chem exam 2
1/112
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
113 Terms
boron forms..
3 bonds — contrary to octet rule
boron is found in
boric acid — ant killer
borax — detergent
nitrogen & its oxidation states
(-3 to +5) — very reduced to very oxidized
ex.
-3 NH₃ (ammonia) gained electrons
0 N₂ (gas in air) neutral
+1 N₂O (laughing gas) slightly oxidized
+2 to +4 NO, NO₂ (smog) reactive gases
+5 NO₃⁻ (nitrate) very oxidized
haber process
process where ammonia is made from H2 and N2 at low temp with a catalyst
plants cant use N2 so it must be converted to NH3
nitrogen is found in…
explosives, fertilizers, laughign gas, viagra, smog, and preservatives
the 3 allotropes of carbon
C60: forms spherical or tube-like structures
graphite: sp2 hybridized
diamond: sp3 hybridized
carbonate (CO3²- )
an inorganic form of carbon that is an essential part of carbon cycle involving the greenhouse gas, CO2, a diprotic acid, and limestone, CaCO3.
carbon cycle
CO2(gas) → CO2 (aq, after dissolved in water))→H2CO3 (carbonic acid) →HCO3− (bicarbonate) →CO32− (carbonate) →CaCO3(solid, limestone)
phophorus is found in
soaps, toothpaste, fertilizer, and pesticides
phosphorus is extracted from…
rock
Ca3(PO4)2
phosphate is the backbone of…
nucleic acids like DNA
phosphate can cause ______ which causes from increasingly discouraged use in the envrionment
algae bloom
halides
group 17
small radii (atomic radius) = small atom = hold onton their electrons TIGHT
high ionization energy (energy needed to remove an electron)
high electronegtaivity
form -1 anions
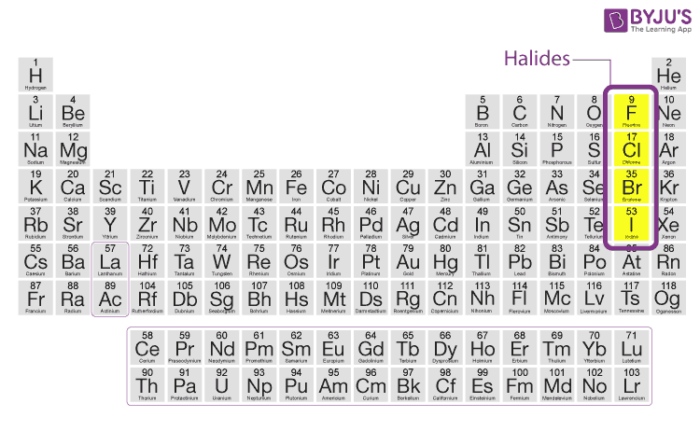
when halides bind with oxygen or hydrogen forming oxides and hydrides, the result is…
oxides and hydrides are acidic!!
ex. hydrochloric acid (HCl), hydroflouric acid (HF),
Flouride’s role in teeth
Fluoride (F–) inserts instead of OH– in tooth enamel to protect from decay.
normal enamel is made of the mineral hydroxyapatite which contain OH
Fl in toothpaste replaces the OH- ion in the mineral → flourapatite mineral → stronger enamel thats more acid resistant
Chloride’s role
manufactured as Cl2
a strong oxidizing agent (takes electrons from other substances)
used in disinfection and sanitation — takes electrons from harmful substances
used to make PVC (polyvinyl chloride) tubing
noble gases are
inert w 2 or 8 electrons — chemically inactive, stable and don’t rlly be in reactions
special uses of noble gases
Helium: in cryogens (extremely cold coolant)
Ar: inert gases to prevent reactions and cryogen
Neon (Ne): used in bright colored lights and signs
What is the most manufactured chemical in the world?
Sulfuric acid (H₂SO₄)
What is sulfuric acid mainly used for?
To solubilize phosphate in rocks for fertilizer production (wet method)
wet method
Ca₁₀(PO₄)₆F₂ + H₂SO₄ → H₃PO₄
used to make phosphoric acid for fertilizers
What does the Claus process produce?
Elemental sulfur (S) from H₂S (hydrogen sulfide — toxic gas)
two-step oxidation produces elemental sulfur from H2S, which is a contaminant in natural gas, methane
What is H₂S and where is it found?
A contaminant in natural gas (methane)
What type of reaction is the Claus process?
Oxidation (two-step oxidation)
contact process
produces Sulfuric acid (H₂SO₄) from sulfur (S)
A four-step oxidation process produces H2SO4 from
elemental sulfur, S.
What is the order of the three processes involving sulfuric acid production and use?
H₂S → S → H₂SO₄ → H₃PO₄
Claus → Contact → Wet (toxic gas → elemental sulfur → acid → fertilizer for use!!)
sulfuric acid is
a strong acid, a strong oxidizing agent, and a strong dehydration agent (removing water).
how much sulfuric acid is used to solubilize phosphate in rcoks by ___ method
50%; wet
solublizeed phosphate (through the ____ method) is used in __________
wet; fertilizers
oxidation vs. reduction
oxidation: loss of electrons
reduction: gain of electrons
OIL RIG
oxidation number
number that shows how many electrons an atom has lost or gained in a compound
if # increases in reaction: oxidation (loss)
if # decreases in reaction: reduction (gain)
oxidation number of a free element is…
0
ex. Mg = 0, Cl2 = 0, O2 = 0
oxidation number of a single ion is…
their charge
ex.: Mn2+ = +2
group 1 metals are +1
alkali metals (group 1)
+1
hydrogen’s oxidation number
+1
UNLESS H2 = 0, or NaH = -1 or H2O = +1
oxygen’s oxidation number
-2
EXCEPT in peroxides (H2O2), it is -1 and if O2 = 0,
periodic table trends of oxidation numbers
group 1: +1
group 2: +2
group 3: +3 or -5
group 4: +4 or -4
group 5: +5 or -3
group 6: -2
group 7: -1
group 17: -1
all oxidation #s must add up to __________ of the molecule or ioon
total charge
oxidizing agent
takes away electrons from others ; cause them to LOSE e
reducing agent
gives electrons to others; causes others to GAIN e
what does it mean when something is plated on another metal?
A solid layer of a metal forms on the surface of another material.
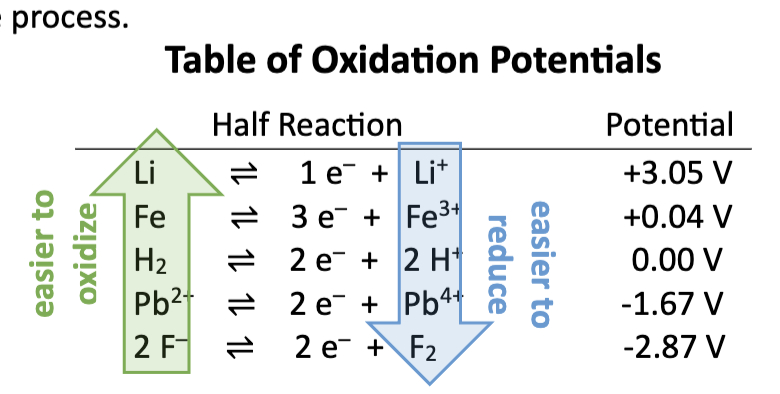
table of reduction potentials (top left)
strong oxidizing agents
As you move UP the table, the species on the left are EASIER TO
REDUCE (stronger oxidizing agents).
easier to reduce (gain e-) and more likely to TAKE others e-
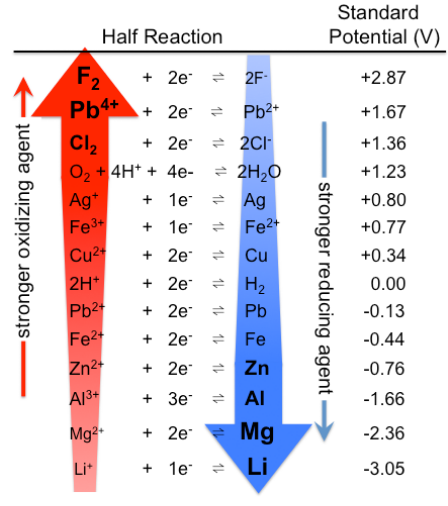
table of reduction potentials (bottom right)
As you move DOWN the table, the species on the right are EASIER TO
OXIDIZE (stronger reducing agents).
less likely to be reduced and more likely to GIVE e-
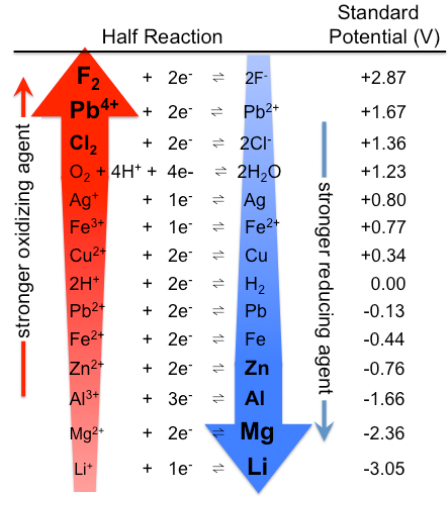
table of oxidation potentials
top left: easier to oxidize; strong reducing agents
bottom right: easier to reduce; strong oxidizing agents
standard cell potential (E°cell)
the maximum voltage of an electrochemical cell under standard conditions (1 M concentration, 1 atm pressure, 25°C)
how storngly a reaction WANTS to happen
if needing to balance an equation before balancing charges, what needs to be done
multiply charge by the number you use to balance
balancing redox reactions in acid
find the deficient O and put the same number of H2O on the deficient side and double the H+ on the oppositie side
balancing redox reactions in base
find the deficient O and put the same number of H2O on the opposide side and double the OH- on the same side
e- are attracted to which side
+ (cathode)
free energy in a battery (voltanic, galvanic)
ΔG = -
spontaneous
K in a battery (voltanic, galvanic)
equilibrium constant
> 1
products favored
** when at equilibrium, its dead battery
E° in a battery (voltanic, galvanic)
+
reduction occurs in
cathode
An Ox, Red Cat
oxidation occurs in
anode
An Ox, Red Cat
e- flow
anode to cathode
cathode in a battery (voltanic, galvanic)
+
electrons are attracted to this side
anode in a battery (voltanic, galvanic)
-
losing electrons; electrons not attrcate dhere
battery (voltanic, galvanic) cell
produces electricity
spontaneous
electrolytic cell
uses electricity; does NOT produce
non-spontaneous
ΔG in an electrolytic cell
ΔG = +
non-spontnaeous
K in an electrolytic cell
K < 1
dead battery
reactants favored
E° in an electrolytic cell
-
e- dont want to flow
anode in an electrolytic cell
+
cathode in an electrolytic cell
-
electrode (anode or cathode) is ALWAYS…
solid
shorthand cell notation
Zn | Zn2+ || Cu2+ | Cu
left side is anode
|| is salt bridge
right side is cathode
What are Pt and Au electrodes and why are they used?
They are inert electrodes — they do NOT participate in the reaction.
when the redox reaction involves only ions in solution (no solid metal naturally available to be the electrode).
Pt/Au just conducts the electrons in and out.
electrochemical cell diagram
the electrons always flow from anode to cathode
electrodes are always solid
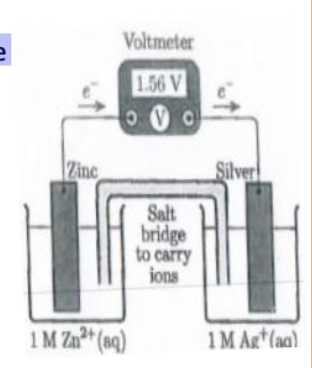
anions go through
oxidation
cations go through
reduction
group I
Li, Na, K
highly reactive
reacts to cold water
group II
Mg, Ca
moderately reactive
react in hot water
Transition Metals
Fe, V
slightly reactive
react in acid
coinage metals
Cu, Ag, Pt, Ag
don’t react, inert and stable
the least reactive metals are located where on the standard reduction potential chart?
in the top right corner (Au is the weakest reducing agent)
the most reactive metals are located where on the standard reduction potential chart?
lower right corner (Li is the strongest reducing agent)
General displacement reaction:
M1 + M2X → M2 + M1X
in which Metal 1, which is more active, replaces Metal 2, which is less active.
standard conditions in the standard reduction potential table
278K, 1 M, 1 atm, pH = 0
how to find oxidation potentials?
find it in the reduction potential table and flip the sign
standard hydrogen electrode (SHE)
every species is measure in voltage in relation to hydrogen
0.0V
found in the middle of the table
if +E° : species easier to reduce than H
if -E° : species is hardre to reduce than H+
coinage metals
Cu, Ag, Au
+ E°
DO NOT REACT
resists oxidation
upper portion of table
famous electrolysis products
anything w a -E°
means it was non spontaneous and went through electrolysis
rust
the oxidation of Fe
when Fe reacts with oxygen and water
how to prevent rusting
To prevent this, you use a metal that is a stronger reducing agent than iron
any metal below iron on the table (more negative E°) will sacrifice itself to protect iron from rusting.
ways rust is prevented
waxing and coating (prevent water and oxygen from reacting w Fe)
sacrifical electrodes w Mg and Al (easy to oxidize— take the place of Fe in oxidation)
stainless steel with Cr and V added to Fe
galvanization (adding a protective coating) with Zn
what 2 elements in stainless steel can be added to Fe to prevent rusting?
Cr, V
atom economy
the efficiency of a reaction by calculating the percentage of reactant atoms that end up in the desired final product
high atom economies = desirable
atom economy equation
MW of DESIRED product atoms utilized/MW of all reactants * 100%
be careful to not include byproducts
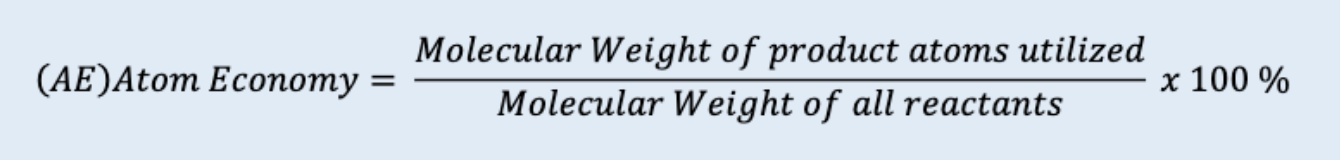
primary batteries
batteries are disposed of after one cycle
example of primary batteries
alkaline battery (duracell)
Zn-C
Zn-air, Zn-Hg (in hearing aids
secondary batteries
batteries that can be reused and recharged; built with consideration to minimize liquid and gas production; contents of reaction are better contained in the battery
examples of secondary batteries
lithium ion in electronics
lead-acid batteries in cars (gas-powered) — recharged by alternator
NiMH batteries: common household rechargebale battery
NiCd: household battery
modern batteries are made with ________ and _____ rather than ____ and _____ to avoid losses of material
solids and paste
liquids and gases
secondary batteries are
environmentally superior and convienient
inefficient batteries produce…
heat NOT work
controlling heat is important for practically and for safety
the problem with lithium ion batteries
they are known to catch fire
not allowed to be shipped as commercial cargo on planes
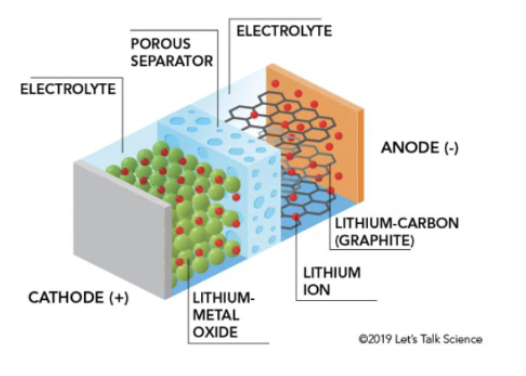
parts of a lithium ion battery
cathode +
lithium metal ocide
electrolyte
porous separator
lithium carbon (graphite)
lithium ion
anode =
lightweight batteries
higher charge density, less dense (ex. lithium)
F
faraday constant (105 C/mole)
key thing when converting E to G
multiply terms together and divide by 1000