Ozone story
1/15
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
16 Terms
define Electronegativity
the ability of an atom to attract bonding electrons in a covalent bond
explain the electronegativity trends
increases across periods and decreases down the group
F, O ,Cl, N, C, H
why are some bonds non-polar
if 2 atoms have the same electronegativity then the e- are shared equally so the bond isn't polar (C-H bonds are considered non-polar)
why are some bonds polar
if two atoms with different electronegativity bond the e- are shared unequally causing an uneven distribution of charge
why are some atoms partially negative
the e- will be closer to atom with higher electronegativity so this atom will be partially negative
why isn't CO2 polar
if the polar bonds are arranged so they point in opposite directions they cancel each other out
define intermolecular forces
forces of attraction between molecules (weak)
Explain instantaneous dipole-induced dipole forces (id-id)
the random movement of all electrons causes them to have a temporary uneven distribution of charge, this then induces a dipole in neighbouring molecules because the electrons are constantly moving the dipole is constantly created and destroyed
how does Mr affect the IDs-IDs
larger molecules have more electrons so are more polar and therefore have stronger IDs-IDs
(this means they have higher melting and boiling points)
how does the shape of the molecule affect IDs-IDS
the more linear the shape of a molecule the closer the electrons can pack together meaning more contact causing stronger IDs-IDs
(this means they have higher melting and boiling points)
how does length of the molecule affect IDs-IDs
the longer the chain the greater the molecule surface contact and more electrons so stronger IDs-IDs
Explain permanent dipole- permanent dipole forces (PDs-PDs)
the partially positive and negative charges on polar molecules cause weak electrostatic forces of attraction between molecules
*happen as well as ID-IDs
*second strongest IMFs
explain the charged rod experiment
if you put an electrostatically charged rod next to a jet of polar liquid it will move towards the rod because the polar liquid have PD-PDs
explain hydrogen bonding
N,F,O are so electronegative they draw bonding electrons away from the H atom because the bond is so polarised and H has a high charge density the atoms form weak bonds with the lone pairs
* very high melting and boiling points
how to draw hydrogen bonding
*partial charges
*lone pair
*correct atoms
*a 180 degree angle
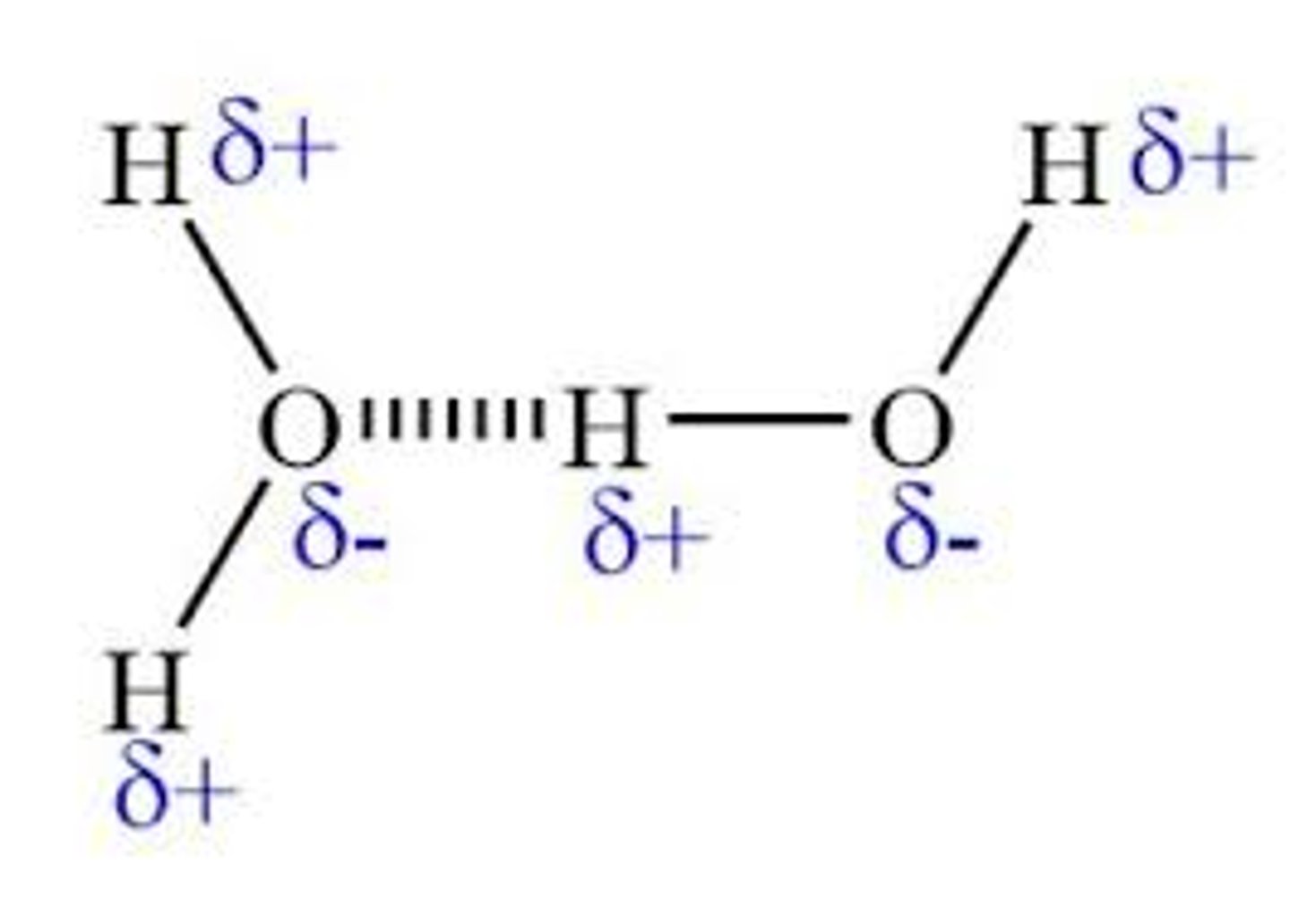
why is ice less dense than water
in ice the water molecules are arranged so there is the maximum amount of hydrogen bonds (lattice) when it melts some of the hydrogen bonds are broken allowing molecules to fill the space