Food Chain
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
Cells
are made of chemical substances: large molecules like proteins, small molecules like water and sugars, and ions like sodium (Na+).
the chemical bonds are made of energy.
All living things take in new chemical substances
microbes and fungi absorbs nutrients through their cell membrane.
plaants absorb CO2 from the air and take in fertilizer in soil water.
Purpose of intake of chemical
1) Matter: new atoms, ions and small molecule are needed to build and fix cells and secreted substance like bone, shells, milk.
2) Energy: can be extracted from molecular bonds for cellular work like pumping ions cross membranes and muscle cell contraction.
The two wastes
1) Matter: Plants give off O2 waste. Organisms that ingest food break down most of the food molecule to extract chemical bonding energy.
2) Energy: as molecular bonds are broken and reformed, there is always some energy lost as waste heat.
Food chain
track of how materials and energy are passed from one organism to the next.
Food Webs
track chemical substances in
Two things that are tracked are matter and energy
cyanobacteria, micro-algae, larger algae.
Aquatic ecosystem have
‘‘Producers’’
Photosynthesizers are called
b/c they are producing organic molecules out of tiny molecules like CO2, water, and fertilizers

‘‘Consumers’’
All other organisms- herbivore, omnivores, carnivores, and detritivores like worms and carbs.

‘‘Decomposers’’
like fungi and bacteria breaks down materials creating. very simple molecular wastes that can directly be reused again by producers they don’t poop.

Autotroph
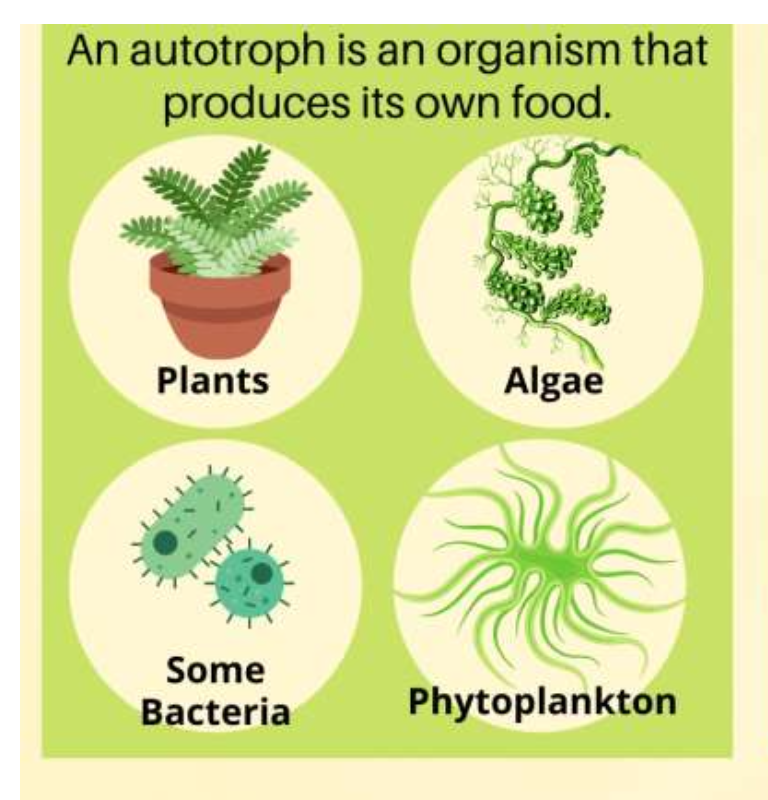
heterotroph

Primary consumers and second consumers
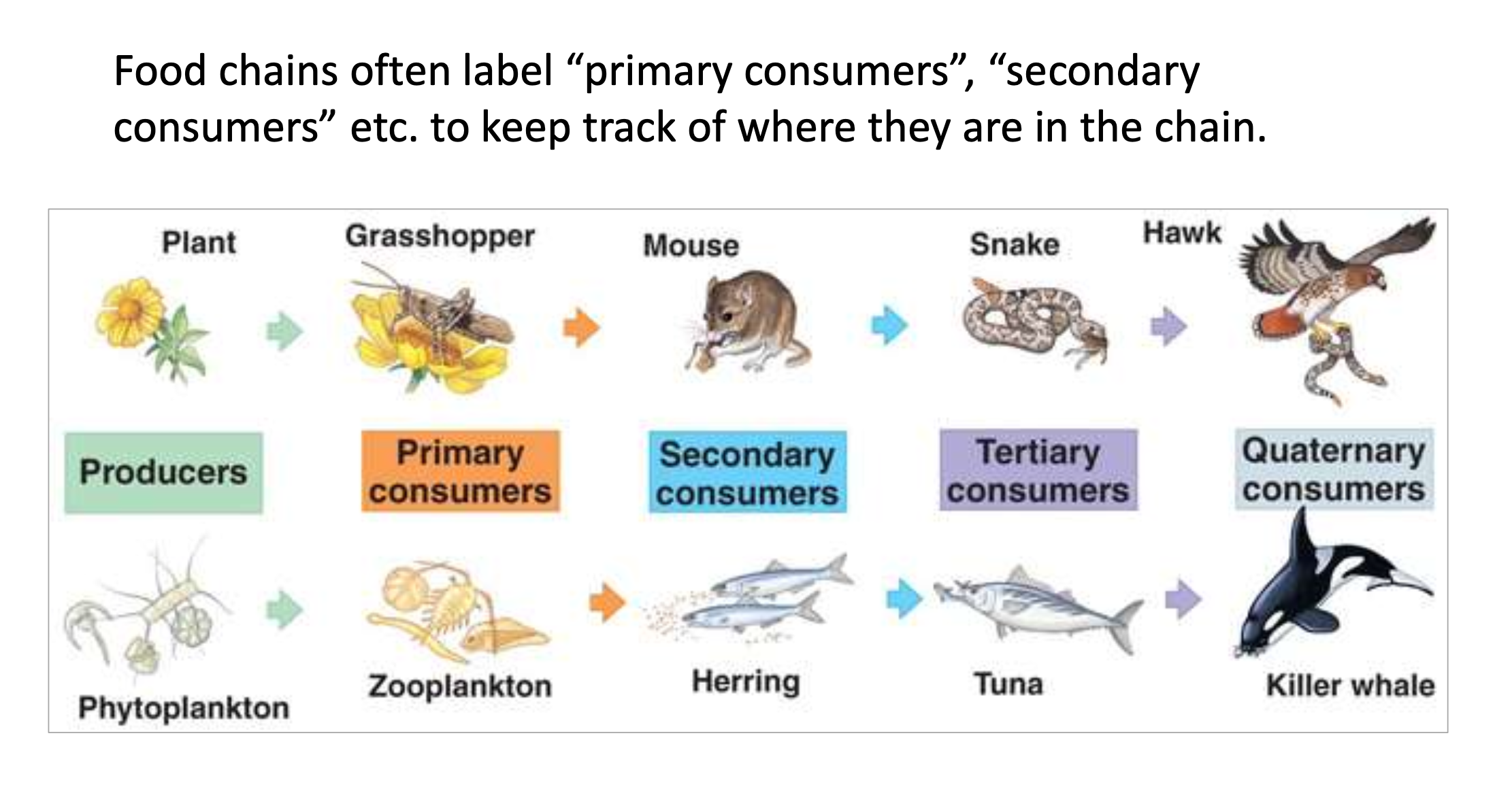
‘‘Trophic level’’
each step or level of the food chain is referred to
‘‘food level’’
keep track of primary consumer and secondary consumer
decomposers
are often omitted from food chain and webs but are the key components of processing everyones waste and dead bodies at each step of the chain, converting molecules Into smaller ones.
Matter and Energy
Tracking ‘‘food’’ through an ecological system involves tracking both
Waste heat
Wasted energy gives off ……. that is why you are warm.
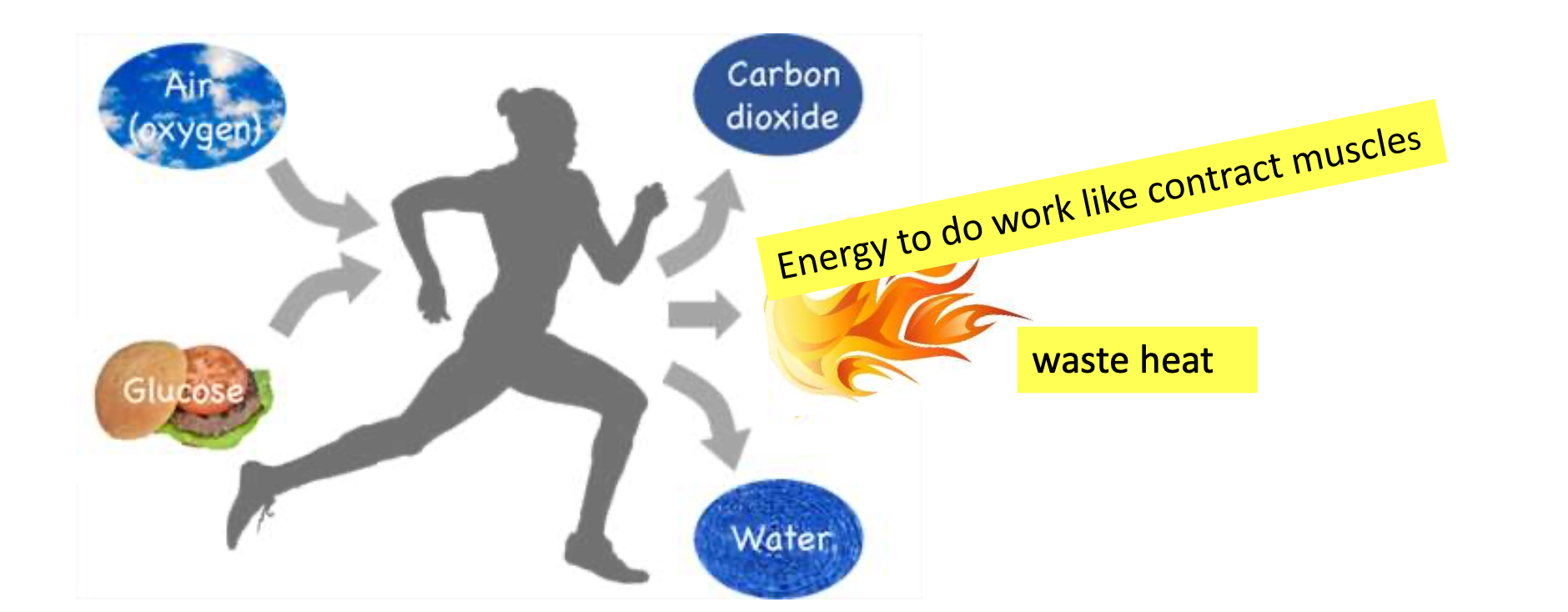
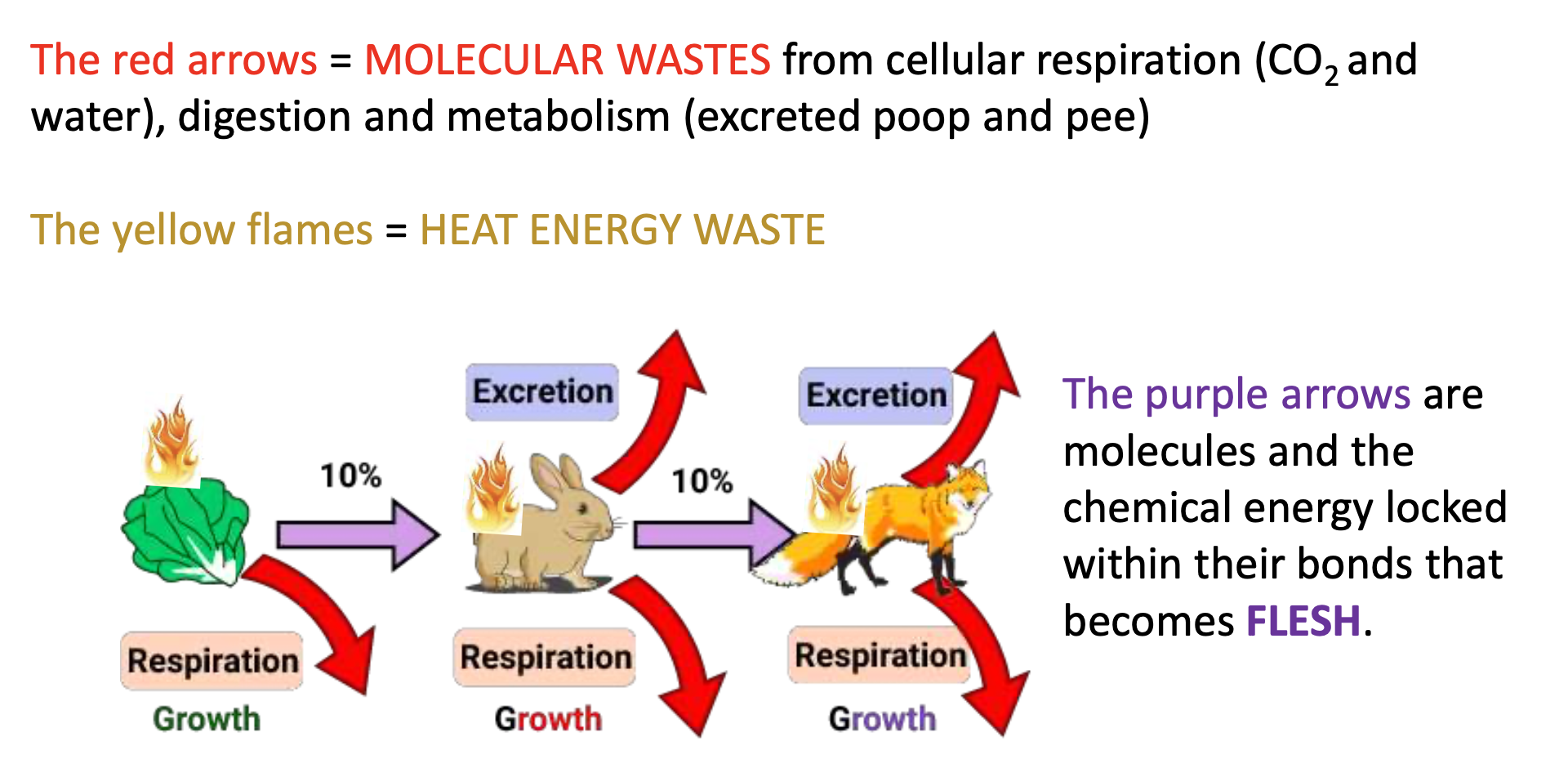
Ex of measured waste molecules and heat energy
10% and 90%
On average only 10% of the food energy stays in the food chain at each step. 90% is lost as waste heat.
Consumers inhale/ excrete moste of the waste atoms and energy, which very little sticks to their body.
Carnivores
The loss of matter and energy at each step of the food chain explains why food chains are so short and why top carnivores are relatively rare.
One Primary consumer example
Grasshoppers eat lots of producers. Grasshopers give off waste molecules and body heats.
One Secondary consumer example
Shrews eat lots of grasshoppers. They give off waste molecules and body heat.
by the time we get to top carnivores, there is not enough food left to support very many of them.
Ex. It takes 10,000 kilocalories of seeds for a growing mouse to put on 1000 kcal of flesh. The mouse burns off the rest.
You can track the 10% rule using energy units also like calories
matters for also considering how many people are trying to be feed and how to conserve enough habitat for other species and other resources.
Cold blooded
Cold-blooded animals like fish, clams, and squid do not burn as much as food as active warm blooded birds and mammals
Biomagnification
another reason to eat lower on the food chain if possible is that fat-soluble pollutant build up in food chain.
Each time a consumer eats food, it is getting all the toxins that build up in their prey bodies.
some toxins like heavy metal stick to fat and oils and are not easily excreted. It builds up over its life time.
some animals are at more risk of absorbing/eating toxins than others depending on where they live and what they eat.
Carbon, nitrogen, phosphorus
the cycle to become different molecules.
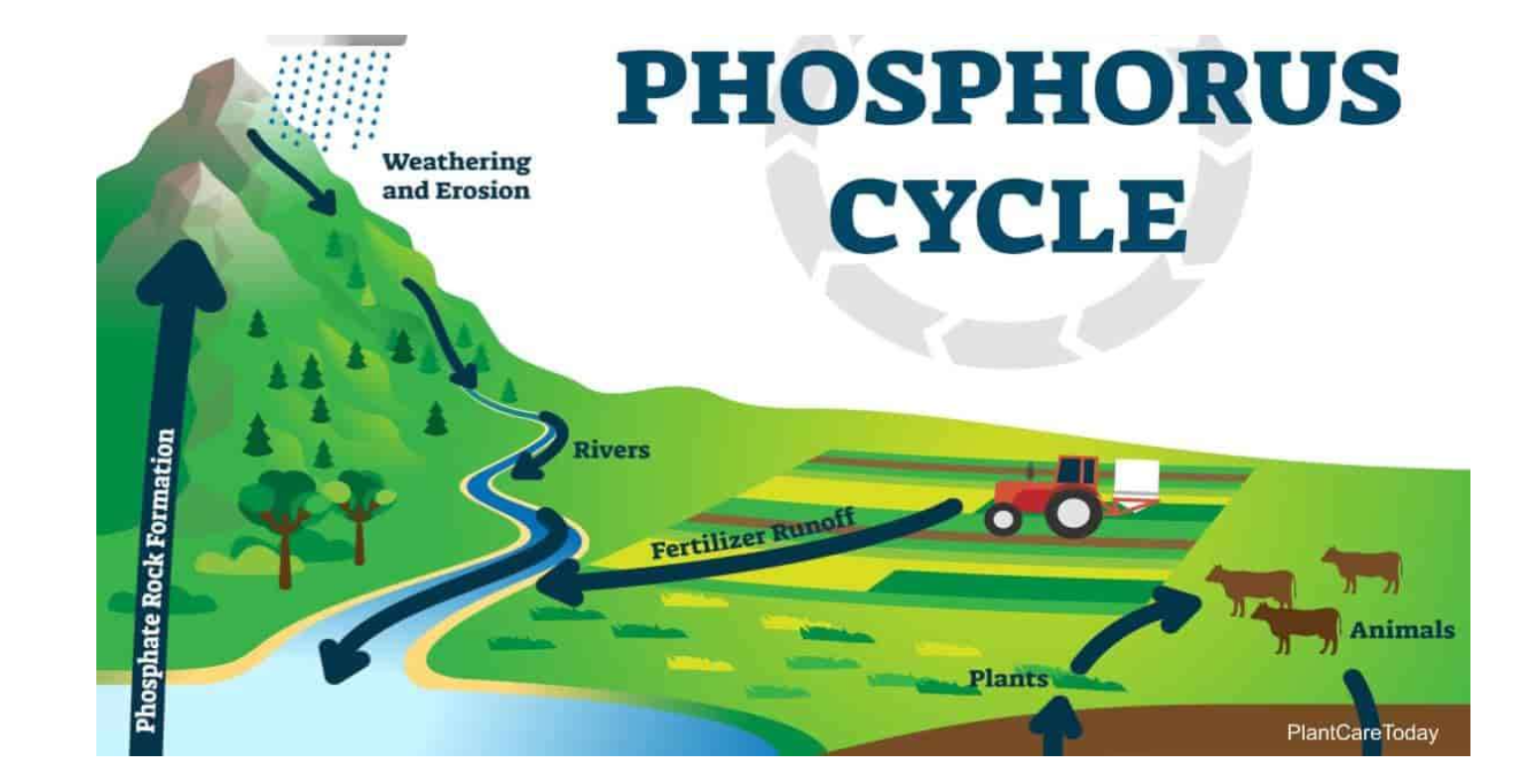
Phosphorus cycle
these atoms.are found in cells, sewage, compost, and some rocks.
describes the transformation and translocation of phosphorus in soil, and water.
not found in gas molecules.
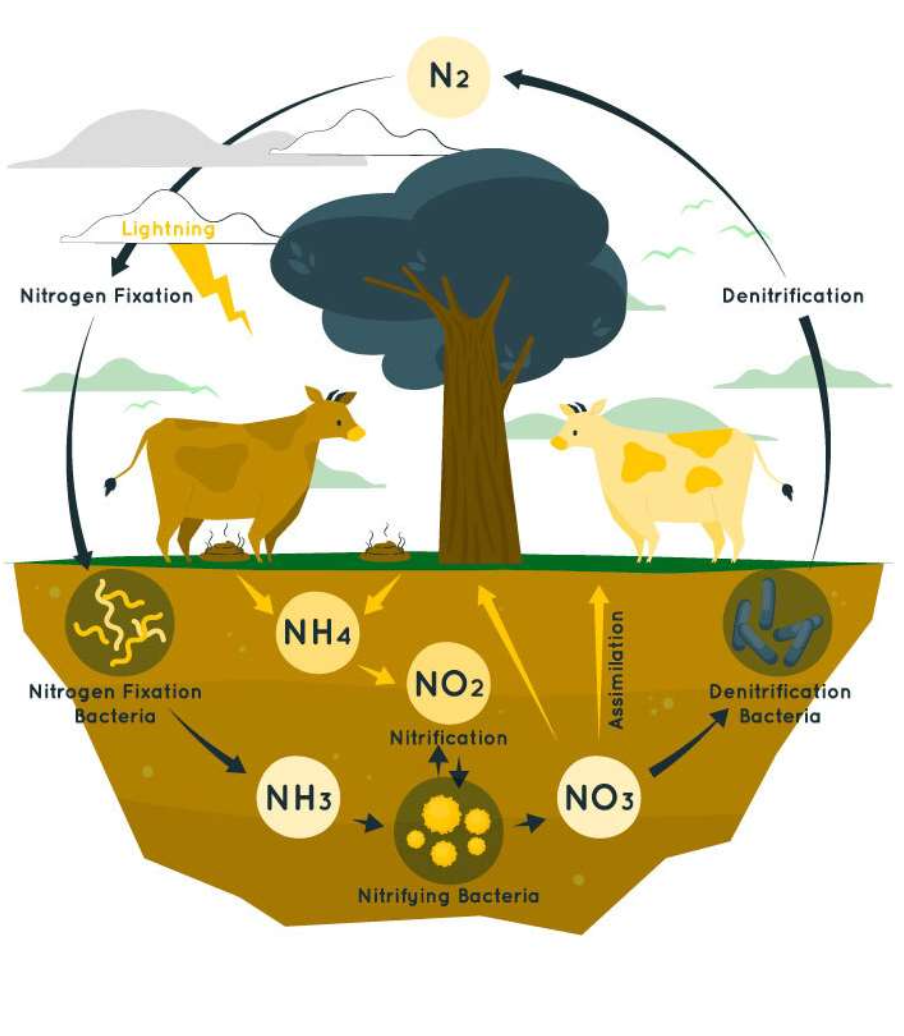
Nitrogen cycle
atoms are found in N2 gas, in cells, in wastes, and various small fertilizer molecules like ammonia.
bacteria converts N2 gas into ammonia
ex. poop, pee, fertilizer,
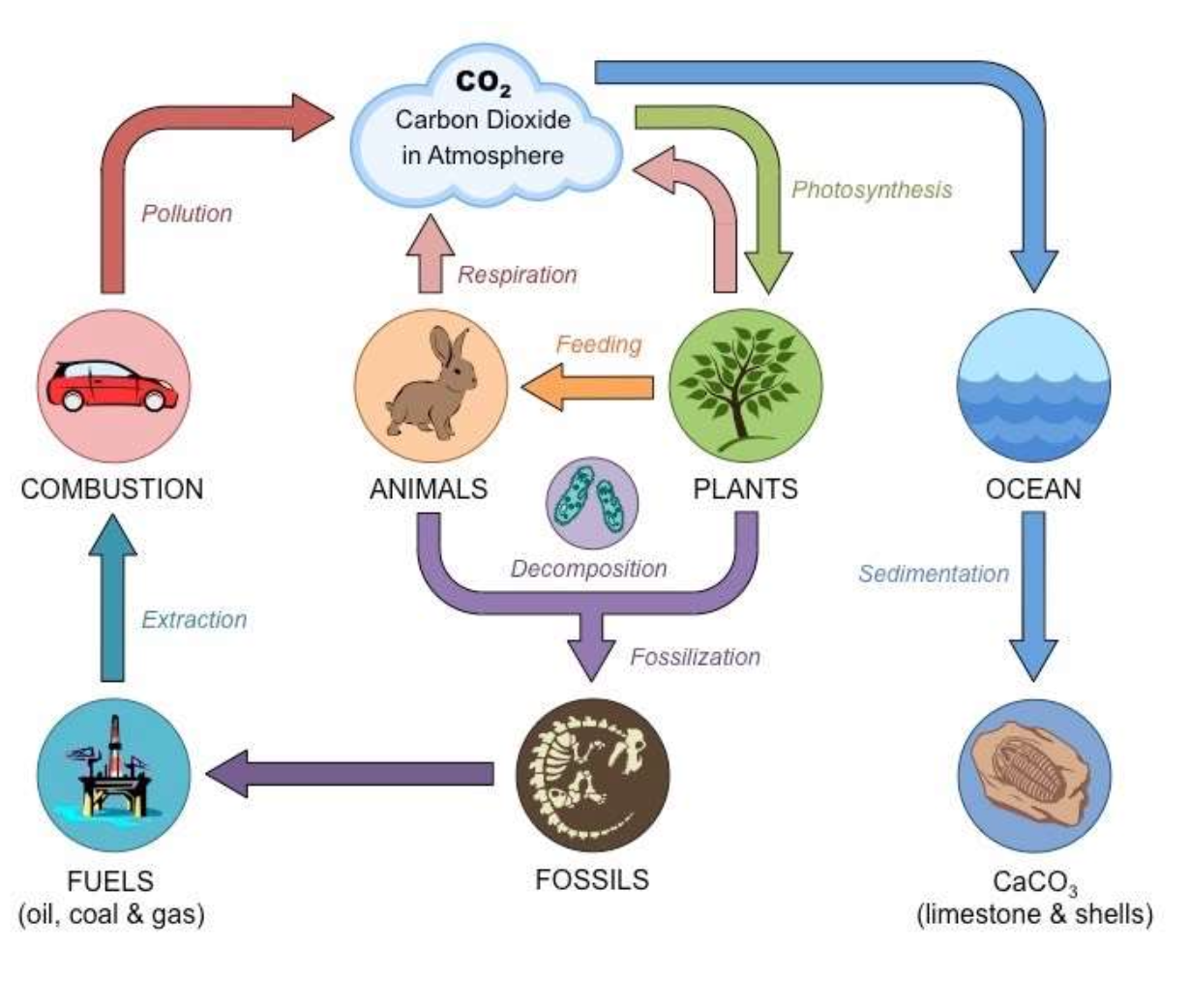
Carbon cycle
Carbons atoms are found in many different molecules such as CO2, methane CH4, carbonate rocks and shells, many biological molecules.
glucose and oxygen
Final end products are
carbon dioxide and water
Two chemical inputs