Functional groups
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
Hydrocarbons
alkane, alkene, alkyne, phenyl/aryl
phenyl/aryl suffix
-yl benzene
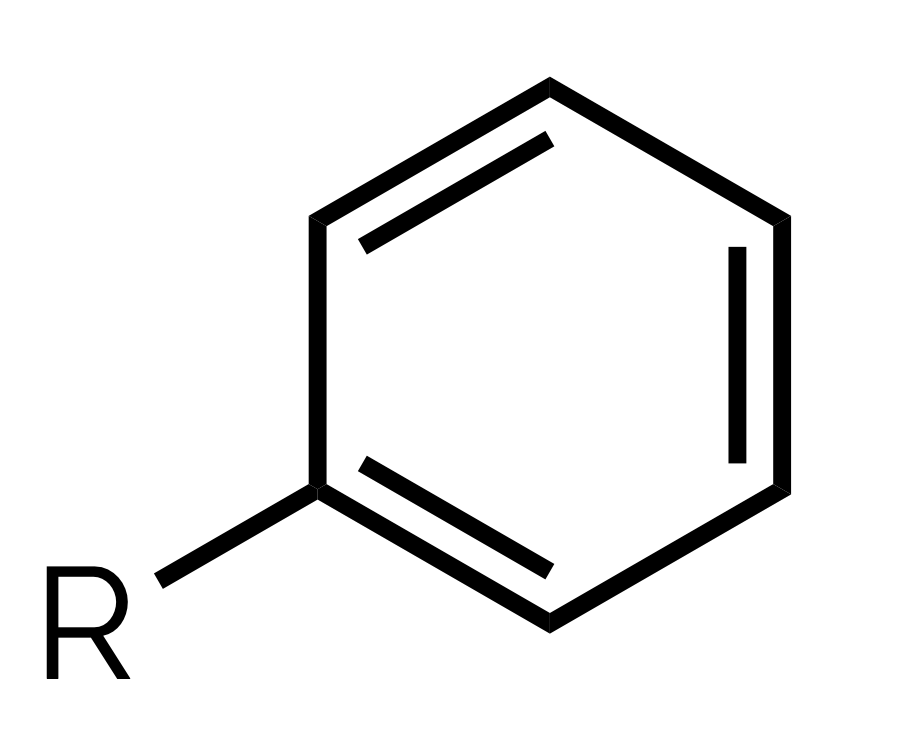
name and suffix
phenyl/aryl, -yl benzene
oxygen based functional groups
alcohol, ether, acetal
ether suffix
-oxy - ane
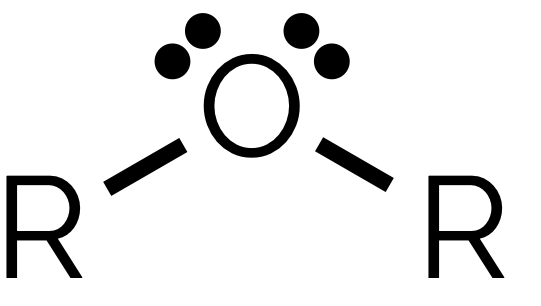
name and suffix
ether, -oxy - ane
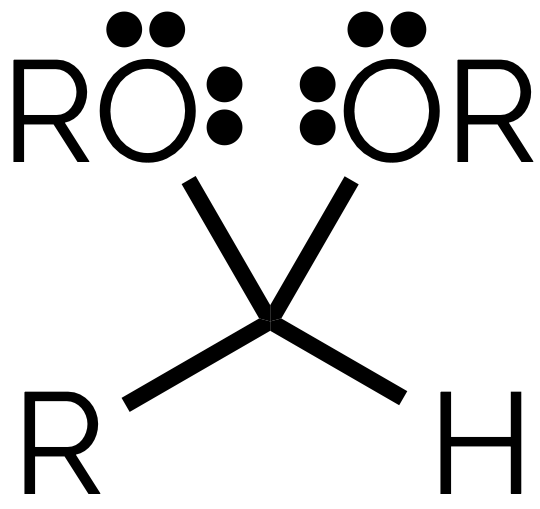
name
acetal
nitrogen based functional groups
amine, imine, nitrile, nitro
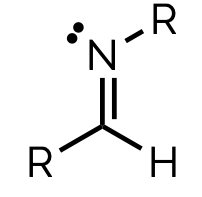
name and suffix
imine, n-alkyl-animine
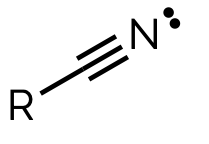
name and suffix
nitrile, -nitrile
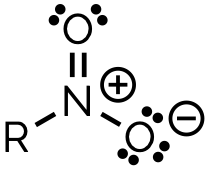
name and suffix
nitro, -nitro
carbonyl based functional groups
aldehyde, ketone, carboxylic acid, ester, acid anhydride, amide, acyl halide
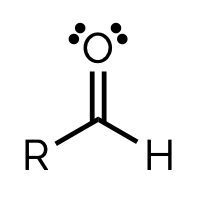
NAME AND SUFFIX
aldehyde, - al
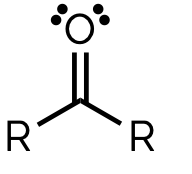
name and suffix
ketone, -one
name and suffix
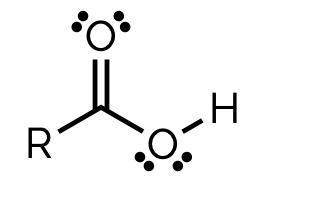
carboxylic acid, - anoic acid
name and suffix
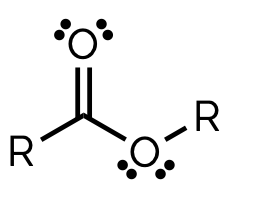
ester, -yl - oate
name and suffix
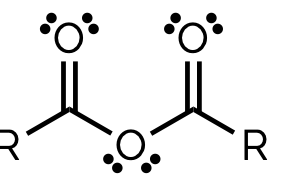
acid anhydride, -oic-anyhydride
name and suffix
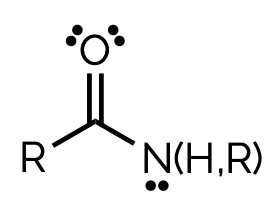
amide, -amide
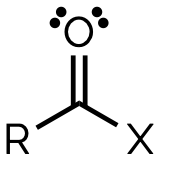
name and suffix
acyl halide, -oyl halide
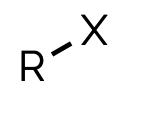
name and suffix
haloalkane alkyl halide, halo-
name and suffix
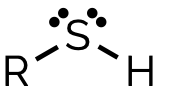
thiol, -thiol
name and suffix
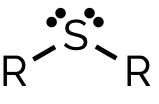
sulfide, -yl -yl sulfide ( -ylthio) -ane
Why is amide rotation restricted
partial double bond character
Why is carboxylate ion stable
charged delocalisation over two oxygen atoms
Which functional group contains C=O bonded to nitrogen
amide
Which functional group contains two carbonyls joined
acid anhydride
Which functional group replaces -OH with Cl
acyl chloride
Which functional group replaces -OH with -OR
ester
Which functional group contains -COO⁻
carboxylate ion
Which functional group has C=O in middle
ketone
Which functional group has C=O at end of chain
aldehyde
Most stable resonance structure
Has full octets minimal charges and charges on correct atoms
Effect of delocalisation
Increases stability and changes reactivity
Condition for delocalisation
Requires overlapping p orbitals
Delocalisation
Electrons spread over multiple atoms lowering energy and increasing stability
Resonance hybrid
Real structure is a combination of all resonance structures
Amide shape
Trigonal planar and flat due to sp² hybridisation
Carboxylic acid property
Acidic because it can donate H⁺
Carboxylate stability
Stable due to delocalisation of negative charge over multiple atoms
Why carboxylic acids are more acidic than alcohols
Conjugate base is stabilised by delocalisation
conjugate base
acid - proton
Ester vs carboxylic acid
Ester has C–O instead of O–H
Acyl chloride reactivity
Highly reactive due to electronegative chlorine and polar bonds
Acid anhydride reactivity
Reactive due to two carbonyl groups and electron withdrawal
Amide structure
nitrogen donates lone pair creating partial double bond with carbonyl
Amide bonding
C–N bond has partial double bond character
structural isomer
same atoms connected differently, DBE must stay the same
Diasteromer
cis/trans or change some but not all stereo centres
enantiomer
change one or all sterocentres or reflect