9. // R3.2 Equilibrium
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
Dynamic Equilibrium
“steady state“, not stopped reaction
Macroscopic properties don’t change
Microscopically, forward and reverse processes occur simultaneously at the same rate
The amounts of reactants and products
remain the sameOnly in closed system!
The Equilibrium Law // The Equilibrium Constant
aA + bB ⇋ cC + dD
At a given temperature the ratio of the concentration of the products raised to the power of their molar coefficients to the concentration of the reactants raised to the power of their molar coefficients is a constant, Kc.
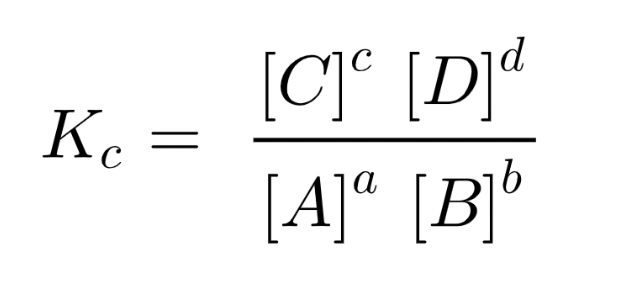
Common equilibrium reaction
Dissossication reactions: H2CO3+H2O ⇋ HCO3- + H3O
CO2 (g) ⇋ CO2 (aq)
CO2(aq)+ H2O (l) ⇋ H2CO3
Ionic product constant for water, Kw
10-14 =[H+][OH-]
Equilibrium Position
Position of the equilibrium
Kc < 1 reactants side / towards left
Kc = 1 equal amounts of reagents and products
Kc > 1 products side / towards right
Closed system
Neither matter nor energy can be lost or gained from the system.
Macroscopic properties remain constant.
If the system is open, some of the products from the reaction could escape and equilibrium would never be reached .
CEC - Reverse the reaction
Kc^-1
CEC - Halve Coefficiants
√Kc
CEC - Double the coefficients
Kc^2
Reaction Quotient, Q
If the reaction is NOT in equilibrium, the reaction quotient can be used to find out the direction of the reaction
Q < Kc The reaction will produce more products / forward reaction is favored
Q = Kc The reaction is in EQUILIBRIUM
Q > KcThe reaction will produce more reagents / reverse reaction is favored
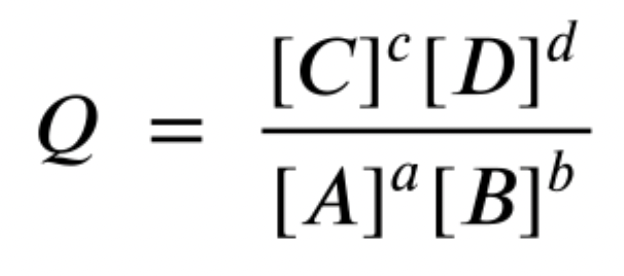
Le Châtelier's principle
If a change is made to a system that is in equilibrium, the balance between the forward and reverse reactions will shift to offset this change and return the system to equilibrium.
Factors affecting the position of equilibrium
Concentration of product or reactant
Pressure
Temperature
Adding a catalyst
Heterogeneous equilibria
Heterogeneous equilibria
Chemical systems at equilibrium involving substances in multiple distinct phases (e.g., solid-gas, solid-liquid). The equilibrium constant ( or ) expression excludes pure solids and liquids because their concentrations remain constant
Homologous equilibria
Reactions with very large or slow K
Practically irreversible.
K >> 1, = forward reaction is favoured so strongly that the extent of the reverse reaction becomes negligible.
K << 1 = only the reverse reaction will be observed while the forward reaction will not proceed to any noticeable extent.