Aging and Disease 1
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
Biological aging and how it differs from disease
the gradual, progressive decline in function across body systems caused by cellular and molecular changes. It is a natural process, while disease is abnormal and results from specific pathological conditions.
the hallmarks of aging
• DNA damage – Accumulation of mutations and breaks.
• Gene expression errors – Altered control of which genes are active.
• Telomere shortening – Loss of protective chromosome ends.
• Protein dysfunction – Loss of protein shape/function over time.
• Impaired cell death – Damaged cells persist instead of being removed.
• Energy production decline – Mitochondria become less efficient.
• Inefficient cell communication – Signaling pathways weaken.
• Metabolic imbalance – Disruption in nutrient and energy processing.
• Stem cell exhaustion – Reduced capacity to repair and renew tissues.
cell turnover rates vary among tissues and how this changes with aging.
Different tissues replace cells at very different rates, and aging slows this turnover, reducing tissue maintenance and repair.
examples of tissues with high and low cell turnover
High turnover: gastrointestinal epithelium (3–5 days), blood cells
Low turnover: neurons and lens cells (last a lifetime)
the impact of aging on tissue repair and regeneration.
Slower cell replacement leads to reduced healing, weaker tissue function, and increased vulnerability to damage.
how DNA damage occurs and accumulates with age.
DNA damage occurs during replication (mismatched bases, insertions, deletions) and accumulates because repair systems become less efficient with age.
sources of DNA damage
Exogenous (external): chemicals, UV/IR radiation.
Endogenous (internal): reactive oxygen species (ROS), replication errors, spontaneous chemical reactions
how reduced DNA repair efficiency contributes to genomic instability.
Repair pathways (BER, NER, HR/NHEJ, MMR) become less effective, allowing mutations to accumulate and destabilize the genome.
gene expression and explain how aging alters epigenetic control
the process by which DNA instructions are used to make proteins.
Controlled by epigenetic regulation — chemical tags on DNA and histone proteins control which genes are turned on or off.
aging causes epigenetic errors, leading to incorrect gene activation or silencing.
Consequences:
• Loss of essential proteins.
• Production of harmful proteins.
• Example: Silencing of tumor suppressor genes can lead to cancer.
What are Epigenetic Alterations?
Changes in DNA methylation and histone modifications that alter gene expression without changing the DNA sequence.
With age:
• Global methylation decreases (DNA becomes overall less methylated).
• Local methylation increases at some regions (e.g., silencing important
genes).
• Histone modifications shift (acetylation and methylation patterns change). These changes affect oncogenes and tumor suppressor genes, altering cell behavior.
Consequences:
• Transcriptional noise: Genes are turned on/off incorrectly.
• RNA processing errors.
• Impaired DNA repair.
• Chromosomal instability.
• Increased risk of aging-related diseases, including cancer.
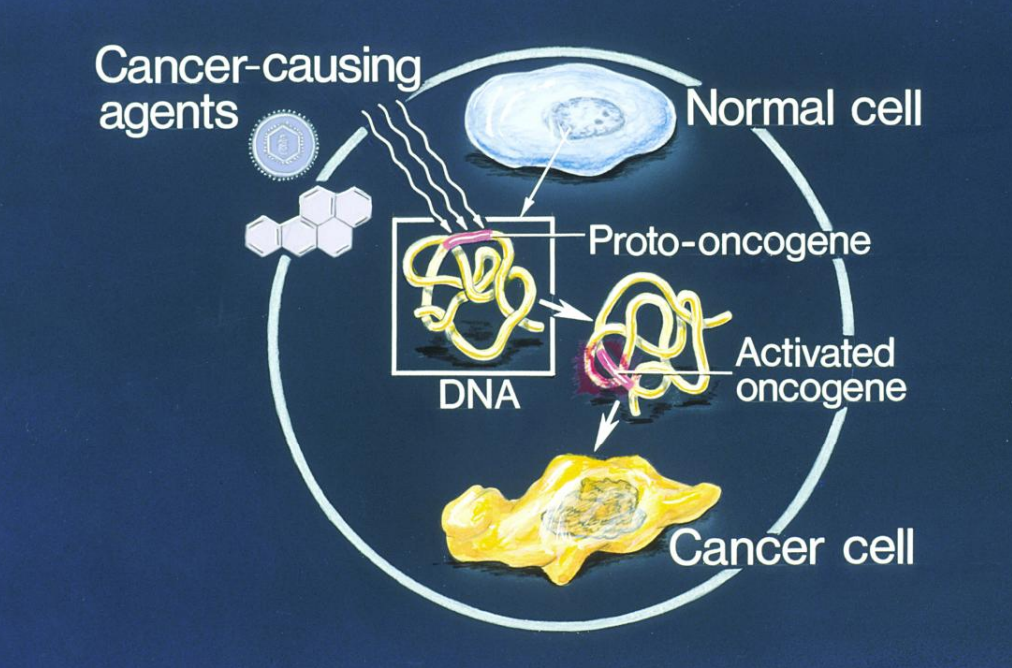
Gene Alterations: Proto-Oncogenes ("Gas Pedal")
• Normal role: Promote healthy cell growth and division.
• Cancer link: Become oncogenes when activated by:
• Mutation
• Insertion
• Gene fusion
• Gene amplification
• Result: Overactive growth signals → uncontrolled division.(excessive growth)
Gene Alterations: Tumor Suppressor Genes ("Brakes")
• Normal role: Prevent uncontrolled growth, repair DNA, or trigger cell death.
• Cancer link: Inactivated by:
• Mutation
• Deletion
• Promoter DNA methylation (epigenetic silencing)
• Result: Loss of growth control → unchecked division.
• Future therapy direction: Restore or mimic tumor suppressor function.
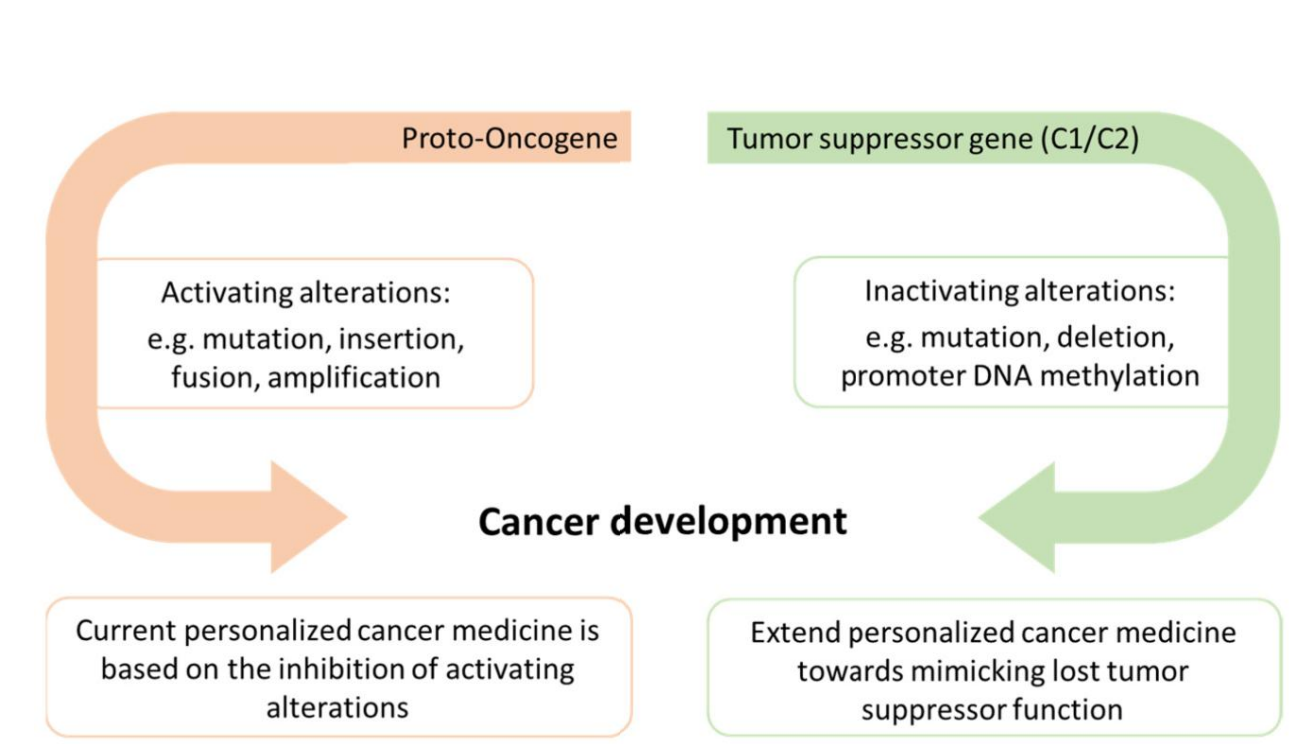
activation of proto-oncogenes and inactivation of tumor suppressor genes.
Activation of proto-oncogenes causes excessive growth, while inactivation of tumor suppressors removes growth control.
Telomeres
• Protective DNA-protein caps at the ends of chromosomes.
• Prevent chromosome ends from fraying or fusing with each other
→ Prevent DNA damage during cell division
What Happens with Age?
• Every time a cell divides, telomeres shorten.
• Eventually, they reach a critical length and can no longer protect chromosomes.
This can cause:
• Chromosome instability.
• Improper DNA replication.
• Fragmentation or unwanted additions to chromosomes.
• Rarely, damaged cells survive with mutations, which can lead to
cancer.
Consequences of Short Telomeres
• Cell death or senescence (aging).
• Pro-inflammatory state (cytokine release) that can harm nearby cells.
They shorten with each division until cells reach a limit and stop dividing.
Role of Telomerase
• Maintains cell health and longevity.
maintains telomere length, but its activity declines with age.
Telomerase: Effect of Chronic Stress
• Increases cortisol, which decreases telomerase activity.
• Leads to faster telomere shortening.
protein synthesis, folding, and degradation maintain cellular function.
Cells constantly produce, correctly fold, and degrade proteins to maintain proper function.
how aging affects protein turnover and leads to toxic protein accumulation
Normal Protein Turnover
• Proteins have a limited lifespan and must be recycled regularly.
• Cellular systems remove old or damaged proteins to maintain efficiency.
Effect of Aging
• The body’s ability to eliminate old proteins declines.
• Ineffective proteins accumulate over time.
Toxic Protein Build-Up
• Unremoved proteins can aggregate and disrupt cell function.
• In Alzheimer’s disease, beta-amyloid proteins form plaques in the brain, killing nerve cell
key pathological proteins in Alzheimer’s disease
• Amyloid-β (Aβ) plaques: Extracellular protein aggregates that trigger downstream damage.
• Tau aggregates / Neurofibrillary tangles (NFTs): Abnormal tau proteins disrupt microtubule stability and axonal transport.
APP (Amyloid Precursor Protein)
Imbalance in production/clearance contributes to plaque formation
Free radicals
Definition
Any molecule capable of independent existence that contains an unpaired electron in an atomic orbital.
• Highly unstable and reactive.
• Can donate or accept electrons → act as oxidants or reductants.
Main Effects on Cells
• Attack macromolecules → lipids, DNA, proteins.
• Leads to cell damage and loss of homeostasis.
• Contribute to oxidative stress and disease progression.
oxidative stress occurs when they accumulate.
Consequences
• Lipid peroxidation → membrane damage.
• DNA damage/mutations → possible carcinogenesis.
• Protein modification → loss of function.
senescence
Cells may stop dividing
apoptosis
Cells may die
Tau aggregates/ neurofibrillary tangles (NFTs)
Abnormal tau proteins disrupt microtubule stability and axonal transport.
• Synaptic dysfunction: Loss of communication between neurons.
• Neuronal loss: Particularly in memory and thinking centers like the hippocampus and cortex.
Vitamin E
protects cell membranes by neutralizing free radicals
• Fat-soluble vitamin.
• Main antioxidant protecting polyunsaturated fatty acids in cell membranes from peroxidation.
• Works by donating an electron to free radicals, stabilizing them and breaking the chain reaction of lipid damage.
Mechanism in Membrane
1. Free radical attacks unsaturated fatty acid in phospholipid bilayer.
2. Vitamin E donates an electron to neutralize the radical.
3. Prevents further lipid chain oxidation
Apoptosis and with age
programmed cell death that removes damaged or unnecessary cells.
Damaged cells accumulate, increasing the risk of dysfunction and disease.
cellular senescence
Senescent cells stop dividing but remain active; “zombie cells” release harmful signals
how senescent cells contribute to chronic inflammation and disease progression
• Release inflammatory molecules that damage nearby healthy cells.
• Spread inflammation and accelerate tissue degeneration.
• Accumulate with age, contributing to chronic inflammation and diseases such as cancer and arthritis.
• Removing senescent cells in lab animals has been shown to improve tissue function and slow aging effects
the role of mitochondria in ATP production and how this declines with age
the “powerhouses” of the cell, generating ATP through cellular respiration.
• As cells age, mitochondria become less efficient and more prone to errors in energy production
Consequences of Mitochondrial Dysfunction
↓ ATP production → energy shortage in high-demand cells (e.g., neurons, muscles)
how mitochondrial damage leads to increased reactive oxygen species (ROS)
Damaged mitochondria leak reactive oxygen species (ROS) — unstable oxygen molecules that can damage DNA, proteins, and lipids
Consequences of Mitochondrial Dysfunction
↑ ROS → oxidative stress and cellular damage.
the relationship between NAD⁺, SIRT1, and HIF-1α in mitochondrial regulation.
NAD⁺ activates SIRT1, which helps maintain mitochondrial function, while HIF-1α regulates metabolic responses under stress.
• NAD⁺ (nicotinamide adenine dinucleotide) levels decline with age.
• SIRT1, a NAD⁺-dependent enzyme, normally keeps HIF-1α (Hypoxia-Inducible Factor-1α) under control.
• When NAD⁺ decreases, SIRT1 activity drops → HIF-1α becomes overactive, disrupting communication between the nuclear and mitochondrial genomes.
• This creates a pseudo-hypoxic state, even when oxygen is available, reducing oxidative phosphorylation (OXPHOS) efficiency.
how loss of mitochondrial–nuclear coordination contributes to aging and disease.
Poor communication between mitochondria and the nucleus disrupts energy production and cellular regulation.
disorders associated with mitochondrial dysfunction
Neurodegenerative diseases such as Alzheimer’s.
Describe the role of DNA methylation and histone modification in gene regulation.
DNA methylation and histone modifications control gene accessibility; aging causes global methylation decrease and local increases, disrupting gene regulation.