Recombinant and synthetic DNA techniques 2 (still needs lecture4)
1/23
Earn XP
Description and Tags
(*) means not gonna be assessed on (but extra reading ofc)
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
24 Terms
When you digest a plasmid with a single restriction enzyme, how do you make sure that the insert (gene of interest) gets ligated into it rather than just re-ligating to itself (recircularization)?
Use a high insert:vector ratio to make it more likely
Treat the vector with phosphatase to remove 5’ phosphate groups from the vector (but not the insert)
Use two different enzymes at the same time, so the ends are not compatible
what are the pros and cons of using heat shock for transformation of a plasmid? (is treated w calcium chloride b4)
-low efficiency (decreases as plasmid size increases)
-most commonly used method for routine work
what are the pros and cons of using electroporation for transformation of a plasmid?
-medium efficiency (less affected by plasmid size)
what are the pros and cons of using bacteriophage to transduct packaging into?
-high efficiency
-most commonly used during construction of gene libraries
-only suitable 4 cells growing in a culture
describe the process of selection?
-plasmids contain a selectable marker (usually resistance to an antibiotic) (e.g. pUC19 and amp^R gene)
-only cells that taken-up the plasmid can grow on media w the antibiotic (part of PLASMID not ligated insert)(has own promoter)
describe microinjection
DNA injected into cell via glass needle
-mainly used to produce transgenic animals
describe ballistics (gene gun)
microscopic (1 micrometer) particles of gold/silver/ tungsten coated w DNA & fired into cell
high pressure helium gas (100-600psi) used to fire
used for difficult targets (some types plant/ cells in living animals)
describe infection with recombinant viruses
-use vectors based around adenoviruses, retroviruses etc
-high efficient, useful in gene therapy or cultured cells
-saftey issues for containment
what are essential features of plasmid vectors
-an origin of replication
-one or more unique RE sites (multiple in tandem= multiple cloning site/ polylinker)
-a selectable marker (usually an antibiotic resistance)
-(ideally has a way of distinguishing recombinant from non-recombinant plasmids- screening)
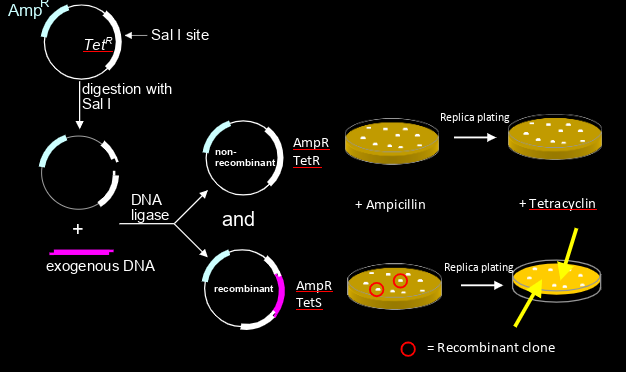
how can you use incisional inactivation of TetR (other resistance is amp)
-plate on ampicillin
-then replica plate to see resistance to tetracyclin
-colonies that don’t survive on tetracyclin are recombinants (cuz gene was inserted into tetracyclin resistance gene)
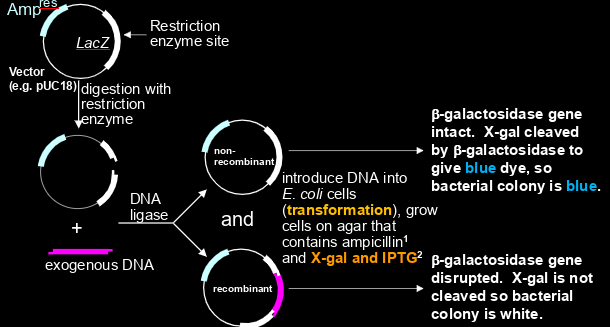
how can you use pUC18 (inactivation of lacZ) for screening?
-grow on agar w ampicillin
-select white colonies because b-galactosidase gene is disrupted, so X-gal is not cleaved = no blue dye produced
-depending on plasmid, lacZ has to maybe be induced (depending on if lacI repressor gene is expressed or no)
how can the ccdB gene be used for screening
-ccdB gene encodes DNA gyrase inhibiting toxin so is lethal to cell (unless has F plasmid that has anti-toxin)
-if ccdB disrupted by insert (in multiple cloning site) then cells will survive, only recombinants survive
what are other ways to screen w/o insertional inactivation?
-can extract and digest plasmid w REs -> run on gel to look for band right size
-do PCR w insert-specific primer - a small peice colony placed in small volume of water & heated to 95deg 5min
then some of the water used as template in PCR reaction (successful amplification= plasmid is recombinant)
how to do plasmid DNA extraction (basic, no detail)
-uses more rapid annealing of plasmid DNA compared to gDNA after denaturation
-cells harvested from culture
-cells lysed in alkaline solution
-renatured in acidic solution (gDNA larger, less contrained= doesn’t renature in time)
-gDNA & proteins extracted via phenol/chloroform
-pDNA precipitated (isopropanol) & washed (70% ethanol, removes impurities)
-pDNA dissolved in water w RNAse & check on gel
what is the pET system
uses combo of lac repressor & T7 RNA polymerase
GOI controlled by lace operator & T7 promoter
T7 RNA polymerase = under control of lac operator + lac promoter in chromosome of host cell
lacl gene encoded in chromosome & plasmid = keeps lace repressor lvls high
adding in IPTG allows expression T7 RNA polymerase (also clears lac operator)
T7 RNA polymerase binds T7 promoter & drives transcription of GOI
after using the pET system, how can you purify the recombinant protein?
-transgene (insert) expressed as fusion protein w 6xHIS tag (make sure remove stop codon b4 tag ad that the tag seq is ‘in-frame’)
-interact w nickle in chromatography column
-HIS-tagged protein eluted from the column
what are some useful features of bacterial expression vectors
-epitope tags 4 purification/ detection via western blot (e.g. His tags/ Hemagglutinin)
-solubility tags
-cleavage sites (can cut off tag from target protein- e.g. TEV protease)
-signal peptides for periplasmic localisation
what is a verry strong inducible promoter
TAC promoter (hybrid of the trp promoter & lace promoter) (inducible w IPTG too)
incorporates PRIBNOW BOX (for transcription) & Shine-Dalgano seq (ribosome binding site for translation)
using bacteriophages for replication- what is a phagamid? (*)
plasmids incorporating phage ssDNA origin of replication (insert DNA is incorporated into phage genome)
what is a cosmid (cloning vector) (*)
hybrid of plasmid and bacteriophage gamma cos-sites
what are cosmids used for (*)
larger inserts than plasmids (~100kb)
replicates & handled like a plasmid
can package DNA into phage particles
what are BACs and YACs (cloning vectors) (*)
bacterial artificial chromosome, yeast artificial chromosome
what are BACs used for? (*)
-for v large DNA fragments (~300kb)
-based on ‘F’ fertility conjugation plasmid
what are YACs used for? (*)
-very large DNA fragments (~1mb)
has centromeric and telomeric sequences & an origin of replication