Ochem Test 3 carboxylic reactions
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
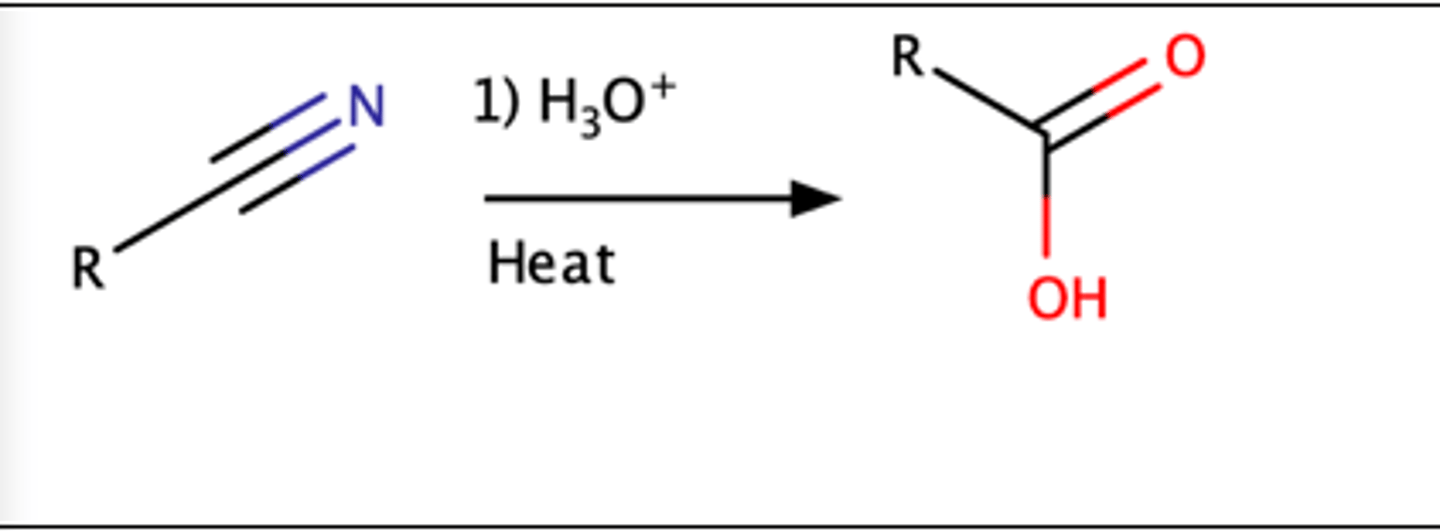
Nitrile (R-CN) + Acid (aq) =
o Carboxylic acid
o SN2 reaction
o Tertiary alkyl halides = no rxn
Mechanism pg: 904
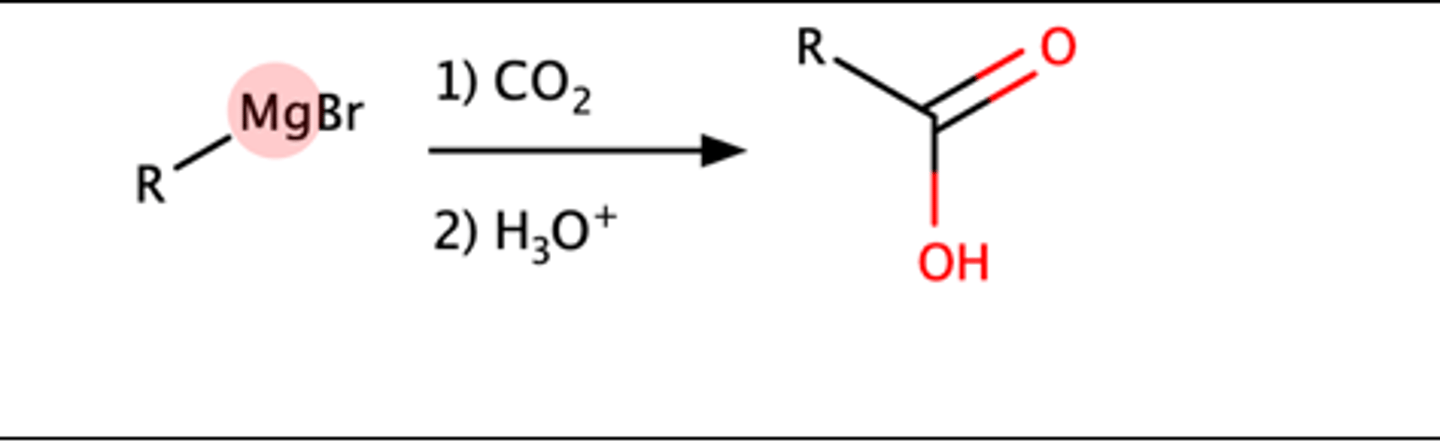
R-MgBr + 1)CO2, 2)H3O =
Carboxylic acid
Mechanism pg: 905
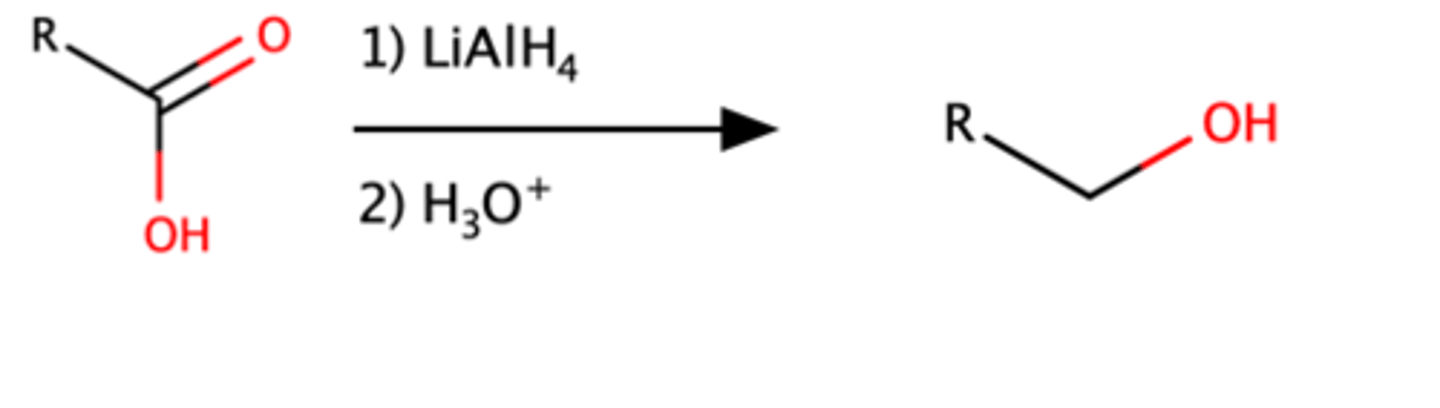
RCOOH + 1)LiAlH4, 2)H3O =
oAlcohol
Mechanism pg: 905-906
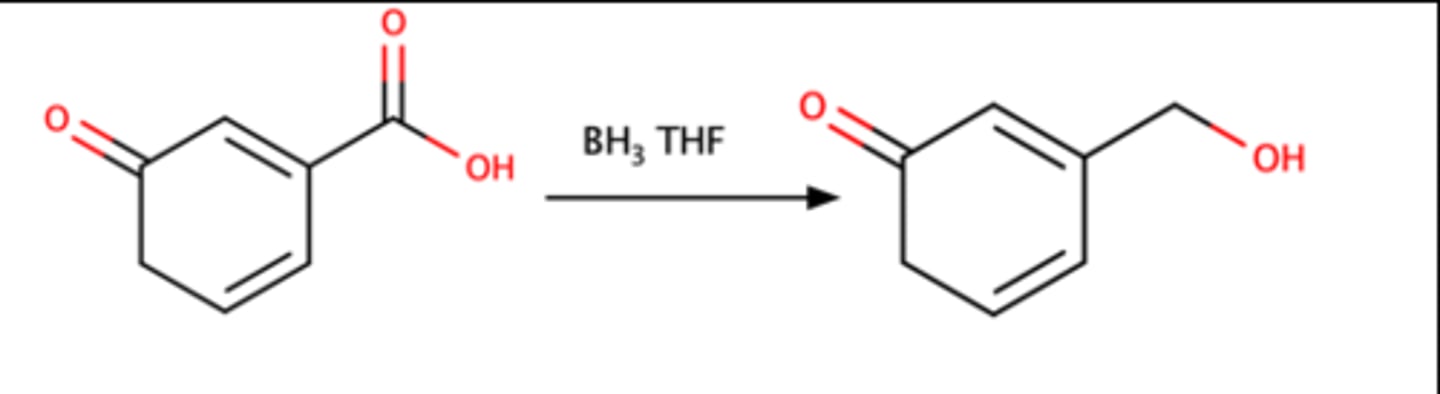
RCOOH + 1)BH3 THF =
Alcohol
Borane reacts selectively with a RCOOH when another C=O is present
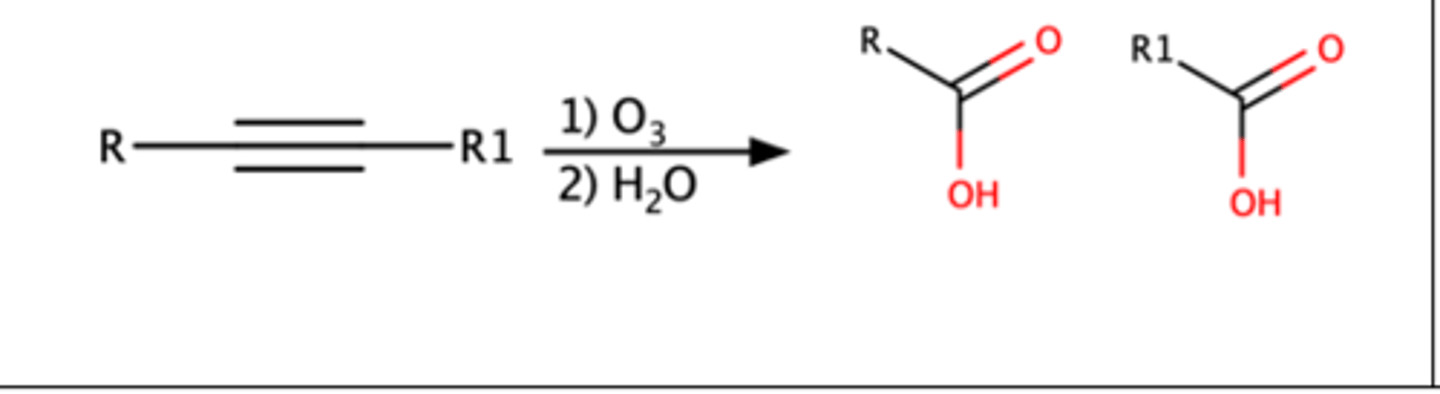
Alkyne + 1)O3 2)H2O
Triple bond will break forming two carboxylic acids
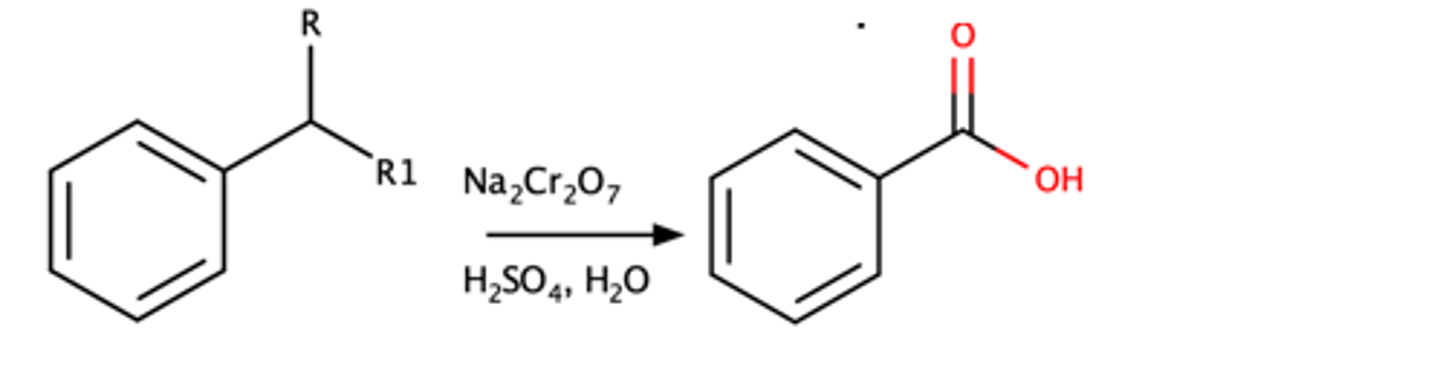
(Alkyl group connected to Aromatic ring) + Na2Cr2O7, H2SO4, H2O
Alkyl group on aromatic ring will be completely oxidized into a carboxylic acid
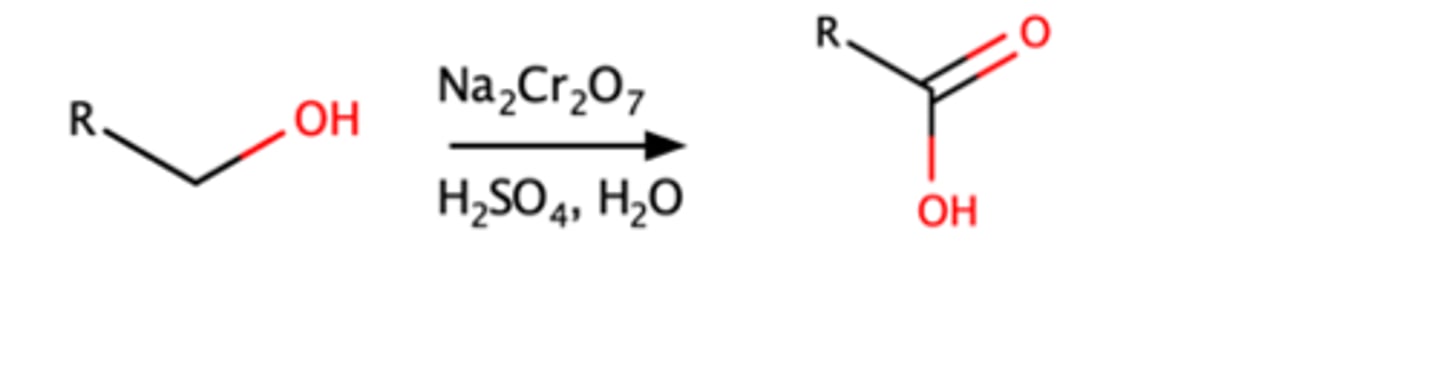
primary alchohol + o Na2Cr2O7, H2SO4, H2O
Strong oxidizing agents will oxidize primary alcohols into carboxylic acids
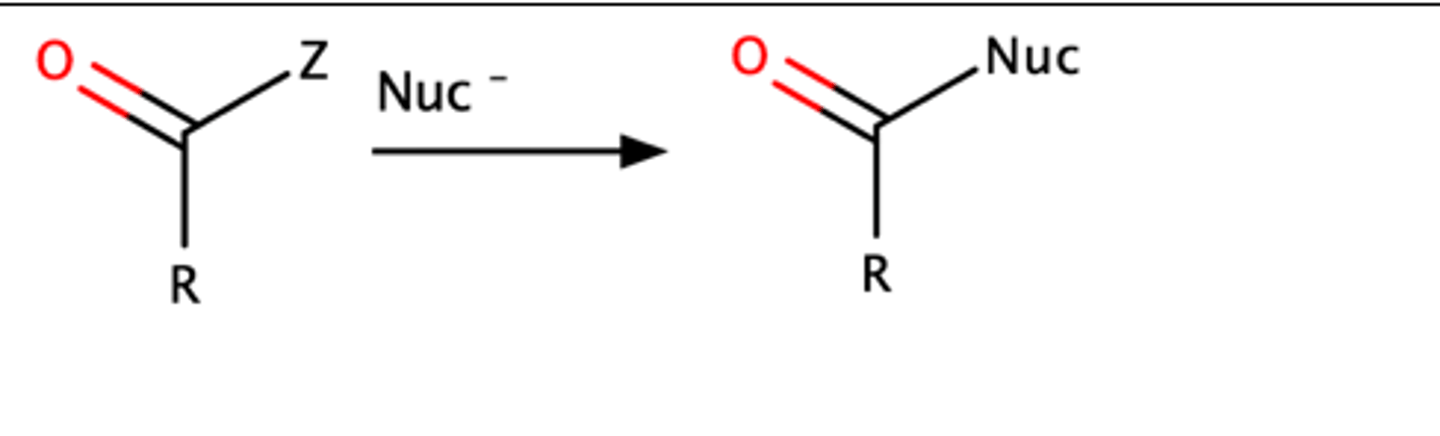
Nucleophile + carboxylic acid derivative: (Cl, O, OR, NH2)
Nucleophile attacks carboxylic acid derivative, loss of leaving group "Z"
"Z" must be a heteroatom (Cl, O, OR, NH2)
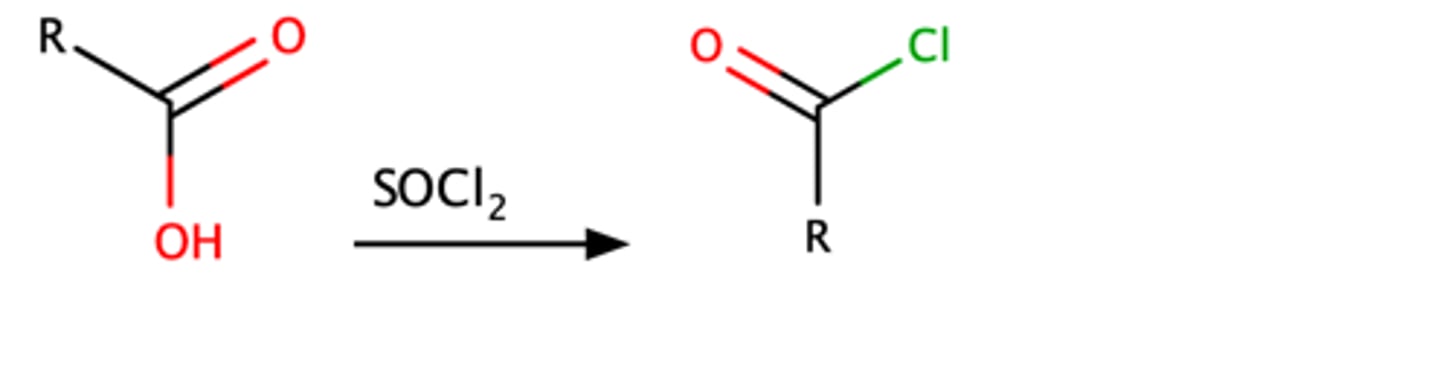
RCOOH + SOCl2
o RCOCl
o 2 step mechanism
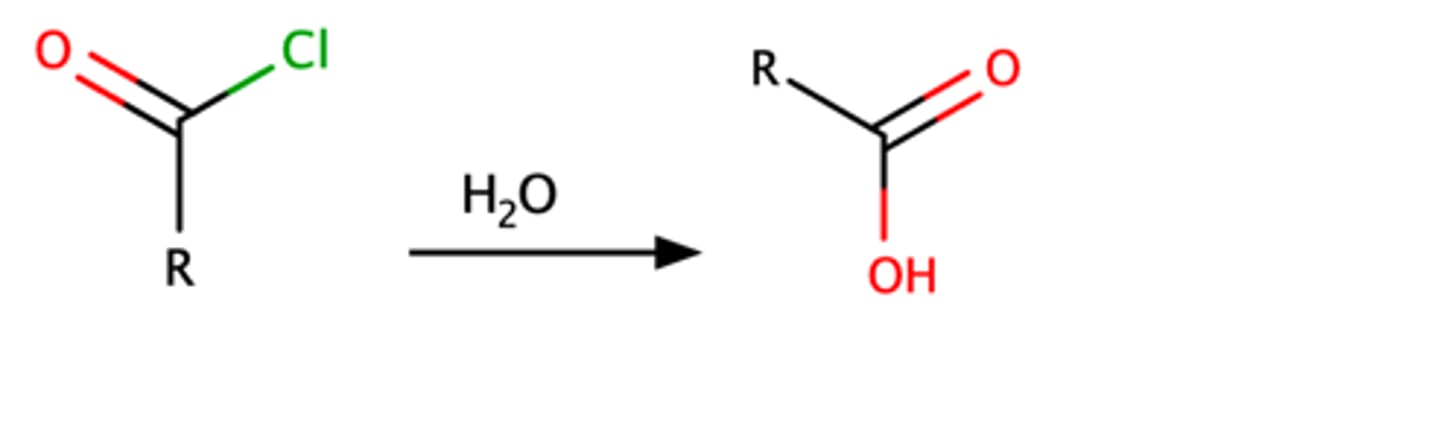
RCOCl + H2O
RCOOH
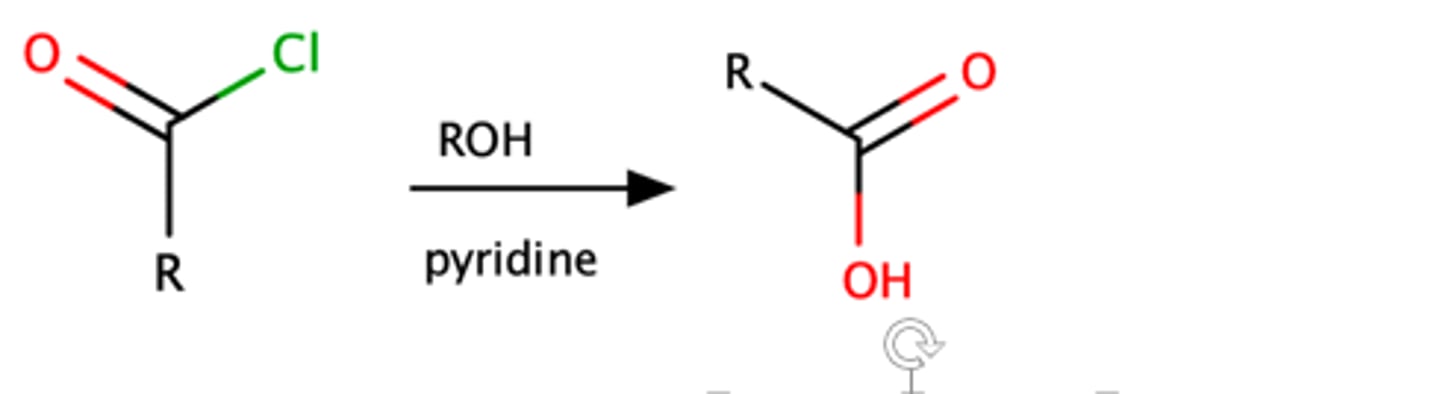
RCOCl + ROH, py
RCOOH
Steric effects
Mechanism pg: 918
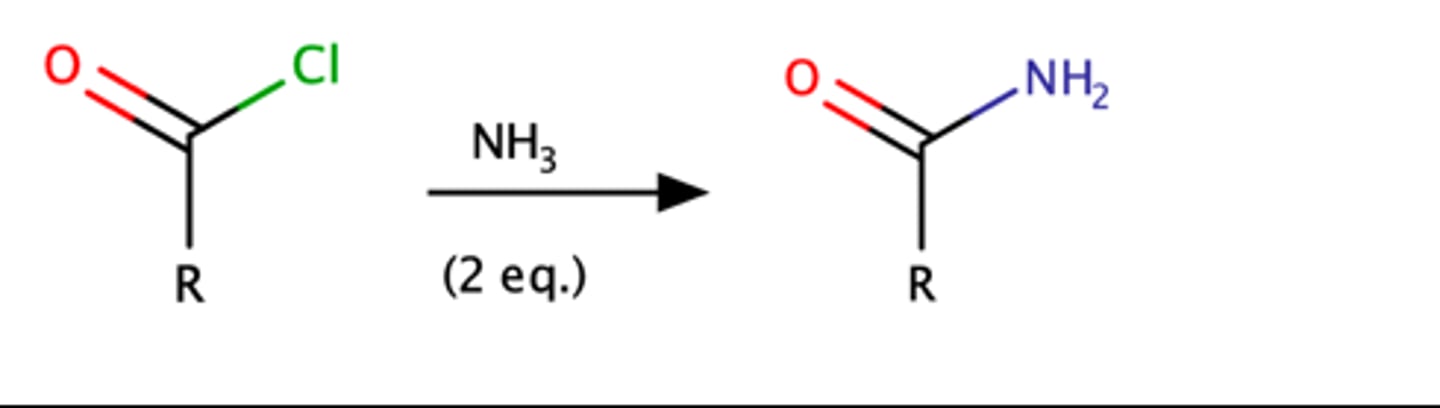
RCOCl + NH3 (2 eq.)
o Rxn. can also occur with RNH2 and R2NH to produce N-substituted amides
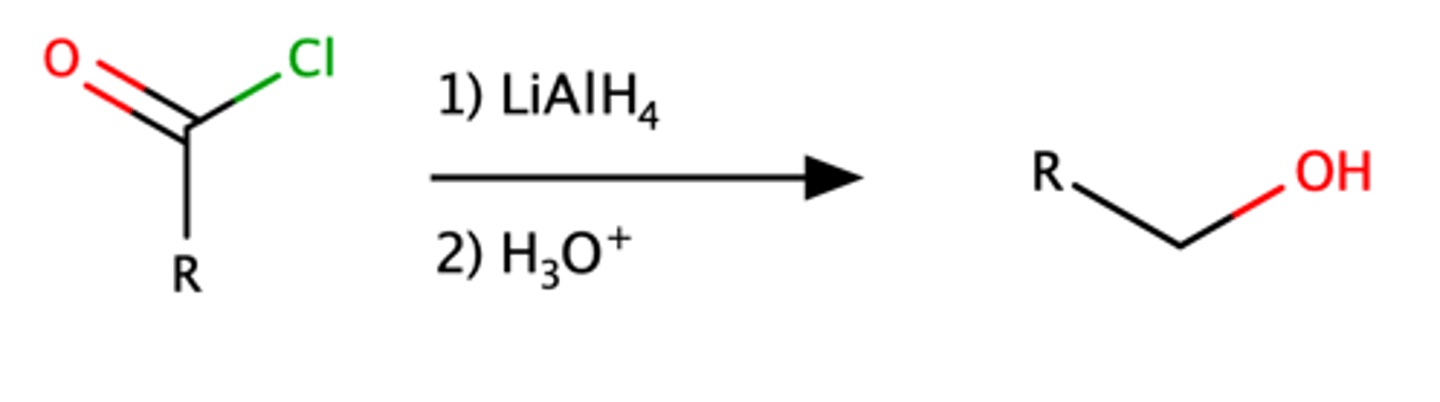
RCOCl + 1)LiAlH4 2)H3O
Alcohol (or aldehyde)
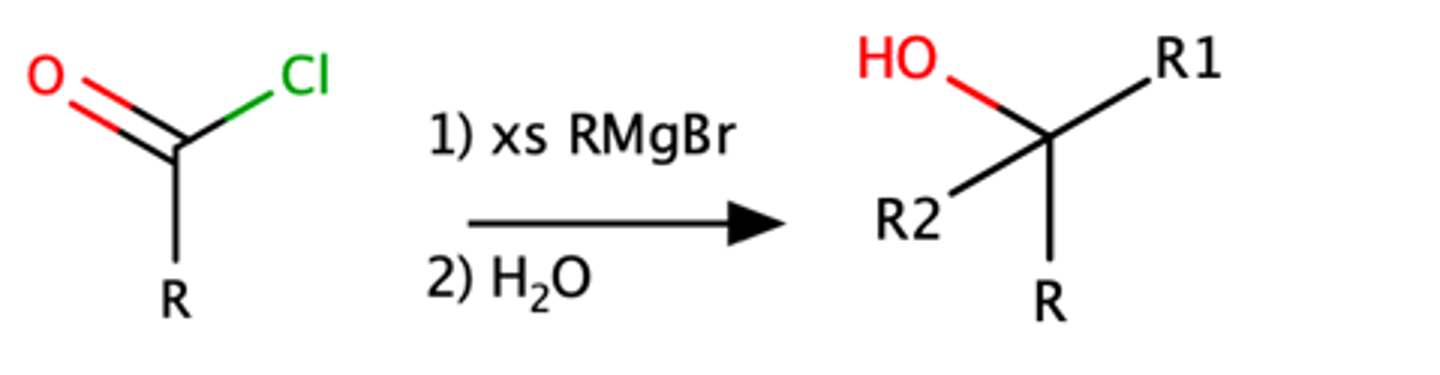
RCOCl + RMgBr
Alcohol with R substituents
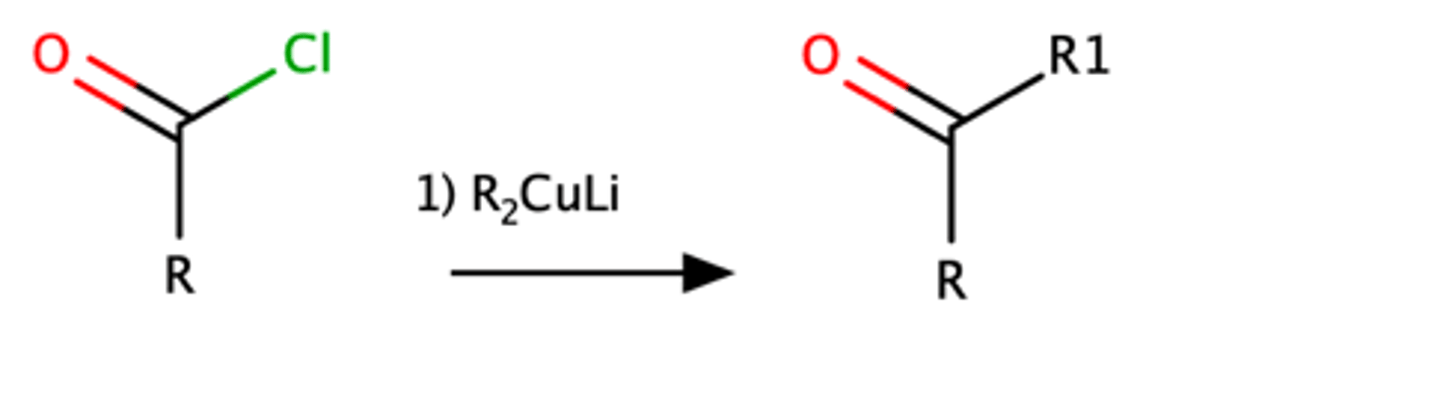
RCOCl + R2CuLi
Ketone with R substituents attached.
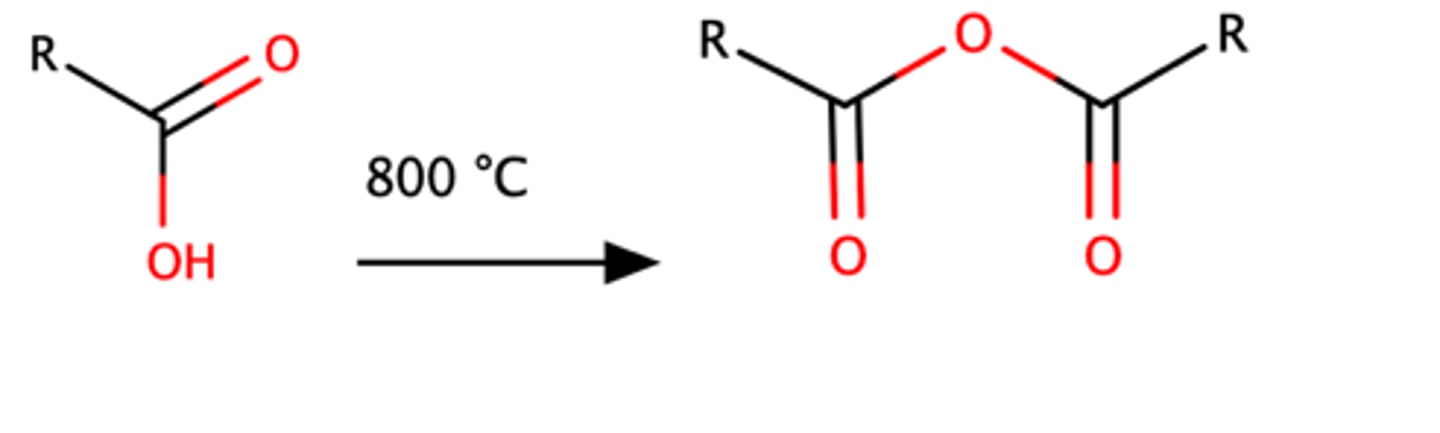
RCOOH + 800C (heat)
Carboxylic acid can be converted into acid anhydrides with excessive heating (800 °C)
This method can only work for acetic acid!!!
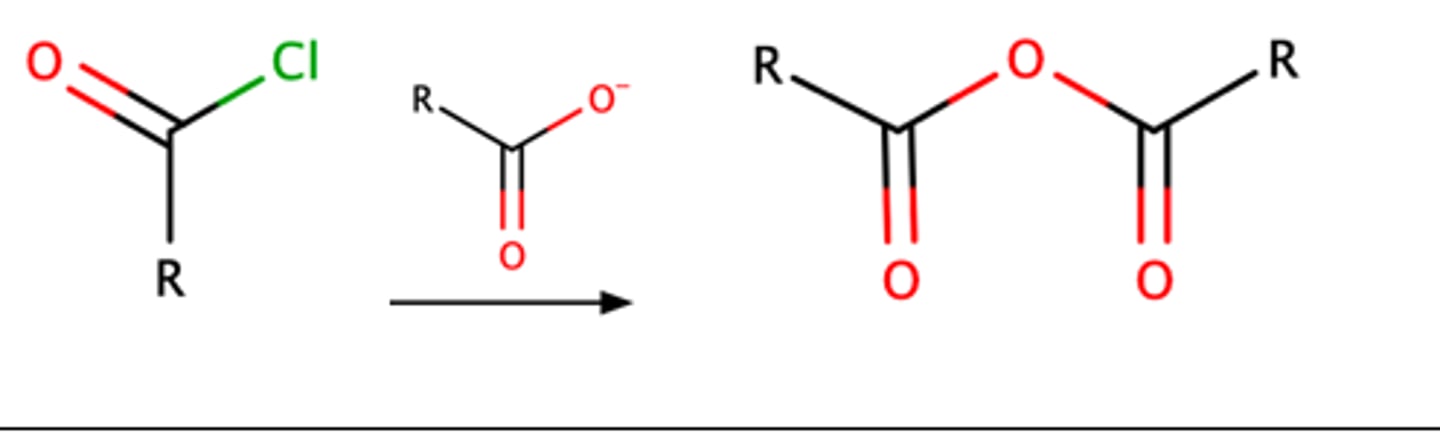
RCOCl + RCOO-
acetic anhydride
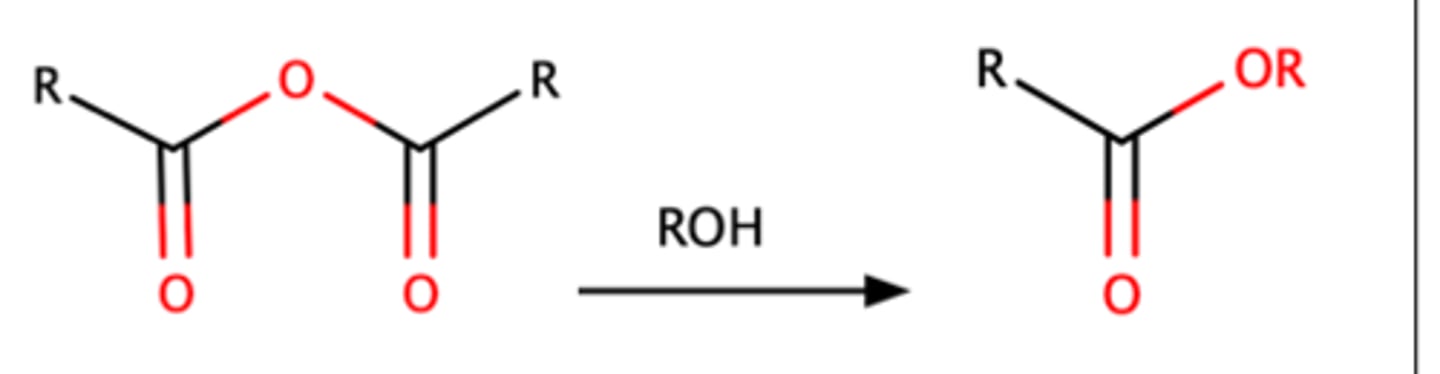
RCOOOCR + ROH
Acetylation of acetic anhydride with an alcohol can form an ester with the R group attached.
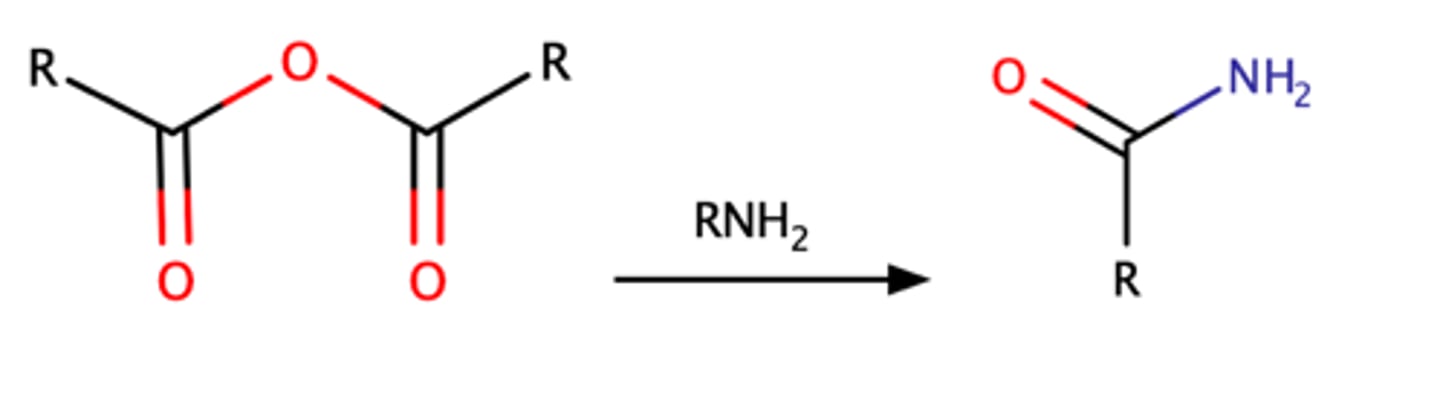
RCOOOCR + RNH2
Acetylation of acetic anhydride with an amine can form an amide
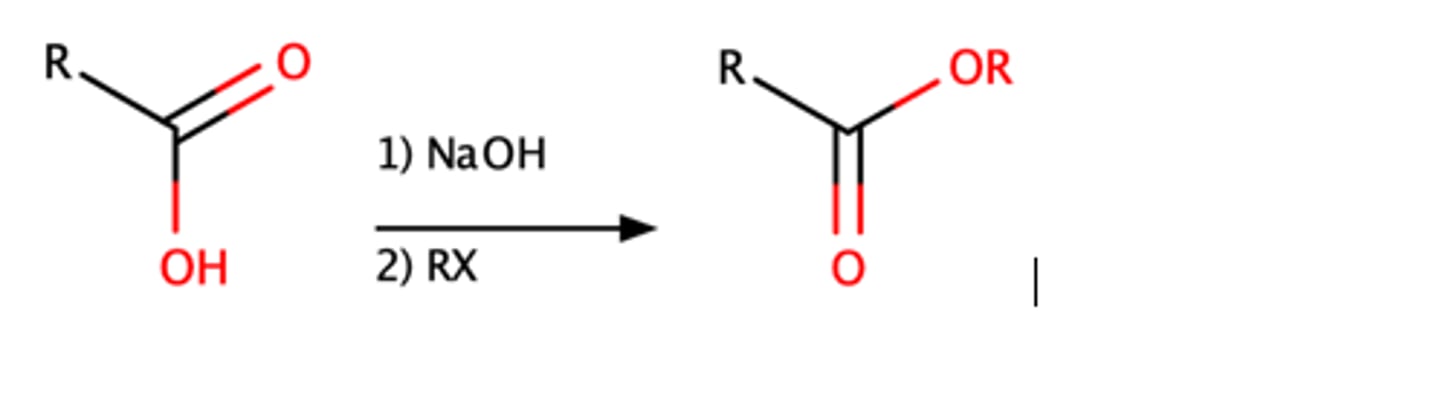
RCOOH + 1.) NaOH 2.) RX
o Strong base with alkyl halide will convert a carboxylic acid into an ester
o SN2 reaction
Tertiary alkyl halide = no rxn
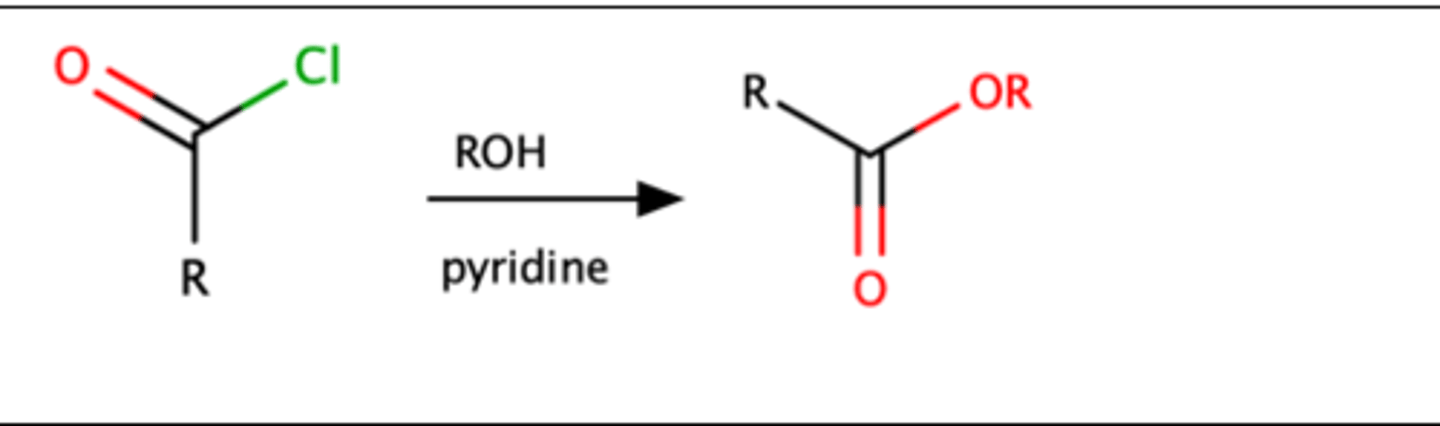
RCOCl + 1.) ROH 2.) pyridine
Acid chloride + ROH = Ester
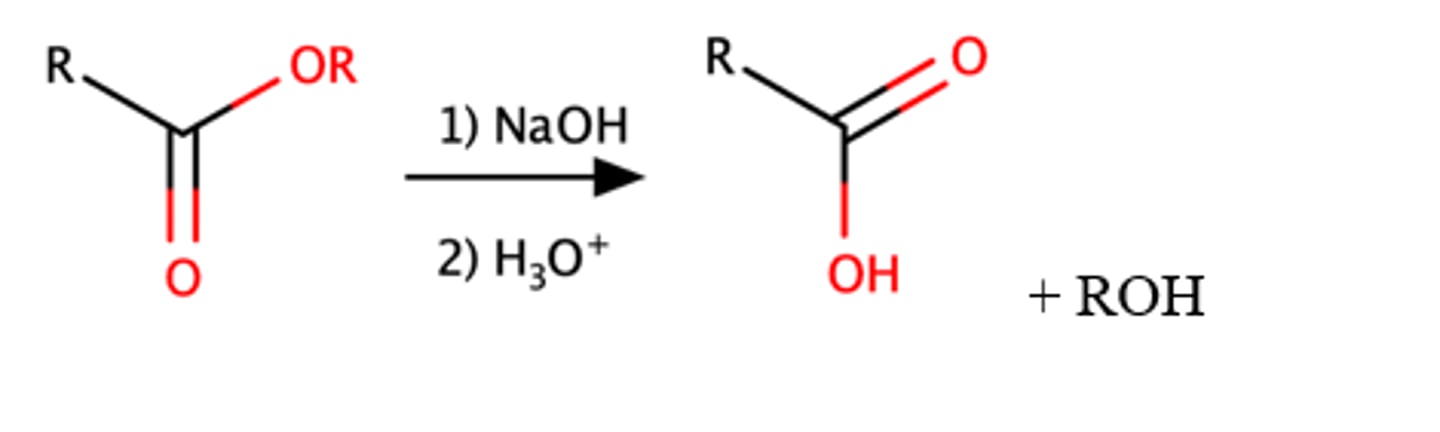
Ester + H3O
Carboxylic acid
This is the reverse of Fischer esterification
Mechanism pg: 928
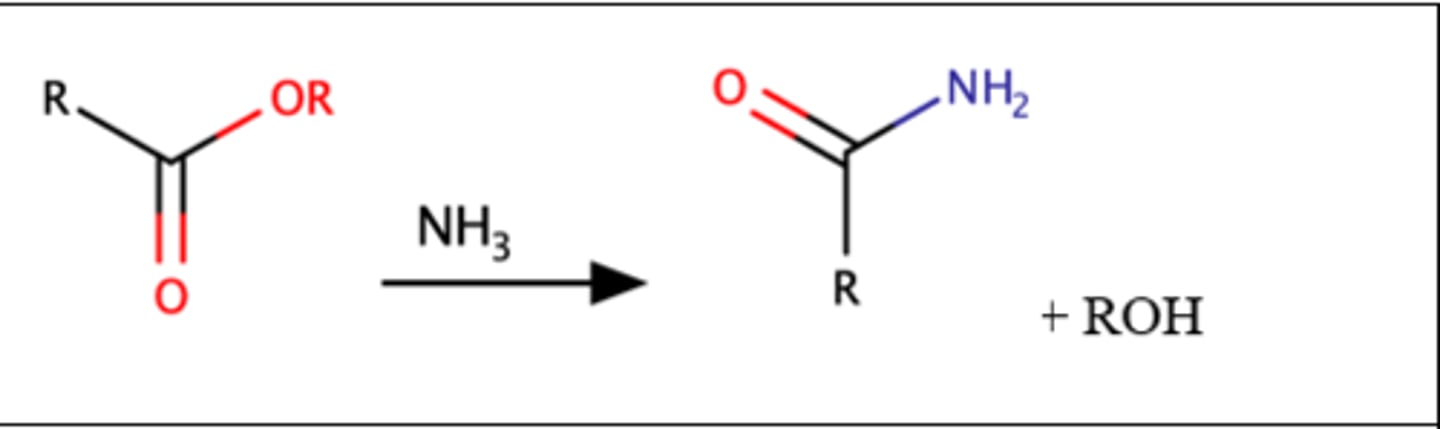
RCCOH + NH3
Ester + amines = Amides
Ester + LiAlH4
Alcohol
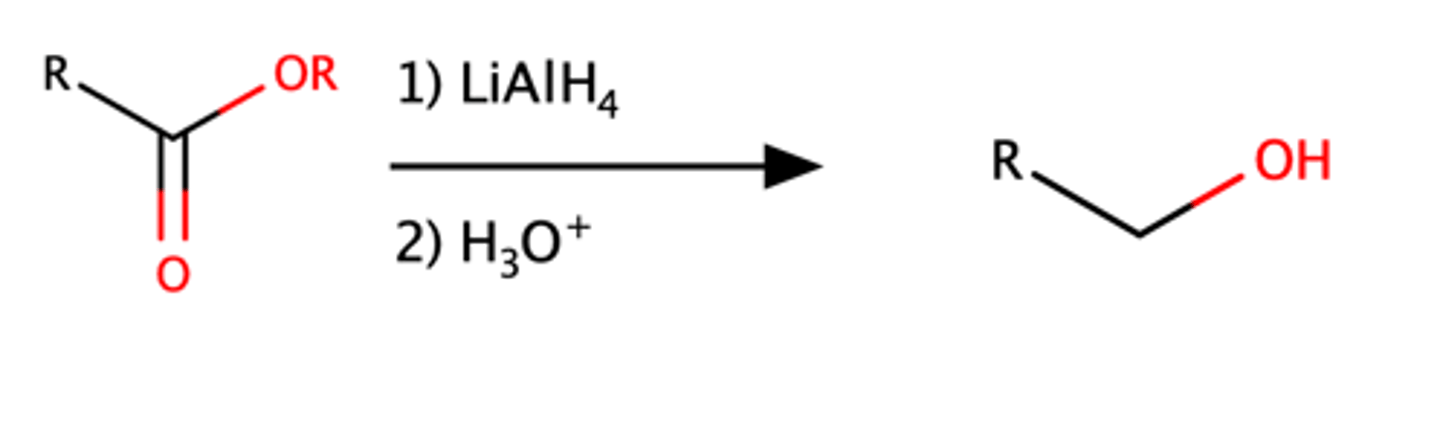
Ester + RMgBr
Alcohol with R groups attached.
Same mechanism as Acid Chloride with Grignard reagent
Mechanism pg: 931
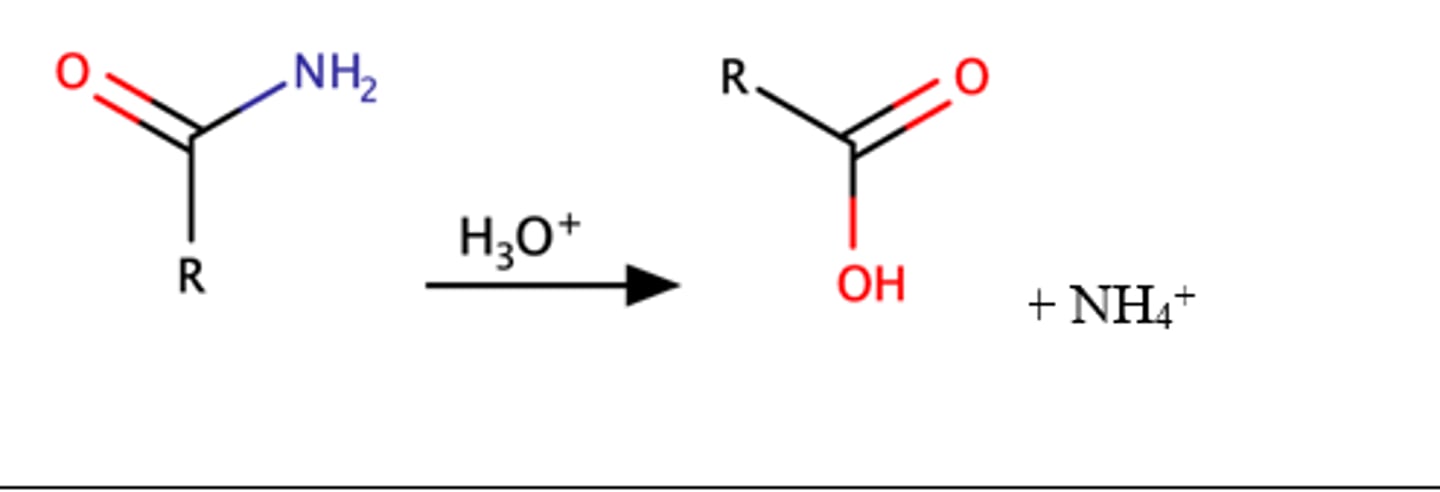
RCONH2 + H3O
Carboxylic acid
Reaction is slow and requires heat
Equilibrium favors formation of the products, reaction is irreversible
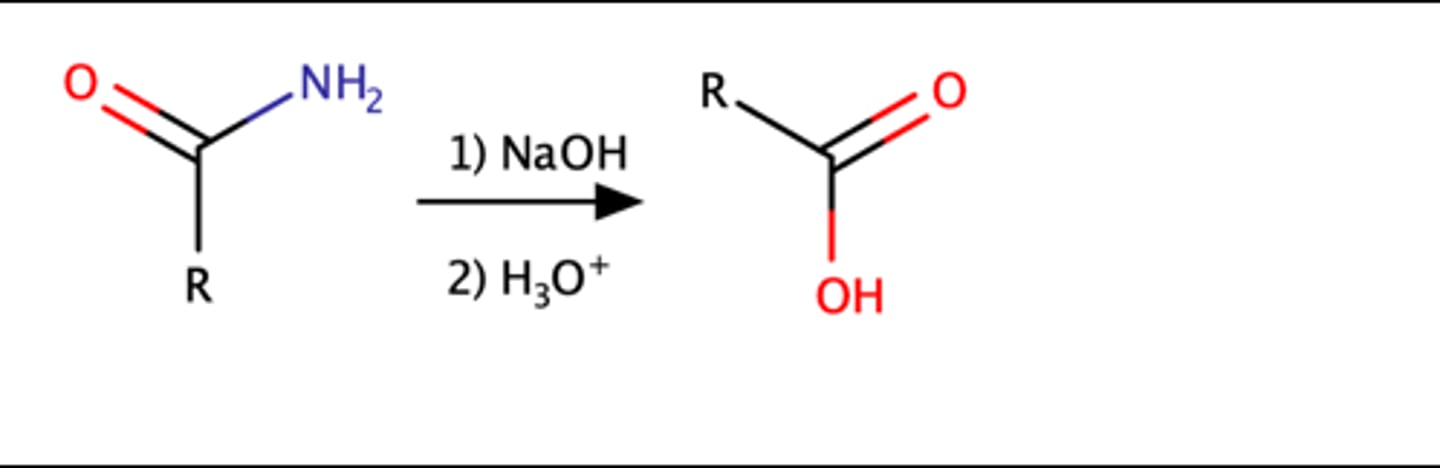
RCONH2 + 1.) NaOH 2.) H3O
Carboxylic acid
Amide + reducing agent
amine
Carbonyl group is completely removed
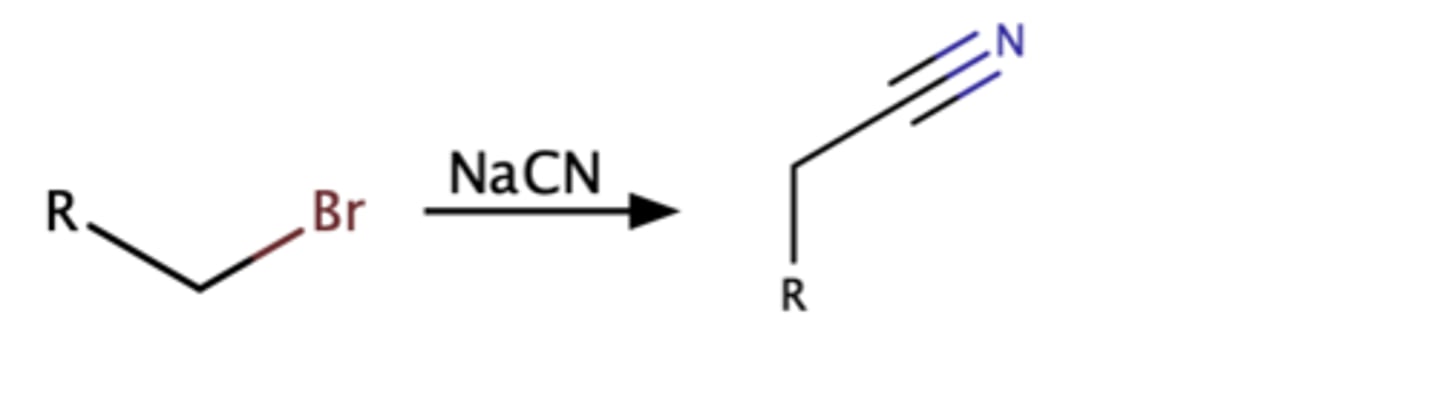
Alkyl halide + cyanide ion
Nitrile
SN2 reaction
Tertiary alkyl halides
no rxn.
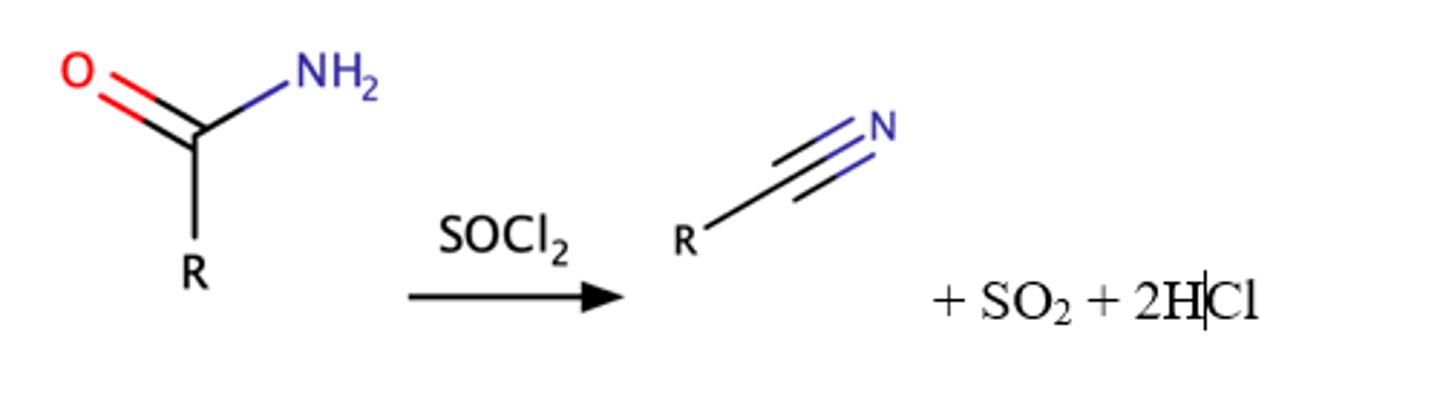
RCONH2 + SOCl2
Dehydration of Amides to produce a Nitrile
Mechanism pg: 936
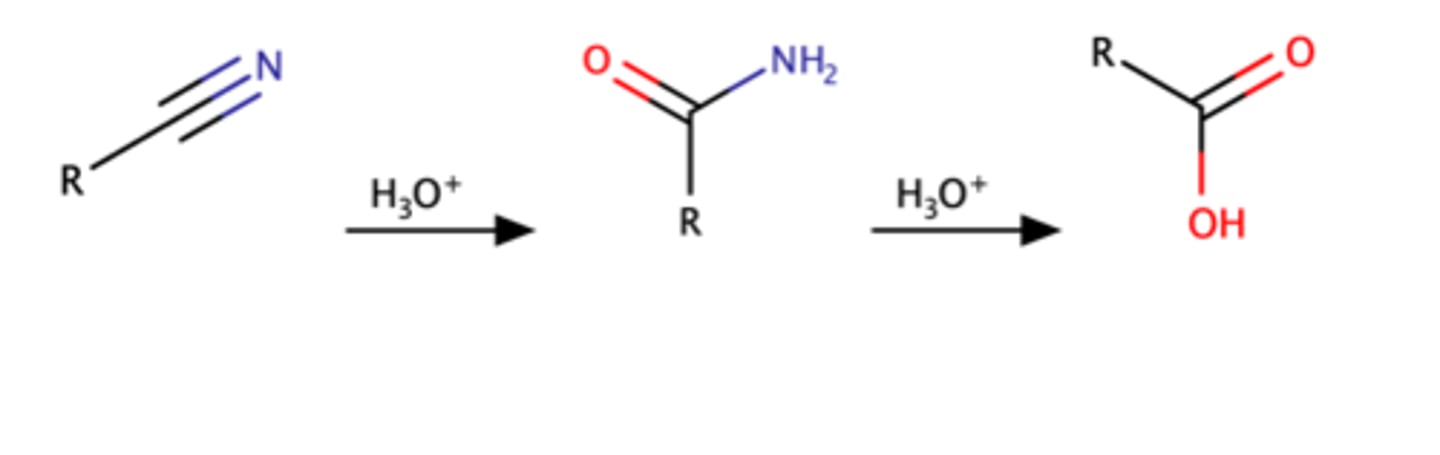
RCN + H3O
Nitrile = amide = carboxylic acid
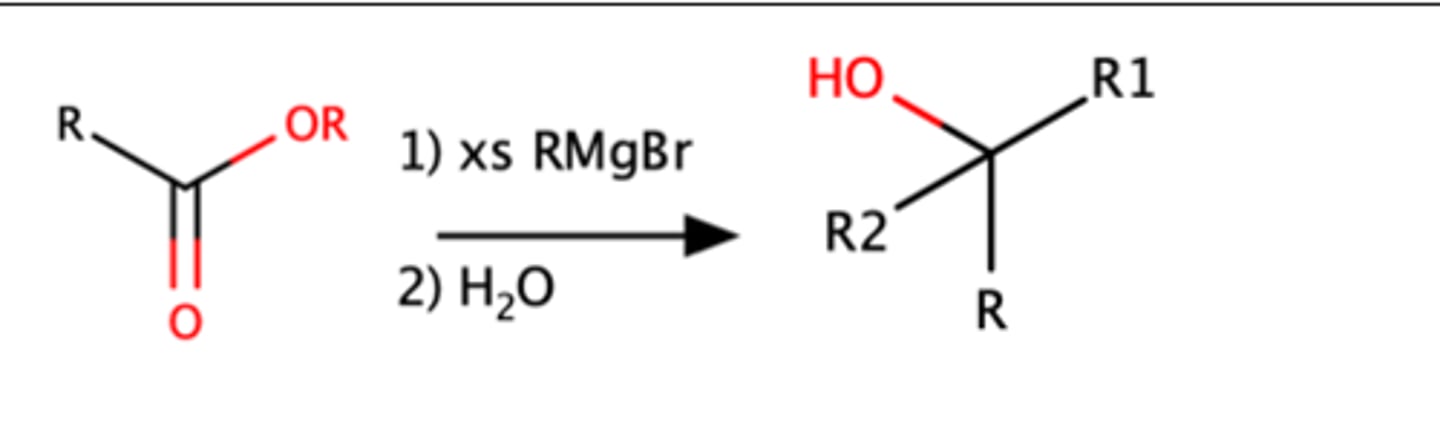
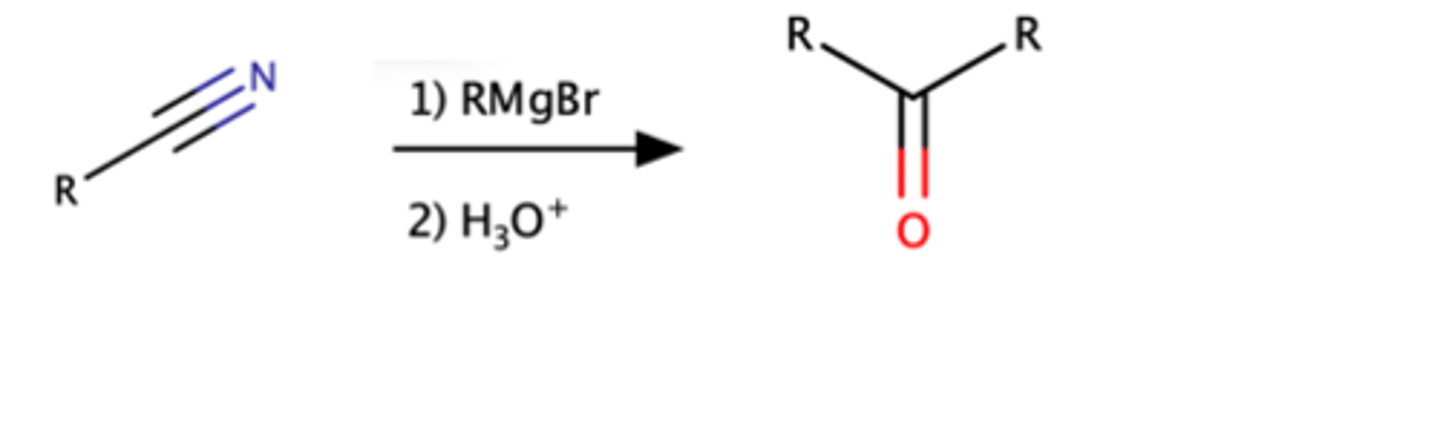
Nitrile + 1.) RMgBr 2.) H3O
Ketone
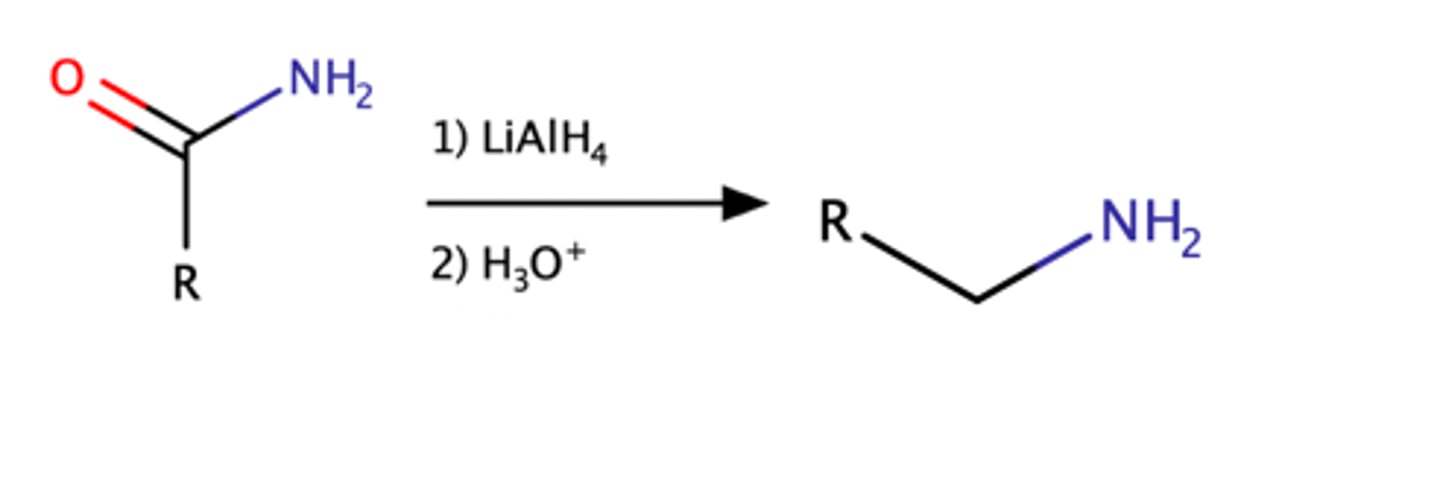
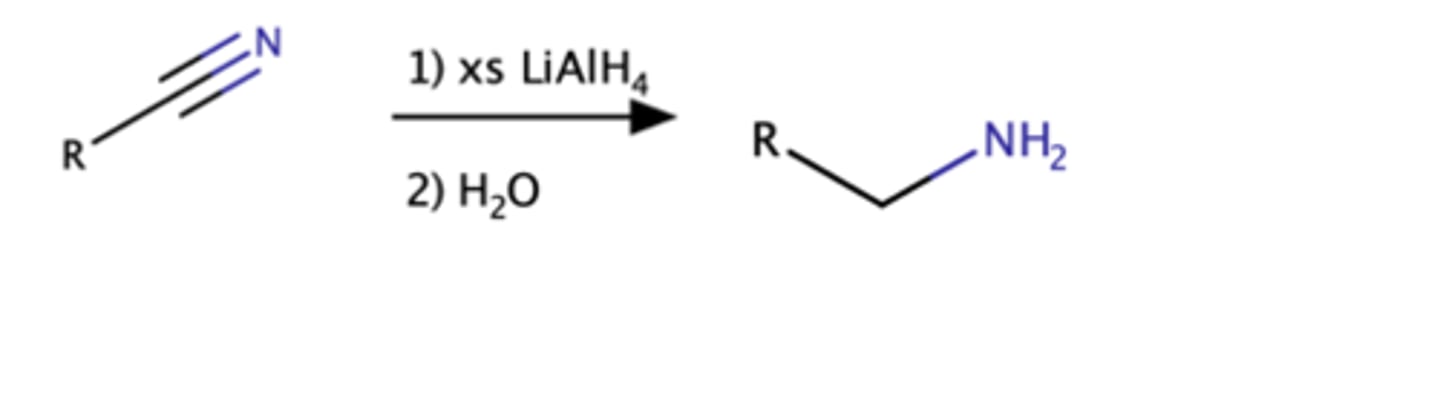
Nitrile + LiAlH4, H2O
amine
What factors effect reactivity to nucleophiles?
steric effects and # of R groups. The more R groups the LESS reactive
Which is more reactive towards nucleophiles aldehydes or ketones?
aldehydes
Aldehyde
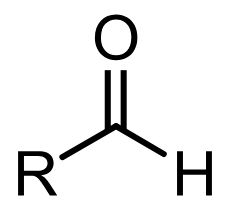
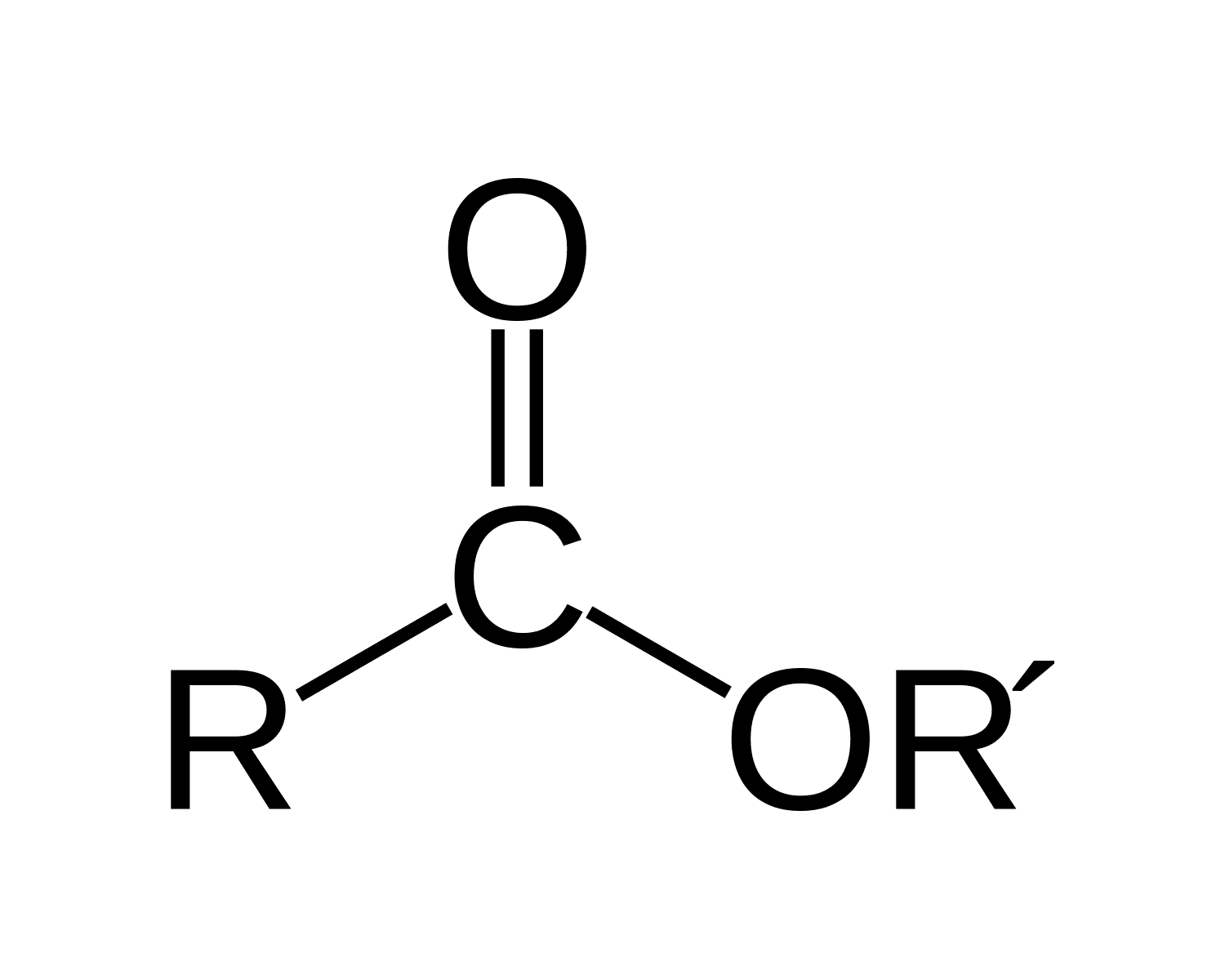
Esther
Mechanism of Hydration
H3O+ donates hydrogen to ketone
Double bond donates a pair to oxygen
The remaining H2O attaches to the carbonyl carbon
Hydrogen leaves the water and gets attached to a floating water molecule
Remaining structure has two OHs with trans stereochemistry
Cyanohydrin formation
When a HCN or KCN attaches to carbonyl carbon and double bond gives back to oxygen
Oxygen gets Hydrogen from nitrile
End product is OH and CN attached to the carbonyl carbon
Imine formation
Amine attaches to carbonyl carbon and double bond feeds oxygen
H attaches to the Oxygen
Imine
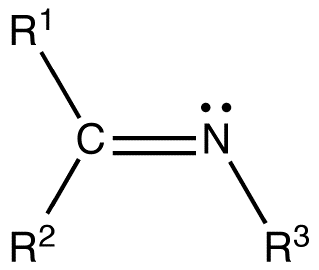
What happens to natural carbinolamine at high ph?
It is deprotonated
What is the alpha carbon
The carbon closest to the carbonyl carbon
Enamine
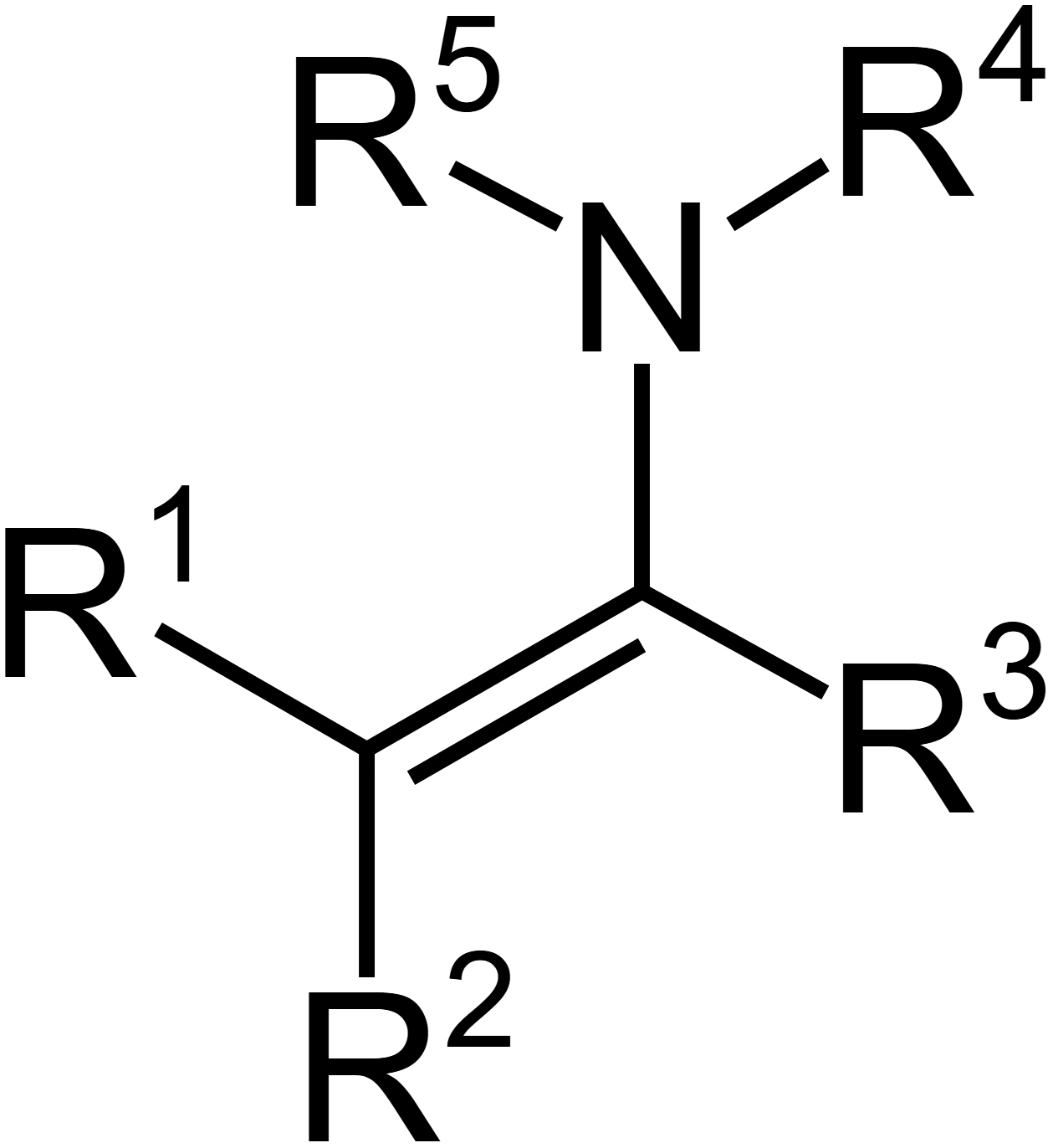
What happens during enamine formation
alpha carbon donates hydrogen bond to create double bond between carbonyl carbon and alpha carbon
Wolf-Kischner reaction works for?
aldehydes and ketones ONLY
Wolf-Kischner reaction mechanism
N4H2/KOH catalyst
Imine
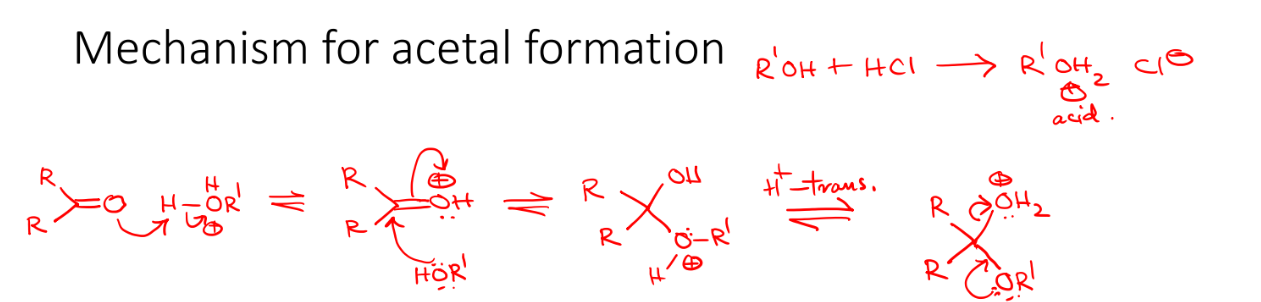
Acetal formation
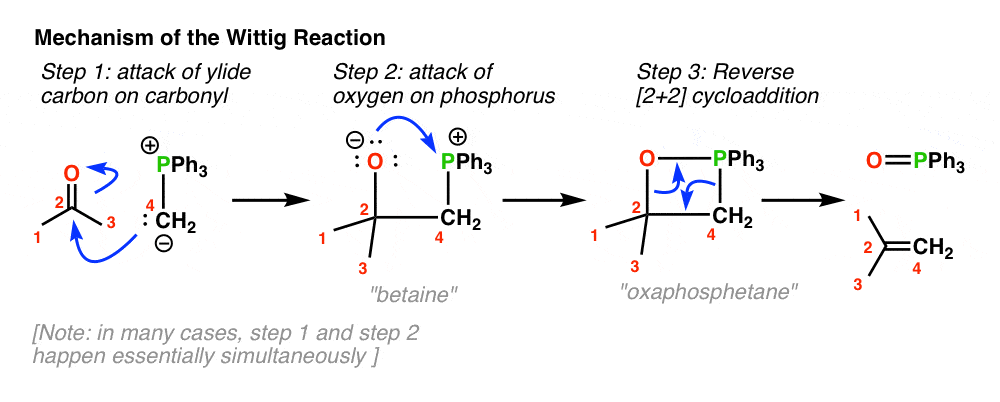
Wittig reaction
Ylene and Yline
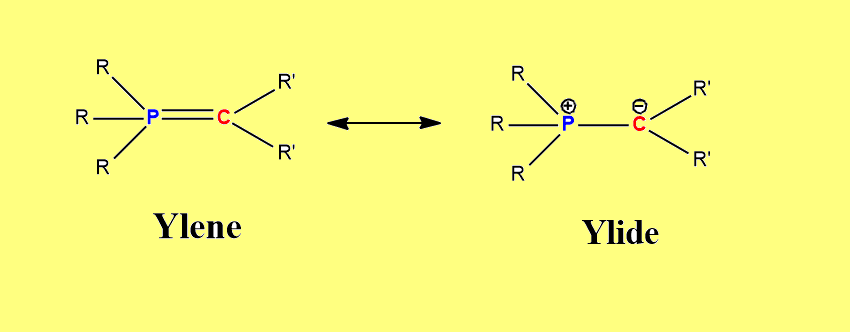
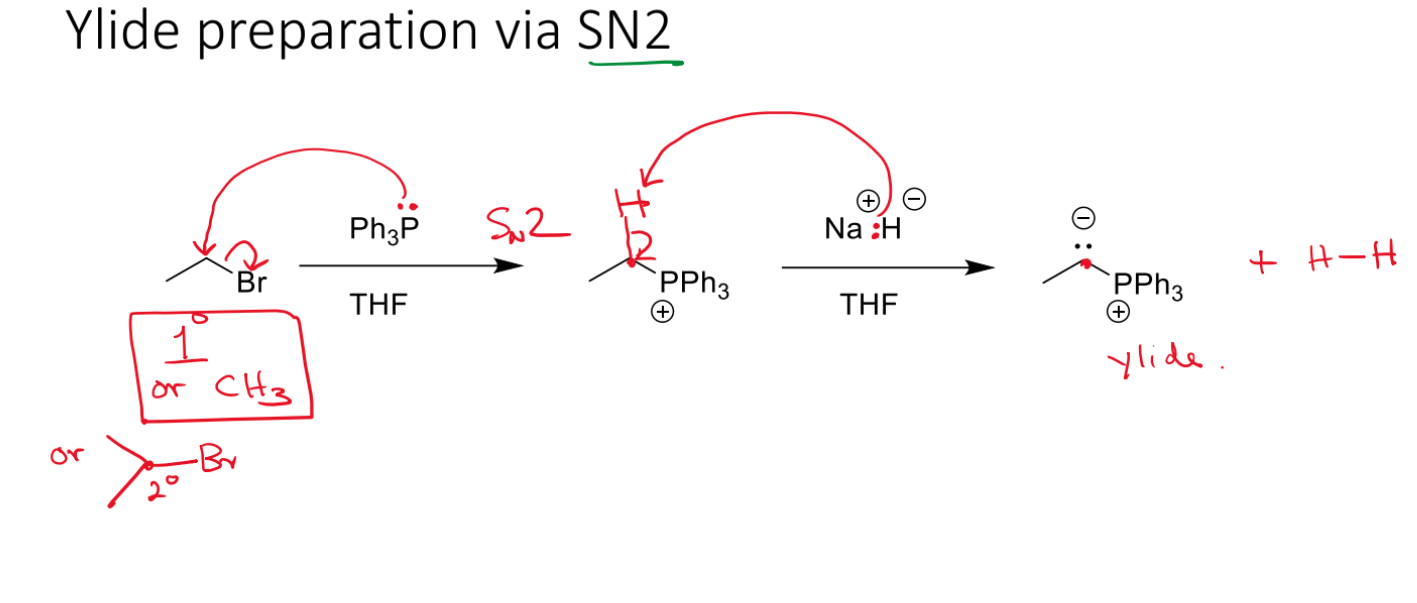
Ylide prep
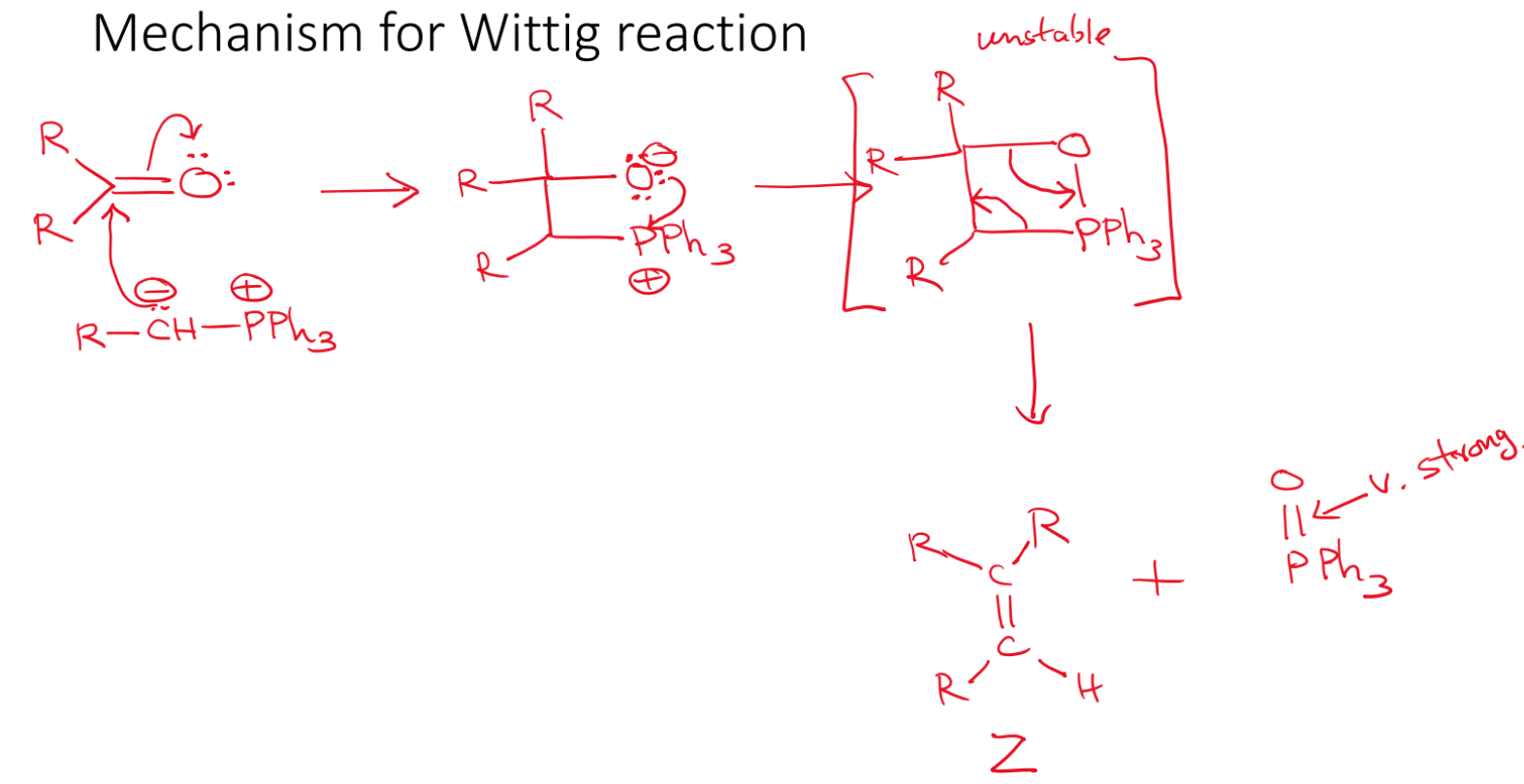
Wittig reaction
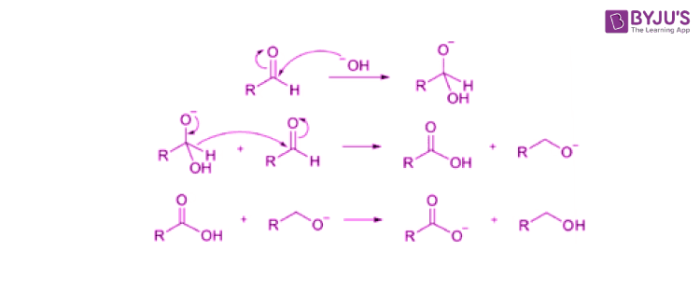
Cannizzaro reaction
Requirements for cannizzaro:
Carbonyl MUST be aldehyde
The alpha-carbon of the aldehyde must NOT have a hydrogen atom