGO AROUND THE CELL
Dynein and Kinesin are ATP binding motor proteins
Dynein moves to the minus end
Kinesin moves to the plus end
Assembly and stabilization of microtubules drives movement of chromosomes captured by the ends of microtubules
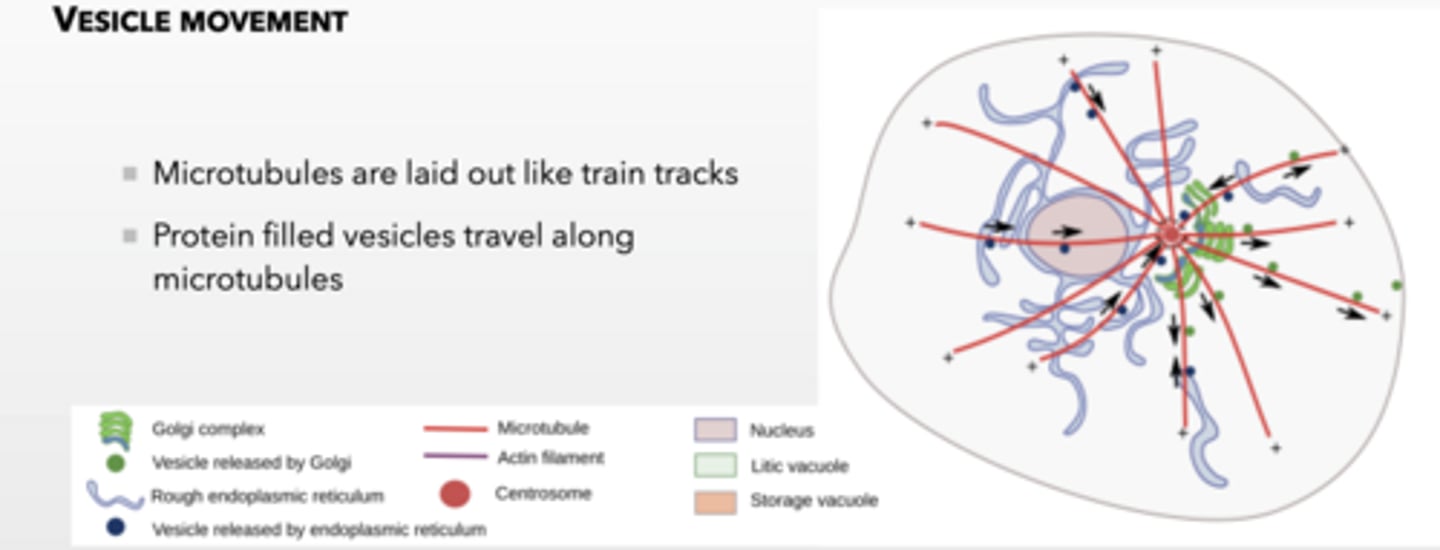
MICROTUBULES ARE USED TO TRANSPORT CARGO AROUND THE CELL
Microtubules are laid out like train tracks
Protein filled vesicles travel along microtubules
MICROTUBULES ARE USED TO TRANSPORT CARGO VESICLES AROUND THE CELL using these proteins
Vesicle movement feet
feet
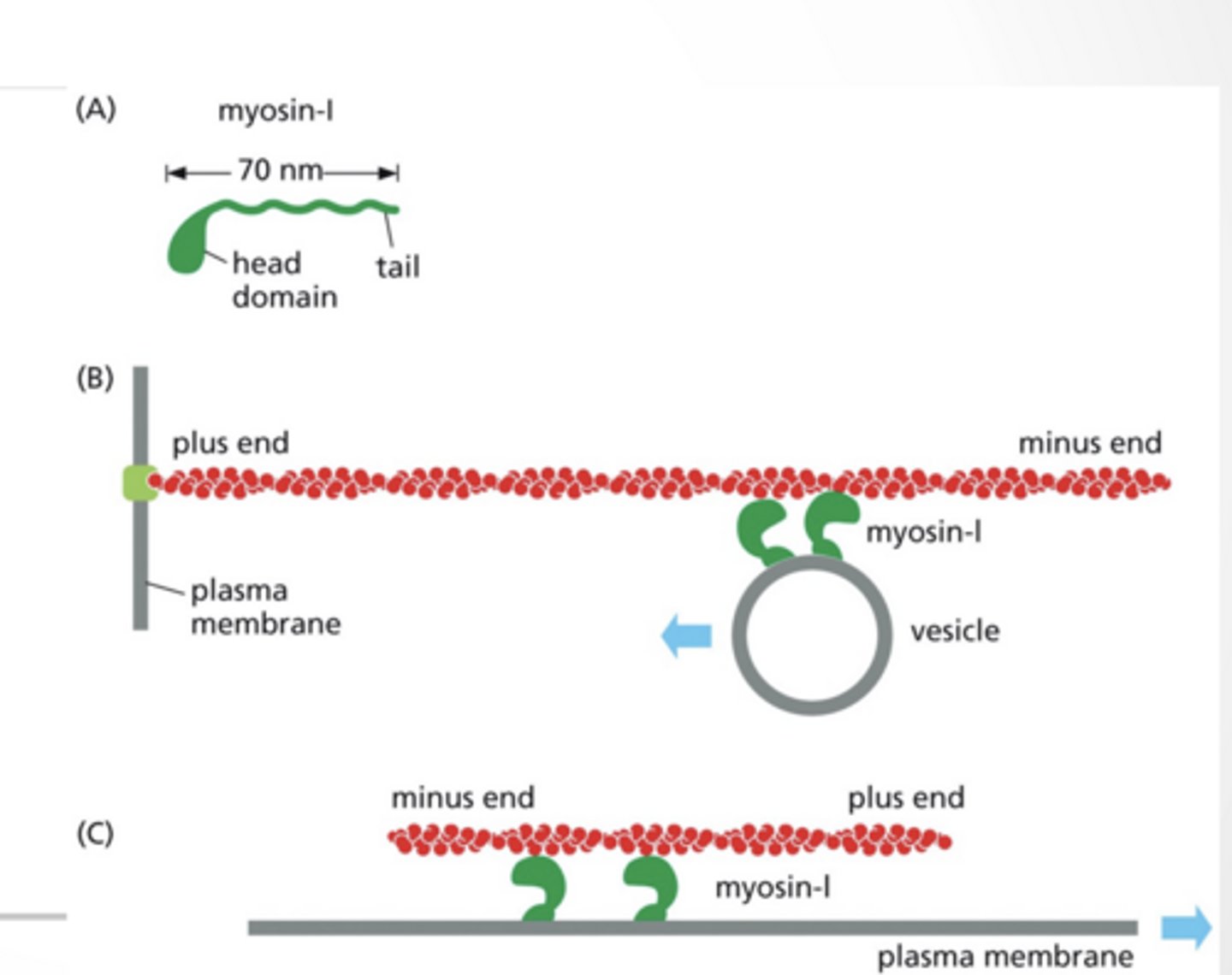
MYOSIN IS A MOTOR PROTEIN FOR ACTIN
Myosin-I is a simple motor protein with 1 head
Move vesicles along a fixed actin filament
Myosin can be fixed and move an actin filament
toward the + end of actin
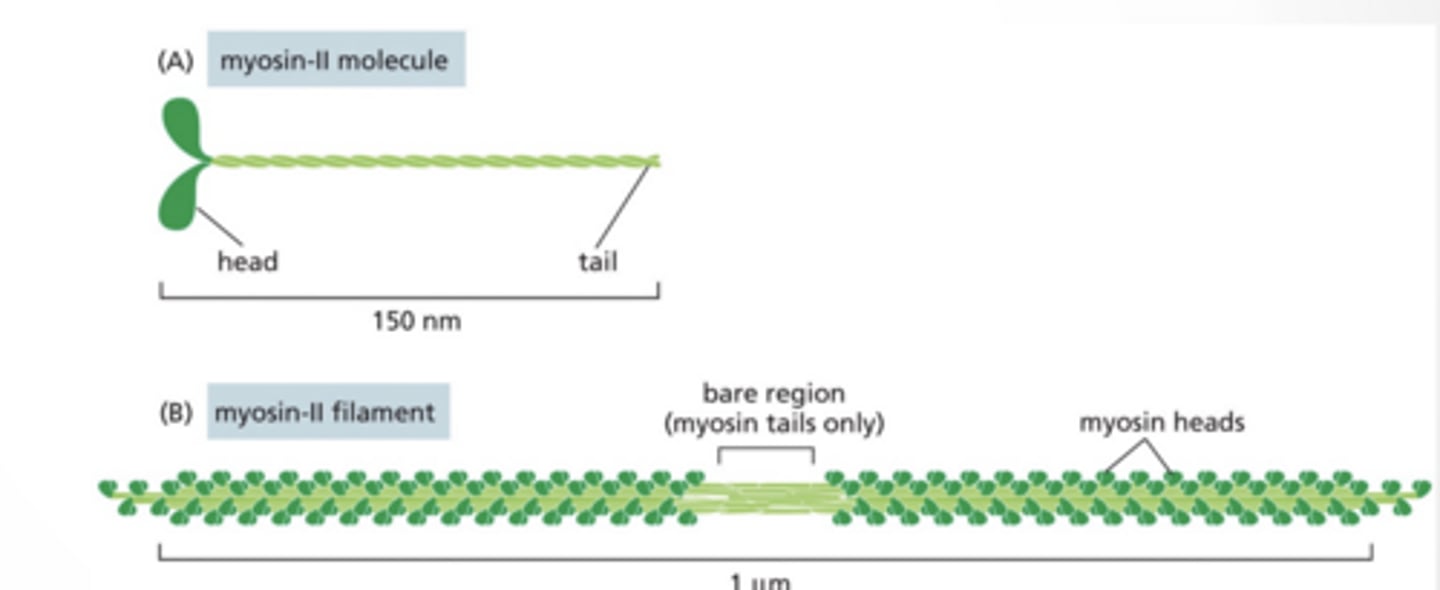
MYOSIN-II HAS TWO HEADS AND CAN FORM FILAMENTS
2 heads (instead of 1)
Forms thick filaments
Used for muscle contraction
FIXED MYOSIN-II MOVES ACTIN FILAMENTS
Sliding filament model
Myosin is fixed in place
Actin filaments slide past it
Myosin pulls actin toward the center
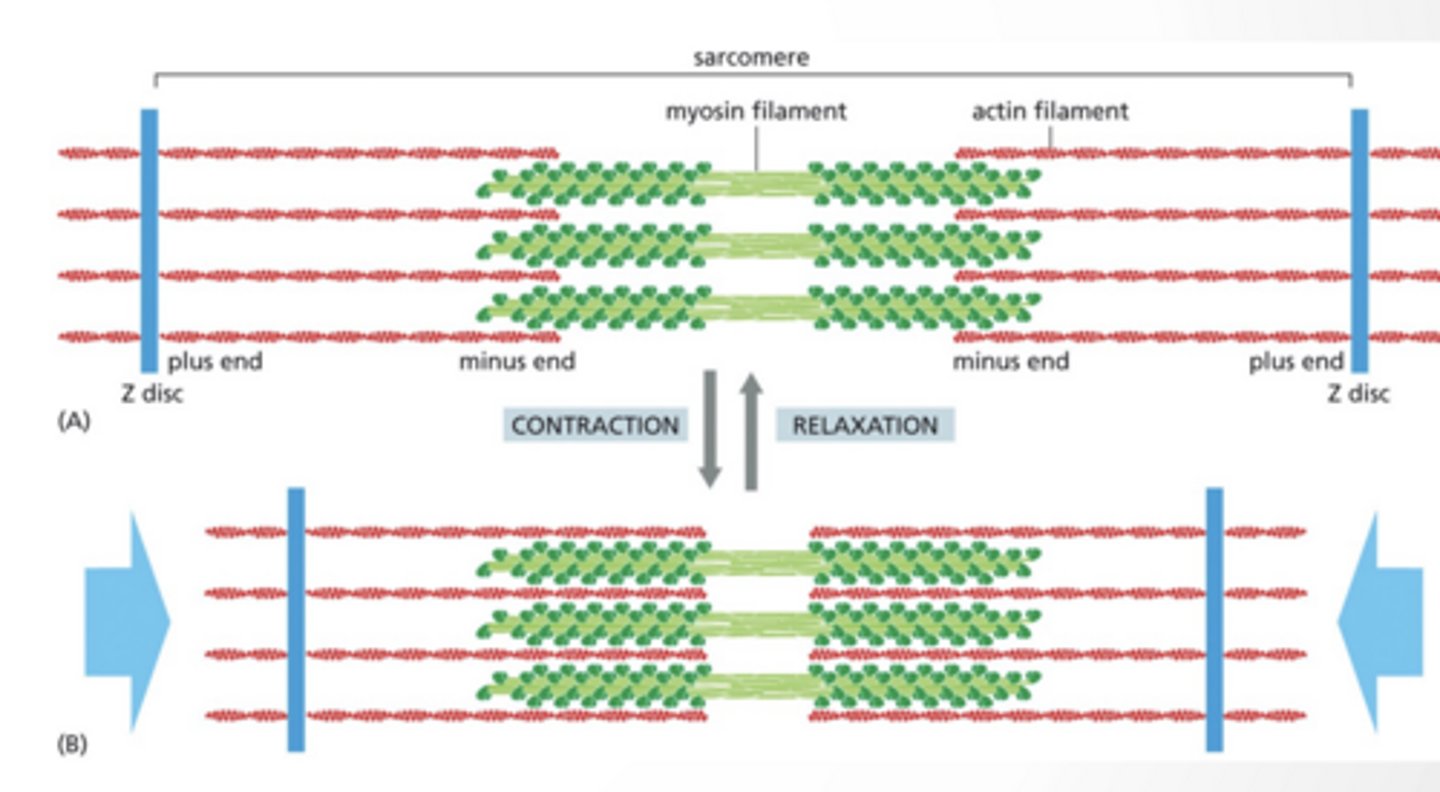
MYOSIN AND ACTIN FORM UNITS CALLED THE SARCOMERE
The sarcomere is a contractile unit
Myosin remains in place and actin is pulled together
Cellular contraction is the Z-discs pulling together
Look at diagram
THis is muscle contraction
myosin II
feet
Sarcomere feet
feet
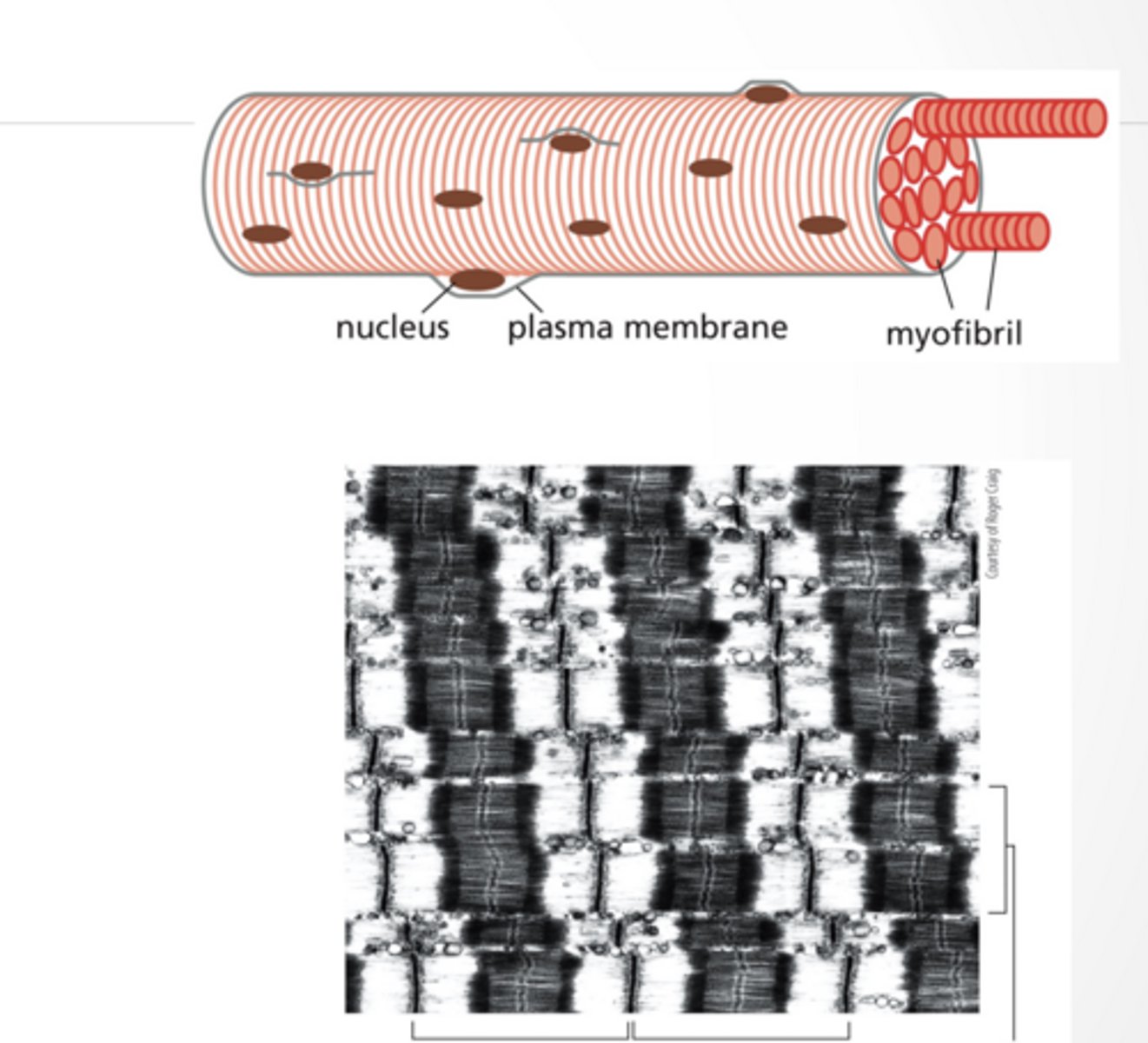
MUSCLE CELLS CONTAIN MANY MYOFIBERS
MUSCLE CELLS CONTAIN MANY MYOFIBERS
Skeletal muscle cells are large multinucleate cells
Form by fusion of smaller cells
Myofibers are many sarcomeres laid end to end
Connection occurs at the Z-disc
MYOSIN AND ACTIN DYNAMICS
MYOSIN AND ACTIN DYNAMICS
ATP binding releases the myosin from actin
ATP hydrolysis shifts the myosin arm into a new conformation
ADP bound myosin binds to actin
Dissociation of ADP from myosin leads to the "power stroke" where myosin moves actin along
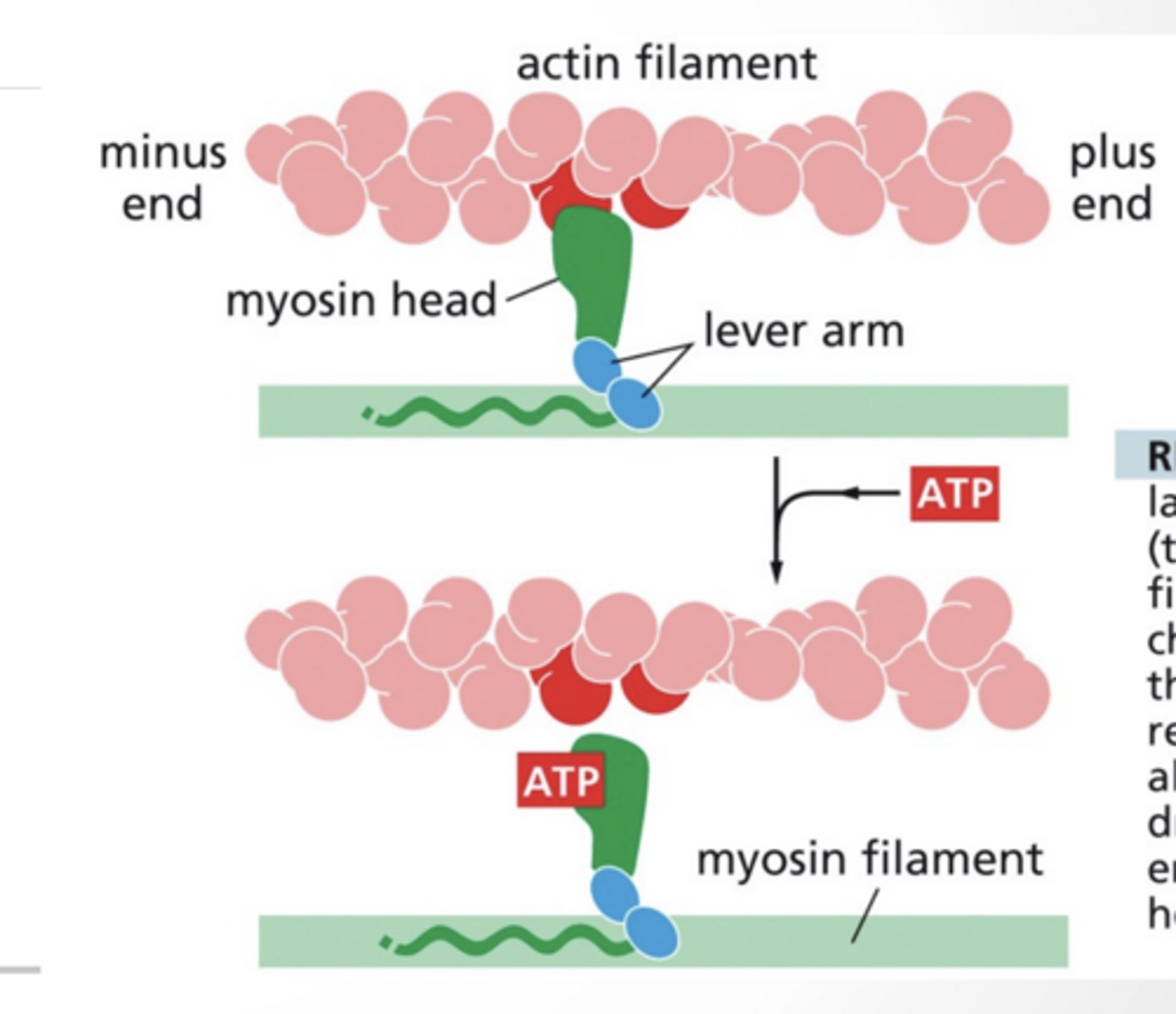
next step feet
feet
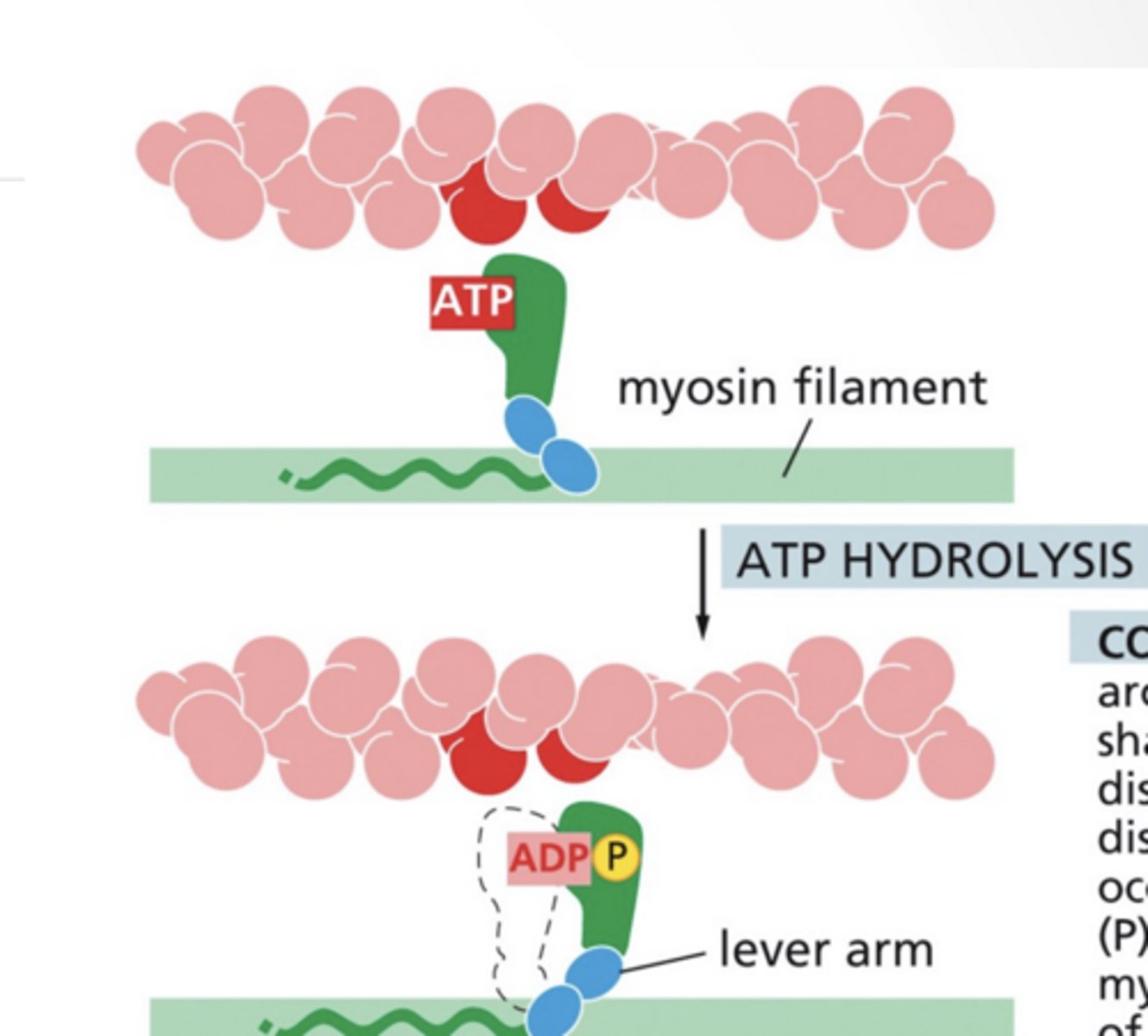
Step 3
feet
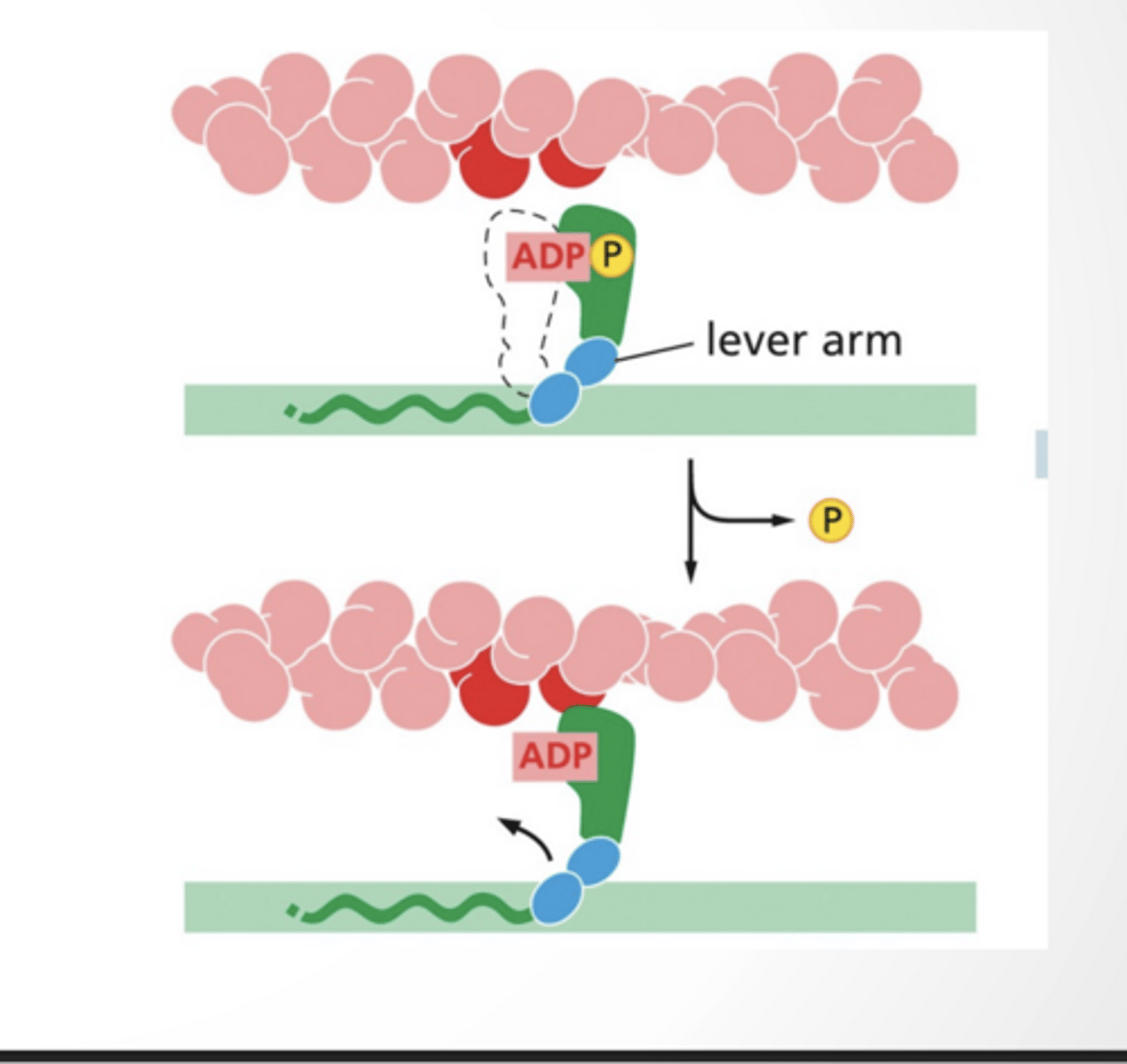
4 feet
feet
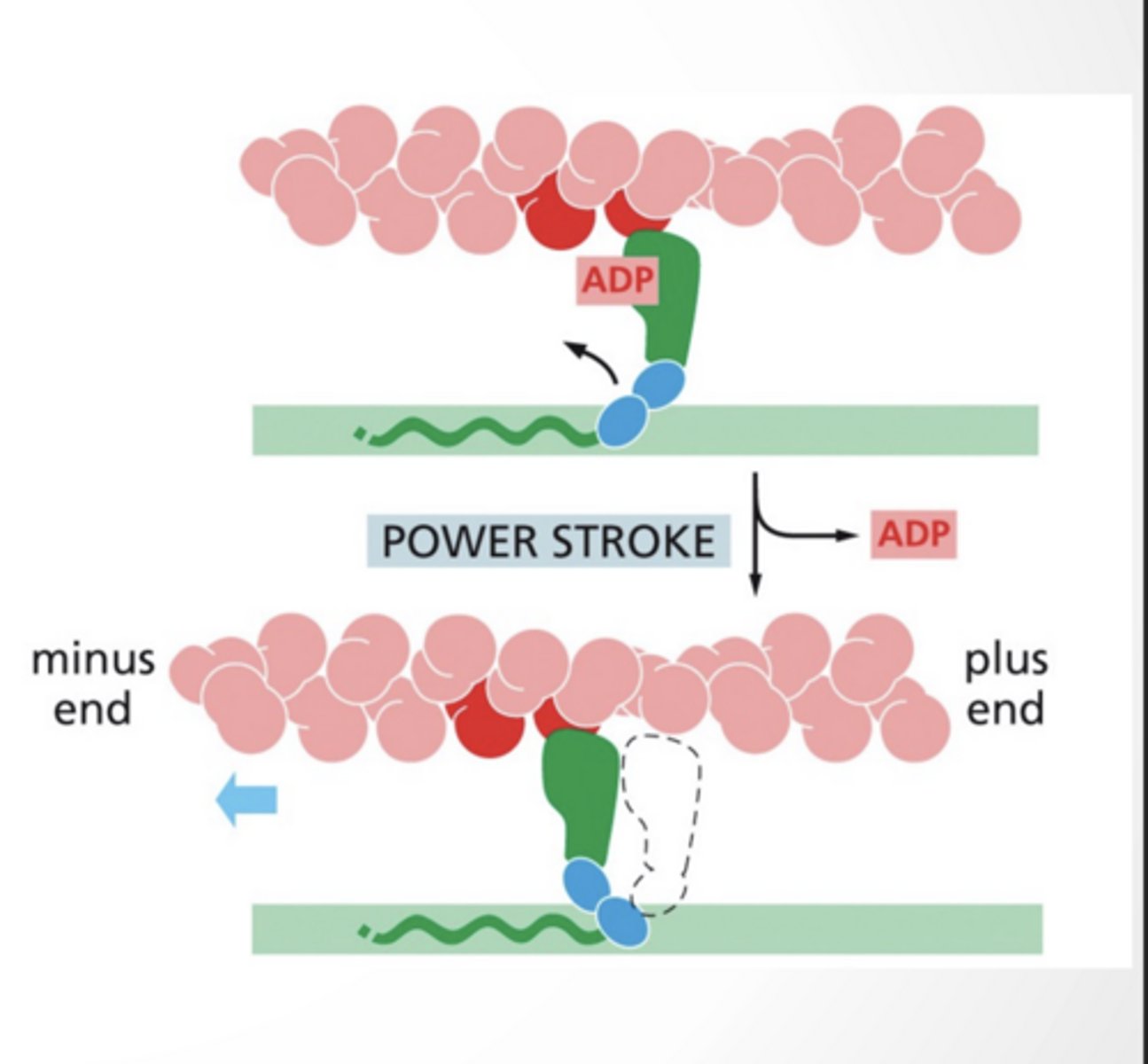
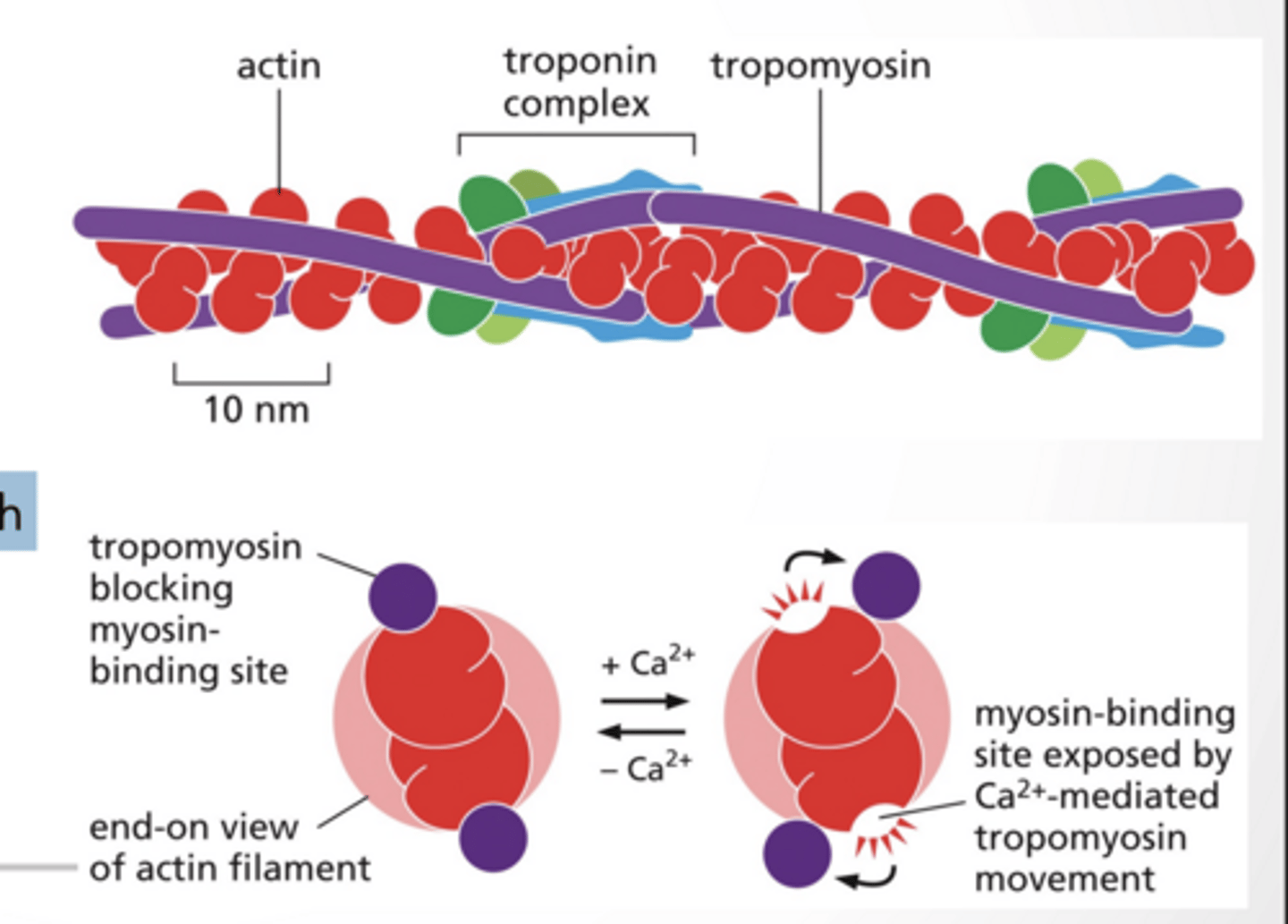
MYOSIN BINDING IS FACILITATED BY CALCIUM
Tropmyosin blocks myosin binding sites during low cytosolic calcium
Myosin binding sites are exposed with high cytosolic calcium levels (mediated by calcium )
CALCIUM IN THE CELL
Calcium levels in the cytosol are VERY low Small changes in Ca+ levels make a big difference
even a tiny increase = HUGE signal
It acts like a trigger molecule
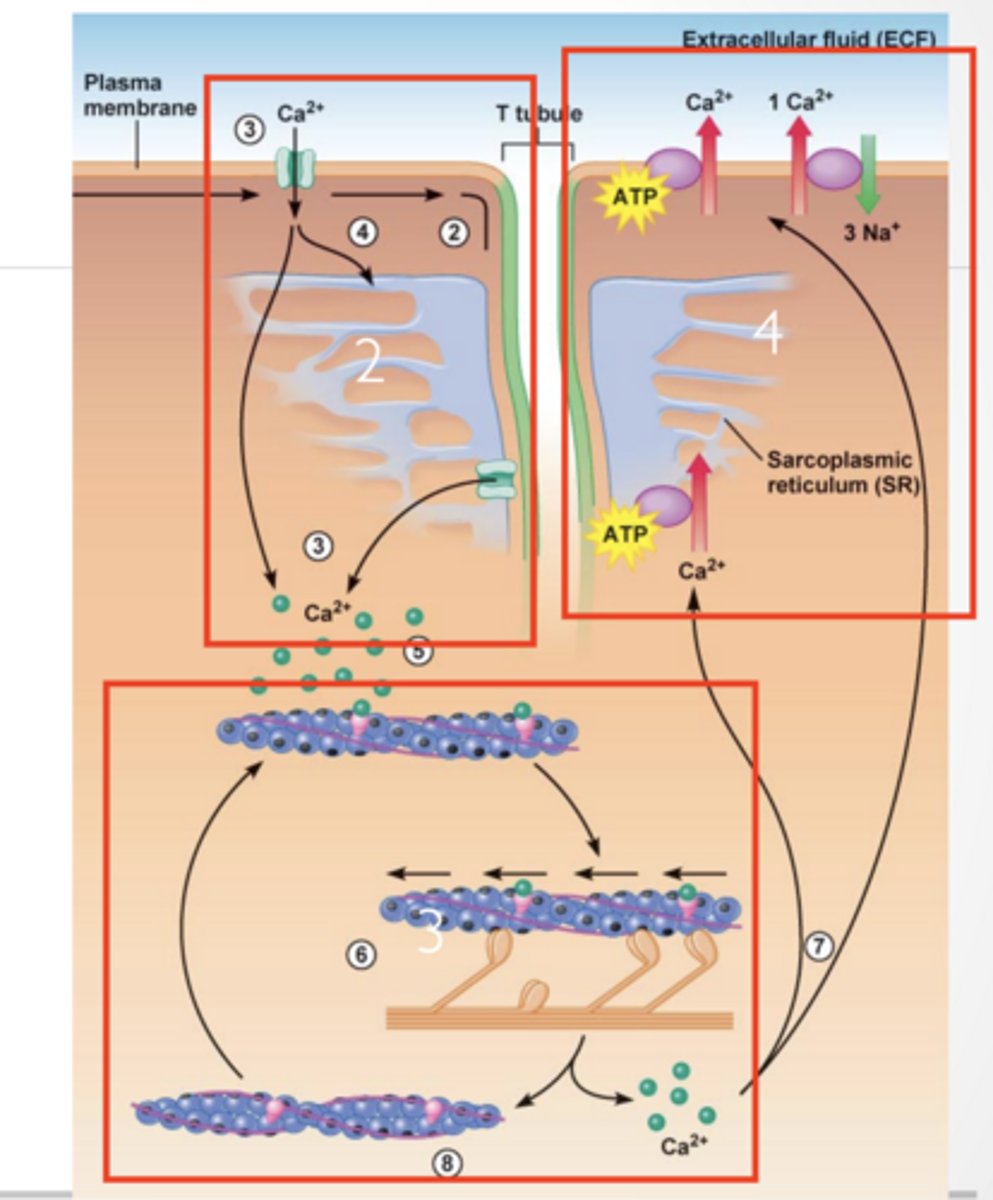
CALICUM IN MUSCLE CONTRACTION
1. Ca2+ is released from Sarcoplasmic reticulum
2. Ca2+ activates myosin/ troposmyosin contractile mechanism . Ca²⁺ binds troponin Tropomyosin moves OUT
Myosin can bind actin
3. ATP is used to quickly remove Ca2+ from cytosol
Contraction stops → muscle relaxes
CALICUM IN MUSCLE CONTRACTION feet
feet
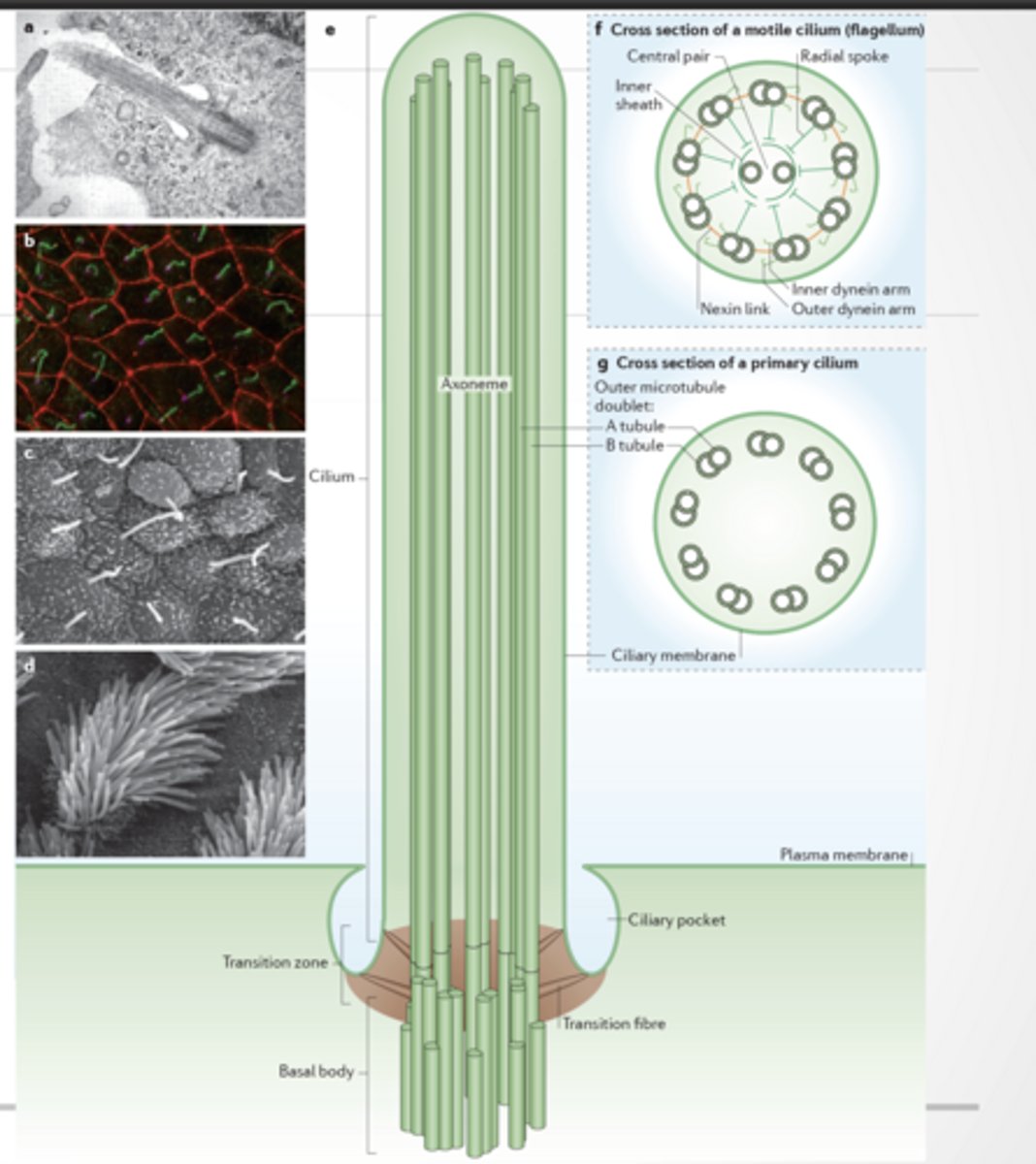
CILIA
Organelle made of membrane, cytosekelton, and motor proteins
9+2 arrangement :
9 outer doublets
2 central microtubules
Has dynein arms → movement - motile
9+0 arrangement - non-motile
Used for motion, environmental sensing, and other
No central pair
No proper dynein-driven motion
Mostly sensory (like antenna)
A cilium = tiny hair made of microtubules + motor proteins
MOVEMENT
Unattached microtubles will slide
Attached (has linking proteins) and microtubles will bend
Movement feet
feet
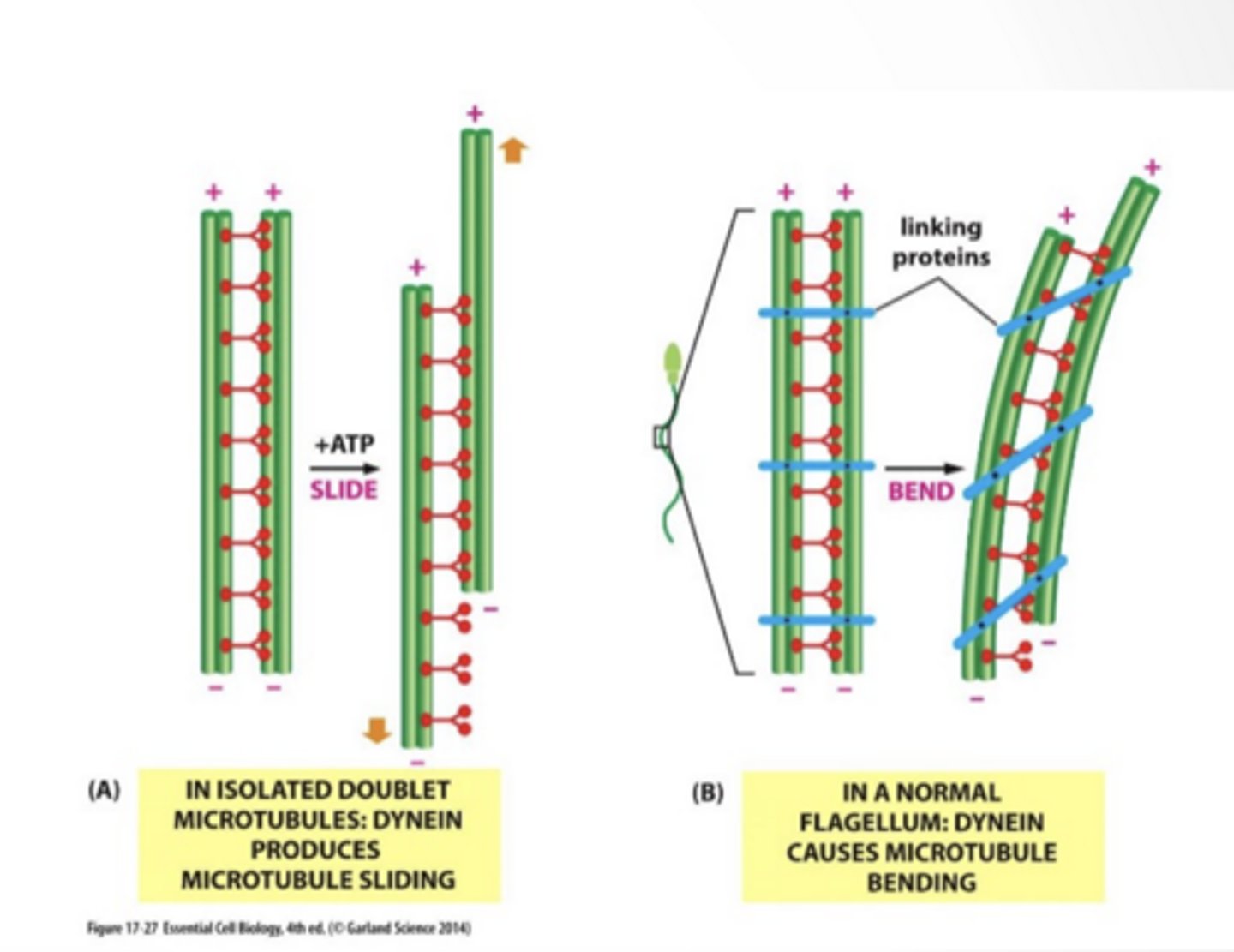
MOVEMENT
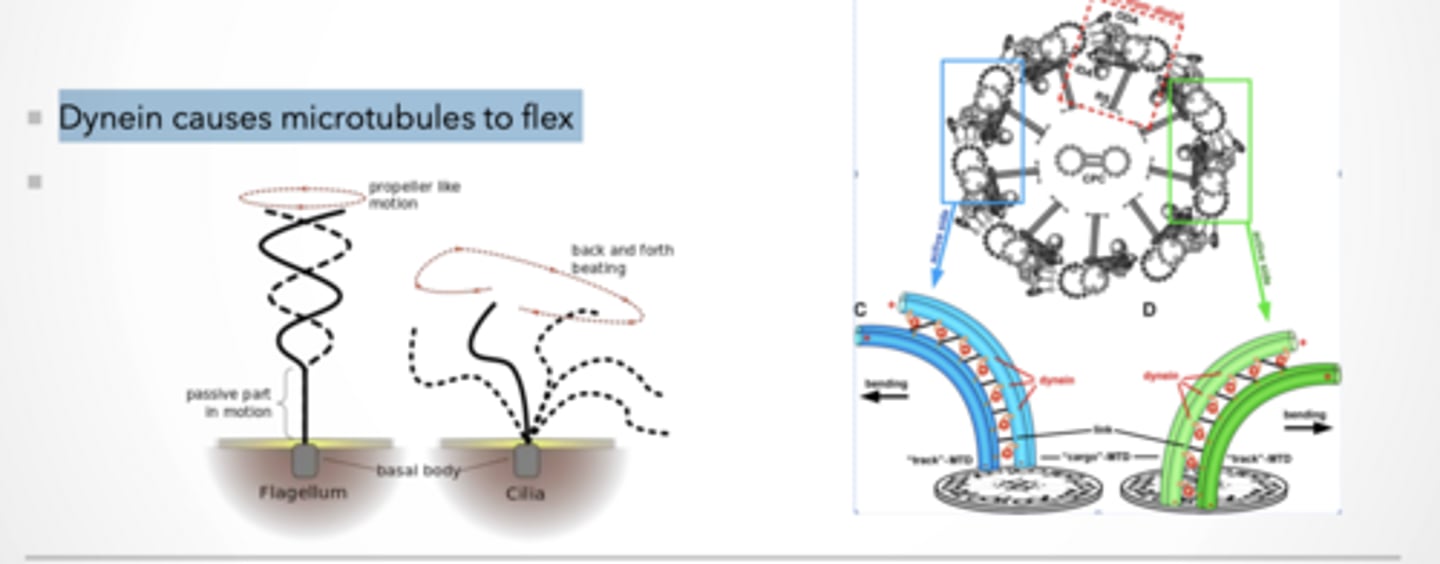
Dynein causes microtubules to flex
Dynein walks → causes sliding
One microtubule tries to slide past the other
If nothing stopped it → everything would just slide apart
But they are CONNECTED (links)
So instead of sliding freely...
Sliding gets converted into BENDING
Flagellum
Spiral / propeller motion
Example: sperm
Cilia
Back-and-forth beating
Like rowing
Same structure (microtubules + dynein) BUT different coordination patterns
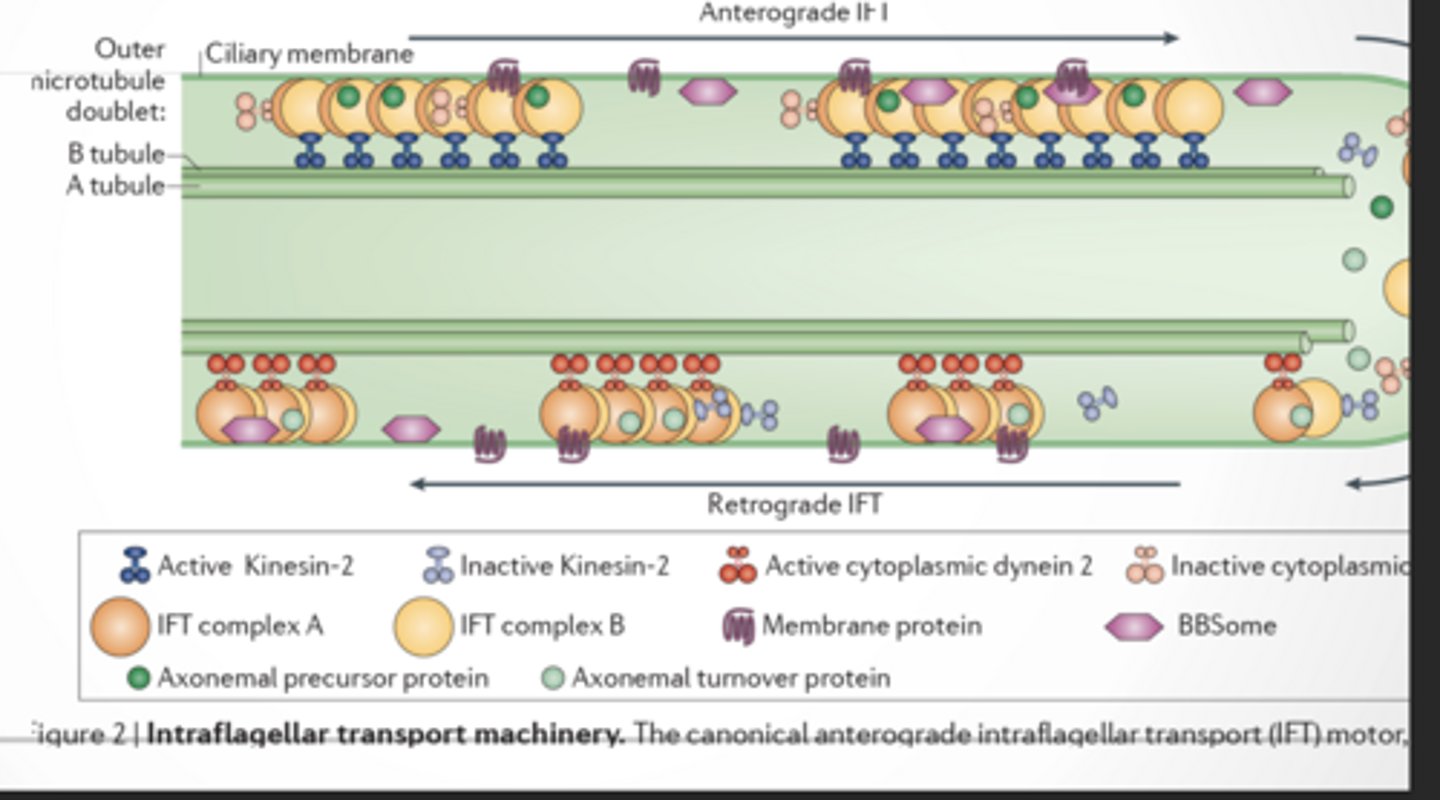
TRANSPORT
This is how cilia are built and maintained
Cilia can’t make proteins inside themselves
So they need a delivery system
ANTEROGRADE TRANSPORT (→)
Base → Tip (outward) Motor: Kinesin-2
BUILDS the cilium
Carries:
Tubulin (microtubule building blocks)
Structural proteins
Membrane proteins
RETROGRADE TRANSPORT (←)
Tip → Base (inward) Motor: Dynein 2
CLEANS + RECYCLES
Carries:
Broken proteins
Waste
Used parts
CASE SUMMARY
Case Summary
Julia (31) & Robert (32) trying to conceive for 22 months
Regular menstrual cycle (29 days)
Prior pregnancy = ectopic → left fallopian tube removed
Robert: normal erectile function, no testicular injury
Robert has asthma
Female ovulation = normal
Male = likely normal
BIG RED FLAGS:
Ectopic pregnancy
Embryo implanted outside uterus (usually fallopian tube)
Required removal of left fallopian tube
Fertilization happened BUT embryo got stuck in tube
Something wrong with transport
Missing fallopian tube
Microtubules Microfilaments And intermediate filaments
Microtubules
Structure:
9+2 in cilia
Function:
Cilia/flagella movement
Vesicle transport
Mitotic spindle
Fertility:
Move sperm (flagella)
Move egg/embryo (cilia)
Defect → infertility
Microfilaments (Actin)
Structure:
Thin actin filaments
Polar
Function:
Cell movement
Muscle contraction
Vesicle transport (myosin)
Fertility:
Acrosome reaction
Sperm function
Reproductive tract contraction
Intermediate Filaments
Structure:
Rope-like fibers
Non-polar
Function:
Structural support
Resist stress
Fertility:
Minor role
Tissue integrity
Table of sperm and defects
Microtubules
present in sperm ? YES
Defect = No sperm movement (flagella fails)
Microfilaments
YES
defect= Fertilization issues
Intermediate filaments
YES
defect = Weak structure
Microtubules (cilia)
Cilia line fallopian tubes and they Beat to move embryo
TEST RESULTS
Robert’s sperm was fine
Focus shifts to female
Julia has chronic cough her whole life
Lungs:
Cilia clear mucus → mucus builds → cough
Fallopian tubes:
Cilia move embryo → embryo stuck → ectopic
SAME PROBLEM
Cilia Structure
Normal = 9+2
Julia = 9+0 (missing center pair)
Without central pair , No coordinated beating
Diagnosis
Primary Ciliary Dyskinesia (PCD)
Genetic disorder
Cilia don’t move
“Primary ciliary dyskinesia results from defective microtubule structure (9+0 instead of 9+2), causing loss of coordinated ciliary movement, which leads to impaired mucus clearance and failure of embryo transport, resulting in chronic respiratory issues and infertility.”
INTRACELLULAR SIGNALING PATHWAYS ARE A SERIES OF EVENTS
1.Transmit signal into cell
2. Amplify signal (Production of second messenger)
3. Integrate other signals (Fan in processes)
4. Distribute signals (Fan out processes:)
5. Output
TRANSMIT SIGNAL INTO CELL
Direct permeability
signal molecule (first messenger) enters cell
gated channels
signal is transduced
Transmembrane receptors
signal is transduced
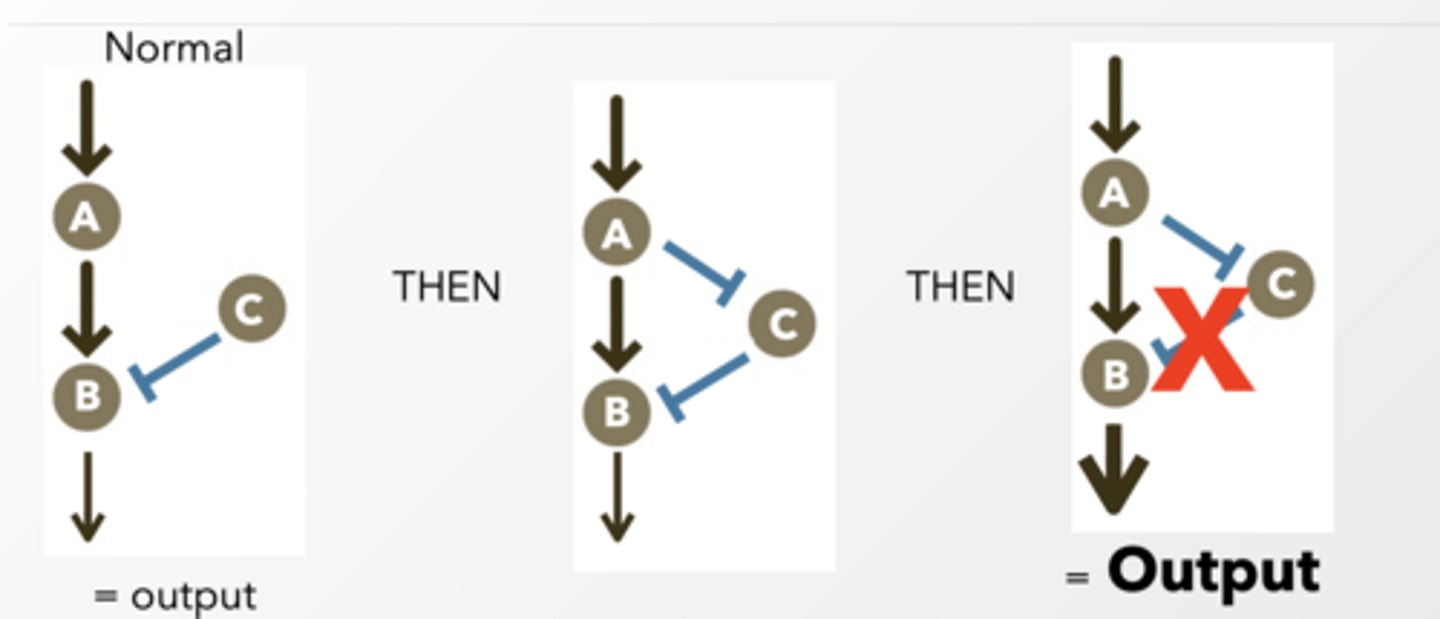
Feedforward feet
self explanatory
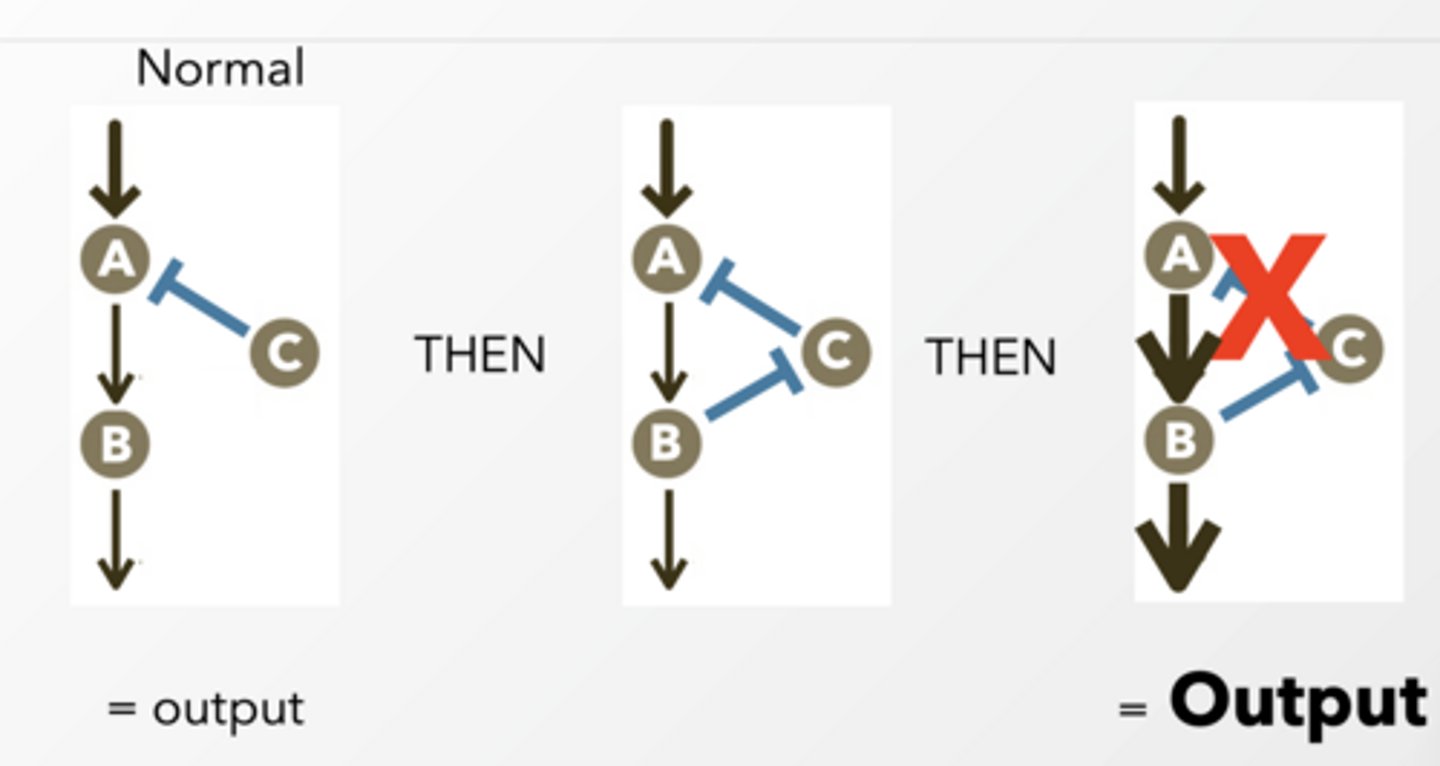
FEEDBACK DISINHIBITION feet
same thing
SIGNAL TRANSDUCERS
G-protein coupled receptors
Enzyme linked receptors
Ion channel receptors
Nuclear receptors
HOW DO G-PROTEINS WORK
Signal molecule activates GPCR
GPCR acts as a GEF for alpha subunit (GTPase) of trimeric G-protein
Parts of complex have different activities
Alpha subunit is activated
Beta/gamma subunit separate and become active
Alpha and beta/gamma alter effector proteins
GPCR can activate more G-proteins
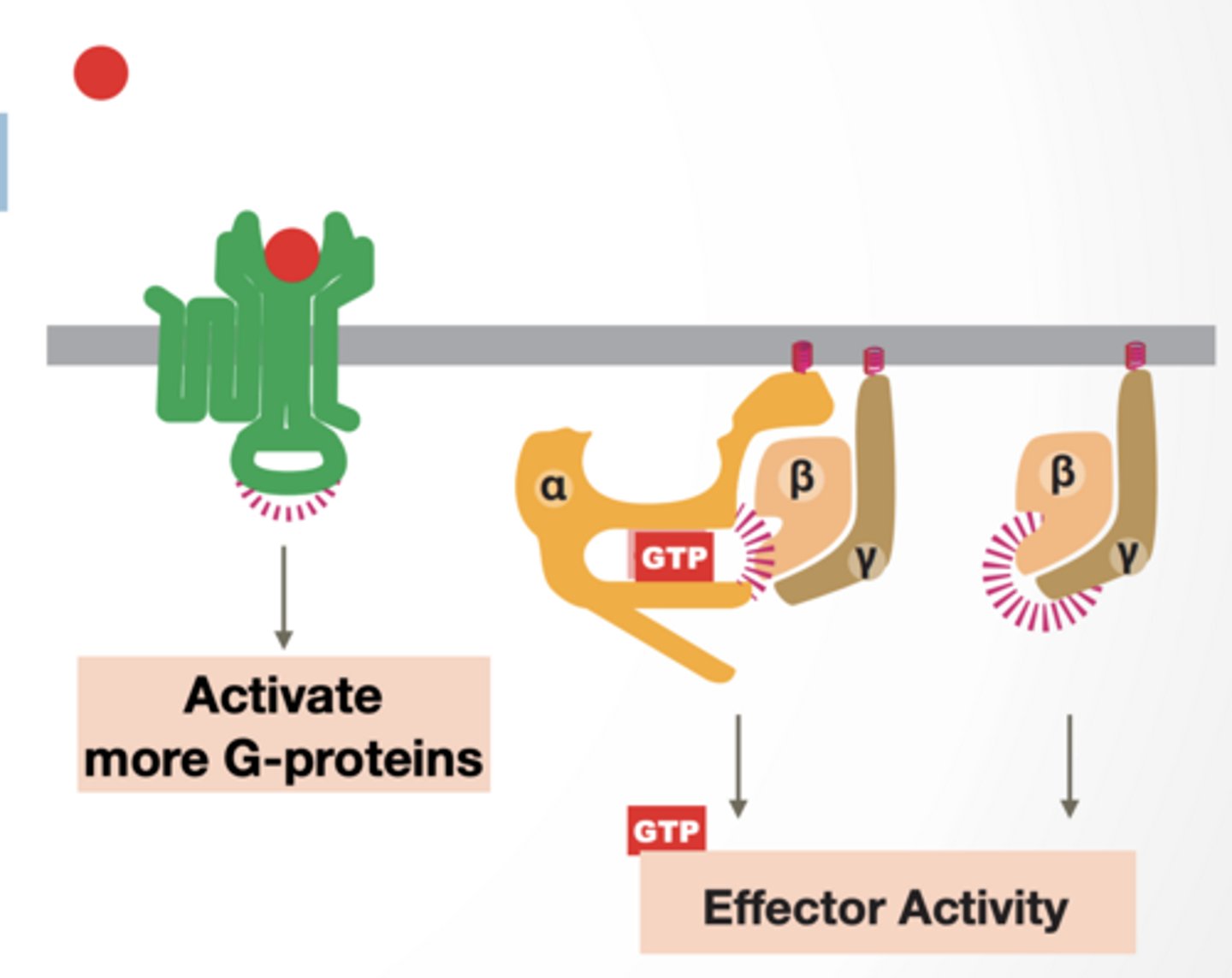
DIAGRAM 1
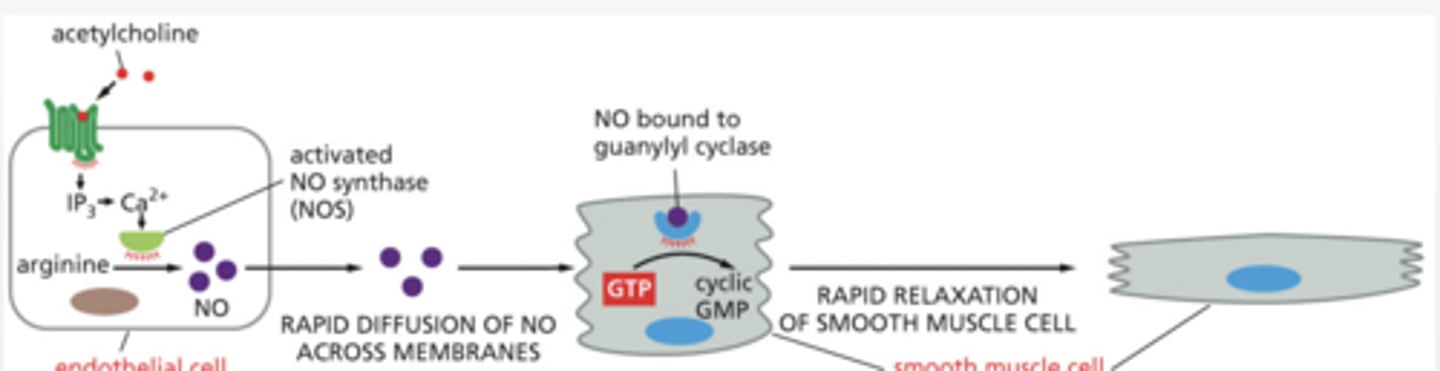
the Nitric Oxide (NO) → cGMP → Smooth Muscle Relaxation pathway
STEP:
1. Ca²⁺ increases in endothelial/neuron cell
2. Ca²⁺ binds calmodulin (CaM)
3. CaM activates nitric oxide synthase (NOS)
4. Arginine converted to → Nitric Oxide (NO)
STEP 2
NO enters smooth muscle cell
Guanylyl cyclase inactive → active
NO diffuses into smooth muscle cell
NO activates guanylyl cyclase (GC)
GC converts GTP → cGMP
cGMP activates PKG (protein kinase G)
PKG = enzyme that phosphorylates proteins
Effects\
Lower cytosolic Ca²⁺
Desensitization to Ca²⁺
↓ Ca²⁺ release from ER
↑ Ca²⁺ sequestration (stored)
↑ Ca²⁺ export out of cell
↓ Ca²⁺ entry
RESULT:
LOW Ca²⁺ inside cell
Low Ca²⁺ →
Myosin cannot contract
Smooth muscle RELAXES
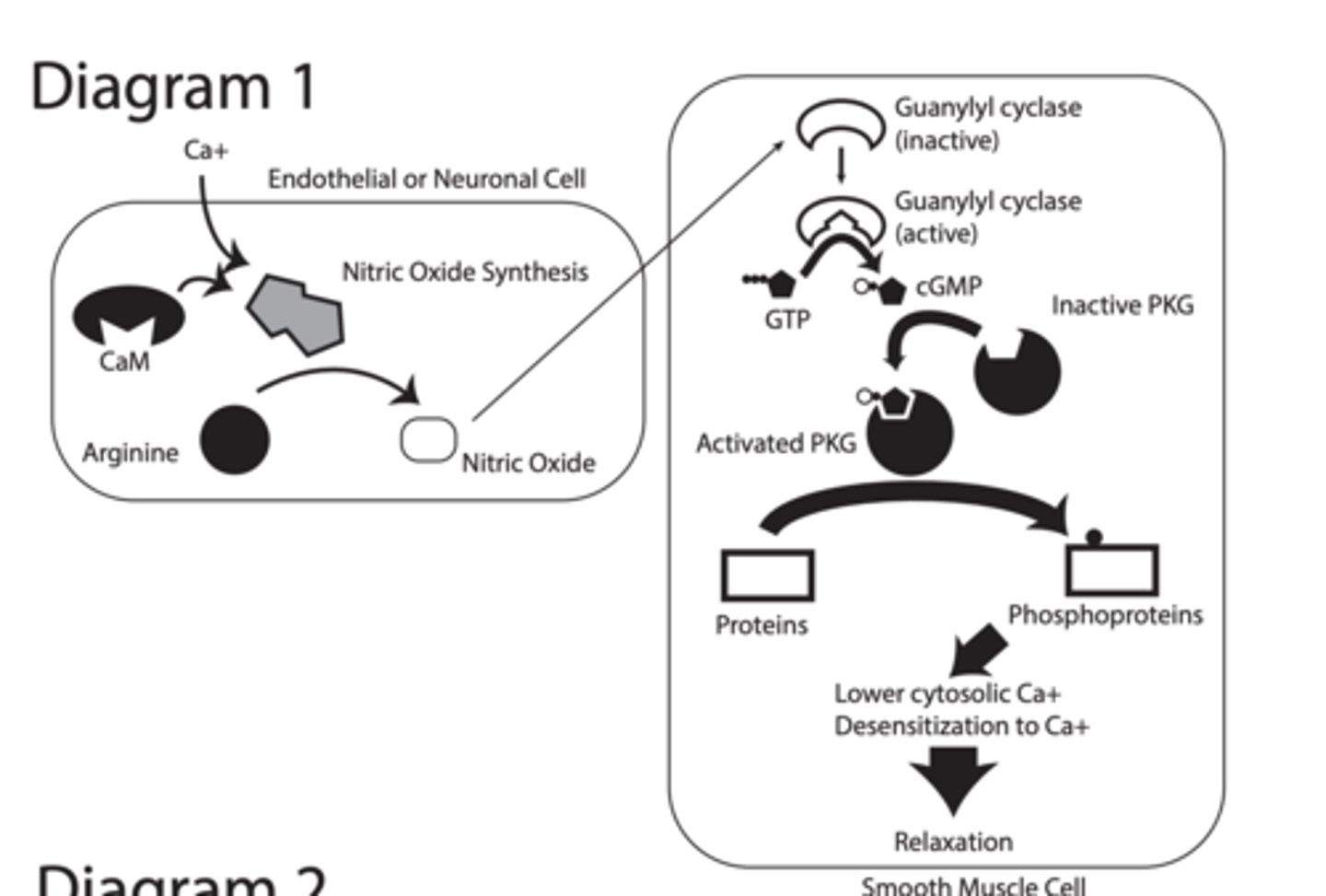
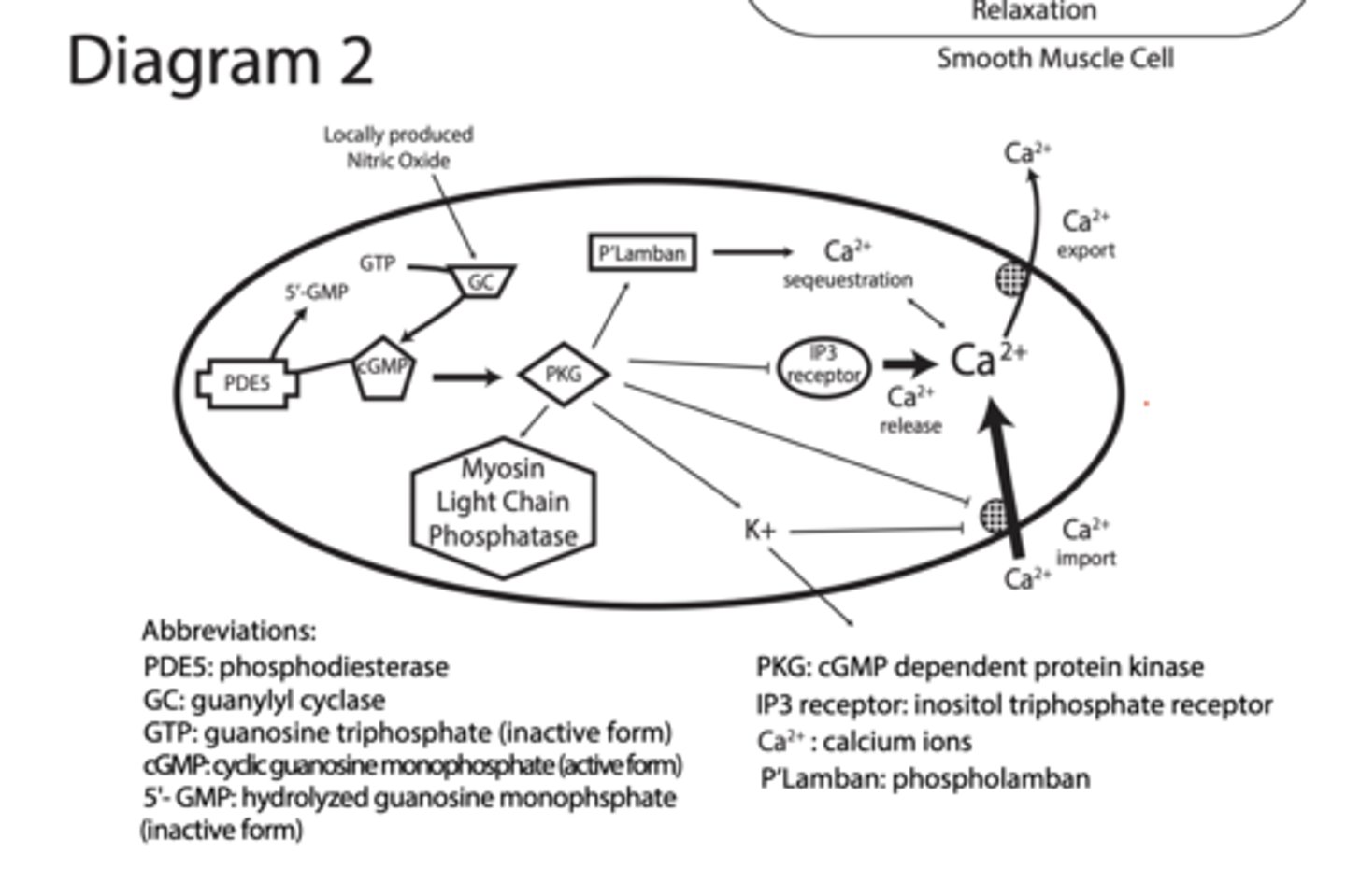
Diagram 2
1. NO → GC
NO activates guanylyl cyclase
2. GTP → cGMP
GC converts GTP → cGMP
PDE5 (phosphodiesterase) converts cGMP → GMP
Turns OFF the signal
3. cGMP → PKG
cGMP activates PKG
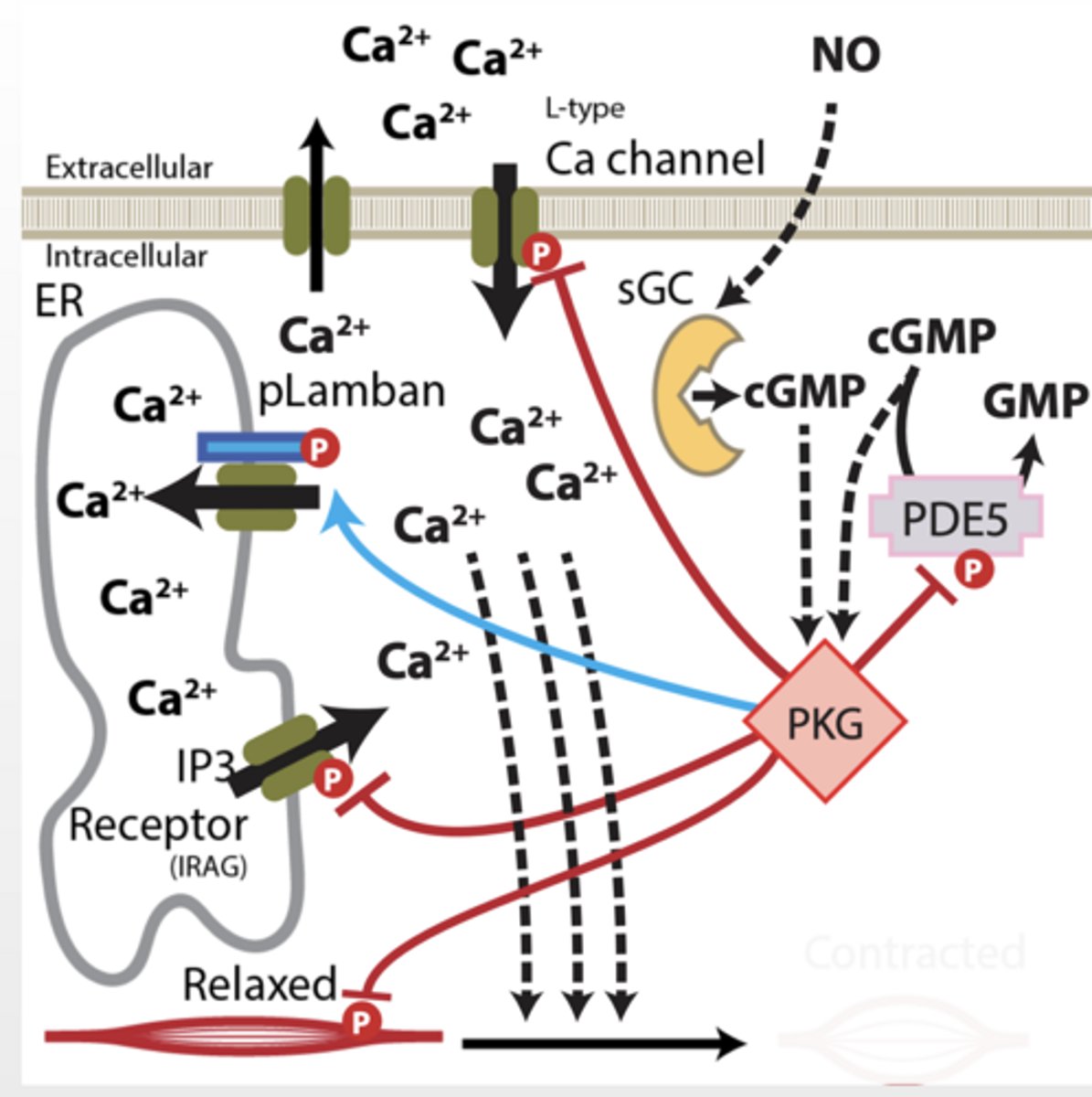
4. PKG TARGETS (VERY IMPORTANT)
PKG TARGETS (VERY IMPORTANT)
A. Phospholamban (PLB)
PKG phosphorylates PLB
→ Ca²⁺ pumped into ER
↓ cytosolic Ca²⁺
B. IP3 receptor
PKG inhibits Ca²⁺ release
Less Ca²⁺ from ER
C. Blocks Ca²⁺ channels (membrane)
↓ Ca²⁺ entry
D. Ca²⁺ export pumps
↑ Ca²⁺ pumped OUT
E. Myosin light chain phosphatase
Activated → dephosphorylates myosin
prevents contraction
F. PKG activates K⁺ channels
“PKG activates potassium channels, causing hyperpolarization of the membrane, which closes voltage-gated calcium channels and reduces intracellular calcium levels."
PKG causes:
↓ intracellular Ca²⁺
↓ Ca²⁺ sensitivity
↑ myosin relaxation
RESULT = SMOOTH MUSCLE RELAXATION
BC Ca2+ binds tropomyosin which inhibits myosin from binding actin
CASE SETUP (DOG ED)
Dog has ED
Signalling pathway controls this
NO → cGMP → PKG → ↓ Ca²⁺ → relaxation
Smooth muscle relaxation
BLOOD FLOW IN CASE STUDY
Normal contracted muscle
reduced blood flow, no erection
cGMP pathway activated relaxed muscle increased blood flow, erection present
RECEPTOR LOCATION
Intracellular receptor
If pathway doesn’t work:
Not enough NO
Not enough cGMP
Too much PDE5 (breaks cGMP)
Result:
Muscle stays contracted
Blood can’t flow
cGMP BASICS
Guanylyl cyclase creates 2nd messenger cGMP
NO signals guanalyl cyclase to make cGMP very important molecule in many cellular functions
Guanylyl cyclase creates cGMP from GTP
cGMP activates down stream targets
cGMP activates:
PKG (main one)
Ion channels
Other proteins
Somewhere in this pathway is broken:
NO production
Guanylyl cyclase
cGMP production
PKG
Ca²⁺ regulation
PDE breaks down cGMP
VIAGRA
Dog is perscribed Viagra
Viagra = PDE5 inhibitor
Effect:
Blocks cGMP breakdown
Keeps cGMP HIGH
If Viagra works →
Problem is NOT upstream (NO/GC still works)
Problem is likely cGMP breakdown
GROUP THE PATHWAY INTO PARTS
START OF PATHWAY:
Nitric Oxide (NO)
Guanylyl cyclase (GC)
cGMP
Ca²⁺ CONTROL ZONE From PKG
Ca²⁺ channels (entry)
Ca²⁺ pumps (export)
ER storage (phospholamban)
IP3 receptor (release)
K⁺ channels (indirect effect)
MYOSIN CONTROL
Myosin light chain phosphatase
INHIBITION
PDE Breaks cGMP → stops signal
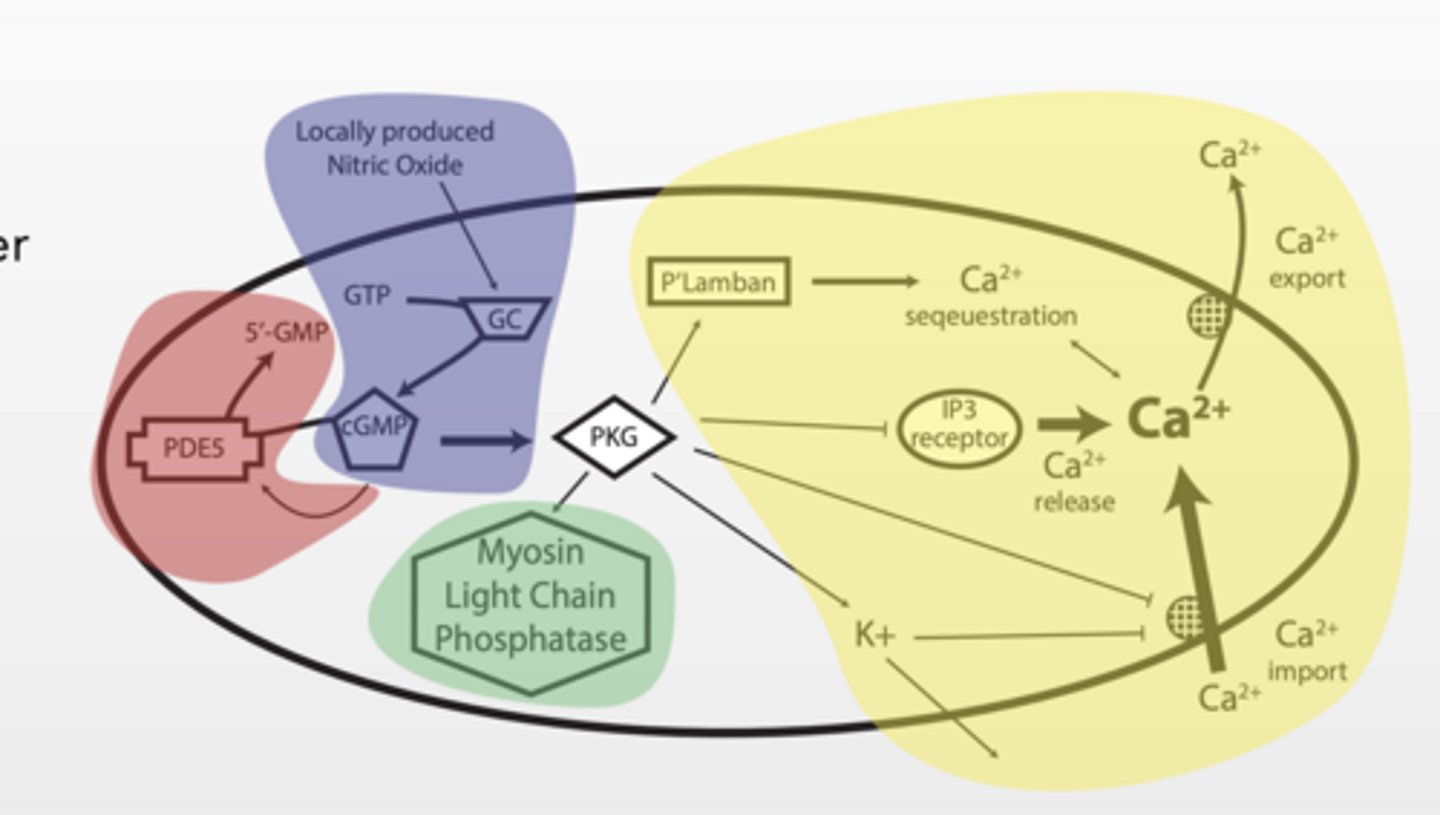
PKG - A "HUB" IN A PATHWAY
Protein kinase G (PKG) aka cGMP dependent protein kinase
Serine/threonine kinase Consensus amino acid binding sequence: R R “X” S/T “X”
What general role does this play?
Fan out or distribution
Already
Ca is keeping muscle contracted
cGMP being degraded by PDE5
1. NO increases cGMP production and activates PKG
2. PKG inhibits Ca release from ER (less Ca)
3. PKG inhibits Ca influx from extracellular environment (less Ca)
4. PKG inhibits myosin complex (no contract)
5. PKG inhibits PDE5 (more cGMP)
6. PKG activates Lamban (Ca sequestration)
sequestration
Sequestration = storing Ca²⁺ somewhere else so it's not free in the cytosol (usually ER)
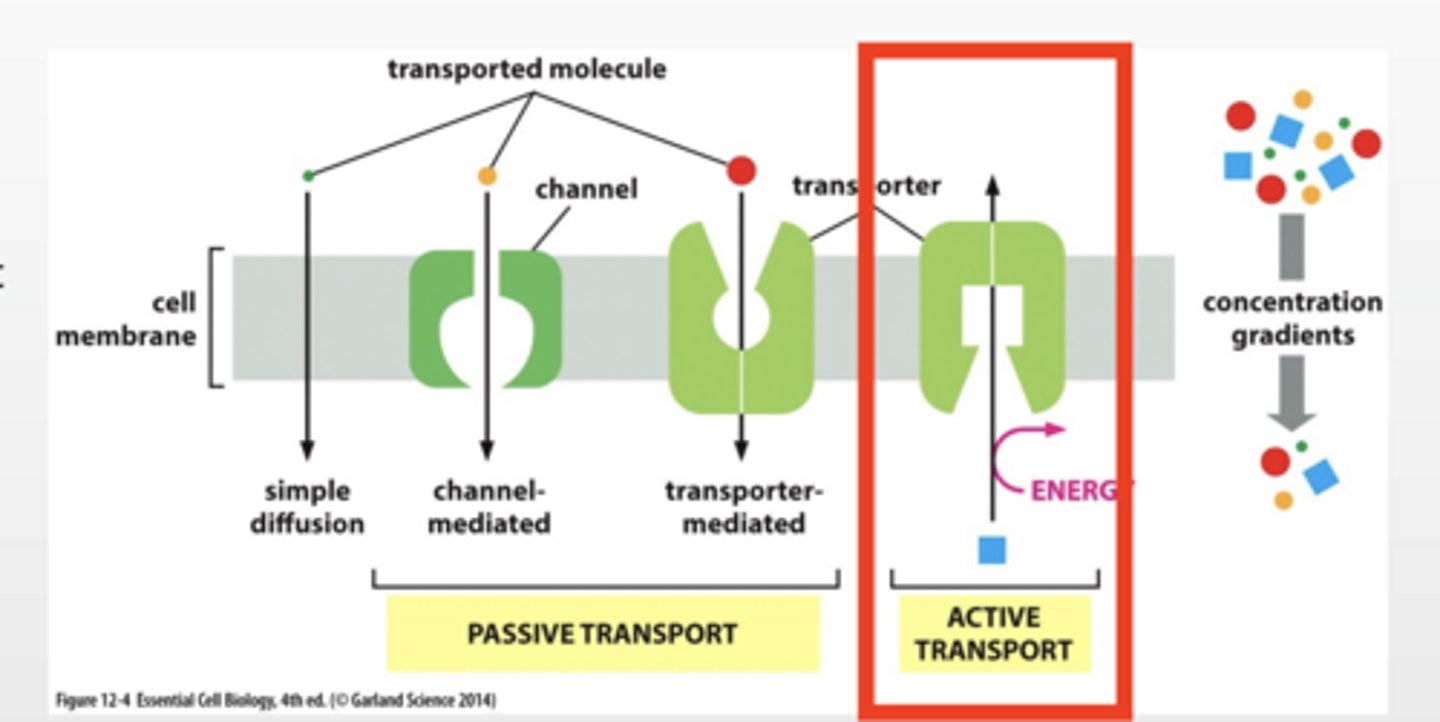
ACTIVE TRANSPORT
Pumps work against concentration gradient
Requires energy input ATP or establish gradient
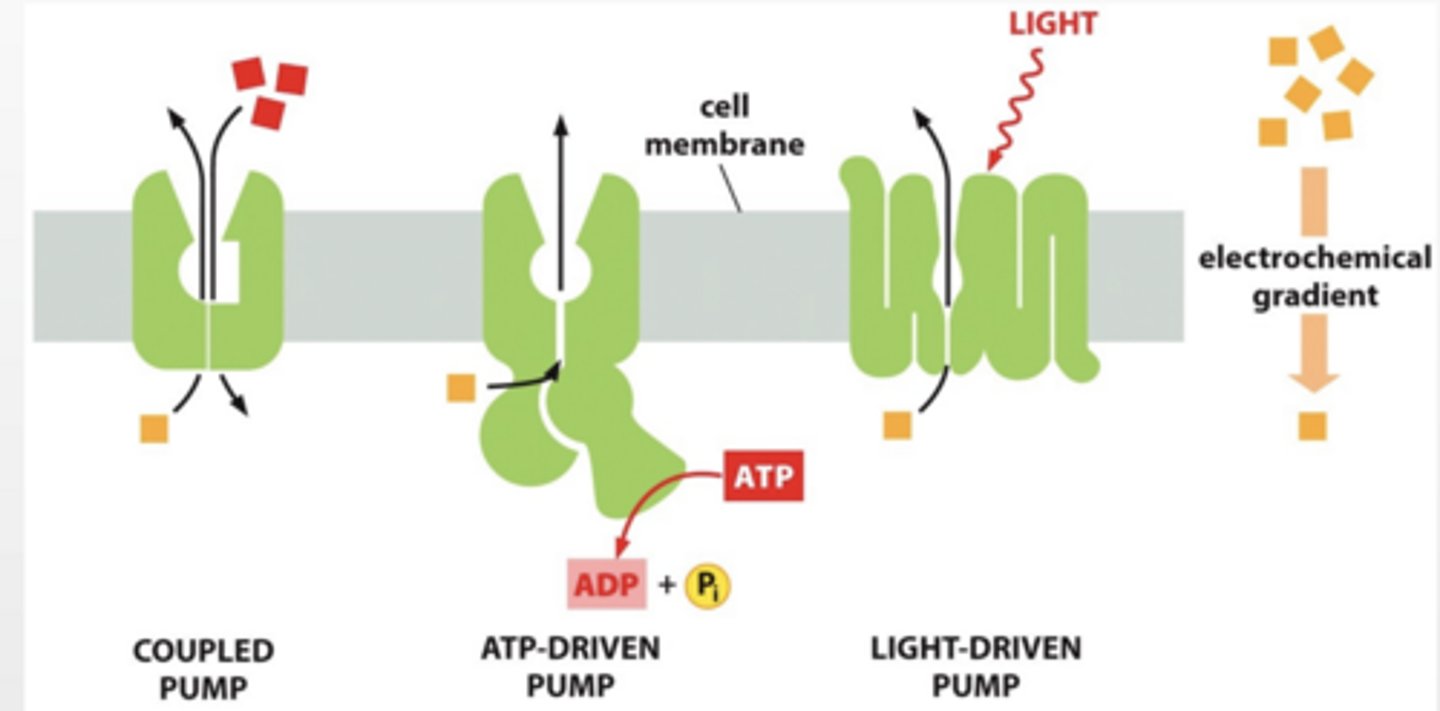
DIFFERENT TYPES OF PUMPS
Coupled pump
ATP driven pump
Light driven pump
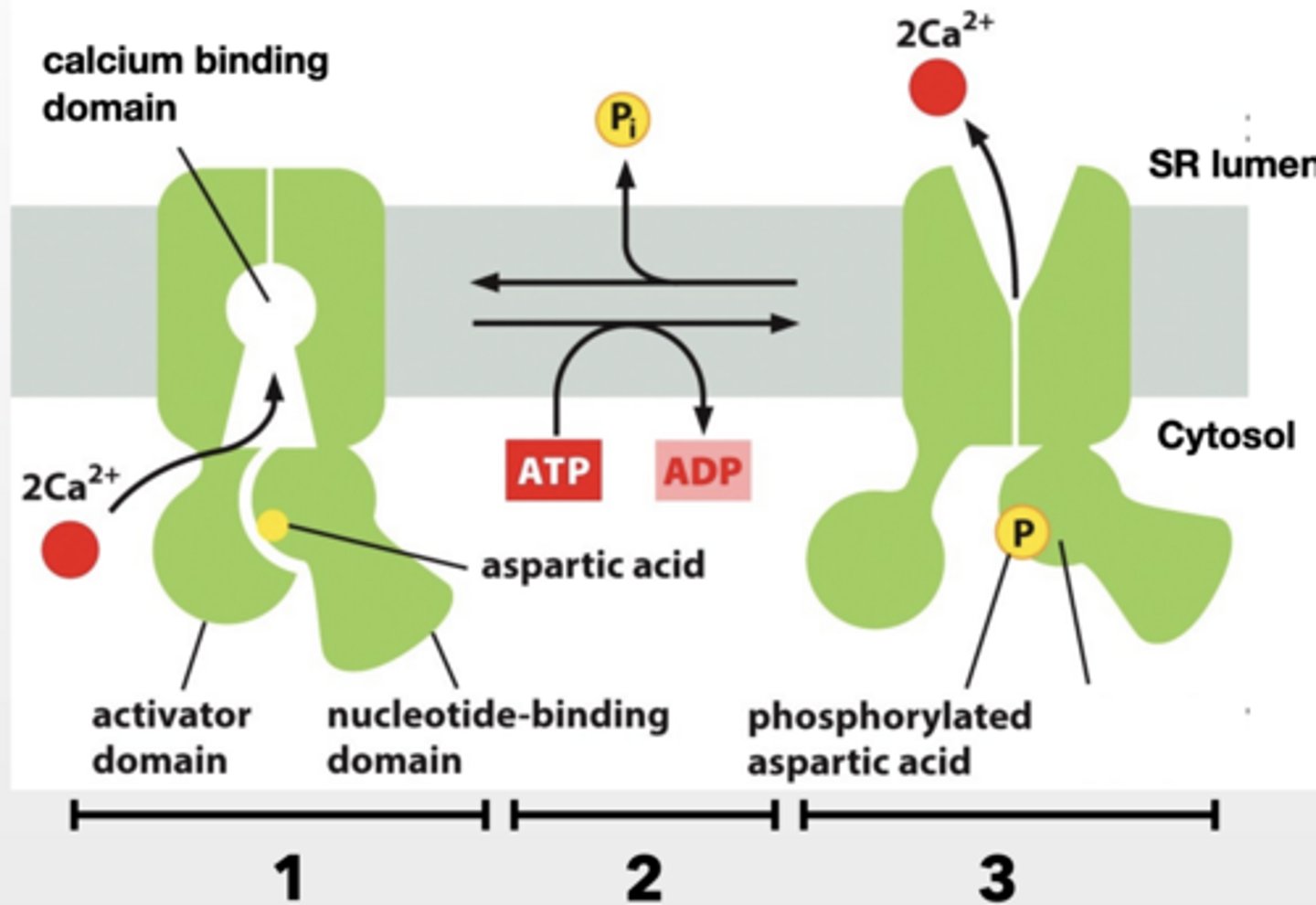
CA+ PUMPS STORE CA+ IN THE SACROPLASMIC RETICULUM (SR) OR ENDOPLASMIC (ER)
Muscle release Ca+ from SR to contract muscles cells
Ca+ pump recovers that Ca+ back to the SR
1. Cytosolic Ca+ binds to pump
2. ATP is used to phosphorylate pump (aspartic acid on pump) and cause conformational shift
3. Shift eliminates Ca+ binding sites and Ca+ is released into SR.
RELAXATION
BLOOD FLOW CONNECTION
NORMAL:
Low blood flow
cGMP ACTIVE:
Relaxed
Increased blood flow
active transport feet
feet
3 types feet
feet
3 TYPES OF SIGNAL TRANSDUCERS
G-protein coupled receptors (GPCRs)
Enzyme-linked receptors
Ion channel receptors
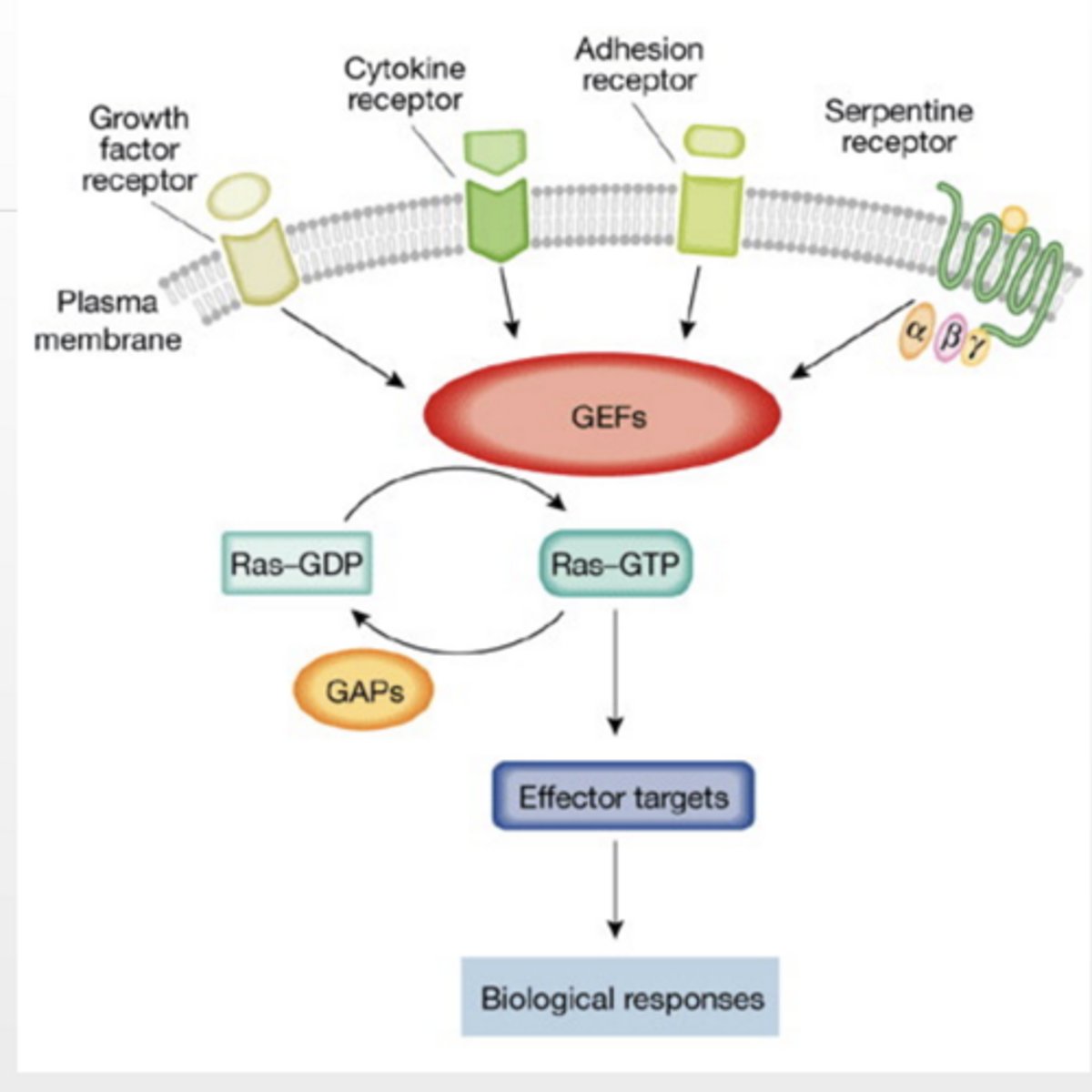
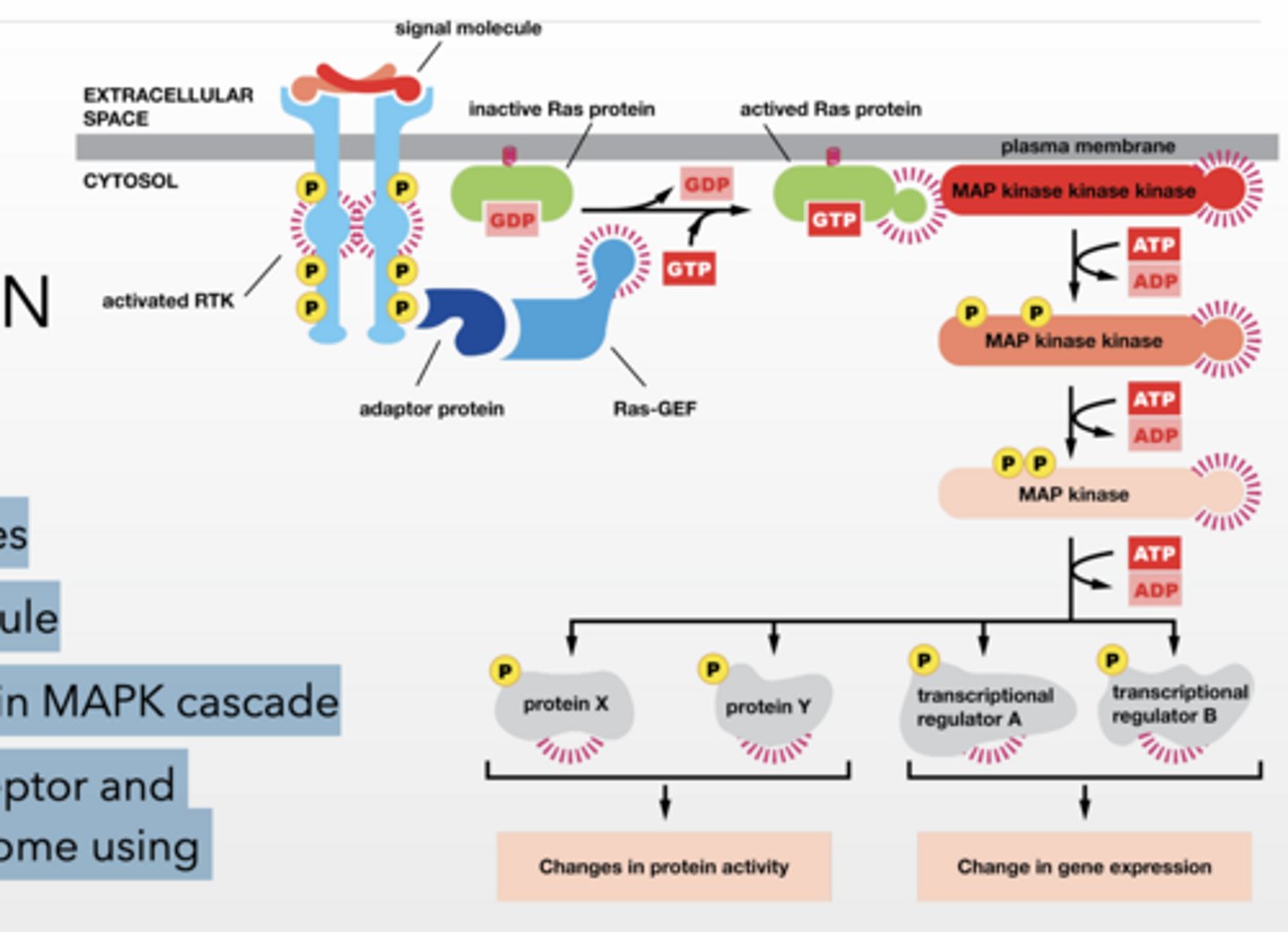
Ras GTPase PATHWAY RECEPTOR TYROSINE KINASES/Enzyme linked
Receptor Tyrosine Kinases
phosphorylation occurs on tyrosine
Ligand (signaling molecule) binds and induces dimerization of RTKs
Cross phosphorylation occurs
Phosphorylation sites are binding sites for other proteins
10-20 different molecules
Ligand induces dimerization and phosphorylation
Adaptor protein binds to RTK
Ras-GEF binds to adaptor protein
localizes GEF to membrane
Ras-GEF facilitates Ras activation
Ras-GEF = turns Ras ON (GDP → GTP)
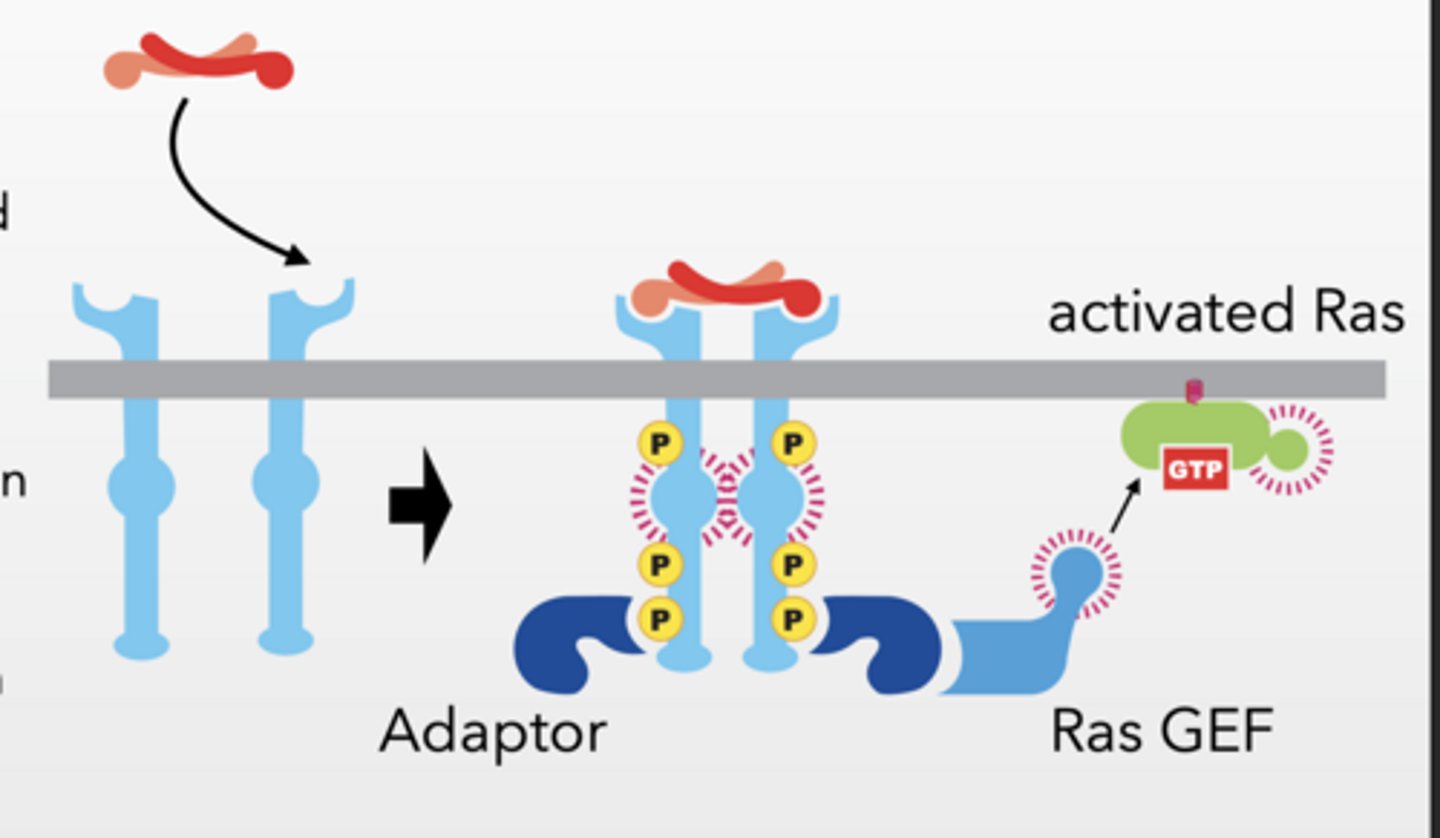
GEF
Guanine Nucleotide Exchange Factors (GEFs) are proteins that activate monomeric GTPases (like Ras) by swapping bound GDP (inactive) for GTP (active), triggering cellular signaling pathways.
Monomeric refers to single-subunit GTPases, or "small G-proteins," which operate as molecular switches rather than heterotrimeric complexes.
RAS GTPASE, GEFS AND GAPS
Ras is a monomeric GTPase
trimeric GTPase proteins are part of the GPCR pathway Most RTKs GTPases are monomeric
Ras-GAP:
turns Ras OFF (hydrolyzes GTP → GDP)
Ras activated by by many different factors
Ras has basal level of activity
Activate GEFS to activate Ras
GAPs and natural hydrolytic activity inactivate
30% of cancers contain mutant Ras that is constitutively active
Plants don’t have an equivalent family of GTPase principles are the same
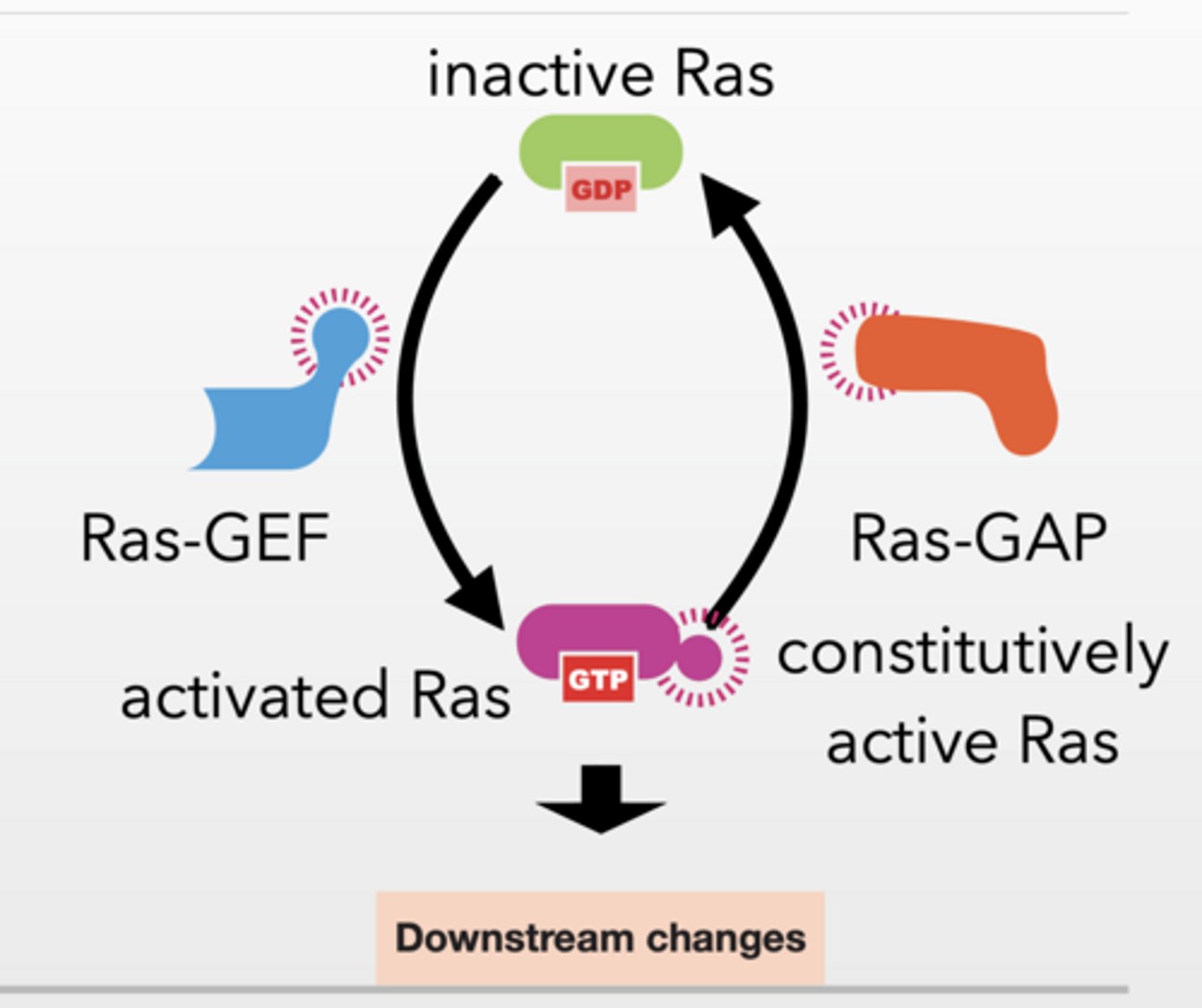
Feet of this bs
feet
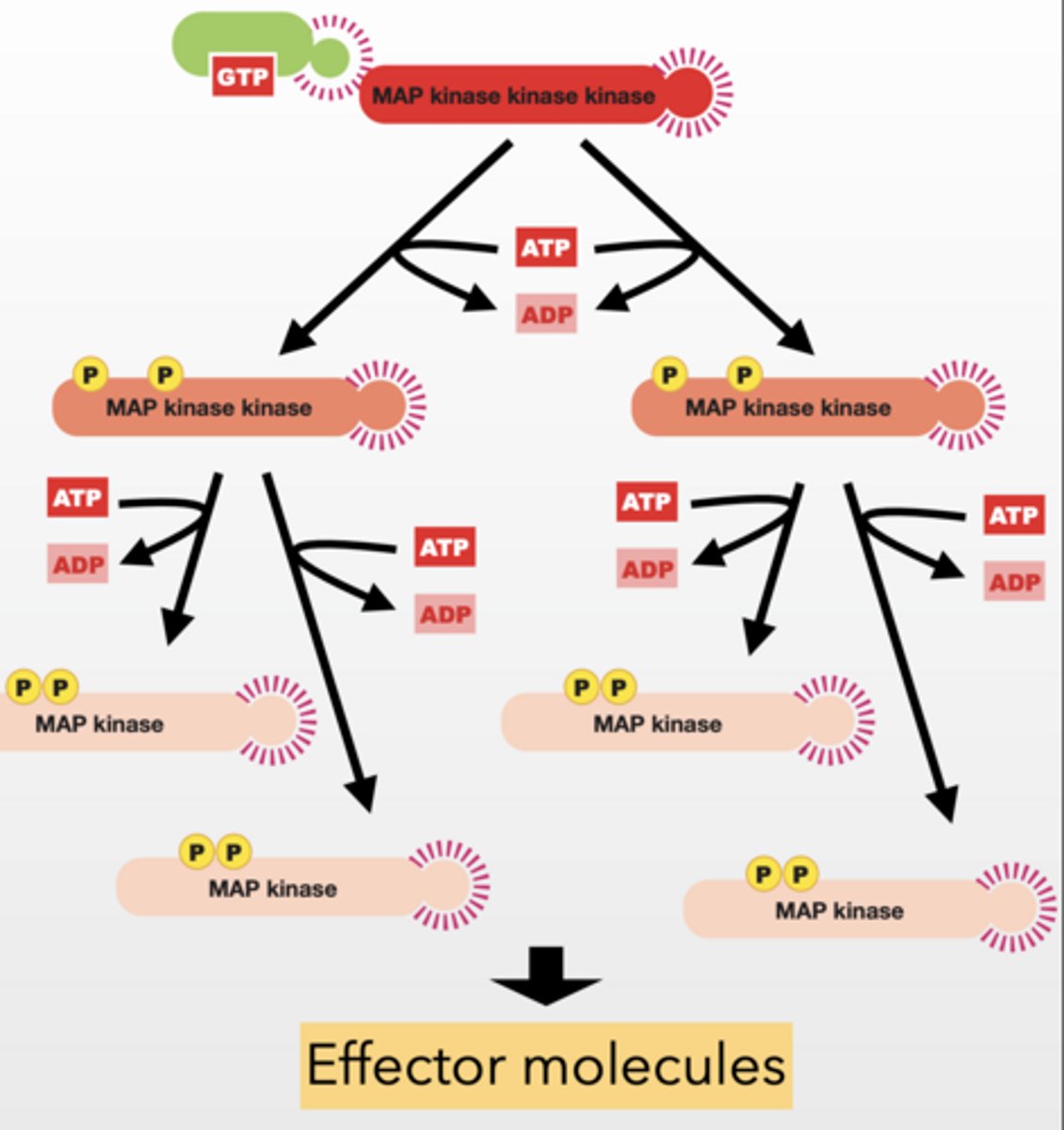
MAP Kinase Cascade
MAP kinase kinase kinase (MAPKKK) -
phosphorylate and activate MAP kinase kinases
Signal Amplification
Feedback regulation
Ras TURNS ON MAPKKK
MAP kinase kinase (MAPKK) - phosphorylate and activate MAP kinases
MAP kinase (MAPK) - phosphorylate effector molecules
Results in Changes in cellular function
Signal Amplification
Feedback regulation
Two ways
Fast
protein X, Y → immediate activity changes
Slow
transcription factors → gene expression
Used by many processes in the cell
. NGF - neuronal cell growth
PGDF - wound healing
Highly mutated in cancer
HOW DOES THE CELL TURN THIS OFF?
Tyrosine phosphotases Remove signal molecule Feedback regulation in MAPK cascade Internalization of receptor and degradation by lysosome using endocytosis
Whole thing feet
feet
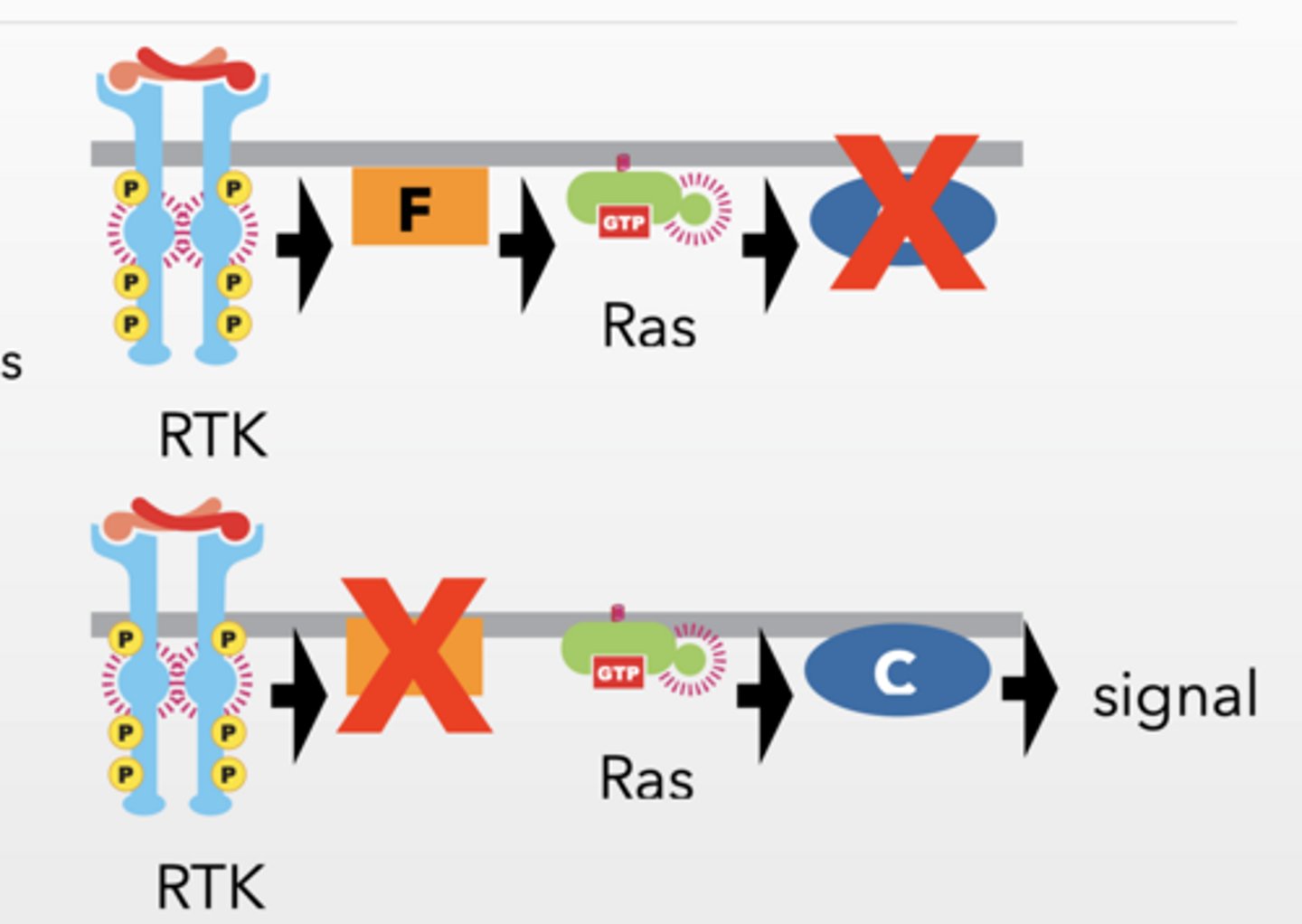
Theoretical pathway order RTK → F → Ras → C → MAPK → cell division
RTK → F → Ras → C → MAPK → cell division
WITH CONSTITUITIVE Ras
Mutation stops signaling
Protein is REQUIRED
Overactive upstream rescues THEN
Protein is UPSTREAM
Overactive upstream does NOT rescue THEN
Protein is DOWNSTREAM
Constitutively active upstream proteins bypass upstream defects but not downstream defects.
More info
CELLS CAN RESPOND DIFFERENTLY TO THE SAME SIGNAL
Acetylcholine induces a different response in salivary cells and skeletal muscle cells
same ligand can activate different kinds of Gproteins depending on the cell
MULTIPLE SIGNALING PATHWAYS CAN BE STRUNG TOGETHER
CELLS INTEGRATE MULTIPLE SIGNALS
G-PROTEINS AND GTPases WORK EVERYWHERE for MOSTLY GROWTH
GTPases (like Ras) are used in MANY pathways
Cellular signaling pathways are interconnected networks that integrate multiple signals to produce context-dependent response
G-protein coupled receptors Pathway Step 1
G-PROTEIN COUPLED RECEPTORS (STRUCTURE)
7 transmembrane spanning helices
Family of proteins includes Rhodopsin (light activated photoreceptor)
signal binding happens deep inside pocket created by helices
controls cAMP and Ca+ among other second messengers
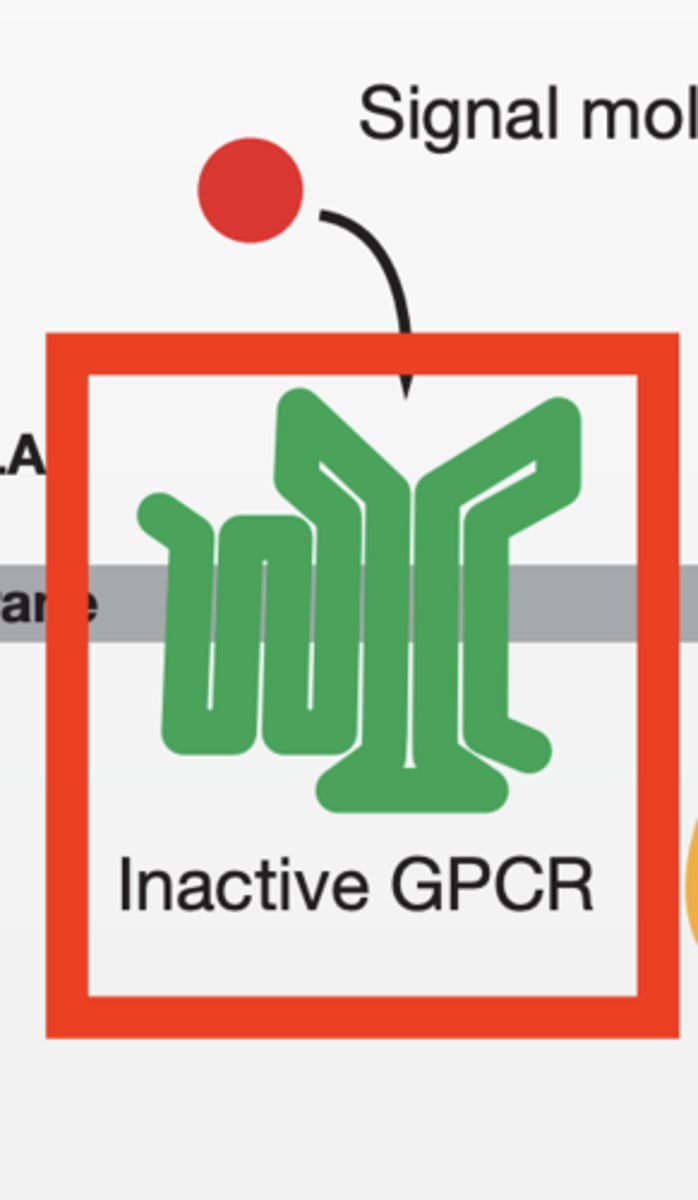
Step 1: No signal
GPCR = inactive
G protein = GDP-bound (OFF)
STEP 2: Signal binds
ligand binds GPCR
GPCR changes shape
Step 2
AFTER SIGNAL BINDS
GPCR becomes active
All subunits of G protein are inactive and complexed
THEN
GPCR facilitates GTP exchange in alpha
GTP bound alpha is active and separate from beta/gamma
G-PROTEIN SUBUNITS ACTIVATE OTHER PROTEINS
G-proteins are specific for a set of receptors or targets
3 protein subunits alpha, beta, gamma subunit separate and become active
Alpha works alone beta/gamma are complexed
Alpha and beta/gamma bind to proteins to alter effector activity - often regulate 2nd messenger
Gi - inhibitory and Gs excitatory/activating
Alpha hydrolyzes GTP into GDP and beta/gamma rejoin inactive complex - occurs in ~ 1sec
GDP for alpha = off
GTP = on
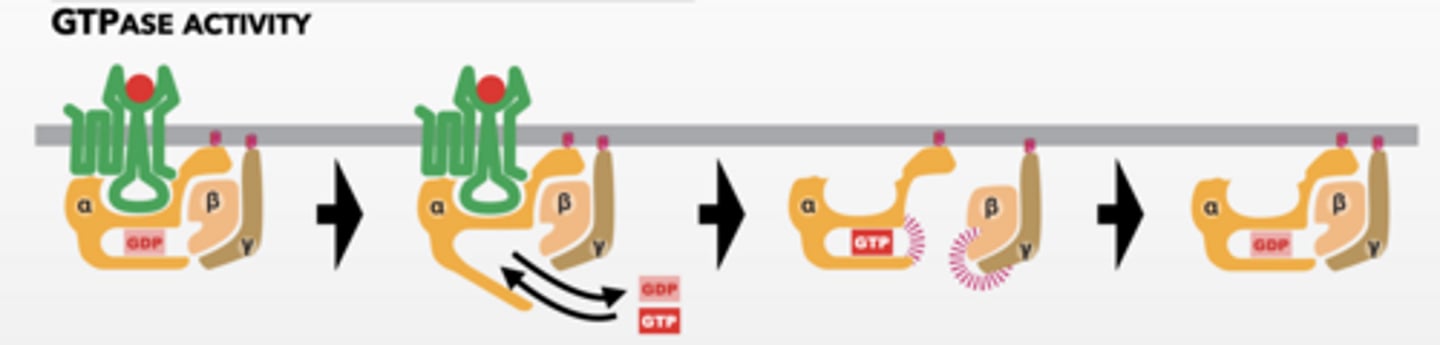
Step 3 of this
After activation:
α (with GTP) goes and activates enzymes (like adenylyl cyclase)
βγ can ALSO activates (like ion channels)
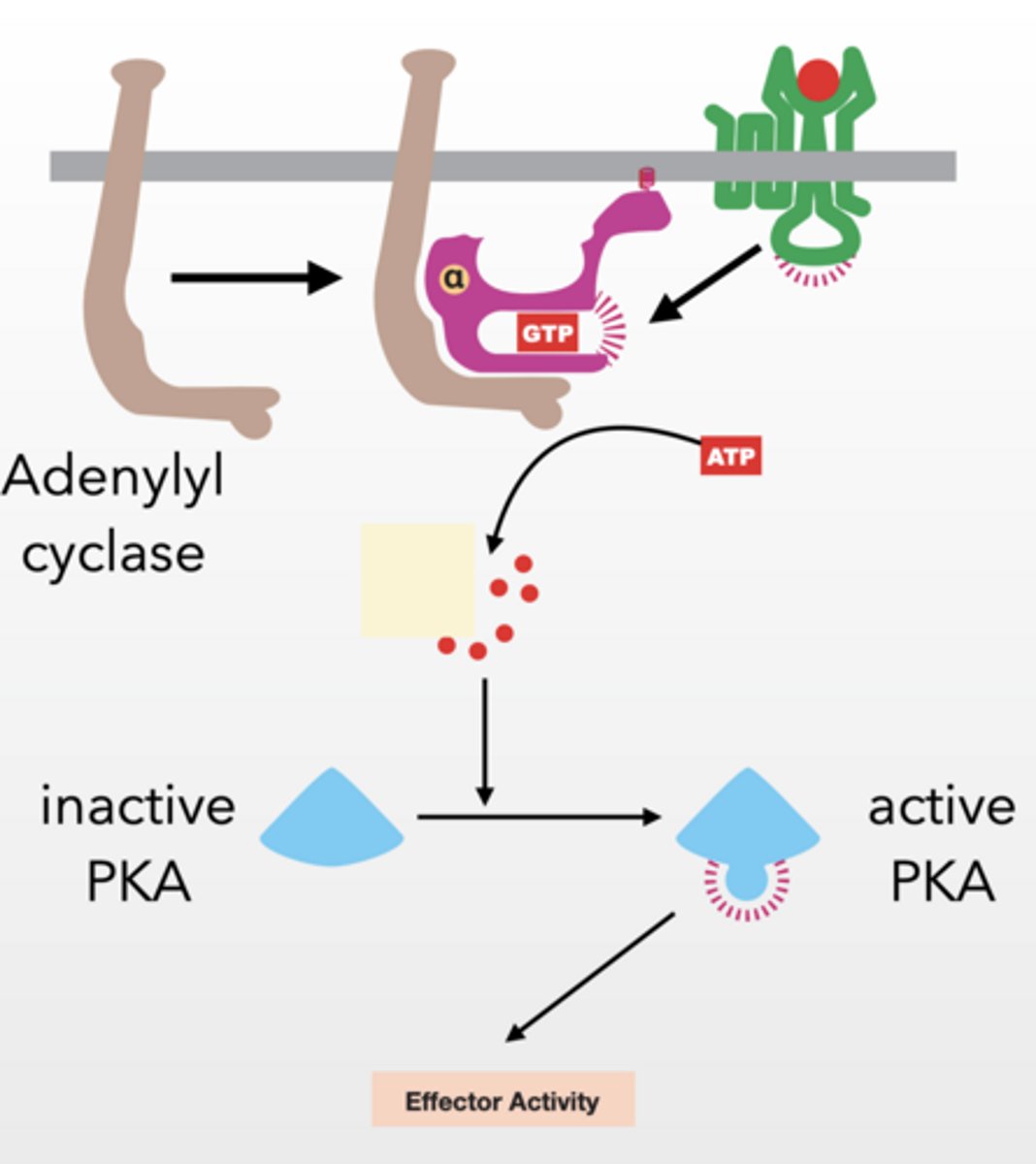
PKA - PROTEIN KINASE A
Alpha subunit activates adenylyl cyclase
Gs type g-protein because it stimulates adenylyl cyclase
Adenylyl cyclase → makes cAMP
cAMP activates PKA by releasing it from a regulatory protein
cAMP similar to cGMP, highly regulated second messenger
PKA prolific protein kinase
PKA → phosphorylates proteins → cell response
serine/threonine kinase
cAMP is degraded by a phosphodiesterase (PDE) (just like cGMP) keeping cellular concentrations low
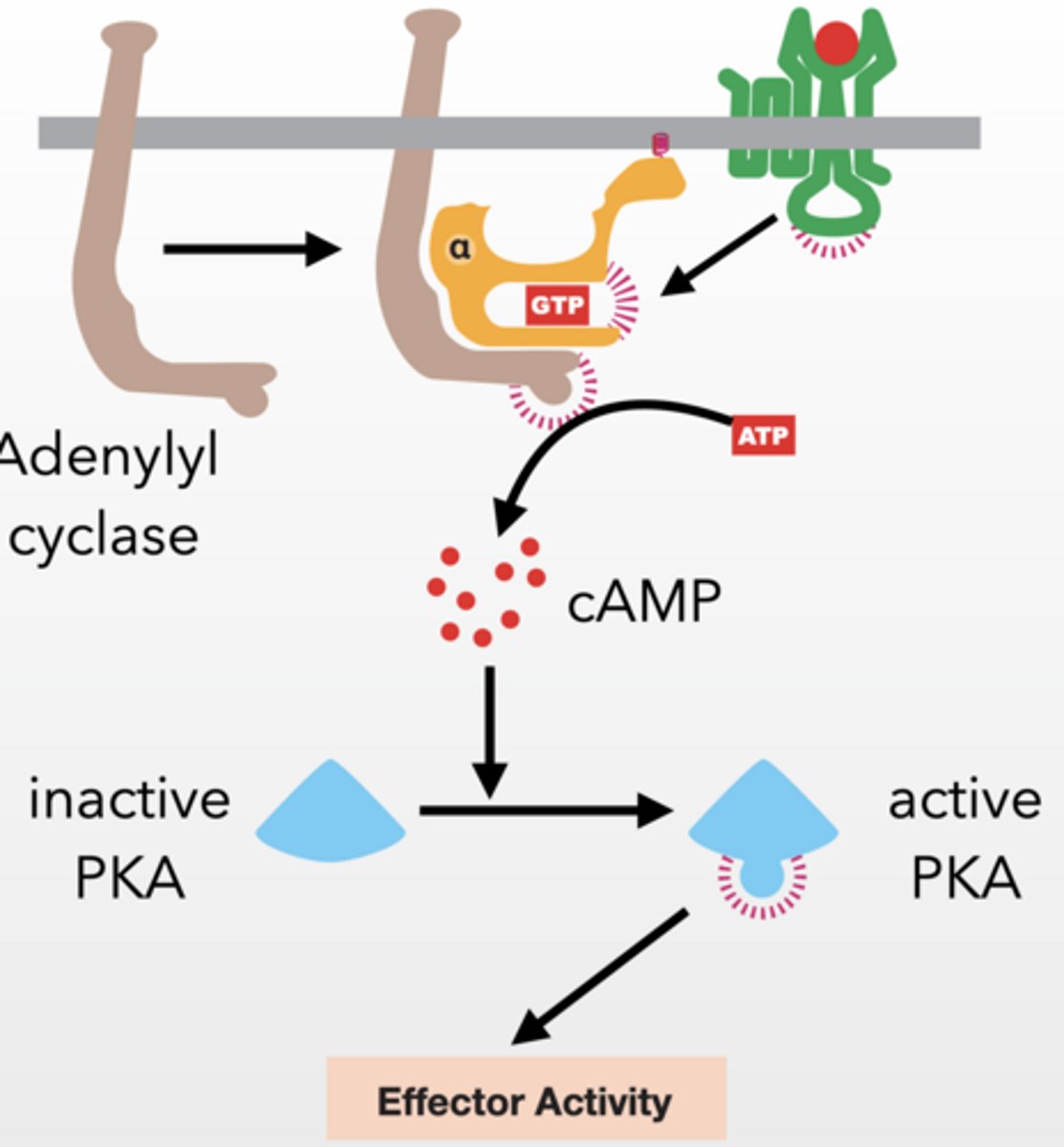
PKA effector response
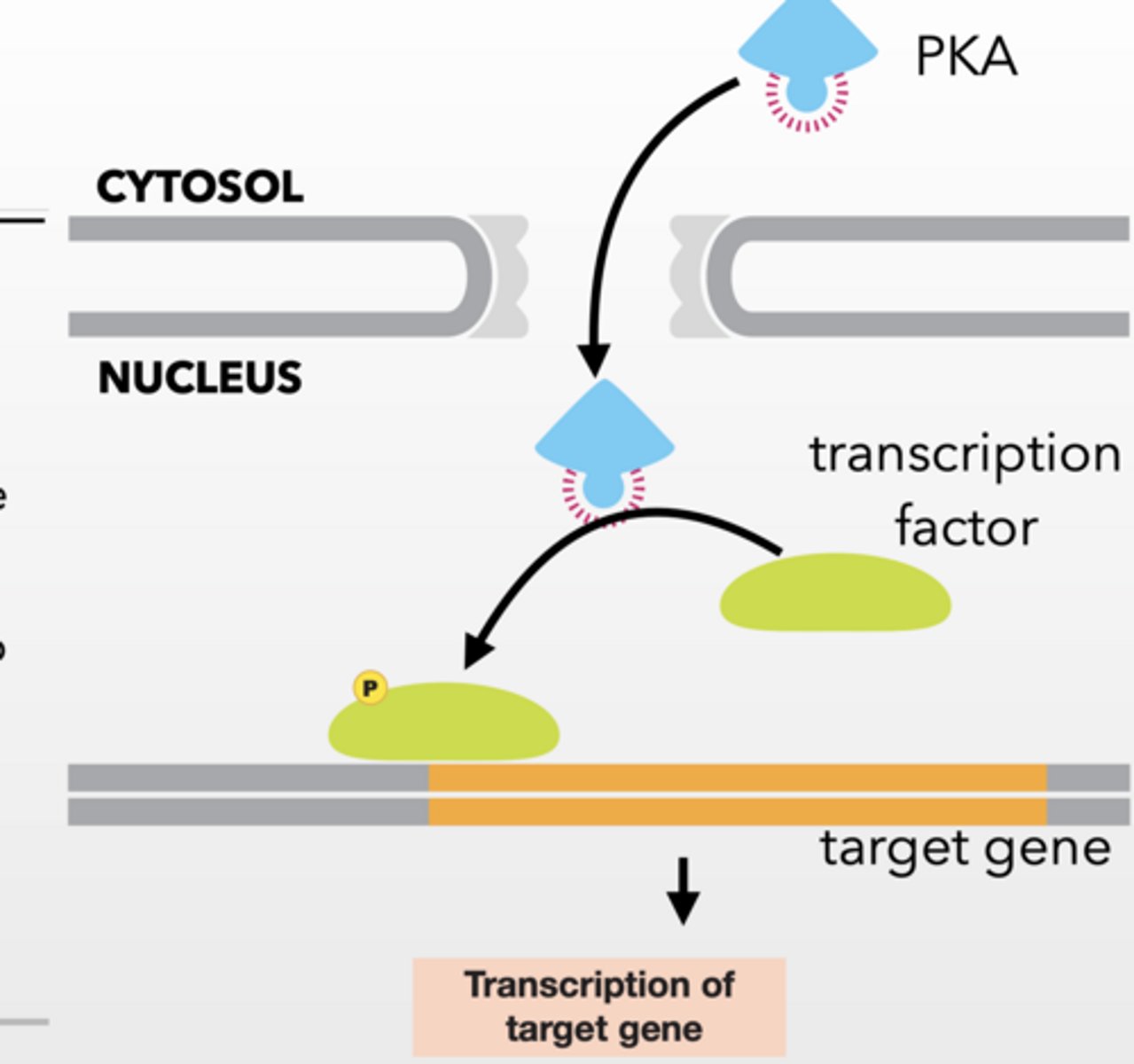
SLOW EFFECTOR RESPONSE
Activated PKA can be transported to the nucleus
Transcription is regulated in response to PKA phosphorylation of target proteins
Slow Response
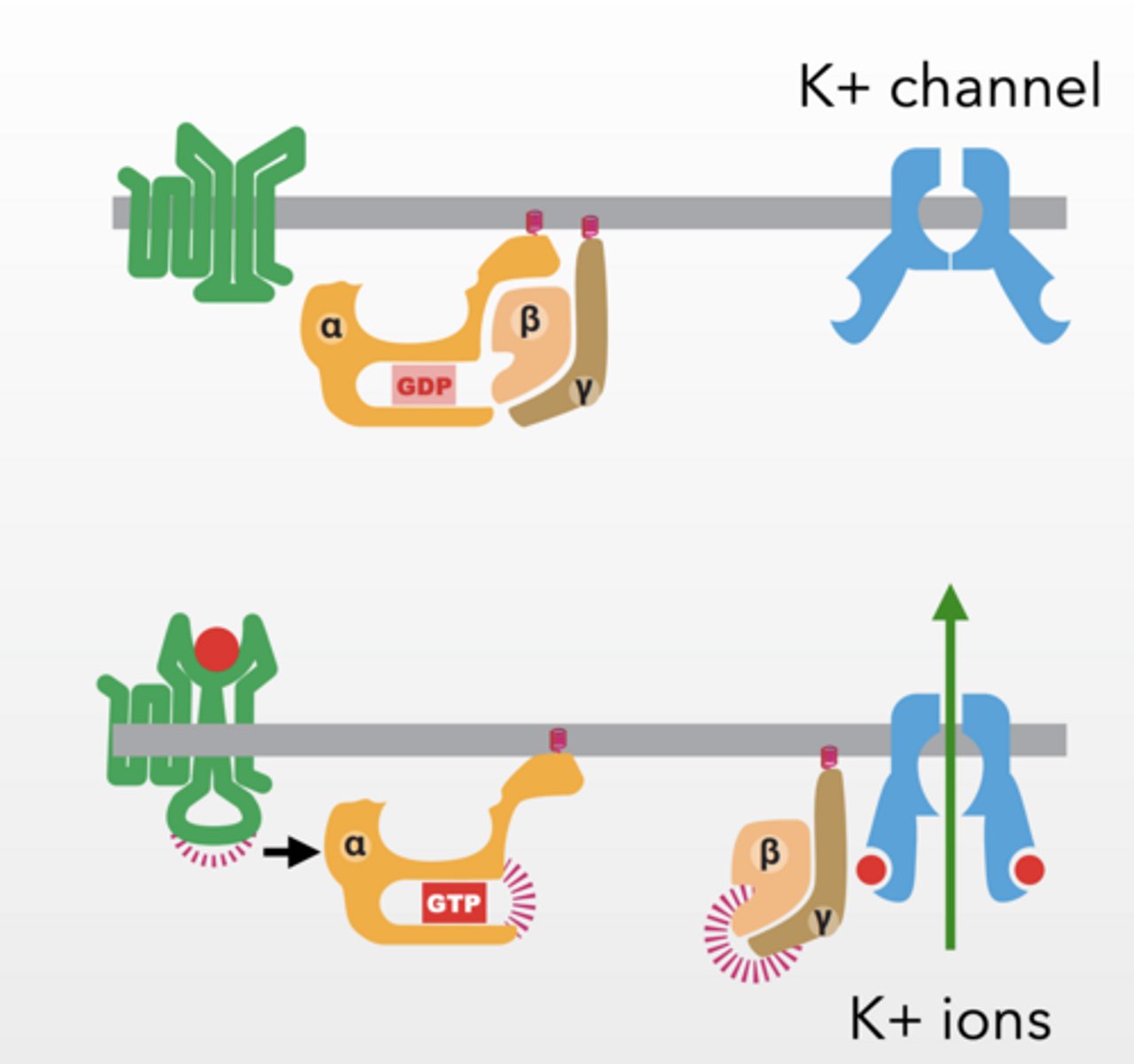
HOWEVER
G proteins can effect ion channels to have immediate effect
Beta/gamma interact with interacts with K+ channel and allow K+ to flow across membrane
Gi G-protein inhibits muscle contraction in the heart by disrupting voltage gradient
K+ feet
feet
WHAT WOULD HAPPEN IF
you drink caffeine?
cholera bacteria invade the body and produce cholera toxin?
activate a Gi subunit of g-protein that inhibits adenylyl cyclase?
You drink caffeine?
Caffeine inhibits the PDE that degrades cAMP to AMP
PKA remains active for a longer period of time
More effector activity
cholera bacteria invade the body and produce cholera toxin?
Toxin causes a constitutively active alpha subunit
Adenylyl cyclase stays active for a longer period of time making more cAMP
PKA stays active for a longer = increased effector activity
activate a Gi subunit of g-protein that inhibits adenylyl cyclase.
Adenylyl cyclase is prevented from activating
all downstream processes slow or stop
GS ACTIVATES , GI inhibits
Cell division
CELLS DIVIDE AND FORM 2 DAUGHTER CELLS
Cell cycle varies in time
DAUGHTER CELLS ALSO INHERIT GENE EXPRESSION PATTERNS AND DNA MODIFICATION STATES
CELL CYCLE Not drawn to scale
M phase much shorter
G1 much longer
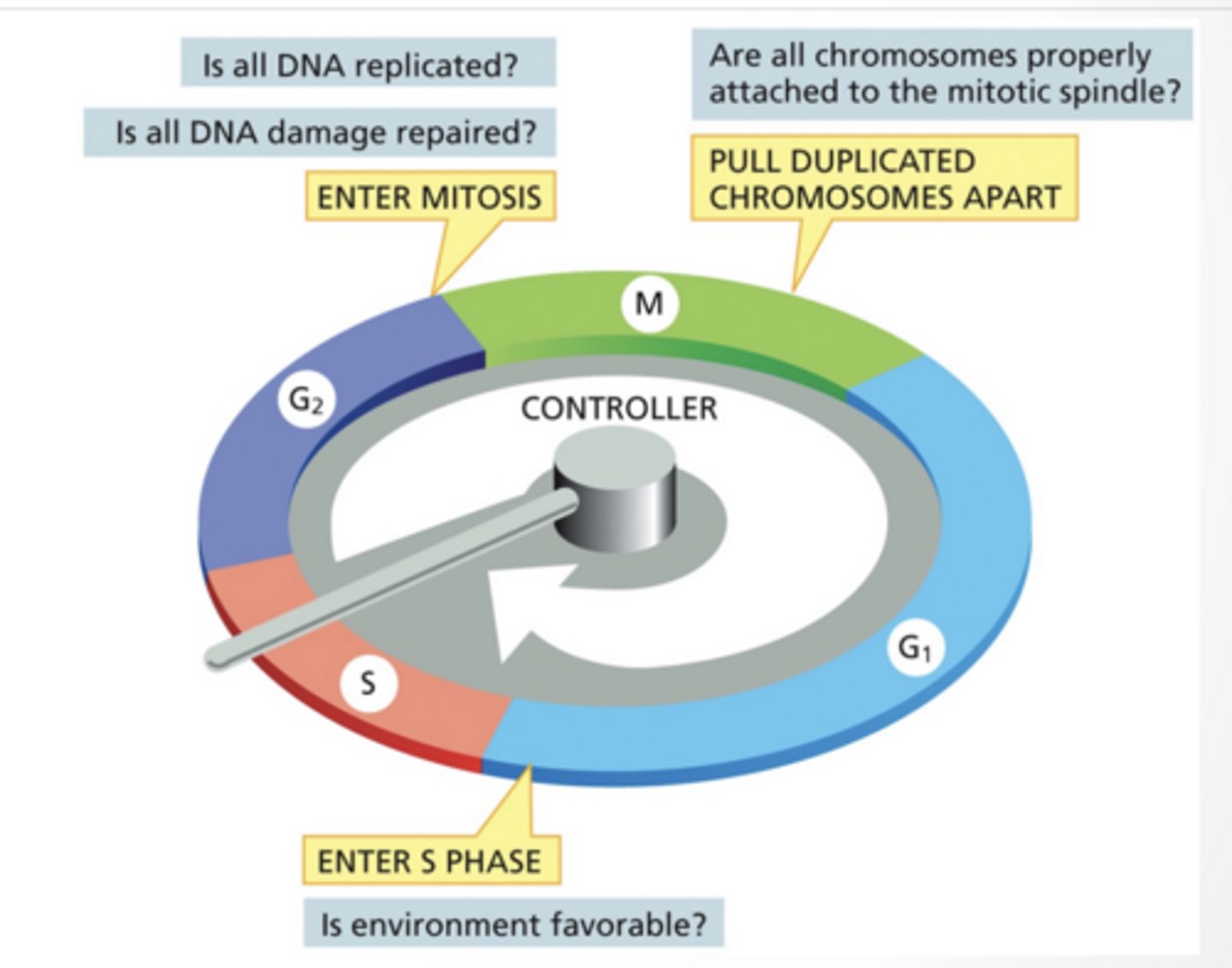
CHECK POINTS CONTROL WHEN A CELL ENTERS A DIFFERENT PHASE
Think of these as "if/then" statements
If DNA is fully replicated cell cycle can proceed
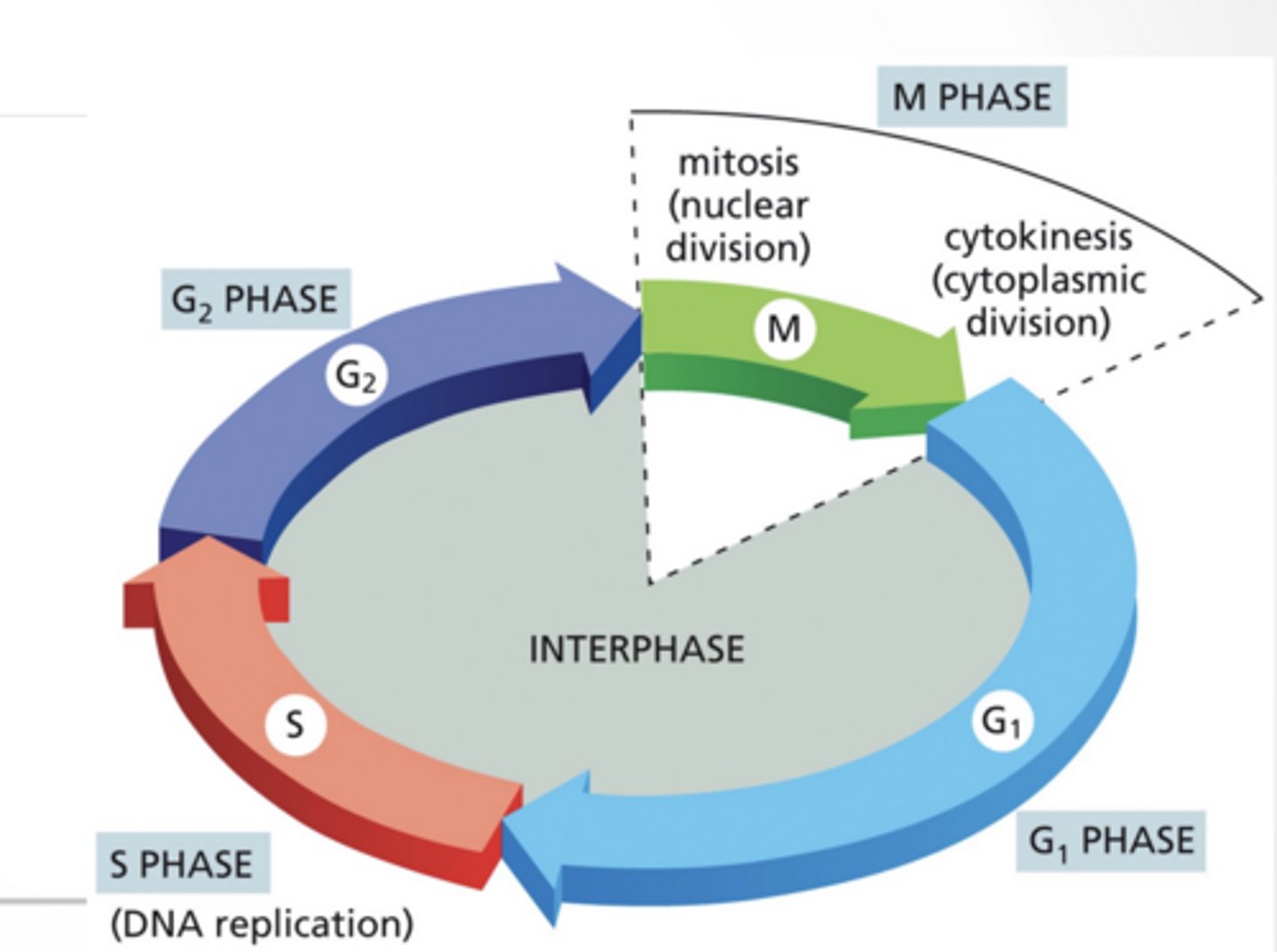
The cycle:
G1
S → copy DNA
G2
M → divide
M phase :
1. Prophase
Chromosomes condense and become visible as sister chromatids. The mitotic spindle begins forming as centrosomes move apart.
2. Prometaphase
The nuclear envelope breaks down. Spindle microtubules attach to chromosomes at kinetochores and begin moving them.
3. Metaphase
Chromosomes align at the center of the cell (metaphase plate). Each chromatid is attached to opposite spindle poles.
4. Anaphase
Sister chromatids separate and are pulled toward opposite poles. This ensures each daughter cell gets identical chromosomes.
5. Telophase
Chromosomes arrive at the poles and decondense. New nuclear envelopes form around each set, creating two nuclei.
6. Cytokinesis
The cytoplasm divides via a contractile ring. This produces two fully separate daughter cells.
CDKS
CDKS INiTIATE SPECIFIC STEP IN THE CELL CYCLE
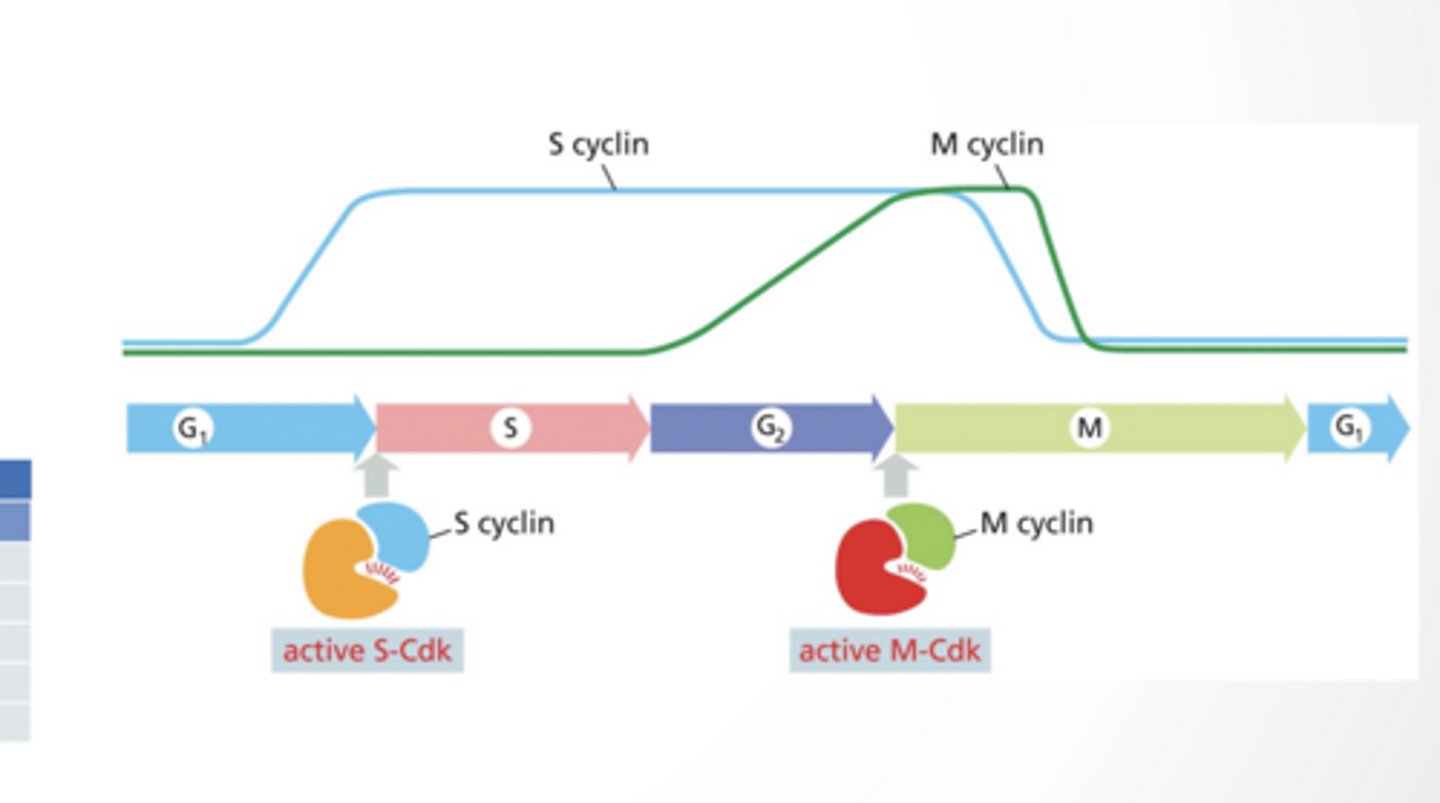
Cyclin dependent kinase :Regulatory protein, cyclin, must bind to CDK for CDK to become active
CDKs phosphorylate specific proteins to initiate the next phase
Amount of CDK changes through cycle
SPECIFIC CDKS FUNCTION AT EACH PHASE
G1 cyclin/CDK
S cyclin/CDK
M cyclin/CDK
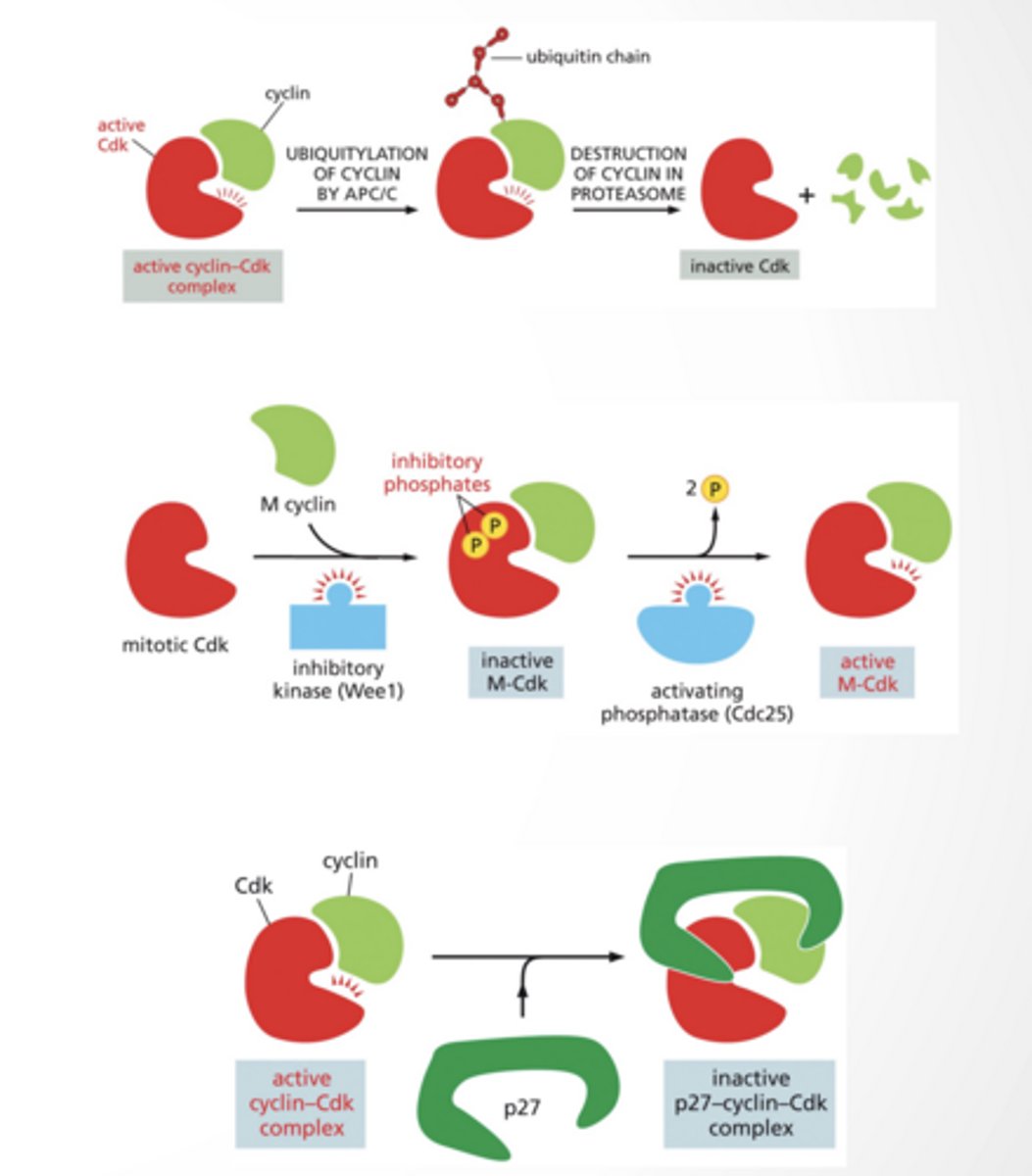
CDK inhibition
CDK INHIBITION
Different ways to inhibit CDKs
Ubiquitination by APC/C --> degradation in proteasome
Dephosphotylation
Binding of inhibitor (like p27)
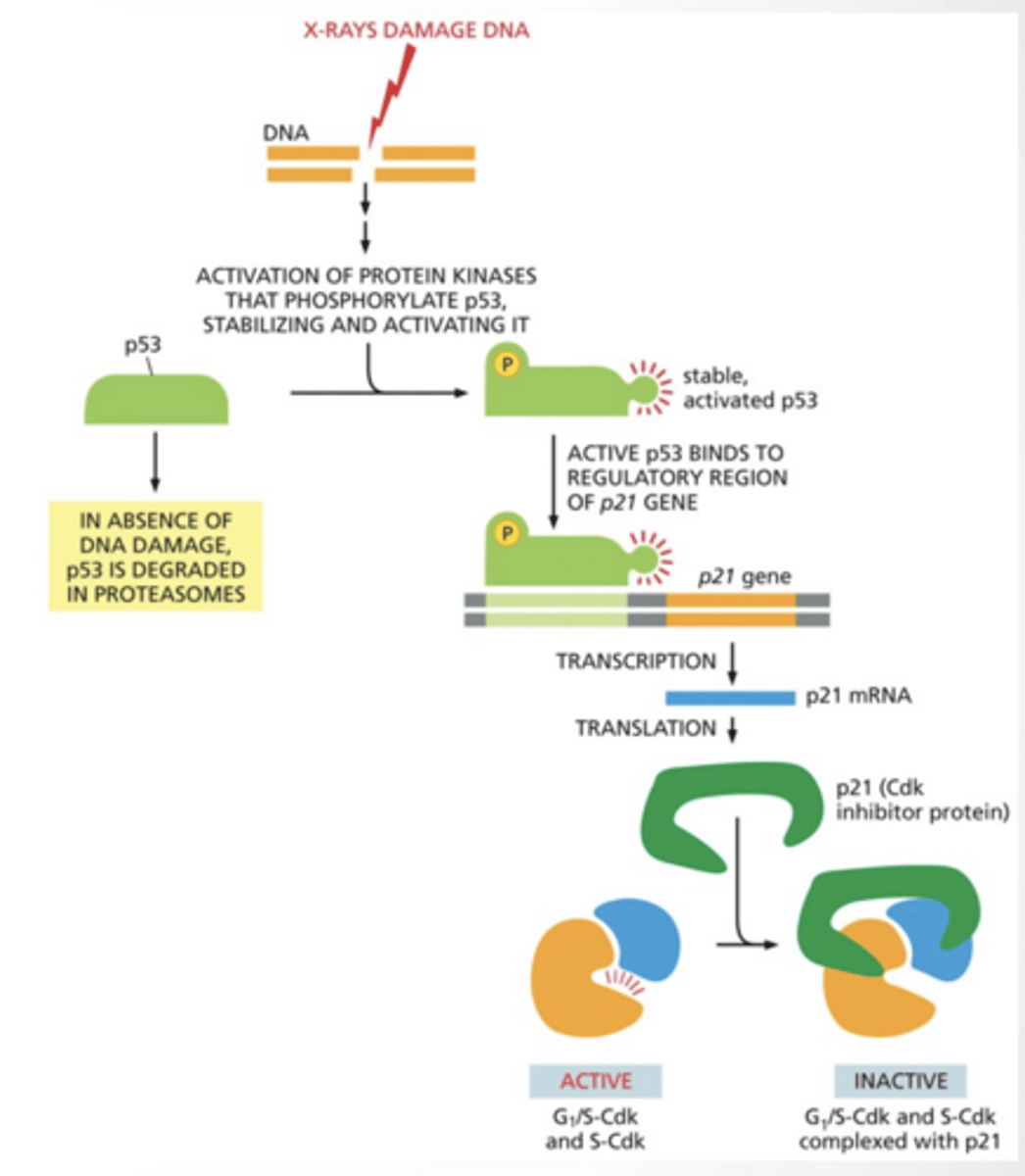
P53 IS A TUMOR SUPRESSOR
Tumor suppressor - prevents tumors by stopping the cell cycle if something is wrong
p53 gets phosphorylates by protein kinase and binds to p21 gene and activates it.
p21 inhibits G1/S CDK
SELECTED EVENTS FROM PHASES OF THE CELL CYCLE
PHOSPHORYLATION CONTROLS NUCLEAR MEMBRANE BREAKDOWN
membrane breaks apart
MICROTUBULES BIND TO THE KINETOCHORE
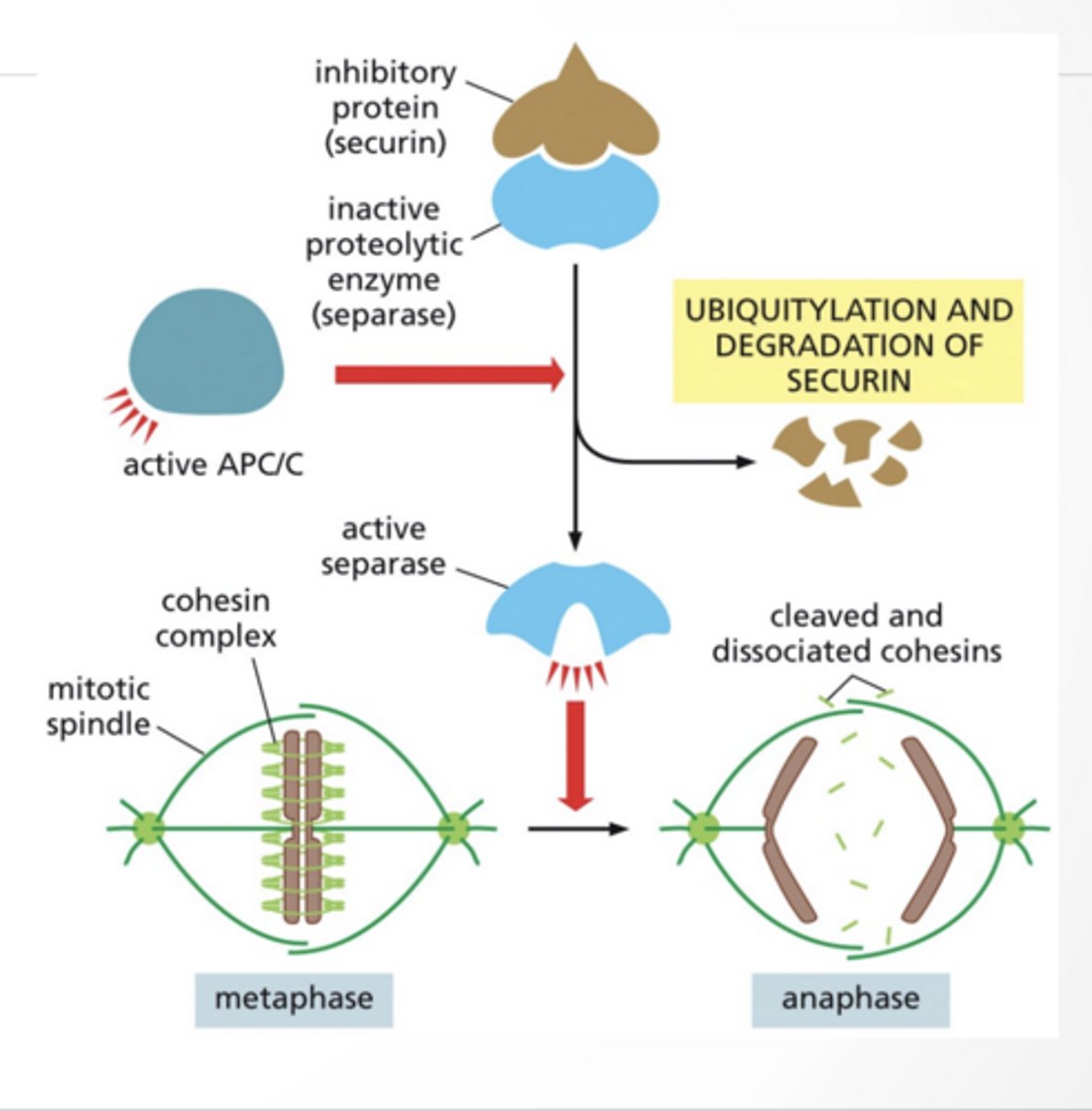
ANAPHASE REGULATION
ANAPHASE A: chromosomes are pulled poleward
APC/C complex:
ubiquitinates securin
securin gets destroyed
separase becomes active then cuts cohesin (the glue holding chromatids)
chromosomes separate
CELL SIZE
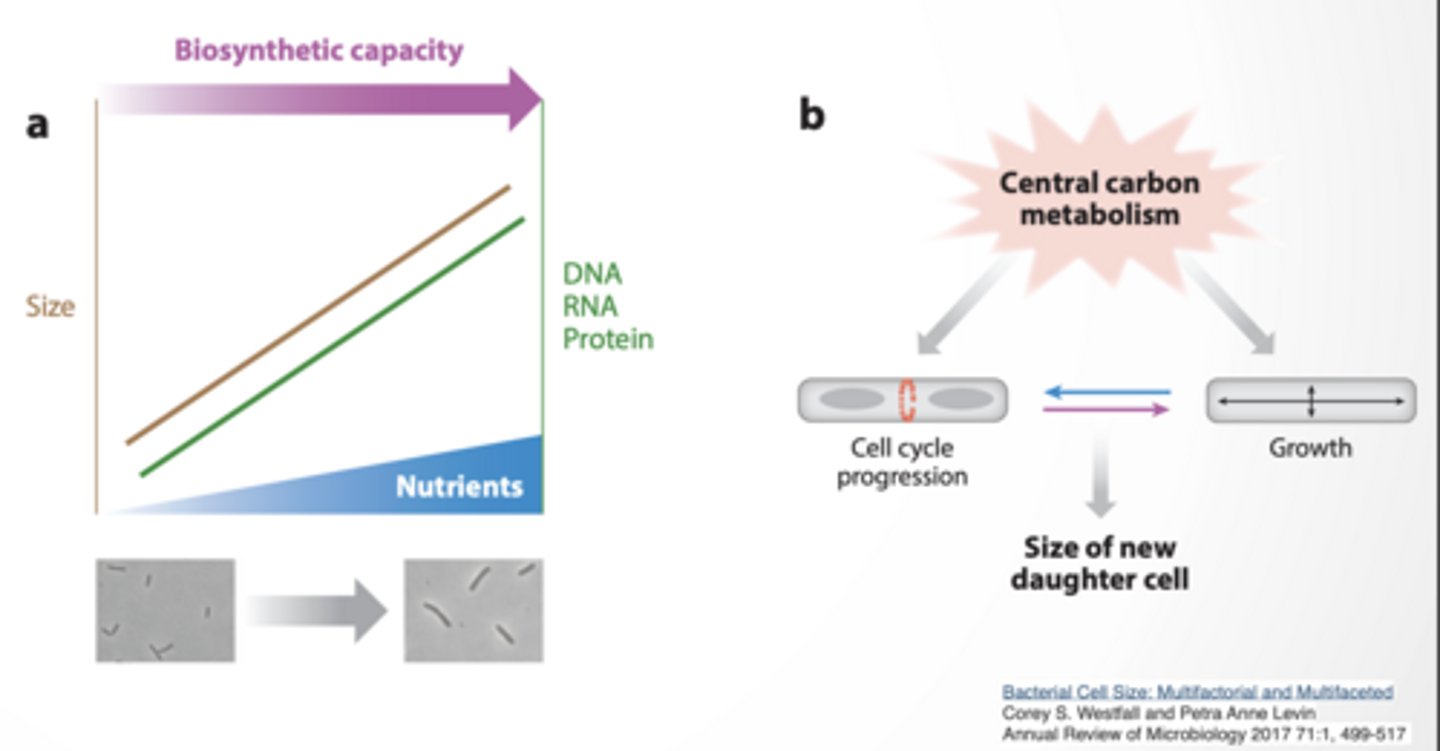
BACTERIAL CELL SIZE SEEMS TO DEPENDENT ON NUTRIENT AVAILABILITY
Ratio of DNA/RNA/protein to cell size remains constant Timing of cell division vs growth determines size of daughter cell
If cell divides EARLY → small cells If it grows LONGER → bigger cells
EXTRACELLULAR SIGNALS CONTROL GROWTH IN LARGER ORGANISMS
Saturation level of growth factors can control cells size
Paracrine
nearby cells send signals
Local