ALL IB HL BIOLOGY
1/569
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
570 Terms
Water as the medium for life
a. the first living cell originated in aqueous solutions (water with substances dissolved in it), called the primordial soup, the solution provided the ideal conditions for chemical reactions to occur (water acted as a solvent, dissolving molecules allowing them to interact which led to the formation of more complex molecules which are essential to life). during the formation of a cell, a small volume of water became enclosed in a membrane and chemical reactions between solutes within the membrane formed structure and life began to develop. water also kept out harmful ultraviolet radiation from the sun.
b. a medium is something in which processes can occur, life’s processes take place in aqueous solutions. the temperature of water is so it is the medium for life.
i. metabolism: reactants and products of most chemical reactions in living organisms are dissolved in water, which allows the reaction of hydrolysis and digestion take place in aqueous solutions.
ii. growth: cells must absorb water by osmosis to increase in size.
iii. reproduction: sperm swim to the egg through water.
iv. water minimises temperature fluctuations are evaporated by water in the others.
v. movement: aquatic organisms swim or drift in water and the pumping of blood and sap transports substances dissolved in water.
vi. response to stimuli: nerve impulses are movements of dissolved ions and water and hormone transport in blood.
vii. excretion: urine is an aqueous solution of waste products and the excretion of gases requires a moist surface.
viii. homeostasis: blood plasma and tissue fluid are aqueous solutions that are regulated to form a stable and ideal internal environment for cells.
Hydrogen bonds as a consequence of the polar covalent bonds within water molecules
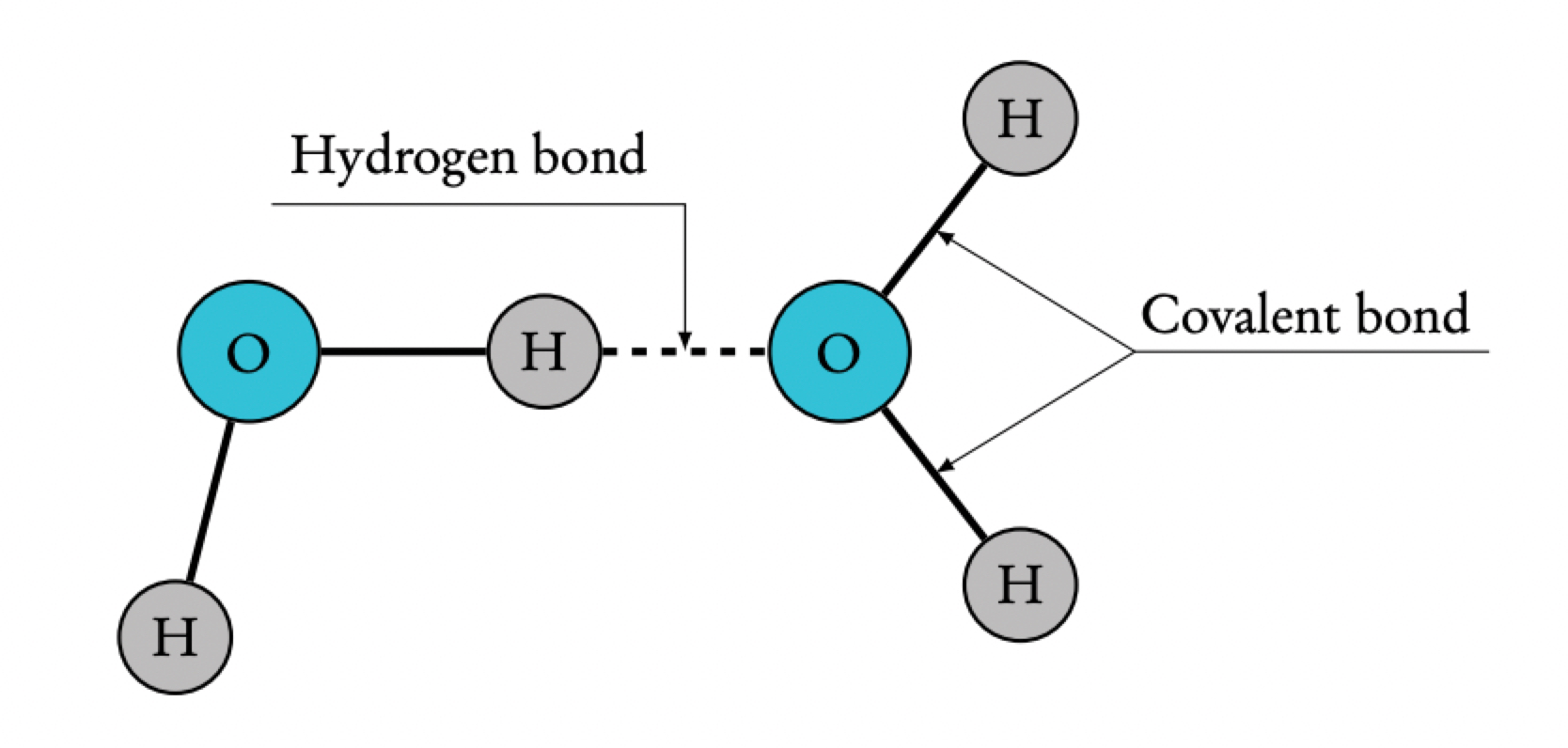
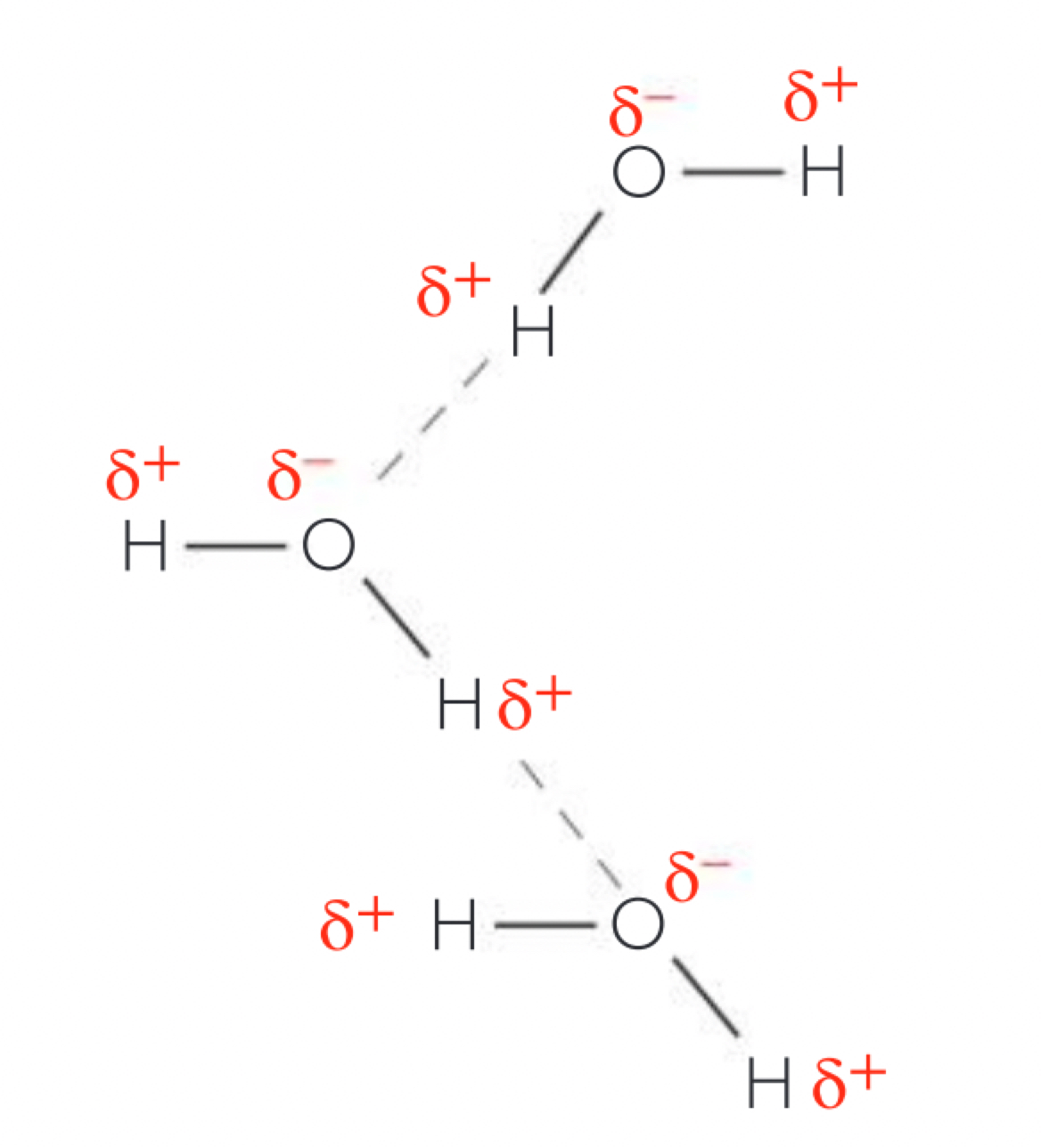
a. the hydrogen and oxygen atoms in a water molecule are joined through covalent bonds where the atoms share a pair of electrons. the hydrogen nucleus is less attractive to the electrons than an oxygen nucleus (more electronegative)(oxygen has more protons) causing the electrons to not be shared equally so the hydrogen atoms have a partial positive charge (δ+) and the oxygen atom a partial negative charge (δ-) because some of the electrons density has success, this causes the molecule to have polarity.
b. the bonds between oxygen and hydrogen atoms within a water molecule are intramolecular, but the electrostatic attraction between the positive and negative parts of another molecule is called a hydrogen bond (ephemeral attraction) individual hydrogen bonds form in water but water molecules are small so relatively large numbers of the bonds form in a volume of water.
Cohesion of water molecules due to hydrogen bonding and consequences for organisms
a. cohesion occurs when molecules of the same type are attracted to each other
b. water molecules stick to each other (cohere) because of the hydrogen bonds that form between them
c. plants use cohesion to move water up the xylem vessels, against gravity. the movement begins with transpiration (evaporation of water from the stomata). this creates a negative pressure/tension in the water column as the evaporated water has cohesion to the water in the xylem. this tension pulls water upwards through the xylem as the molecules are all cohesive to each other and all move up collectively to the low pressure area and the water moving upwards is replaced by ground water moving into the root system.
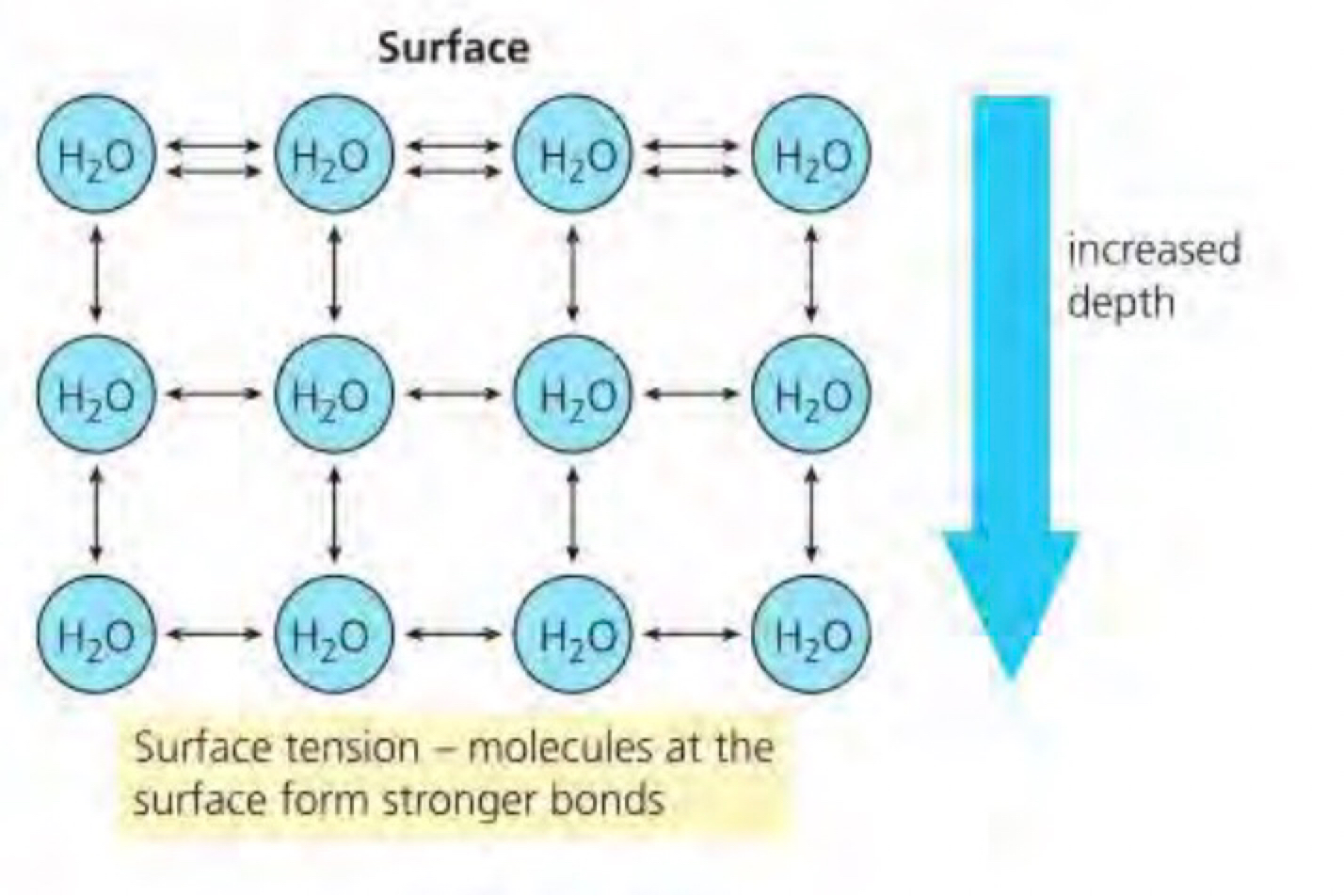
d. at the surface of a body of water the molecules are pulled inwards by hydrogen bonds with neighbouring molecules below and beside (but not above). the surface acts as an elastic membrane that shrinks to the minimum possible area. this is because the molecules on the surface have no hydrogen bonded molecules above and therefore exhibit stronger attractive forces upon them from their nearest neighbours on and below the surface. within the body of water there is no net force on a molecule because the cohesive forces exerted by the neighbouring molecules all cancel out. however for a molecule at the surface there is no inward force since there is no attractive force acting from above. this causes the molecules to arrange themselves to maximise bonding with adjacent molecules forming a hexagonal lattice (in the bulk of water in the water molecules don’t need to compensate for the missing attraction so move more freely). this causes molecules on the surface to contract and resist being stretched or broken. this is called surface tension and is used by many animals as habitats.
Adhesion of water to materials that are polar or charged and impacts for organisms
a. adhesion is the attraction between, unlike molecules due to hydrogen bonding. water molecules can adhere to materials which are hydrophilic which are substances that are attracted to water and can make intermolecular bonds with the water molecules. polar and charged materials are hydrophilic.
b. cellulose in plant cell walls are polar and form hydrogen bonds with water
c. capillary action is the movement of water through narrow spaces even against gravity. capillary action occurs when adhesion is greater than cohesion (creating the meniscus) as the water molecules stick to the tube’s wall and climb upwards, pulling other water molecules along within the xylem. this means the water molecules up the stem are being pulled up adhesion in the xylem keeps the entire column from dropping down within the tube.
the plant cell walls/cellulose pulls it along from the xylem vessels and surrounding tissue keeping the plant cell hydrated (for example in spongy mesophyll after water is evaporated, driving continuous water transport). the water up and adhesion to polar molecules in the soil keeps it from draining downwards in the soil as there are microscopic channels that act similarly to capillary tubes.
adhesive forces are greater in narrow tubes as more water molecules are in contact with the surface, therefore being able to stick and support greater masses of water.
Solvent properties of water linked to its role as a medium for metabolism and for transport in plants and animals
a. a wide range of hydrophilic molecules are able to dissolve in water, any substance which is polar or has charged regions will be soluble in an aqueous solution, water has solvent properties due to its (as polarity) the polar molecules dissolve by forming hydrogen bonds with water ions with one positive charge form electrostatic interactions with the negative pole of water molecules and negative ions with the pos pole
b. most chemical reactions in cytoplasm are catalysed by enzymes - reactions without an aqueous solution in cytoplasm depend on enzymes to proceed at a rate necessary for life and at a temperature tolerated by that cell type. these chemical reactions take place in aqueous solutions as it allows both the enzymes and soluble substrates to move and collide. other solutions can occur but water is the best medium for transporting materials from cell to cell
c. the ability of water to dissolve organic salts, acids and gases provides a medium for transporting materials around in plants and animals and into and out of cells
d. the water contained in the xylem vessels of plants is also an aqueous solution that transports inorganic ions (Na+, K+, Ca2+) upwards from the root systems to the leaves and the stoma for products of photosynthesis to specialised organs
e. the blood plasma in animals (also in humans) is an aqueous solution that transports nutrients, amino acids, glucose and O2 (O2 is recognised by haemoglobin) to specialised organs. other examples include amino acids, glucose and O2 (O2 is recognised by haemoglobin)
f. if a substance is hydrophobic (one in which molecules more strongly attracted to each other than to the non-polar molecules) and are insoluble in water and they can therefore form a barrier to water and hydrophilic substances, the wax on the leaf surfaces and oils on the human skin prevent dehydration. hydrocarbon tails in the centre of cell membranes form a barrier that allows cells to regulate the movement of hydrophilic substances across the membrane. many proteins attached to the membrane have hydrophobic areas are inserted into the hydrophobic layer while their hydrophilic sections extend into the intercellular fluid or cytoplasm to interact with soluble substances.
Physical properties of water and the consequences for animals in aquatic habitats
a. upthrust and force which is the global movement of water is density, density allows organisms with smaller densities to experience significant buoyant forces, reducing energy needed to counteract gravity, ice is much less dense and therefore less buoyant, ringed seals have air-filled lungs to increase their buoyancy and loons have large wings to generate lift, less buoyant and solid bones to increase the weight and to compress air from its lungs and feathers to decrease buoyancy for diving
b. viscosity is resistance to flow and how much energy is needed to change shape of a liquid, water has a greater viscosity than air meaning aquatic animals need to overcome a larger drag force while swimming, to adapt the seals streamline their bodies to reduce drag and their flippers allow efficient propulsion whilst the black-throated loon takes advantage of requiring significant energy and strong and broad feet in the water (webbed feet help them move through the water swiftly as their webbed feet provide a large surface area to push against water, the feet are also located laterally and towards the back end of the body to allow maximum propulsion and reduce drag.
c. thermal conductivity, the ability of a substance to transfer heat, water has higher thermal conductivity than air, this means that water conducts heat away from the body more efficiently, the seals therefore face significant heat loss which they combat with their thick blubbery layers which insulate against heat loss and use a counter-current heat exchange system with blood vessels (minimise heat loss from arterial blood to veins that flow near their tail, where they lose heat) (or water) (or water providing (ice, who rings heat raising sea lumps)), in water the loon relies on feathers that trap insulating air vessels to reduce heat loss, they also have a (oil) near their tail where they oil their feathers to waterproofing
d. specific heat capacity is the amount of energy required to raise the temperature of 1g of substance by 1°C, water has a higher specific heat capacity than air meaning the temperature of water changes less easily, this benefits the animals as they experience less dramatic temperature fluctuations, this also means that in very cold water may be ice and when the water does freeze, the ice floats forming habitats on and below the ice for the seals
Extraplanetary origin of water on Earth and reasons for its retention
a. when the earth was first formed it was far too hot to have liquid water on its surface; over a very long time as the earth cooled, water was brought to earth through many collisions with meteorites with asteroids (that had hydrated minerals)
b. earth began to cool, water was able to condense and the force of earth’s gravity (strong pull due to its size) helped to retain water on earth’s surface and after that, life began to evolve. (the intensity of sunlight due to the distance keeps temp between 0 to 100°C). that theory is supported by the hydrogen isotopes which are similar to the water on earth and found on some asteroids
Relationship between the search for extraterrestrial life and the presence of water
a. a planet that could possibly support life must have liquid water on the planet, therefore must exist in an area of its solar system where the sun is positioned in relation to the planet in the habitable zone or goldilocks zone
b. needs suitable temperatures for liquid water (0°C–100°C) and sufficient gravity to retain its water. earth also developed an atmosphere and magnetic field that protects it from harmful incoming radiation emitted from the sun.
c. considering the great number of stars, it is reasonable to conclude that earth is not the only planet within the goldilocks zone.
d. water is responsible for most of the complex chemical processes without it, life would not occur in the search for extraterrestrial life begins with the search for water.
DNA as the genetic material of all living organisms
a. a key feature that unifies all life is that the molecule used as genetic material in all living organisms is DNA (deoxyribonucleic acid) provides the long-term storage genetic information. every cell in a multicellular organism has the same DNA but each has a different type of cell only uses the genetic information that is appropriate to that cell
b. viruses are not considered living in some definition as DNA is the genetic material, they do not living because they cannot survive without a cell of a living organism and they have no internal biochemistry when they exist as a separate particle (cannot replicate without a host)
Components of a nucleotide
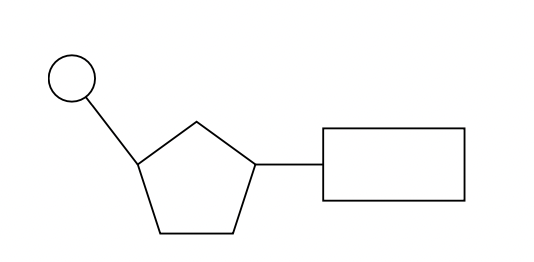
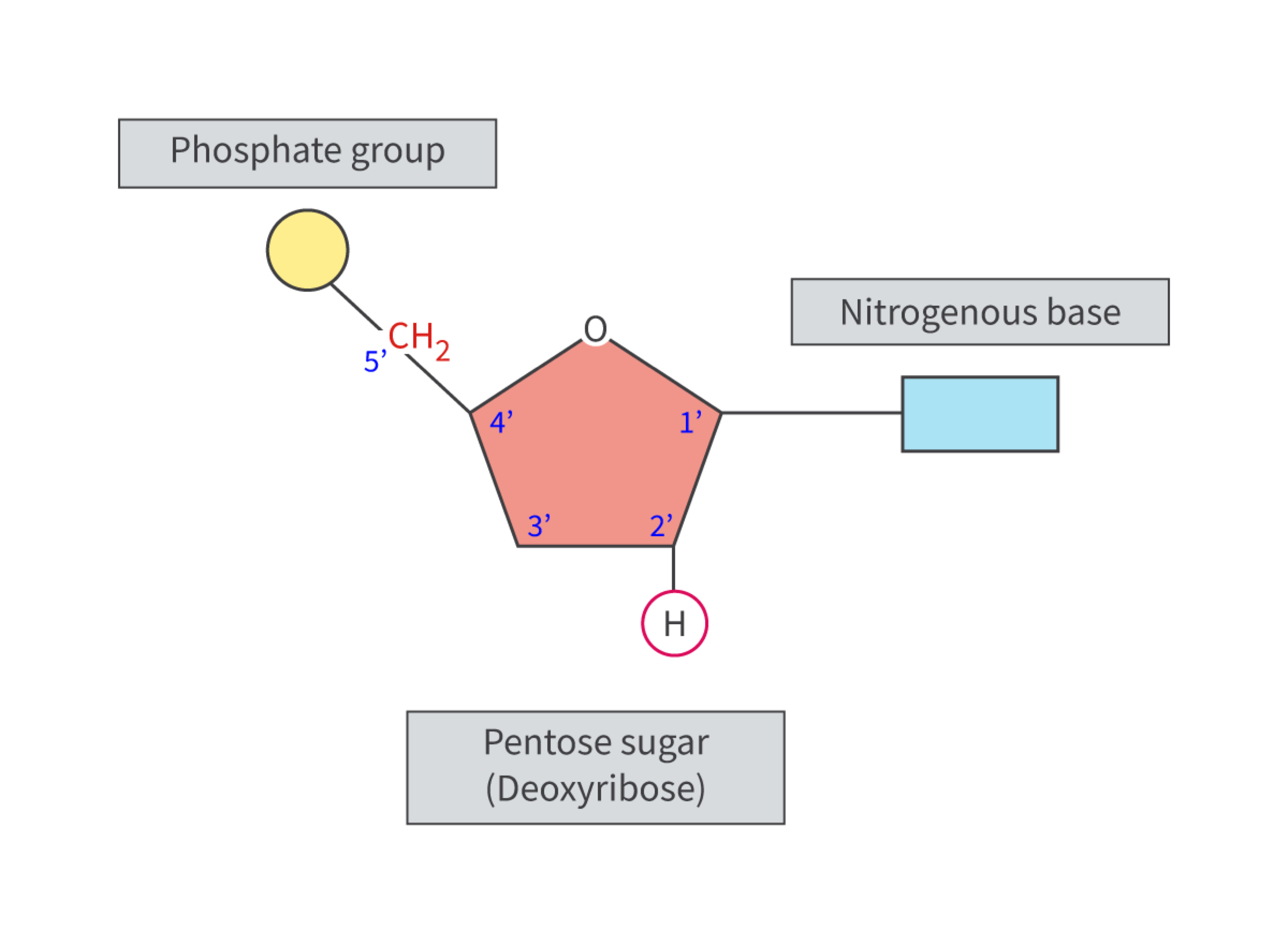
a. DNA and RNA are polymers of nucleotides (subunits), each nucleotide consists of 3 parts: a phosphate group, a five-carbon monosaccharide (pentose sugar) (deoxyribose in DNA and ribose in RNA) and a nitrogenous base. the components are combined by an enzyme-controlled condensation reaction to form a nucleotide; the nitrogenous base to the 1’ carbon atom and the phosphate group to the 5’ carbon atom.
Sugar–phosphate bonding and the sugar–phosphate “backbone” of DNA and RNA
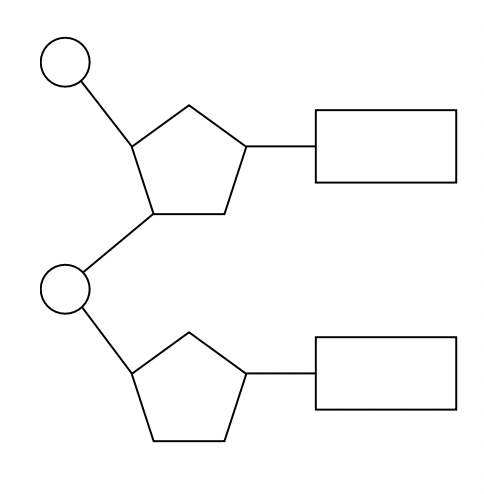
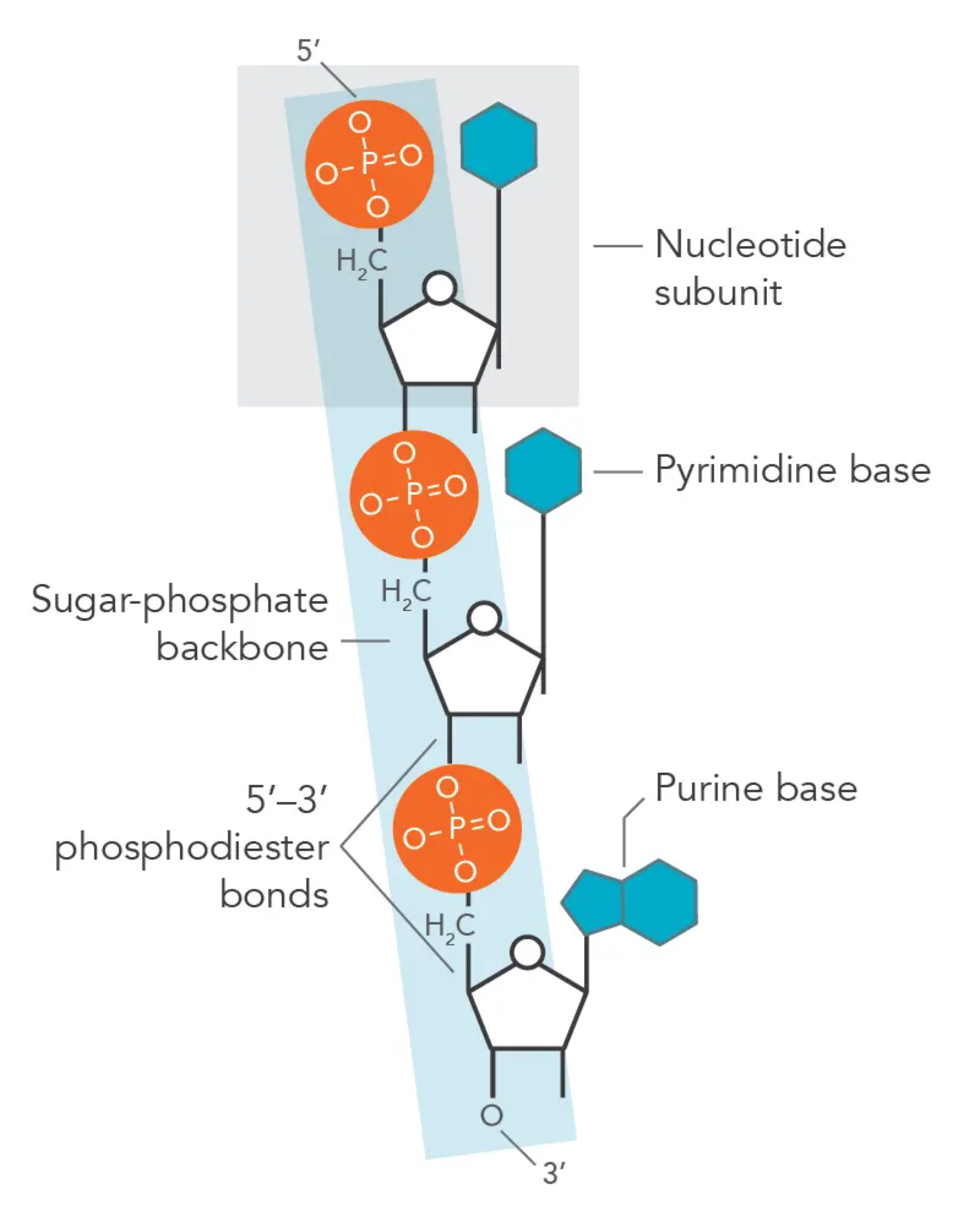
a. the polymers are formed through covalent bonds between the phosphate group of one nucleotide bonds with the 3’ hydroxyl group (-OH) on the pentose sugar of the preceding nucleotide. this is called a phosphodiester bond. covalent bonds are relatively strong and thus nucleic acid polymers are stable structures. this process creates a repeating chain of sugar and phosphate groups forming the backbone of the molecule.
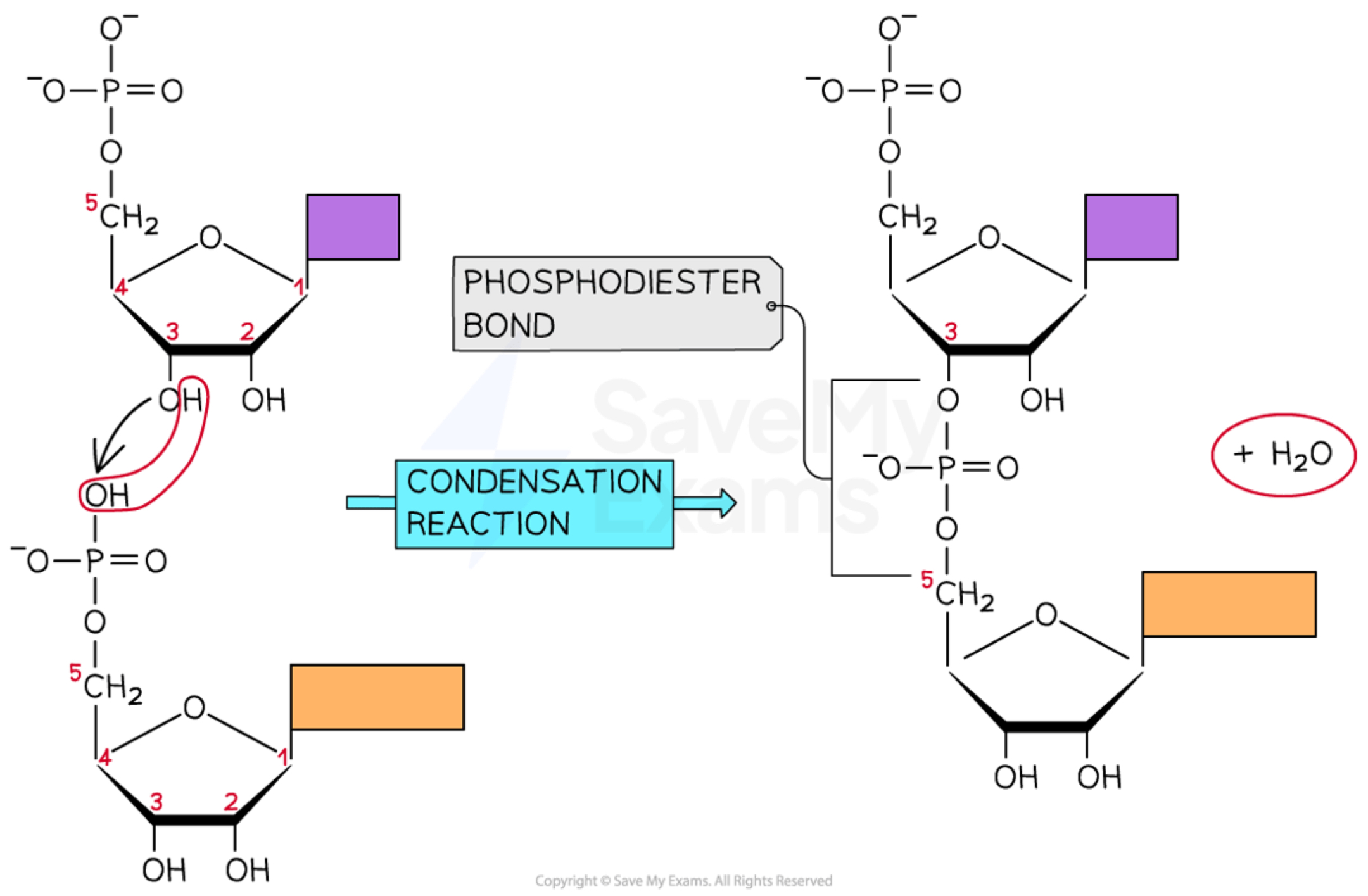
Bases in each nucleic acid that form the basis of a code
a. there are 4 nitrogenous bases but it are found in both RNA and DNA. these bases can be arranged in any sequence which forms the basis of the genetic code
nucleic acid | pyrimidines (-) | purines (+) |
|---|---|---|
DNA | cytosine | adenine |
thymine | guanine | |
RNA | cytosine | adenine |
uracil | guanine |
RNA as a polymer formed by condensation of nucleotide monomers
a. DNA is formed as nucleotides join together by a condensation reaction between the pentose sugar and the phosphate group of 2 nucleotides with water as a by-product. RNA is a single stranded polymer of nucleotide monomers
b. mRNA (messenger RNA) forms in the nucleus and moves out to the ribosomes in the cytoplasm. tRNA (transfer RNA) responsible for transporting amino acids to ribosomes during protein synthesis. rRNA (ribosomal RNA) forms part of the ribosome.
DNA as a double helix made of two antiparallel strands of nucleotides with two strands linked by hydrogen bonding between complementary base pairs
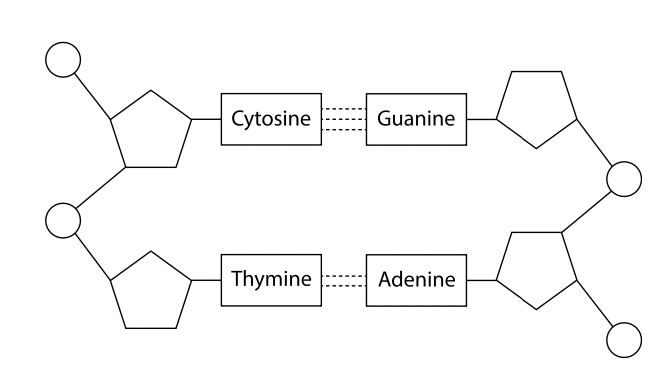
DNA consists of 2 strands of nucleic acids connected by hydrogen bonding between base (will only form hydrogen bonds with one of the other bases, adenine with thymine and cytosine with guanine. they are complementary base pairs). the 2 strands are antiparallel (run in opposite directions, one runs in the 5’ to 3’ direction while the other from 3’ to 5’) as a they are wound together to form a double helix.
Differences between DNA and RNA
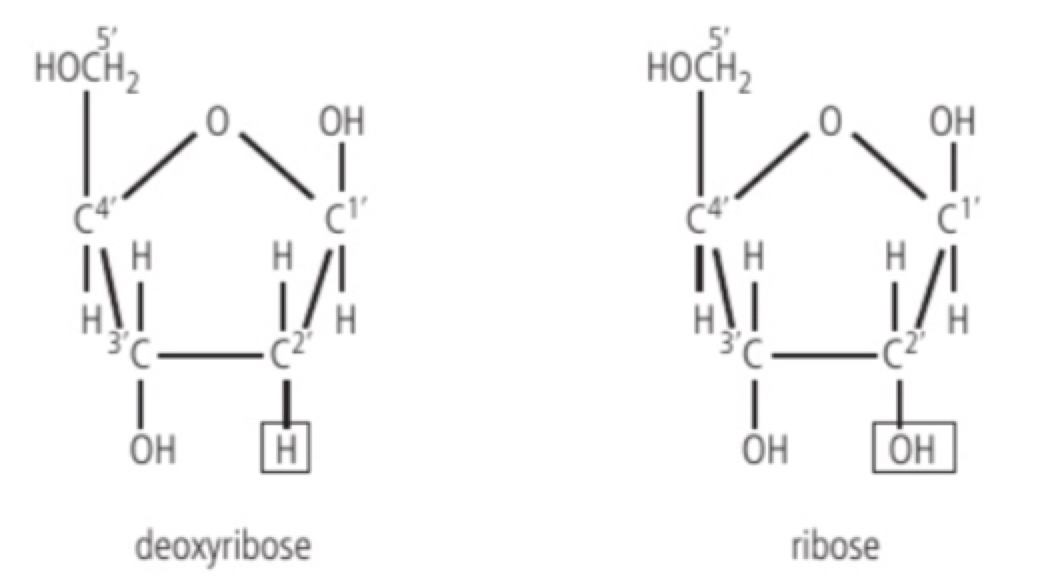
a. DNA: double stranded molecule, all nucleotides contain deoxyribose sugar, thymine is one of the four nitrogenous bases shaped into a double helix and acts as the permanent genetic code of a cell/organism.
b. RNA: single stranded molecule, all nucleotides contain ribose sugar, uracil is one of the four nitrogenous bases, variety of shapes depending on the type of RNA and does not contain a permanent genetic code, except in RNA viruses.
c. ribose has a molecular formula of C5H10O5 while deoxyribose has a formula of C5H10O4 (- ribose has one more oxygen)
d. different types of nucleic acids: mRNA, tRNA, rRNA, ATP
Role of complementary base pairing in allowing genetic information to be replicated and expressed
a. complementary base pairing is based on hydrogen bonding and achieved when purine forms H-bonds with pyrimidine so that only cytosine and guanine form 3 H-bonds and adenine and thymine form 2 H-bonds.
b. this is very important in DNA replication as the sequence of bases on one strand accurately specifies the sequence of bases on the other strand.
c. it is also seen in transcription (RNA can be made from DNA in dividing cells).
d. the base sequence on one of the DNA strands (DNA) carries the base sequence (sense strand - ribosome) translation (= base sequence can be used for the amino acids sequence in a polypeptide. mRNA has a codon and the tRNA has nucleotides using complementary base pairing to create specific amino acids in a polypeptide.
Diversity of possible DNA base sequences and the limitless capacity of DNA for storing information
a. DNA stores genetic information in triplet codons. Because the 4 bases could occupy each position along the DNA strand, the number of possible sequences is 4^n (where n is the number of bases). The shortest DNA molecule in the human genome is about 50 million base pairs so the likely hood of 2 DNA molecules being identical due to random chance approaches 0. Therefore DNA can contain a nearly unlimited amount of genetic information.
Conservation of the genetic code across all life forms as evidence of universal common ancestry
a. Each of the 64 codons of the genetic code indicates either one of the 20 amino acids or the end of the polypeptide. There are many ways of assigning meanings to the codons but all organisms use the same meanings with only minor variations. This universally suggests that all life evolved from the same common ancestor. (The organisms are different because the DNA base sequence are different but if the code to read the sequence is the same, evolution changes the DNA sequence but always continued to use the same mechanism for genetic coding.)
Directionality of RNA and DNA
a. In a DNA molecule the 2 antiparallel strands have a 5’ to 3’ orientation and a 3’ to 5’ orientation, but new strands are synthesised from the 5’ to the 3’ end, this directionality is important when a new nucleic acid strand is formed: when DNA is replicated each strand can act as a template and the new strand will always begin with the 5’-end nucleotide first as the 3’ end has the pentose sugar’s -OH group to which new bases can be linked.
b. The first stage of protein synthesis is transcription where the DNA is unwrapped and the RNA is synthesised using complementary base pairing, it is synthesised using the 5’ end first, this mRNA then pairs with a ribosome and then the tRNA bonded to a specific amino acid bonds with triplets of the mRNA (the process is translation which also starts at the 5’ end of the mRNA)
Purine-to-pyrimidine bonding as a component of DNA helix stability
a. Pyrimidines (thymine and cytosine) are smaller and have a single ringed structure while purines (adenine and guanine) are larger as they have a double ringed structure, to make the double helix a purine always bonded to pyrimidine resulting in there being a constant distance between the 2 strands leading to the stable double helix shape
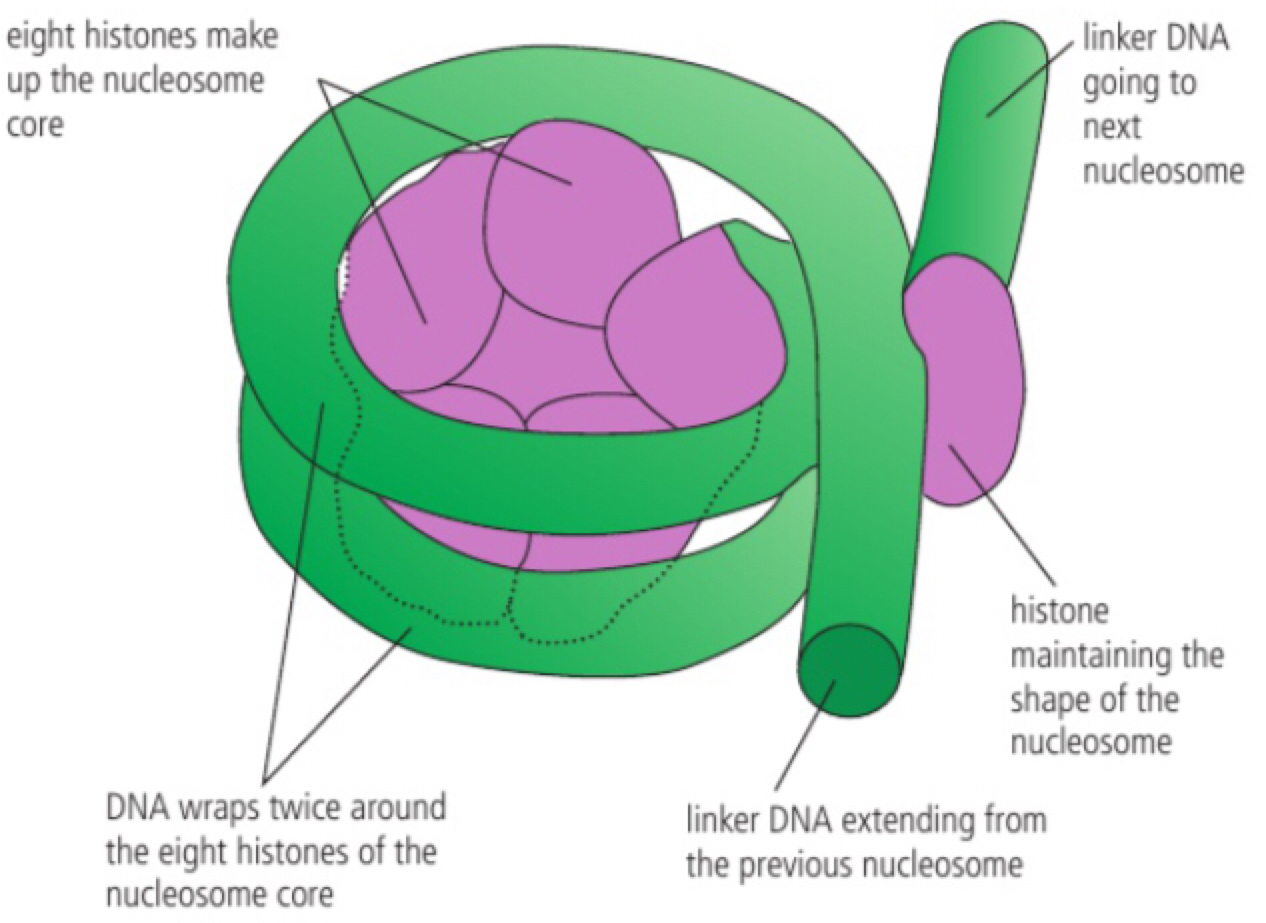
Structure of a nucleosome
a. Due to the length of some DNA molecules to fit more inside the nucleus in eukaryotes, packaging solution has evolved. Within the nucleus there are histone proteins. first the DNA wraps itself around eight of these histone proteins and then an additional histone (H1) helps hold the structure together resulting in a structure called a nucleosome. The DNA that extends from one nucleosome to another is called linker DNA, the nucleosome then stacks to begin coiling around other proteins in a condensed shape called a chromatin (a chromosome is formed)
Evidence from the Hershey–Chase experiment for DNA as the genetic material
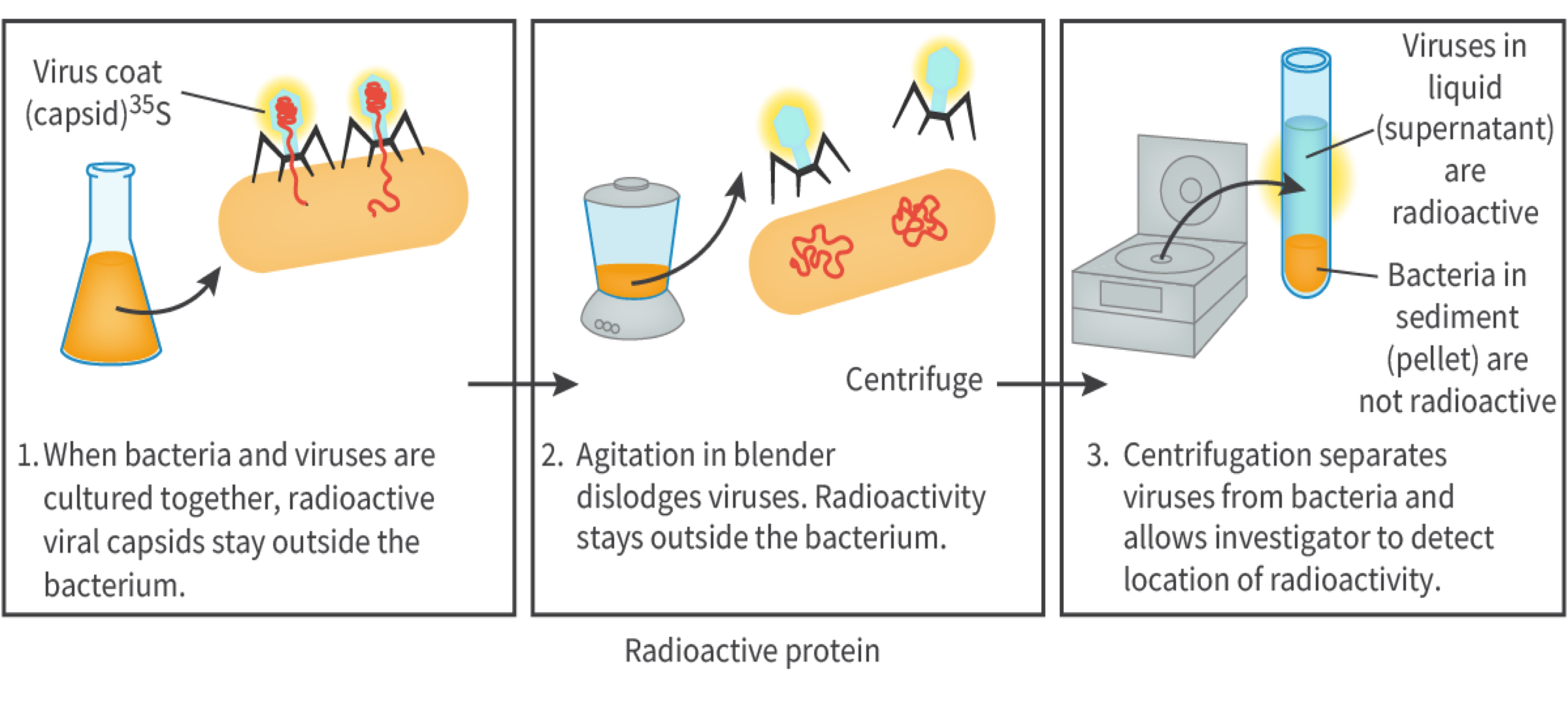
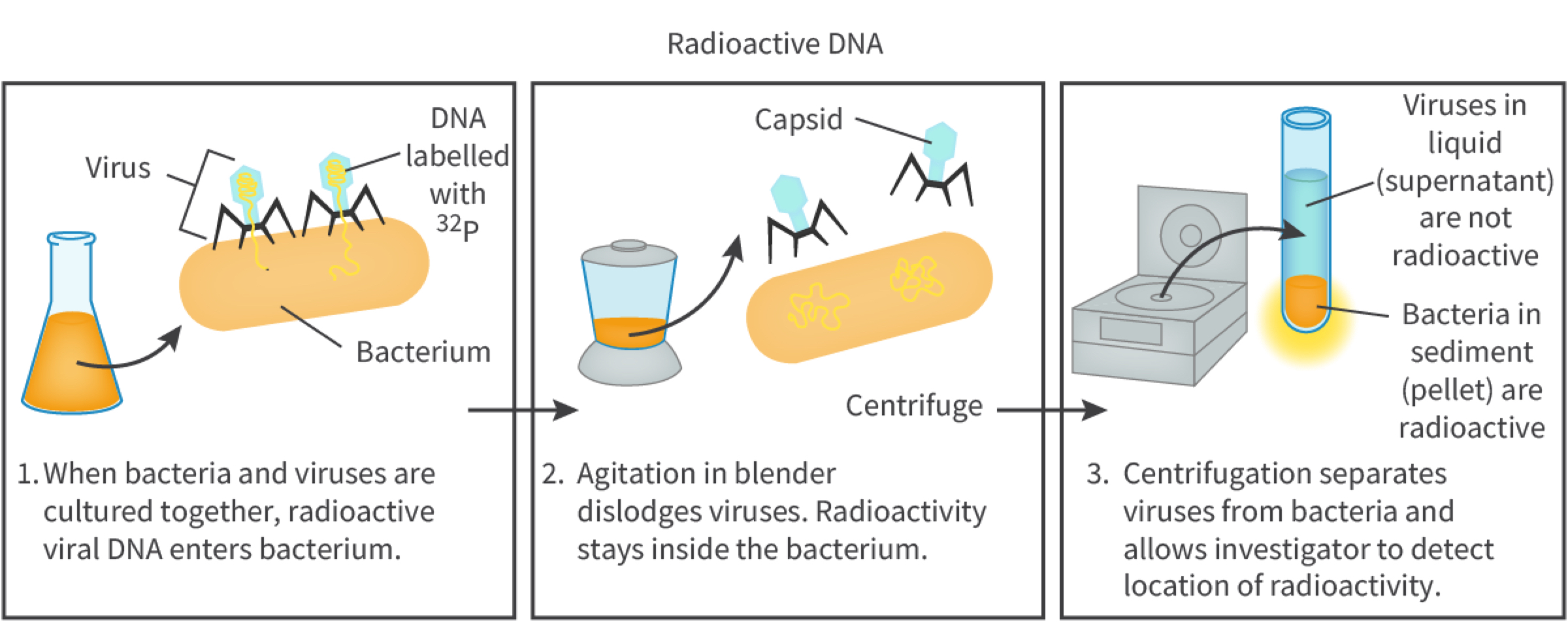
a. Biologists knew some genetic genetics was based on nucleic acids or proteins so in 1951 Alfred Hershey and Martha Chase conducted experiments which confirmed DNA as the genetic material.
The experiment used bacteriophage, T2 virus (a virus which infects bacteria) and the bacterium Escherichia coli. When bacteriophage infects an E. coli, the virus injects only the nucleic acid into the metabolism of the cell. They used radioactive process called radio active isotope labelling (making certain forms of elements that can be detected within molecules). They grew bacteriophage viruses in radioactive phosphorus 32 (these viruses had DNA inside their viral core with detectable phosphate 32) and in radio active sulfur 35 (this was present in the protein outer coat). The viruses produced (DNA does not include sulfur, proteins do) both viruses were allowed to infect the bacterium E. coli. After leaving it for enough time the mixture was agitated in a blender/processor and then centrifuged to separate the solid pellet containing the bacteria from liquid supernatant containing viruses and viral garbage. Sample was used to locate the radio activity: the bacteria infected by the phosphorus 32-labelled bacteriophage had radioactivity detected inside the cell indicating DNA to be the genetic material as the sulfur 35-labelled bacteriophage had no radioactivity inside the cell.
Chargaff’s data on the relative amounts of pyrimidine and purine bases across diverse life forms
He hypothesised in the 1900s that DNA exists as a double helix molecule (comparing the weight of the nucleotides) there is 1:1 ratio of purine to pyrimidine. Chargaff developed a research technique designed to show the proportion of the nitrogenous bases found in DNA – which is paper chromatography. He found that adenine always equals thymine and cytosine always equals guanine, this became known as Chargaff’s rule and it showed that the tetranucleotide theory is false as not all bases were equal in proportion but there are still relative amounts of pyrimidine and purine bases across diverse life forms.
Chemical properties of a carbon atom allowing for the formation of diverse compounds upon which life is based
a. carbon atom consists of six electrons orbiting a nucleus of six protons and 6, 7 or 8 neutrons depending on the isotope. four of the six electrons are in the outer shell, so carbon can form 4 covalent bonds to gain a complete outer shell. this can be done through single and double bonds with other carbon atoms, other non-metallic elements. carbons ability to bond with 4 other atoms allows complex molecular structures to be formed (rings (single or multiple) and branched/unbranched chains) – all these possibilities result in a diverse range of organic compounds (carbohydrates, lipids, proteins and nucleic acids) which form the basis of life, as it is the most versatile block of life on earth is described as carbon-based.
atomic prefixes: kilo 10^3 centi 10^-2 milli 10^-3 micro 10^-6 nano 10^-9
Production of macromolecules by condensation reactions that link monomers to form a polymer
living organisms produce macromolecules from subunits called monomers that instead of using covalent bonds, a chain of 10 or more monomers is a polymer and the monomers are linked together through a condensation reaction (molecules are joined together and a by-product is created from an OH group and a H from the other molecule)
c. examples of this is amino acids → polypeptide (amino acid → protein + water), nucleotides → nucleic acid (phosphate group + pentose sugar + nitrogenous base = nucleotide + 2 water) and monosaccharides → polysaccharides (glucose + galactose → lactose + water or glucose → starch + water)
Digestion of polymers into monomers by hydrolysis reactions
polymers are hydrolysed or broken down into smaller molecules and water is used in the process. hydrolysis reactions are used to digest food. it breaks the covalent bonds between monomers. the resulting monomers are then soluble that are absorbed into bloodstream and excreted by body cells, often after the monomers enter the cell they are built up into macromolecules. it uses the splitting of water to provide the OH group and H (hence the name) which bind to the exposed ends.
e. examples include: polysaccharides → monosaccharides (starch + water → glucose), and polypeptide → amino acids (protein + water → amino acid)
Form and function of monosaccharides
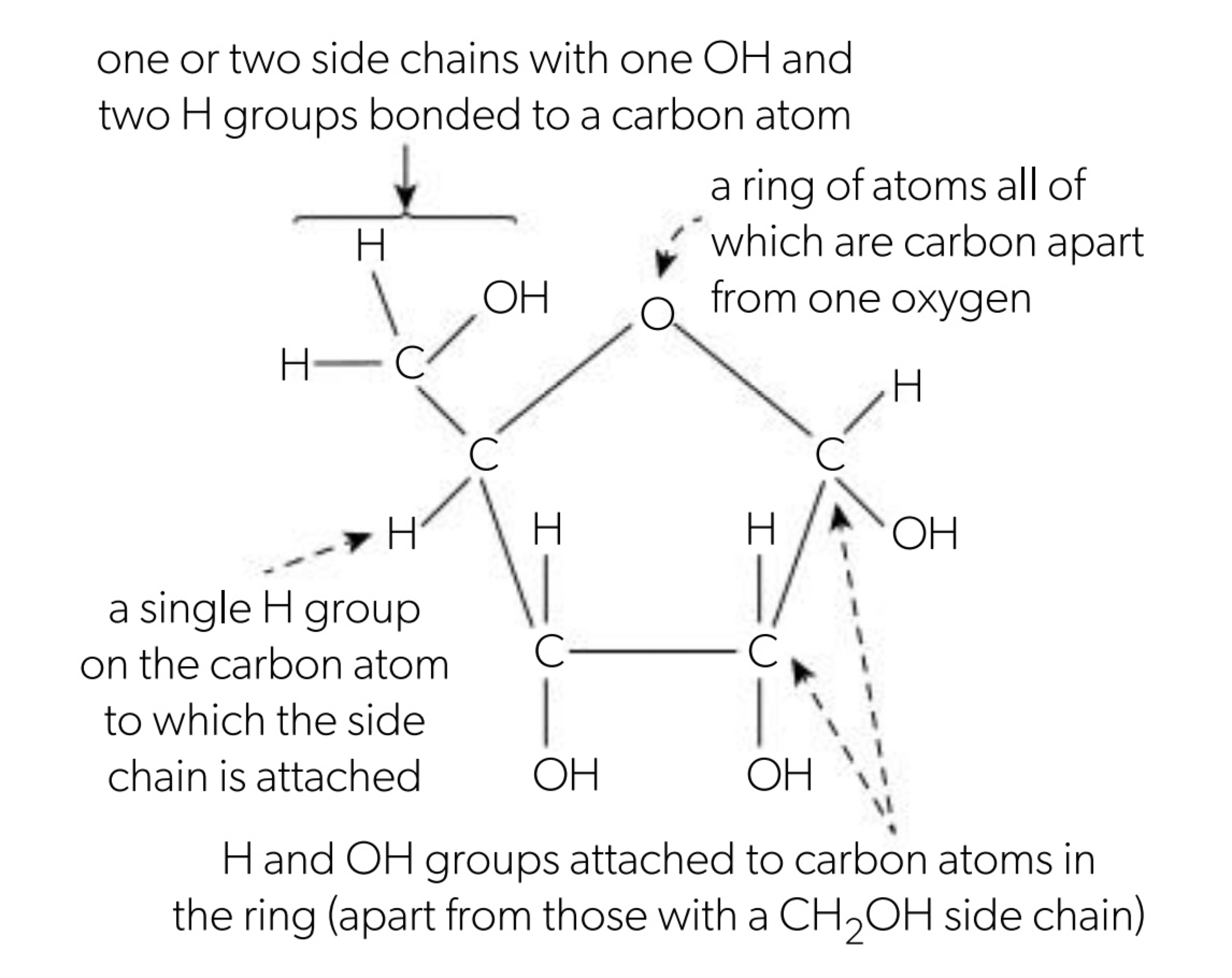
Monosaccharides are sugars that cannot split into simpler sugars so they are monomers (simplest form of carbohydrates). Most living organisms contain monosaccharides that contain 3, 5 or 6 atoms of carbon (trioses, hexoses and pentoses), hydrogen and oxygen in the 1:2:1 ratio (CₙH₂ₙOₙ) (hexose: C₆H₁₂O₆ and pentose: C₅H₁₀O₅).
one of the oxygen atoms un monosaccharides is bonded to carbon only, either by double or 2 single bonds to carbon/s in a ring of atoms. the other oxygens are part of the OH group.
Ribose is a monosaccharide found in pentose monosaccharides and glucose is a hexose monosaccharide. Monosaccharides can be used for structural purposes (cellulose) or for energy (starch). Glucose is functional, group is hydroxyl (alcohol) forms a linear carbon chain in molecule. The covalent bond between oxygen and hydrogen is polar, causing glucose to be a polar molecule due to its polarity it has properties such as molecular stability (should covalent bond break), high solubility in water (dissolves under solvent), easily transportable (soluble in water - circulates blood and fluids between cells) and can yield a great deal of chemical energy (covalent bonds between H atoms) - an oxidation reaction sustaining the glycolysis pathway.
Polysaccharides as energy storage compounds
a. After glucose (glycogenesis) is photosynthesised plants were made to create starch molecules (glucose polymer) to make the starch as compact as possible, plants use alpha 1-4 linkages and alpha 1-6 linkages which structure their α polysaccharides. Amylase and amylopectin enzymes are the common forms of starch in plants, amylase is a straight chain of α 1-4 carbon of the adjoining glucose, amylopectin is a repeated chain of 1-6 linkages and creates branches. A molecule of starch is a very large and compact molecule and is not soluble in water, this means that by storing glucose it can add and store glucose molecules either amylose or amylopectin by condensation reactions. When the plant needs to use the reserved glucose, hydrolysis reactions are used to break glucose molecules away from starch.
b. Glycogen is a polysaccharide made of glucose monomers that are bonded similar to the 1-6 linkages causing it to be very branched in many animals store excess glucose as glycogen and is not readily soluble in cytoplasm and other storage as they would not affect the osmotic balance turning leaves, stored in liver and muscles.
c. Starch and glycogen function well as energy stores because it is coiled and branched and makes them compact so they take up little space in the cell being relatively insoluble and do not draw an excessive amount of water into the cells. Glycogen is the main storage of glucose so easily mobilised when glucose is needed as a food source for the animals. This can be done more rapidly with more branching since there is more ends.
Structure of cellulose related to its function as a structural polysaccharide in plants
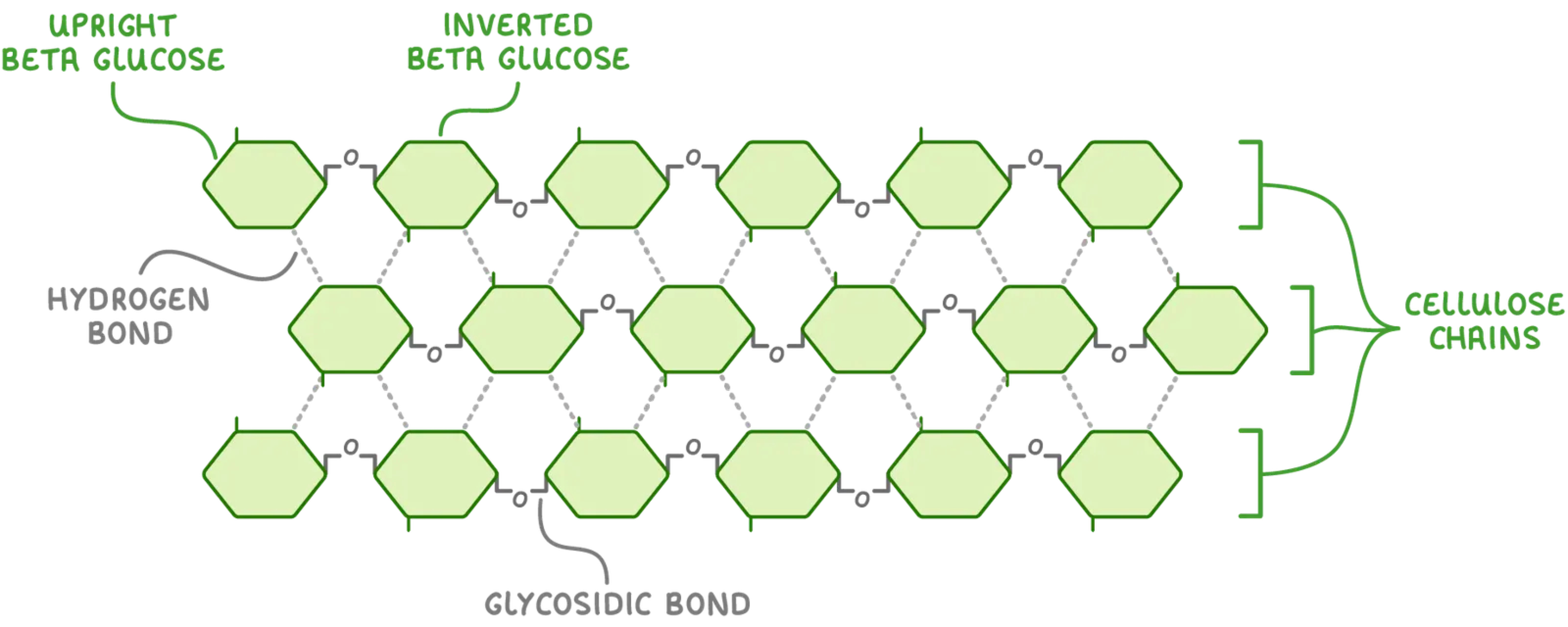
Cellulose is composed of beta glucose molecules (not group to left instead at bottom), linked by beta glycosidic bonds and make up the primary component of the cell walls of plants, the condensation reaction that bonds the beta-glucose molecules requires alternating orientation (every second molecule is upside down) to match the OH group of carbon-1 to a reaction with the OH group of carbon-4, this results in a linear polymer with no branching, this leads to cellulose polymers running parallel to each other in groups bundles and cross-linked with hydrogen bonds to create microfibrils. Cellulose has high tensile strength and insoluble in water, the fibres also allow water and other substances to pass freely into and out of plant cells.
Role of glycoproteins in cell–cell recognition
a. Glycoproteins are proteins with one or more short chains of sugar (oligosaccharides) projecting outward from the membrane and carbohydrate is attached to protein (glycoprotein) or to lipid (glycolipid). Interactions between the oligosaccharides and carbohydrate-binding proteins allow cell-cell recognition. Glycoproteins at the membrane distinguish self from nonself binding the tissue cells to multicellular organisms.
b. Glycoproteins on the surface of red blood cells determine a person’s ABO blood type. Your blood cells can have 2 possible types of glycoproteins on their plasma membranes and they are called antigens because their presence can trigger the immune system. The white blood cells recognise self from non-self, immune system is triggered by the presence of the protein (A/B) that they do not have.
Hydrophobic properties of lipids
lipids include fats, oils, waxes and steroids. lipid molecules contain many non-polar covalent bonds between carbon and hydrogen (hydrocarbons). this means that lipids dissolve well in non-polar solvents but do not dissolve well in water (hydrophobic).
Formation of triglycerides and phospholipids by condensation reactions
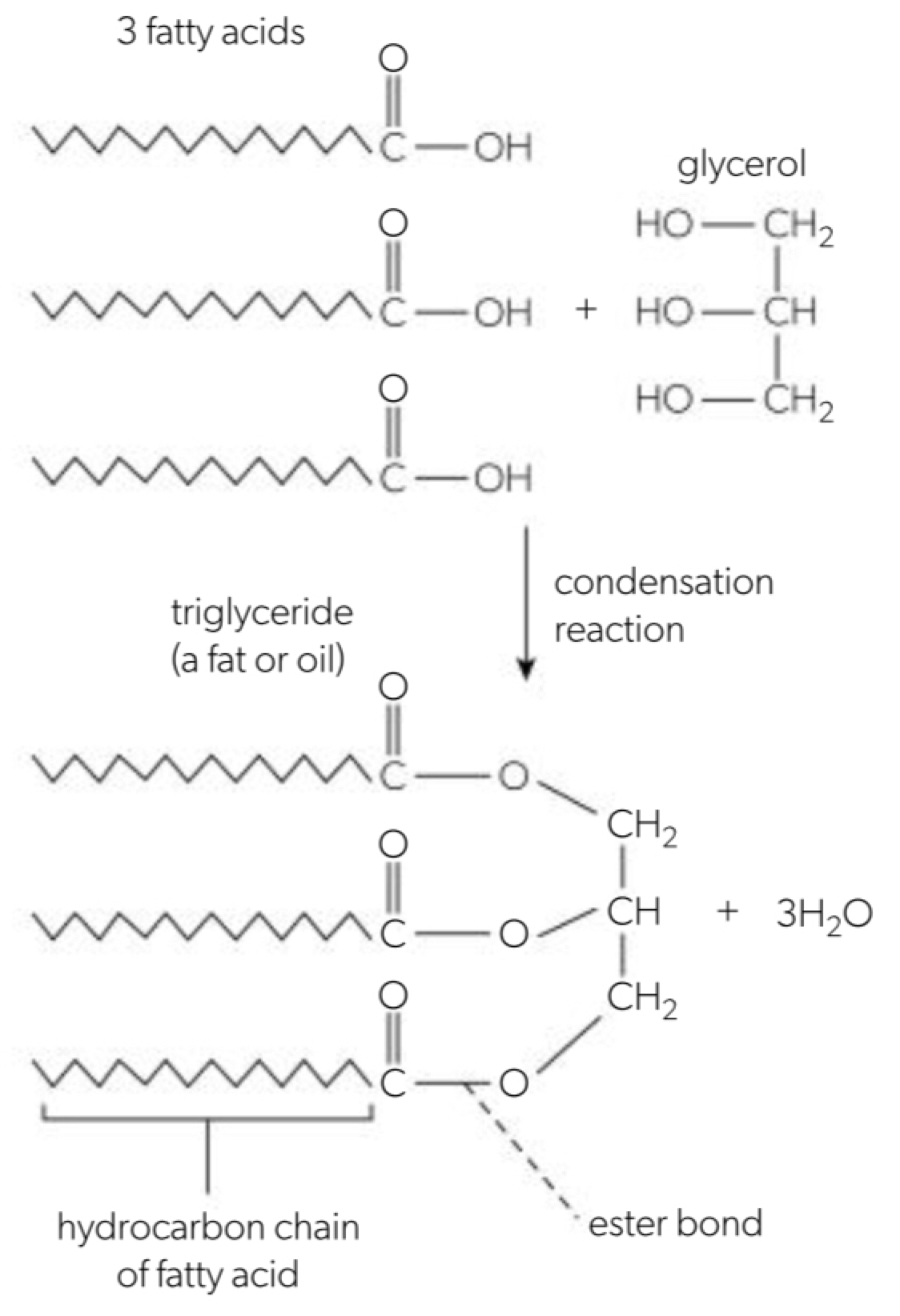
a. triglycerides are made by combining 3 fatty acids and one glycerol molecule through a condensation reaction
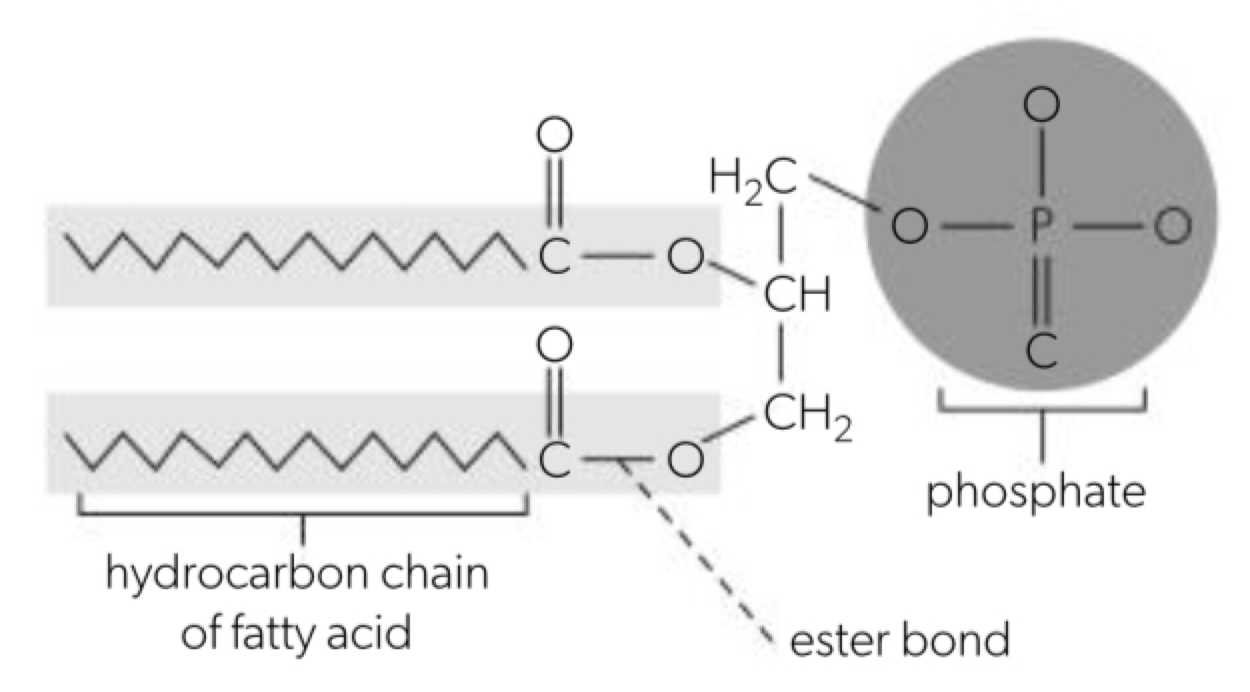
b. phospholipids are formed when a phosphate group replaces one of the three fatty acids
c. formation of a phospholipid or triglyceride: hydroxyl group of the glycerol aligns with the carboxyl group of the fatty acid number 1 and a condensation reaction will occur. a new covalent bond is formed, fatty acid 1 and the glycerol molecule unite. the second fatty acid aligns with the second hydroxyl and undergoes condensation. a new covalent bond is formed and the phosphate group aligns with the third oh group and undergoes condensation reaction. 3 new covalent bonds are formed and a phospholipid/triglyceride are formed.
Difference between saturated, monounsaturated and polyunsaturated fatty acids
a. saturated fatty acids contain single bonds between the carbons meaning all the other carbons bonded are to hydrogen (except for carboxyl group) (saturated with hydrogen) they have a relatively high melting point and are solid at room temperature, hydrogenation may saturate fatty acids making oils solid
b. monounsaturated fatty acids are unsaturated as whether one carbon in the hydrocarbon chain is not bonded to the max (less than one) hydrogens (less than one unsat). triglycerides containing monounsaturated fatty acids have lower melting point than saturated fatty acids and are liquid (oil) at room temp. some animals and many plants store energy in the form of oils.
c. polyunsaturated fatty acids have more than one double bond in the hydrocarbon chain (location and number may vary). triglycerides composed of polyunsaturated fatty acids also have a relatively low melting point and are liquid (oil) at room temp (commonly what are commonly known as oils form).
d. the double bond alters the kink in the hydrocarbon chain so the fats do not pack together neatly so they change from solid to liquid at lower temperatures than other fats and oils are a source of energy in their seeds (commonly unsaturated fatty acids) the energy from stored fats is used by the germinating seedling until it can begin photosynthesis (amongst this is why unsaturated fats tend to generally have a much lower melting point (liquid at body temperature) where a constant supply of energy in the form of food to generate heat to the sea fat in the store in adipocytes as liquid droplets and can be broken down into ATP where it used to power cellular processes.
Triglycerides in adipose tissues for energy storage and thermal insulation
a. adipose tissue is composed of cells that store fat in the form of triglycerides. the quantity of triglycerides stored is determined by an organism’s calorie intake compared to the calories burned. in condensation reactions that form triglycerides, water is produced when an organism eats food and has more calories than the organism will use, the triglycerides are then stored as fat for energy during starvation or when sufficient foods are not available. hydrolysis breaks the stored triglycerides down throughout the body’s tissues and provides (glycerol and fatty acids) energy as needed by forming droplets in the cytoplasm.
c. triglycerides are useful for long-term energy storage because they have more energy than carbohydrates when used in respiration, adipose tissue can be stored in a nearly stable state so energy is not lost over time, they are more efficient energy stores as they release more h and energy for atp than that released from carbohydrates when used in respiration. adipose tissue can be stored in a nearly stable state so energy is not lost over time, they are more efficient energy stores as they release more h and energy for atp than that released from carbohydrates when used in respiration. the body will break it as potential not to be lost. the body then becomes fats and oils used for hair conditioners as they function as thermal insulators reduce loss of body heat to the environment.
Formation of phospholipid bilayers as a consequence of the hydrophobic and hydrophilic regions
a. the phosphate group in a phospholipid molecule creates a polar end (the phosphate group) and a non-polar end (two hydrocarbon chains) that is called amphipathic. in an aqueous solution, the phospholipids form a double layer called a bilayer in which the hydrophobic fatty acid tails orient in towards each other in order to keep away from the aqueous solutions inside and outside the cell and the polar phosphate group arrange themselves to be outside of the bilayer. this is the foundation of the plasma membrane.
Ability of non-polar steroids to pass through the phospholipid bilayer
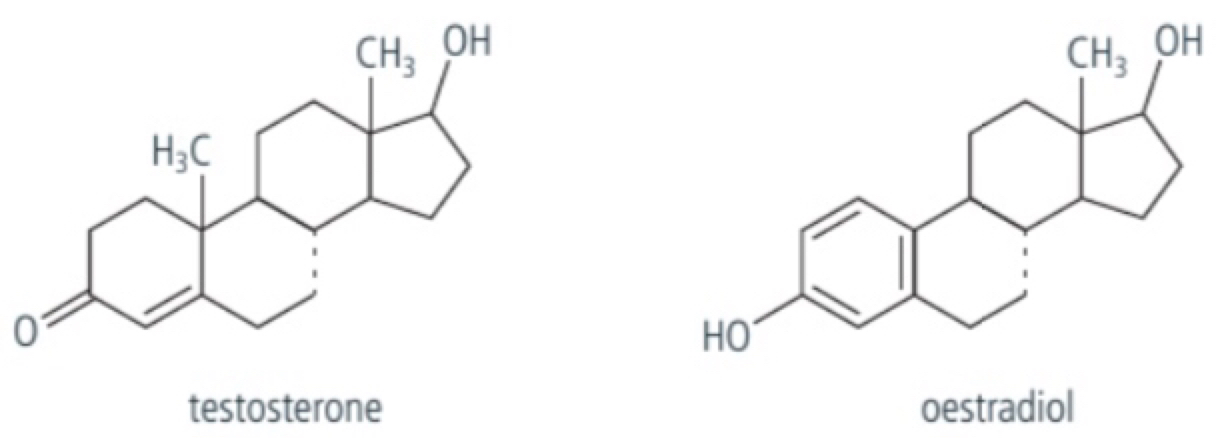
a. hormones are chemical messenger molecules and include a group called steroids which are made from cholesterol which is primarily a hydrocarbon molecule and so are hydrophobic, they have 4 fused rings of carbon atoms (3 with 6 carbon atoms and one with 5). examples include estradiol and testosterone which are hydrophobic hormones and are soluble through the lipid bilayer of cells
Generalized structure of an amino acid
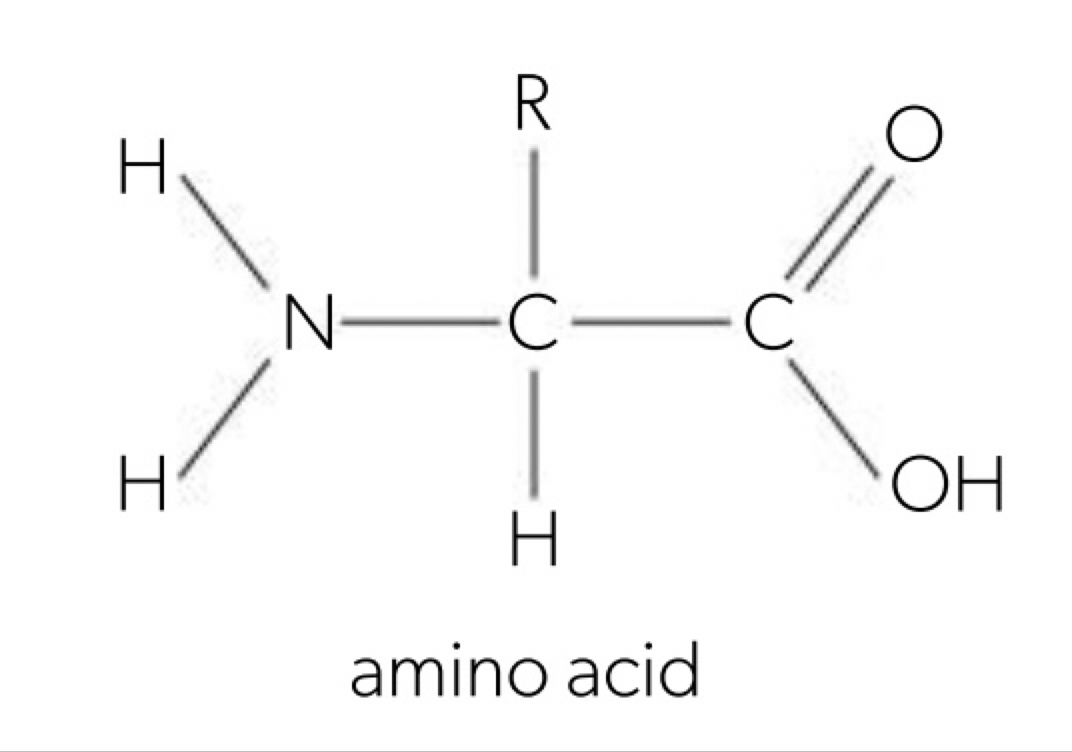
a. amino acids have a central carbon atom with a hydrogen atom, amino group (-NH2), a carboxyl group (-COOH) and a R-group (each different amino acid have a different R-group and so determine the specific chemical properties such as polarity, charge and size).
Condensation reactions forming dipeptides and longer chains of amino acids
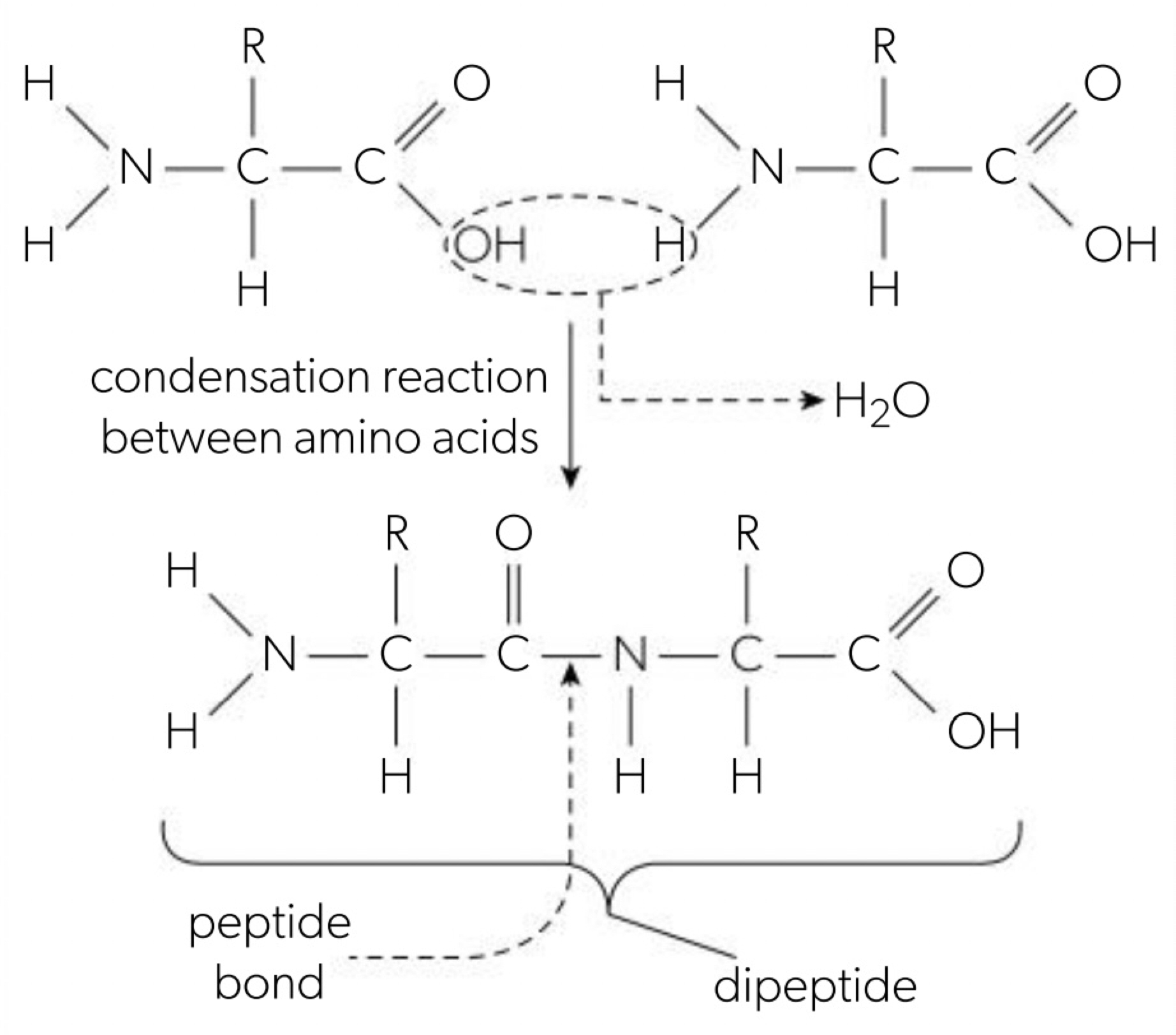
a. amino acids are linked together by condensation reactions between the amino group and the carboxyl group of 2 amino acids called peptide bond (between the carbon and nitrogen) and it forms a dipeptide. polypeptide consists of many amino acids linked by peptide bonds to the N-terminal (amino-terminal) or C-terminal (carboxyl-terminal) and a water molecule is released.
(amino acid 1 + amino acid 2 = dipeptide + water)
Dietary requirements for amino acids
a. cells can synthesise 12 of the 20 amino acids from other amino acids but the 8 we need to come from our diet called essential amino acids and cannot be synthesised.
b. a varied diet that includes different sources of protein will provide all the essential amino acids humans need. in a region diet often meat is a good source of the essential amino acids and a diet of plant-based foods lack one or more essential amino acids so combining different kinds of plant foods can provide a complete amino acid profile.
Infinite variety of possible peptide chains
a. 20 amino acids are coded for by the genetic code and the chains can have any number of amino acids (a few to thousands) in which the amino acids can be in any order creating an almost infinite number of combinations (20^n) (unique proteins making all the variety and diversity of life possible).
b. examples of polypeptides are: haemoglobin, keratin, pepsin, collagen, actin, isomerase, insulin
Effect of pH and temperature on protein structure
a. the function of a protein is dependent on its structure (3D shape of globular proteins) as they have to precise three-dimensional shape resulting from intramolecular bonds between the amino acids including hydrogen bonds.
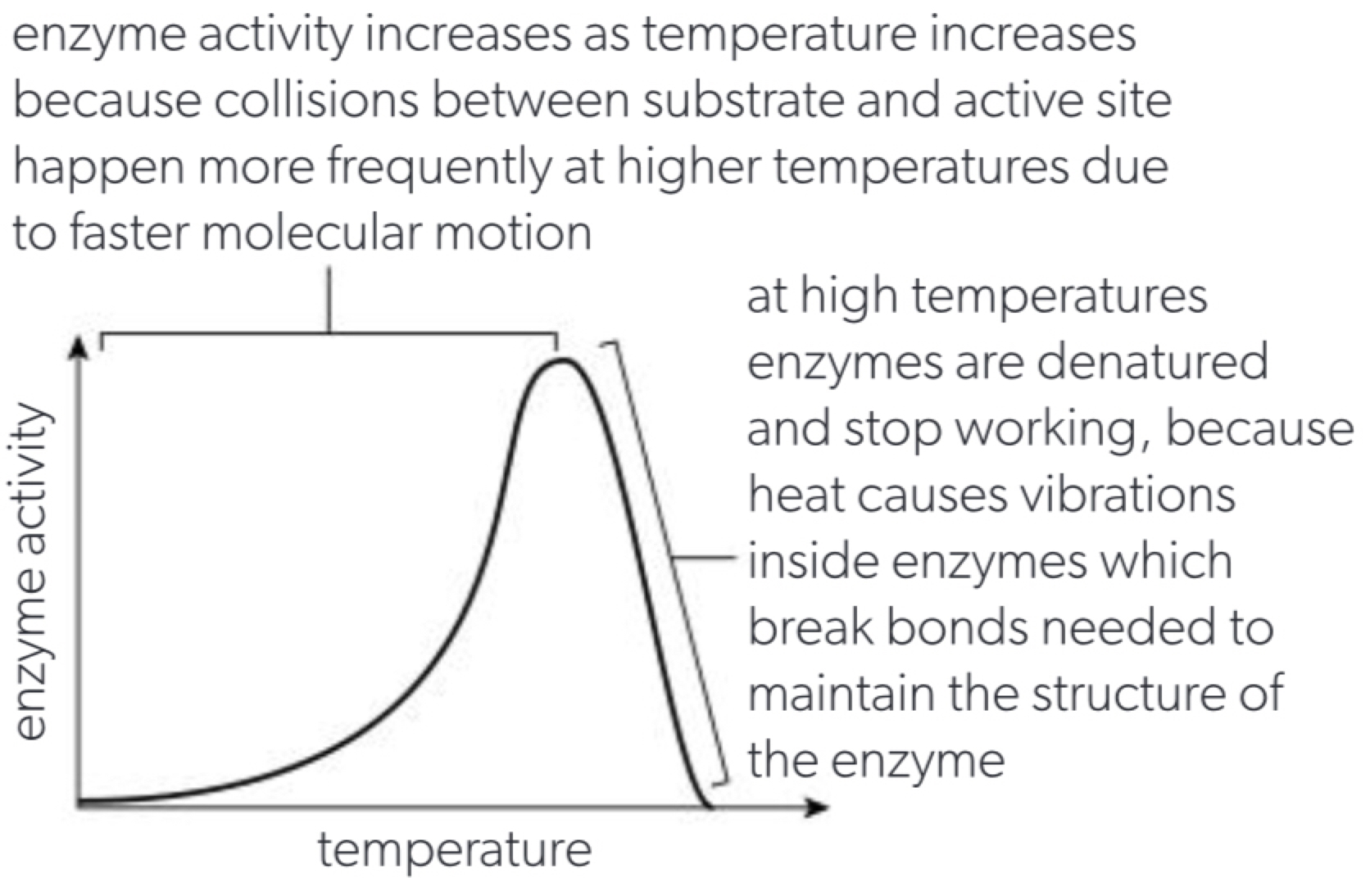
b. at higher temperatures than their physiological optimum puts a lot of stress on the relatively weak hydrogen bonds due to increased molecular motion the result is that the protein loses its shape and results in all of its function called denaturation, as long as the bonds between the amino acids remain intact the protein will reform to its normal shape and function if returned to its optimum temperature.
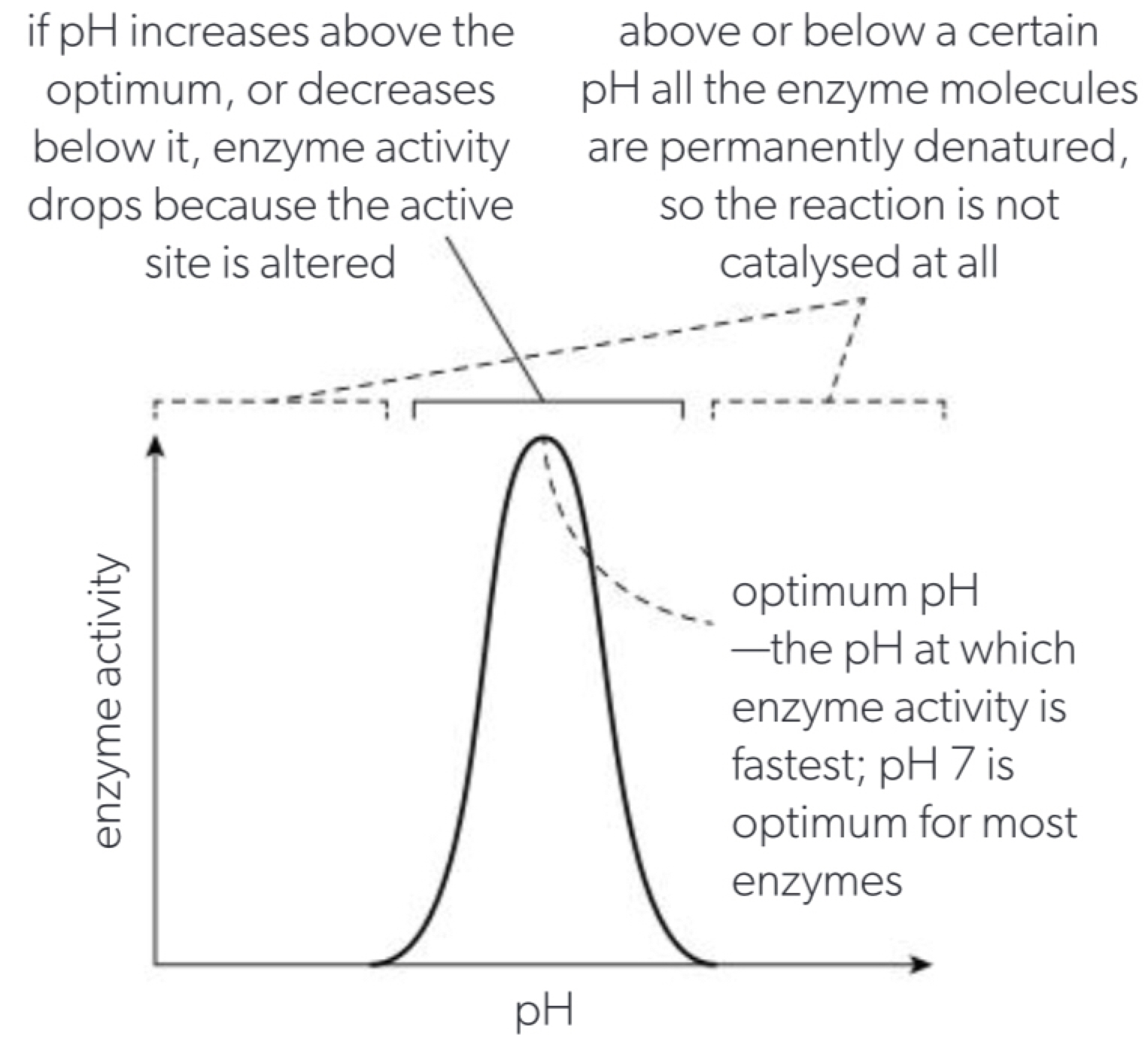
c. every protein also has an optimum pH at which its conformation is normal, if the pH is increased or decreased as far away from the optimum denaturation will occur as H+ ions or OH- ions can prevent normal hydrogen bonding that is usually reversible as long as the underlying polypeptide chain is not damaged.
Chemical diversity in the R-groups of amino acids as a basis for the immense diversity in protein form and function
a. there are 20 different R-groups for the 20 different amino acids. all amino acids have a carboxyl and amino group bonded to a central carbon, when in a neutral aqueous solution both the amino and carboxyl groups ionise because the carboxyl group acts as an acid and donates hydrogen ion while the amino group acts as a base and accepts the hydrogen ion, this results in the carboxyl group having a net negative charge and the amino group a net positive charge so the R-groups contribute to the unique structure characteristics.
b. non-polar R-group is a hydrocarbon only (a), polar R-group contains elements that form a polar covalent bond (O, H, S) (c), polar due to ionisation charge (d); R-group acts as an acid (e) and polar due to ionisation charge (f); R-group acts as a base (g) hydrophobic + polar or charged.
Impact of primary structure on the conformation of proteins
a. DNA contains the genetic code for proteins: controlling the number and sequence of amino acids in a given protein chain as they joined together by peptide bonds called the primary structure of the protein. each named protein is predictable despite their complexity because the gene will be translated into the same sequence of amino acids and it that then determines the three-dimensional shape it undertakes. named proteins is precise and repeatable because each time the gene is used the same sequence of amino acids and it that then determines the three-dimensional shape it undertakes.
b. primary structure: the primary structure of proteins is the linear sequence of amino acids in its polypeptide chain.
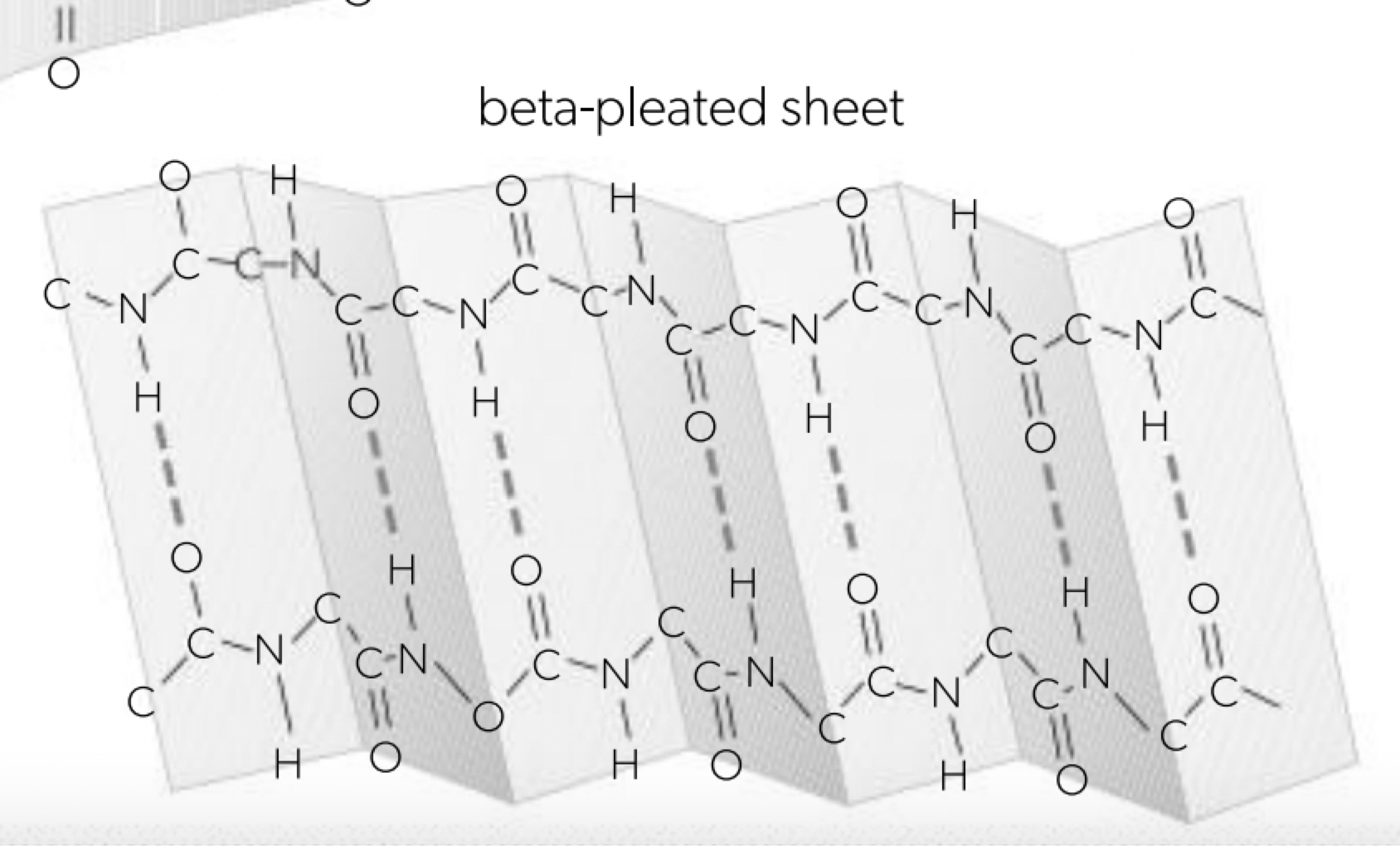
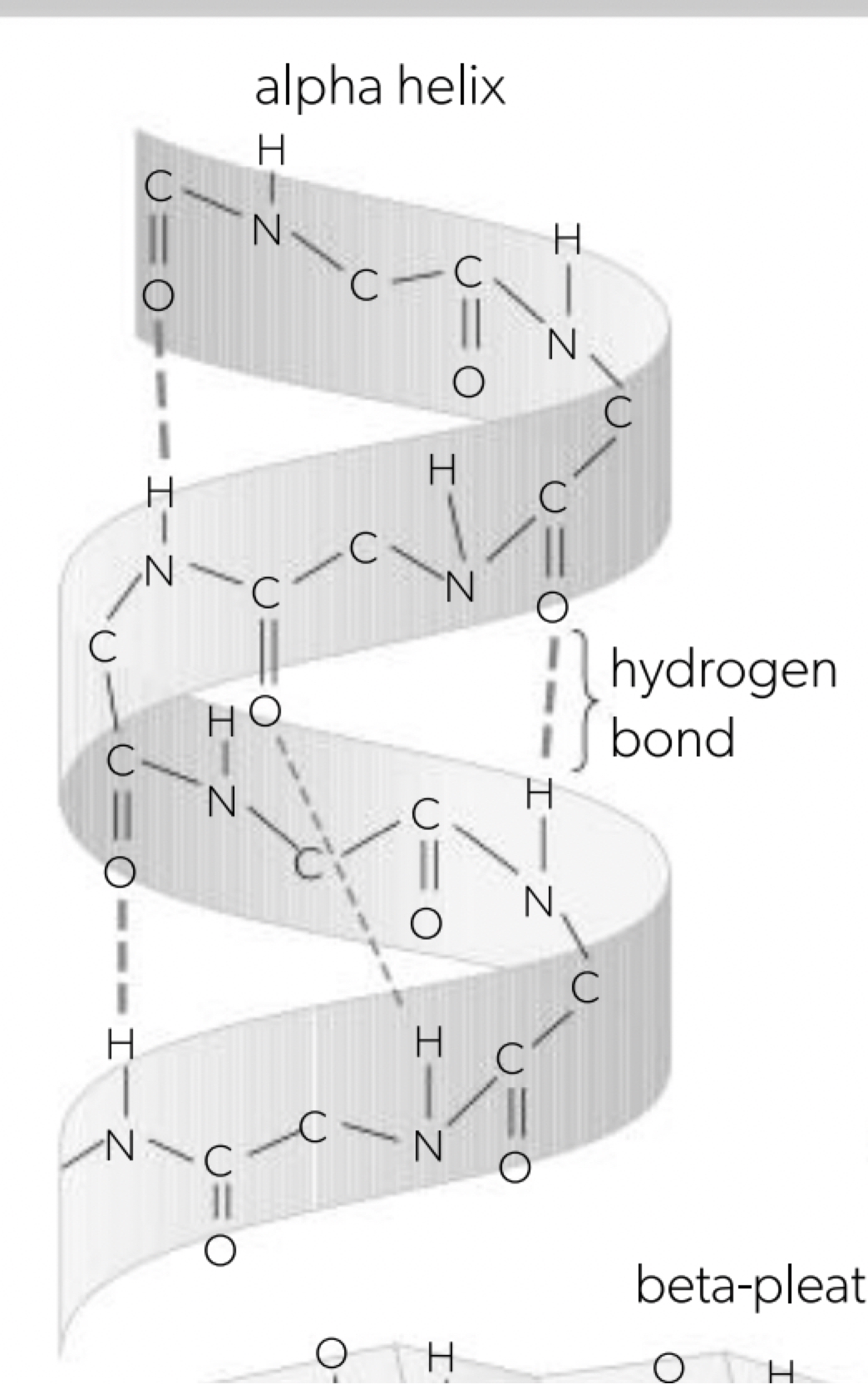
Pleating and coiling of secondary structure of proteins
a. once primary structure has been established the molecule will begin shaping itself, the charges (amino or carboxyl group) result in either beta-pleated sheet or alpha helix structure held together by hydrogen bonding (the R-groups not involved) all the hydrogen bonding is between the adjacent amino and carboxyl residues. where proportions of repeats the protein molecule will take on a shape that maximises the number of hydrogen bonds.
b. secondary structure is the regular repeating patterns that form when the polypeptide chain folds. these patterns arise from interactions between the atoms in the backbone of the protein. alpha (α) helix: coiled spring-like, beta (β) pleated sheet: folded sheet-like.
Dependence of tertiary structure on hydrogen bonds, ionic bonds, disulfide covalent bonds and hydrophobic interactions
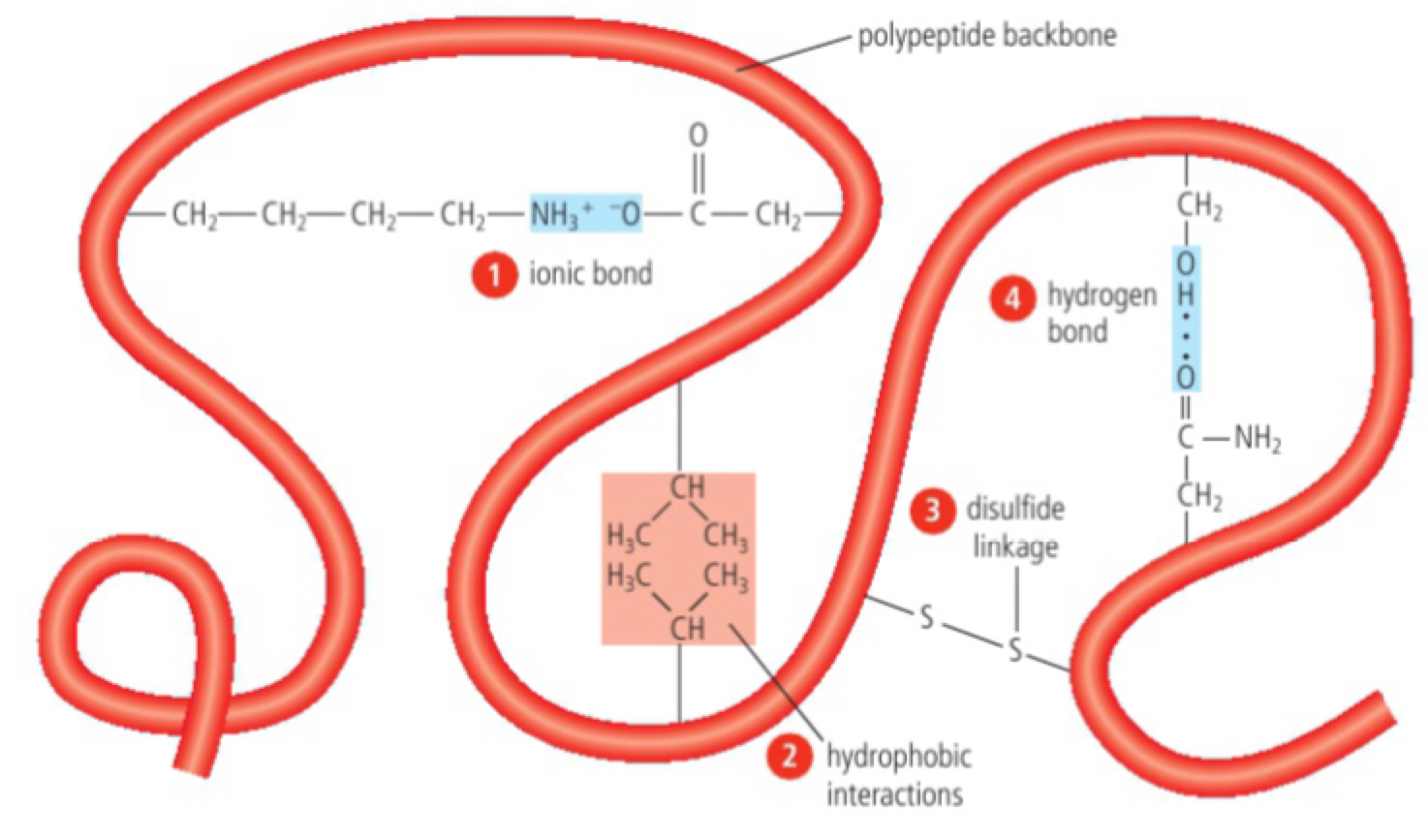
a. tertiary structure - the three dimensional folding of a polypeptide chain that is stabilised by the separate interactions between the side groups (R groups) of amino acids.
b. hydrogen bonds (covalent, requires) will form between amino acids which do not lose the hydrogen atom but become negatively charged in an amino the oxygen will bond
c. hydrogen bonds becoming positively charged. It will be oppositely charged to oxygen. The new form is ionic bond.
d. non-polar amino acids (hydrophobic) fold inwards, the interior of the polypeptide, the interaction is hydrophobic interactions.
e. pairs of cysteine amino acids form covalent bond between themselves within the polypeptide. Cysteine's -SH group donates sulfur atom bonded to hydrogen shown to other non-adjacent cysteine amino acids get close to each other. The H hydrogen can be removed and the sulfur atoms become covalently bonded to each other forming the disulfide bond (strongest in the polypeptide).
f. polar amino acids will form hydrogen bonds but only are only found near the exterior of the polypeptide because their hydrophilic properties (most common).
Effect of polar and non-polar amino acids on tertiary structure of proteins
h. proteins with polar amino acids near the edge and other close to the outside of the cell. This arrangement stabilises it in aqueous solutions. (globular protein with hydrophilic core).
i. proteins with non-polar amino acids on the outer surface are attached to the non-polar core of the membranes so they become integrated membrane proteins.
j. the distribution of polar and non-polar amino acids determines the folding structure of proteins and also where they are located in the cells.
Quaternary structure of non-conjugated and conjugated proteins
a. specific three-dimensional arrangement of multiple polypeptide chains (subunits) in a single functional protein
b. conjugated proteins: proteins that contain non-protein (non-polypeptide) component called prosthetic group such as a metal ion or carbohydrate. An example of this is the haemoglobin which consists of four individual polypeptide chains (2 α chains and 2 β chains) and is associated with a non-protein component called haem, which is a molecule with iron in the centre. Responsible for binding to oxygen in the lungs to transport it in the body.
c. non-conjugated proteins: proteins that consist only of amino acids linked together by peptide bonds. Example: molecule insulin, collagen, insulin is composed of two polypeptide chains linked by disulfide bonds. Collagen is made of three polypeptide chains twisted into a single triple helix, it provides structural support with high tensile strength.
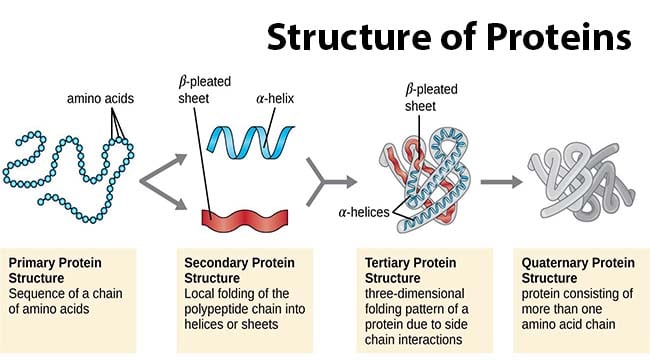
Relationship of form and function in globular and fibrous proteins
a. globular proteins are compact, spherical proteins that are typically soluble in water, their structure results from the folding of the polypeptide chains into a tightly packed 3-dimensional shape.
b. they have a compact and irregular arrangement, transport proteins include an example a globular protein: insulin is released into the bloodstream. Other kinds of proteins bind near which helps the controls in all body reactions. Insulin binds to insulin receptors on the cell surface which have a complementary shape to hormone, when they bind, signal pathways occur, whole activity in the cell opens channels in the plasma membrane that allow glucose to enter the cell.
c. fibrous proteins are structural proteins that have an elongated shape (long and narrow). They are largely composed of repeating structures that make them very strong, provide structural support and stability to cells and tissues. For example, collagen made of chains, must together to form long and tough triple helix structure. The fibres are strong and flexible, resisting forces without breaking, providing structural support to tissues (long, regular, geometric fibrous shapes).
Enzymes as catalysts
a. catalysts speed up chemical reactions without being changed themselves. globular proteins that function as biological catalysts, made by living organisms. they do this by lowering the activation energy required for the reaction. many reactions that are useful to living organisms would be too slow without an enzyme acting as a catalyst; this would slow down life processes such as cell respiration, digestion, photosynthesis and growth.
Role of enzymes in metabolism
a. metabolism is the complete network of interdependent and interacting chemical reactions occurring in living organisms. each chemical reaction is controlled by a specific enzyme and because of its specificity many different integral are required in each living organism, and control over metabolism can be exerted through these enzymes (interdependence).
Anabolic and catabolic reactions
a. anabolic reactions use energy to build complex organic molecules from simple organic molecules by condensation reactions. examples include photosynthesis, protein synthesis and glycogen formation.
b. catabolic reactions break down complex macromolecules into monomers by hydrolysis. examples include digestion and the oxidation of substrates in respiration.

Enzymes as globular proteins with an active site for catalysis
a. enzymes are globular proteins, each enzyme has an active site on its surface where catalysis occurs. the shape of the active site exactly matches the shape of one particular substrate. the active site is composed of a few amino acids brought together by the folding of the polypeptide chain, but it has a three-dimensional shape and chemical properties that cause the substrate to attach via the active site and not each other and they are brought together by the folding of the polypeptide chain in the 3-dimensional structure of the enzyme (tertiary structure is important).
Interactions between substrate and active site to allow induced-fit binding
a. the first stage of enzyme catalysis is binding of the substrate to the active site. they interact with each other during the binding process, before becoming attached to fit the active site, the substrate induces reversible changes in the active site and the active site induces changes in the substrate that facilitate its conversion to the products. the changes affect bond angles, bond lengths and conformation in the substrate and active site achieving complementarity through mutually induced changes. this is called induced fit binding.
b. the original model was the lock and key model of the active site of the enzyme is the complementary shape of the substrate and so fits the substrate precisely. (enzyme lock, substrate = key)
Role of molecular motion and substrate-active site collisions in enzyme catalysis
a. molecules dissolved in water are in constant motion and they randomly collide. the initial fit is between a substrate molecule and the active site of an enzyme called a successful collision, the shape and chemical properties of the active site are complementary to those of the substrate, so when they do collide, enzyme and substrate bind in appropriate orientation. the substrate is chemically attracted to each other and fit together. unlike with large substrate molecules or enzymes by being embedded in membranes, allowing more efficient joining of substrate and active site.
Relationships between the structure of the active site, enzyme–substrate specificity and denaturation
a. the structure of the active site is very precise and attractive structure. the substrate is complementary to it so binding occurs; molecules other than the substrate are not attracted to the active site, making enzymes substrate-specific.
b. enzyme-substrate denaturation, altering enzyme structure at the active site (can temperature/pH reach) so successful collisions can no longer occur. (denaturation changes the tertiary structure, interfering formation of a protein and so create interactions between amino acids within the protein. denatured state follows recurring 3-dimensional shape to change)
Effects of temperature, pH and substrate concentration on the rate of enzyme activity
a. the rate of enzyme activity is how frequently the reaction is catalysed which can be measured by the amount of substrate used up or the amount of product formed per unit time. (units: molar min^-1)
b. temperature affects the rate of molecular movement and therefore the number of substrate + enzyme site collisions per unit time but can also cause denaturation.
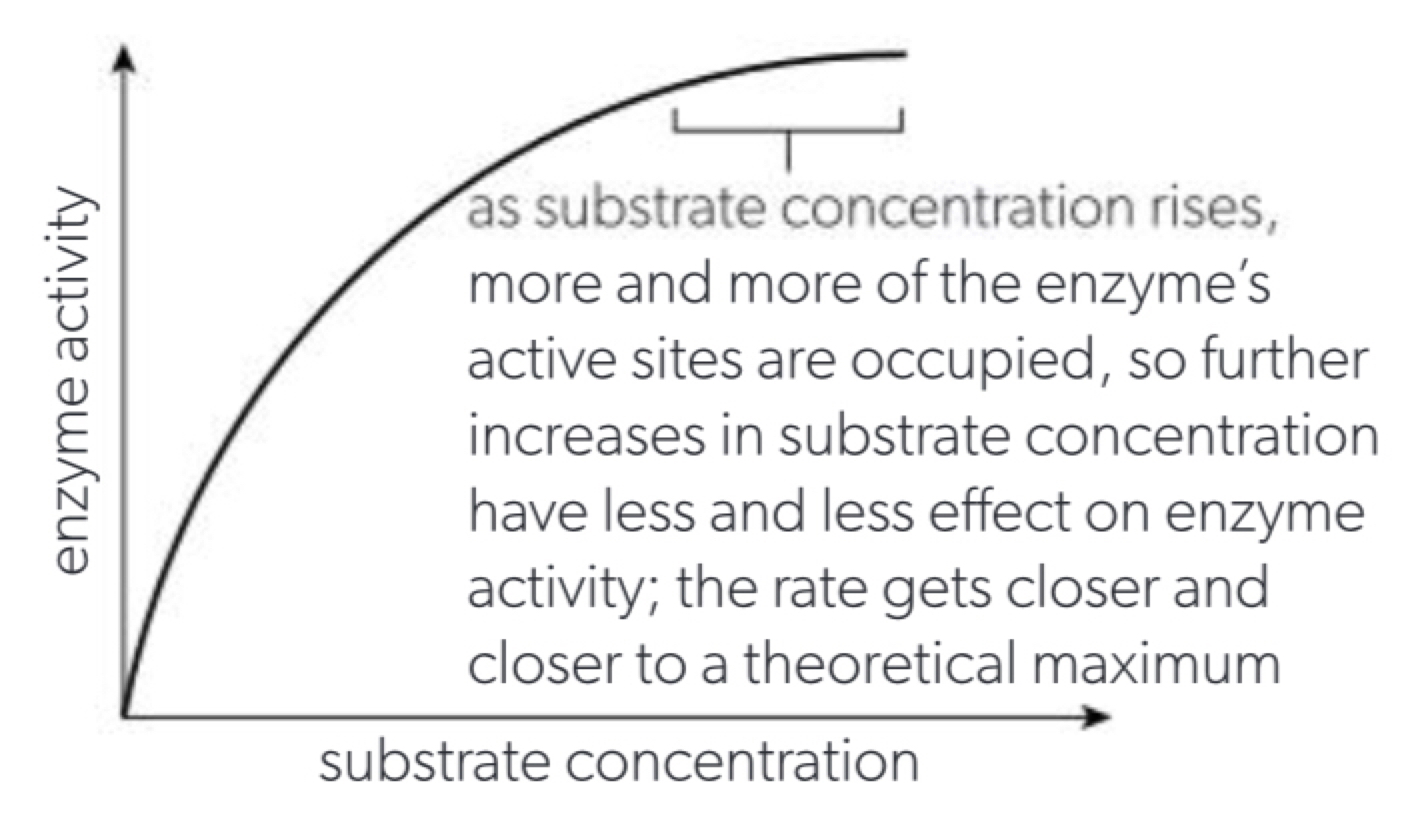
c. substrate concentration affects the number of substrate + active site collisions per unit time but since the enzyme number remains constant and more active sites are occupied (curve not hydrogen bonds)
d. pH affects the ionisation of room and COOH and the NH2 groups which alters enzyme conformations and causes denaturation. If pH is above or below the optimum enzyme activity drops because the active site is altered
Measurements in enzyme-catalysed reactions
f. the rate of reaction catalysed by an enzyme can be measured by measuring either the quantity of substrate or product per unit time (mass or volume)
Effect of enzymes on activation energy
a. to convert reactants to products in chemical reactions, bonds must be broken in the reactant to get to the transition state between the breaking and making of bonds (energy level at a maximum). the amount of energy needed to reach the transition state is the activation energy of the reaction. enzymes reduce the activation energy of reactions by weakening bonds in their substrates so an alternative transition state is reached at a lower energy level enabling more molecules with sufficient energy to overcome the initial energy requirement.
Intracellular and extracellular enzyme-catalysed reactions
a. intracellular enzyme-catalysed reactions: metabolic reactions that take place within the cell and are catalysed by enzymes that are produced by free ribosomes. examples: metabolic glycolysis which takes place in the cytoplasm of the cell and breaks down glucose into 2 pyruvate molecules releasing energy and the krebs cycle which takes place in the mitochondria which produces acetyl-CoA to produce carbon dioxide, energy rich molecules and electron carriers.
b. extracellular enzyme-catalysed reactions: are metabolic reactions that take place outside the cell, and are catalysed by enzymes that are produced by bound ribosomes and secreted outside the cell by exocytosis. examples: starch and protein hydrolysis in the gut; digestive enzymes such as lysosome amylase; proteases are secreted extracellularly by exocrine cells in the pancreas to catalyse reactions.
Generation of heat energy by the reactions of metabolism
a. metabolic reactions generate heat because energy transfer during reactions is not 100% efficient and free thermal energy is converted to heat. this is often useful to living organisms as the heat warms up tissues and speeds up enzyme activity in animals and endotherms (warm-blooded animals, birds etc.) depend on heat generated by metabolism to maintain a constant body temperature. thus, increases the rate of metabolism to speed up homeostasis. (anabolic reactions or endergonic (absorb energy from chemical bonds), the energy is provided by ATP hydrolysis (which releases heat) or by hydrocarbon (anabolic uses heat than catabolic as bonds broken = energy released = dissipates as heat)
Cyclical and linear pathways in metabolism
a. metabolic reactions catalysed by enzymes often occur in sequences which are called metabolic/biochemical pathways; metabolic pathways consist of chains of reactions that convert initial substrate into end product (example: glycolysis). cyclical pathways: both the end regenerate intermediates (example: krebs reaction, lactic cycle and calvin cycle)
b. some reactions are anabolic while substances also in the cycle from intermediates other reactions are catabolic involving splitting to produce substances that exists.
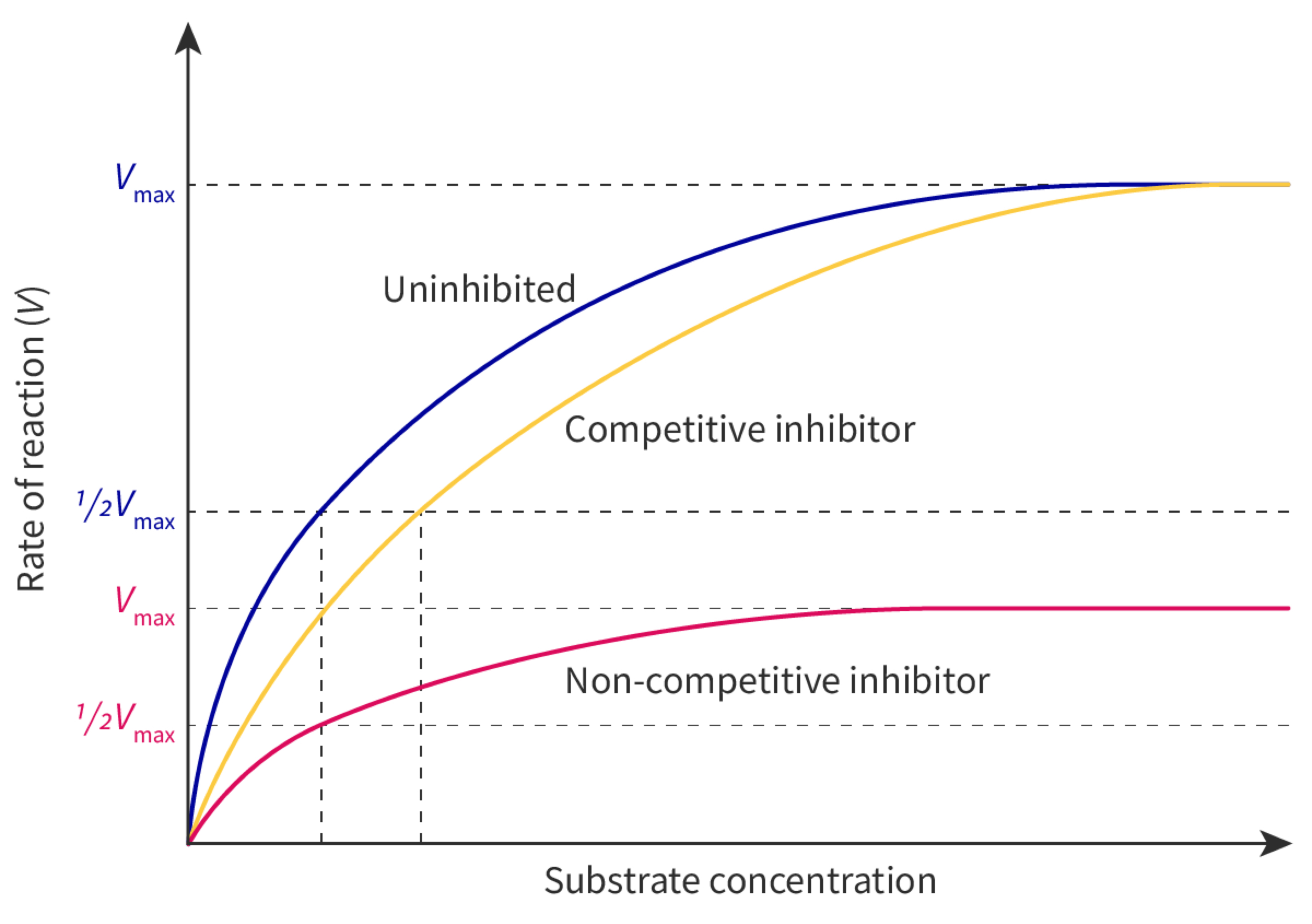
Allosteric sites and non-competitive inhibition
a. all enzymes have an active site on their surface in which the substrate binds many enzymes also have an allosteric site in which a different substance binds. the binding is specific and reversible. the substance that binds to the allosteric site is an enzyme inhibitor. other inhibitors are substances that bind to enzymes and slow down or block enzyme activity when an inhibitor binds to the allosteric site it causes interactions within the enzyme that change its conformation and the chemical properties of the active site are altered. when the inhibitor dissociates from the allosteric site the enzyme returns to its previous conformation and can resume catalysis.
b. because of their chemical differences, the inhibitor does not bind to the active site and the substrate does not bind to the allosteric site. they do not therefore compete with each other so enzyme inhibition using an allosteric site is non-competitive. the substrate cannot prevent binding of the inhibitor, so increasing the substrate concentration cannot overcome the inhibition, even at very high substrate concentrations.
Competitive inhibition as a consequence of an inhibitor binding reversibly to an active site
c. competitive inhibition is a consequence of an inhibitor binding reversibly and similarly to the substrate, both of them can bind to the active site. when inhibitor is bound, the substrate cannot bind and vice versa, binding of the inhibitor is reversible and competitive inhibitors and substrate compete dynamically for the active site, with random molecular movement determining which binds when so it becomes random; the relative concentrations of the substrate or inhibitor therefore determine the chance of each binding.
d. statins are competitive inhibitors of HMG-CoA reductase. this enzyme catalyses the conversion of HMG-CoA(coenzyme A) into mevalonate, the rate-limiting step in the metabolic pathway used to synthesise cholesterol in liver cells. so statins bind to the active site of the enzyme, preventing binding of the substrate and blocking synthesis of cholesterol which helps to lower cholesterol levels reducing the risk of atherosclerosis/ coronary heart disease.
Regulation of metabolic pathways by feedback inhibition

f. metabolic pathways convert an initial substrate to an end product via a series of intermediates and intermediates are converted to the next step by enzyme-catalysed reactions. the end product is useful and must be produced at the appropriate rate so that accumulation is not out which can be achieved by end-product inhibition. the product of the last reaction in the pathway inhibits the enzyme that catalyses the first reaction through non-competitive inhibition meaning that it is reversible and the end product binds at the allosteric site so the substrate can bind again. this means that there is excess of the end product, whole pathway is switched off and intermediates do not build up; and if the levels of end product falls more of the enzymes will be working to catalyse the first reaction and products inhibition is removed (essential amino acid).
g. an example of this is the inhibition of threonine deaminase by isoleucine. it ensures that supplies of isoleucine are replenished when the amino acid has been used up in protein synthesis, but surpluses do not build up.
Mechanism-based inhibition as a consequence of chemical changes to the active site caused by the irreversible binding of an inhibitor
i. some inhibitors binding irreversibly to the active site of a specific enzyme by forming a covalent bond. the inhibitor-enzyme complex is stable and the enzyme can never again function as a catalyst; the reaction is the same as the normal substrate but the enzyme is permanently changed so that it cannot catalyse any more reactions. these are called mechanism-based irreversible enzyme inhibitors.
j. an example is the antibiotic penicillin which binds covalently with serine in the active site of transpeptidases (the enzyme that cross-links peptidoglycan in cell wall in gram-positive bacteria). the cell becomes weak and the bacterial cell target dies because cells do not have cell wall to save strain and bacteria become resistant to penicillin because of substitution mutations; such changes in the amino acid sequence of transpeptidases meaning that penicillin can no longer bind to active site; another example is another antibiotic called penicillamine which attaches to methionine structure of penicillin inhibiting specific targets (to prevent structural make-up of proteins was changed).
ATP as the molecule that distributes energy within cells
a. the basic energy currency in all cells is the nucleotide ATP (adenosine triphosphate). It has the base adenine, ribose and three phosphate groups. Because the phosphate groups are negatively charged, they repel each other resulting in an unstable covalent bond between the 2 called a high-energy bond. These unstable bonds hold large amounts of chemical energy and are easily broken by hydrolysis under a enzyme (energy-releasing).
b. the properties that make ATP ideal as the cellular energy currency include: ATP is chemically stable in the medium pH levels typical of cells, so ATP doesn’t break down and spontaneously release energy. c. it is soluble in water so ATP can diffuse easily in cytoplasm, reaching any part of the cell fast. d. ATP is unable to diffuse through the phospholipid bilayers of membranes so the movement within cells can be controlled (no leakage out of cells). e. ATP can release a quantity of energy (by removal of the last phosphate group) that is sufficient for a wide range of actions within the cell, but with a little that is wasted by transformation to heat. f. ATP can be easily regenerated by adding a 3rd phosphate group to ADP (adenosine diphosphate)
Life processes within cells that ATP supplies with energy
a. active transport across membranes, synthesis of macromolecules (anabolism) and movement in the whole cell (eg cilia or flagellum motion) or cell components (such as chromosome during mitosis/meiosis)
Energy transfers during interconversions between ATP and ADP
a. energy is needed to synthesise ATP from ADP and phosphate - energy is required to synthesise ATP from ADP and phosphate (they are endothermic, nonreversible). this energy can come from a variety of sources including energy extracted from foods (sugar and fat) and transfer of phosphate groups from other compounds in the cell to ADP; the energy is stored during ATP synthesis and released during ATP hydrolysis is delivered to drive many tasks within a cell.
Cell respiration as a system for producing ATP within the cell using energy released from carbon compounds
a. respiration is the part of metabolism in which carbon compounds are oxidised to release energy in the form of ATP. it involves the release of energy from carbon compounds especially glucose and fatty acids (for carbohydrates, balanced eq for using other carbon-containing compounds can also be used). this is the equation for the life-sustaining process of respiration:
C6H12O6 + 6O2 → 6CO2 + 6H2O energy (stored in the high-energy molecules compound). carbon dioxide and water are waste of the reaction process. energy is released so the pathway of cellular respiration is the slow release of energy from the glucose molecules so that ATP can be produced more efficiently.the majority of cell respiration uses oxygen and produces carbon dioxide.
a. it is therefore necessary for oxygen to enter cells through the plasma membrane while carbon dioxide exits the cells together this is known as gas exchange; if gas exchange and cell respiration are interdependent: without gas exchange, cell respiration could not continue because there would be lack of oxygen and harmful excess of carbon dioxide inside the cell; without cell respiration, gas exchange could not continue because the use of oxygen and production of carbon dioxide creates the concentration gradients which are required to drive diffusion.catabolism feeds into glucose via products in respiratory substrates
a. the molecules can already enter glycolysis (as reactants) or carbohydrates through gluconeogenesis (before glycolytic products have to be hydrolysed to sugars and amino acids, then deaminated before entering the respiratory pathway, which requires ATP, lowering the net ATP produced, or into structural molecules when respiratory substrates are limited) (nuclear starvation conditions)
Differences between anaerobic and aerobic cell respiration in humans
a. aerobic respiration (glucose is broken down to pyruvate in the cytoplasm (glycolysis) producing a small amount of ATP and reduced NAD+, pyruvate enters the mitochondrion where it is broken down releasing CO2 and transferring energy to reduced NAD and FAD, electrons from reduced NAD and FAD flow through the electron transport chain in the inner mitochondrial membrane, driving ATP production through chemiosmosis; oxygen acts as the terminal electron acceptor forming water; this is the most efficient pathway, the highest ATP yield; glucose + oxygen → carbon dioxide + water)
b. anaerobic respiration (glucose is broken down to pyruvate in the cytoplasm (glycolysis) producing a small amount of ATP and reduced NAD+; to regenerate NAD+ for continued glycolysis, pyruvate is converted into lactate; this process produces ATP but allows glycolysis to continue without oxygen (anaerobic system) (glucose → 2 lactate + 2 ATP)
c. aerobic uses oxygen, higher ATP yield per each molecule of glucose, carbon dioxide and water waste products; anaerobic does not use oxygen, glucose only sugars can be used, 2 ATPs are produced per glucose, lactate is the waste product and the reaction occurs in the cytoplasm only
d. anaerobic does not use oxygen, glucose only sugars can be used, 2 ATPs are produced per glucose, lactate (lactic acid) are waste products and the reaction occurs in the cytoplasm only
Variables affecting the rate of cell respiration
a. temperature, carbon dioxide/oxygen/glucose concentration and type of cell/organism are used to measure the rate of cellular respiration - because it puts out to the respirometer where it can absorb and the amount CO2 in the air or uptake is calculated from the increase in pH or the potassium hydroxide solution which absorbs it so pressure - only to oxygen; temp must be constant since effects are passive. (v = pi r² h, rate = volume / time)
Role of NAD as a carrier of hydrogen and oxidation by removal of hydrogen during cell respiration
a. oxidation is the removal of electrons, reduction is when hydrogen/with electron is removed from a substrate (during hydrogenation), the substrate has been oxidised while reduction, when can be the gain of electrons or hydrogen to the substrate
b. dehydrogenase enzymes occur together in oxidation reactions
c. nicotinamide adenine dinucleotide (NAD) is coenzyme used by enzymes in cell respiration, it carries out oxidation and reduction (NAD is hydrogen carrier) - when hydrogen is added to NAD molecule, it is reduced and is used to transport it to oxidative phosphorylation/mitochondria electron transport chain, where it is oxidised, dehydrogenation cellular respiration is glucose is catabolised/oxidised loses hydrogen to become CO2 as this happens the reaction is gaining hydrogen atoms as it is reduced to H2O, the movement of electrons, carried by NADH, the conversion of glucose to carbon dioxide is an energy-releasing process. The reduced NAD produces more than carry the energy, role electrons and further respiration (O2/CO2) produce ATP as usable energy.
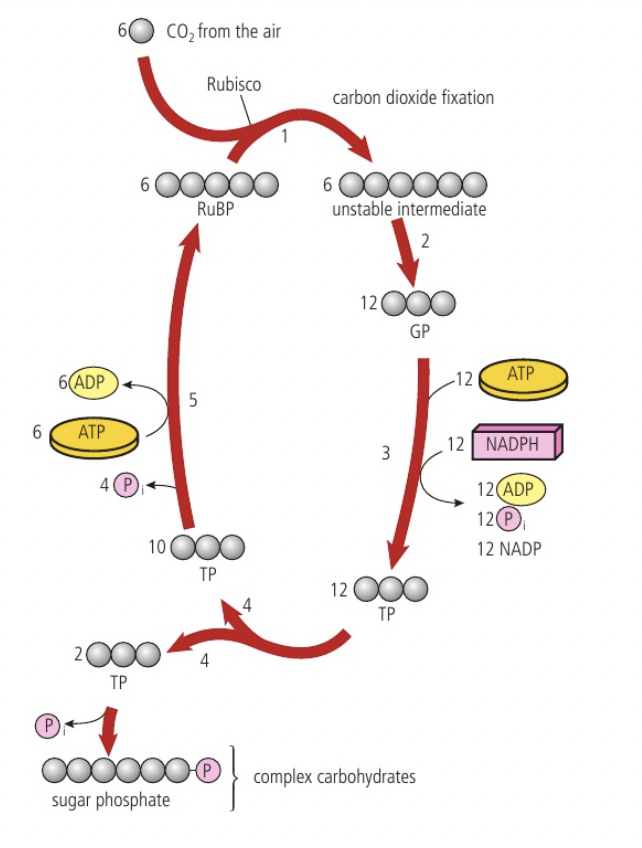
Conversion of glucose to pyruvate by stepwise reactions in glycolysis with a net yield of ATP and reduced NAD
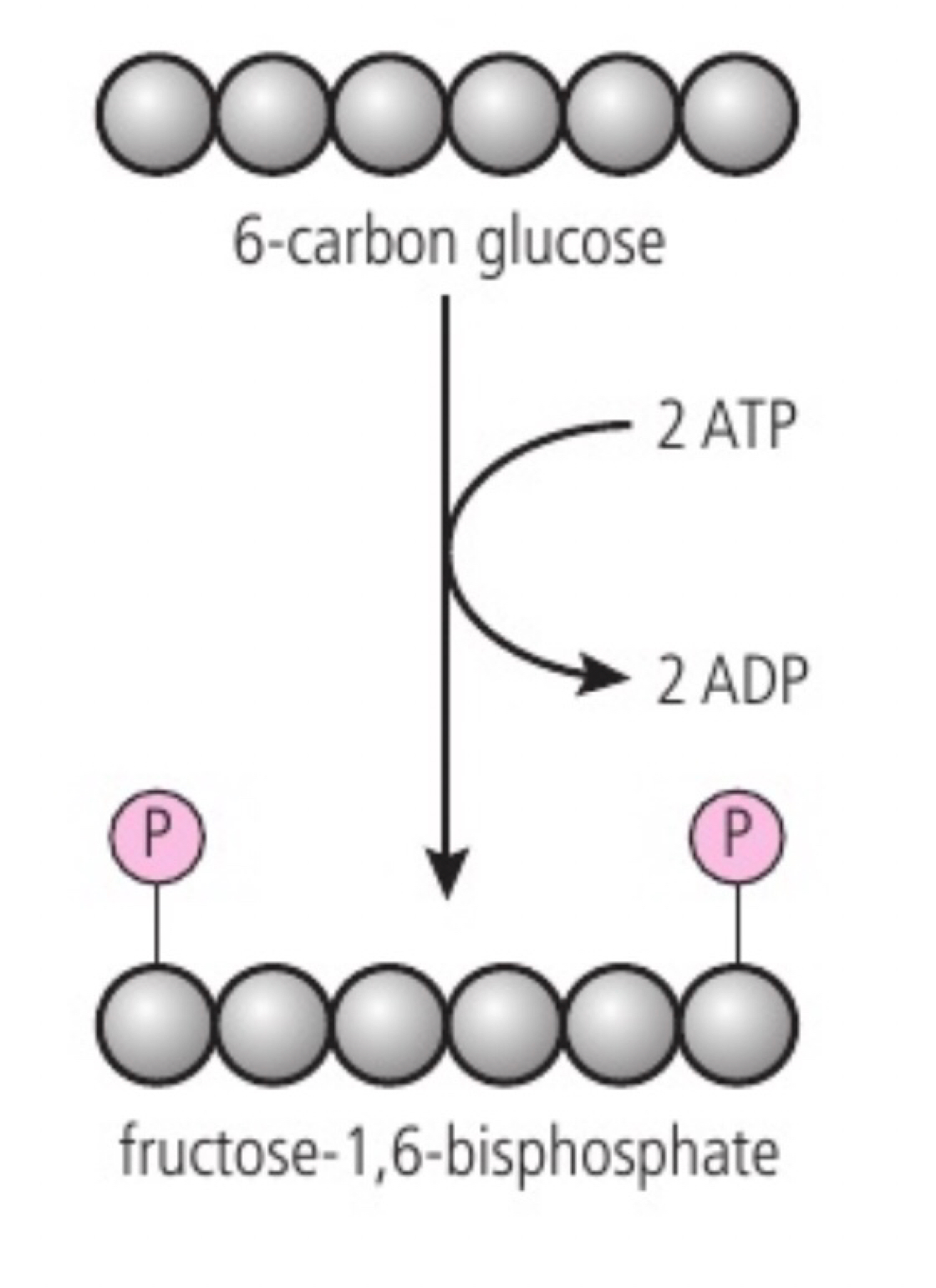
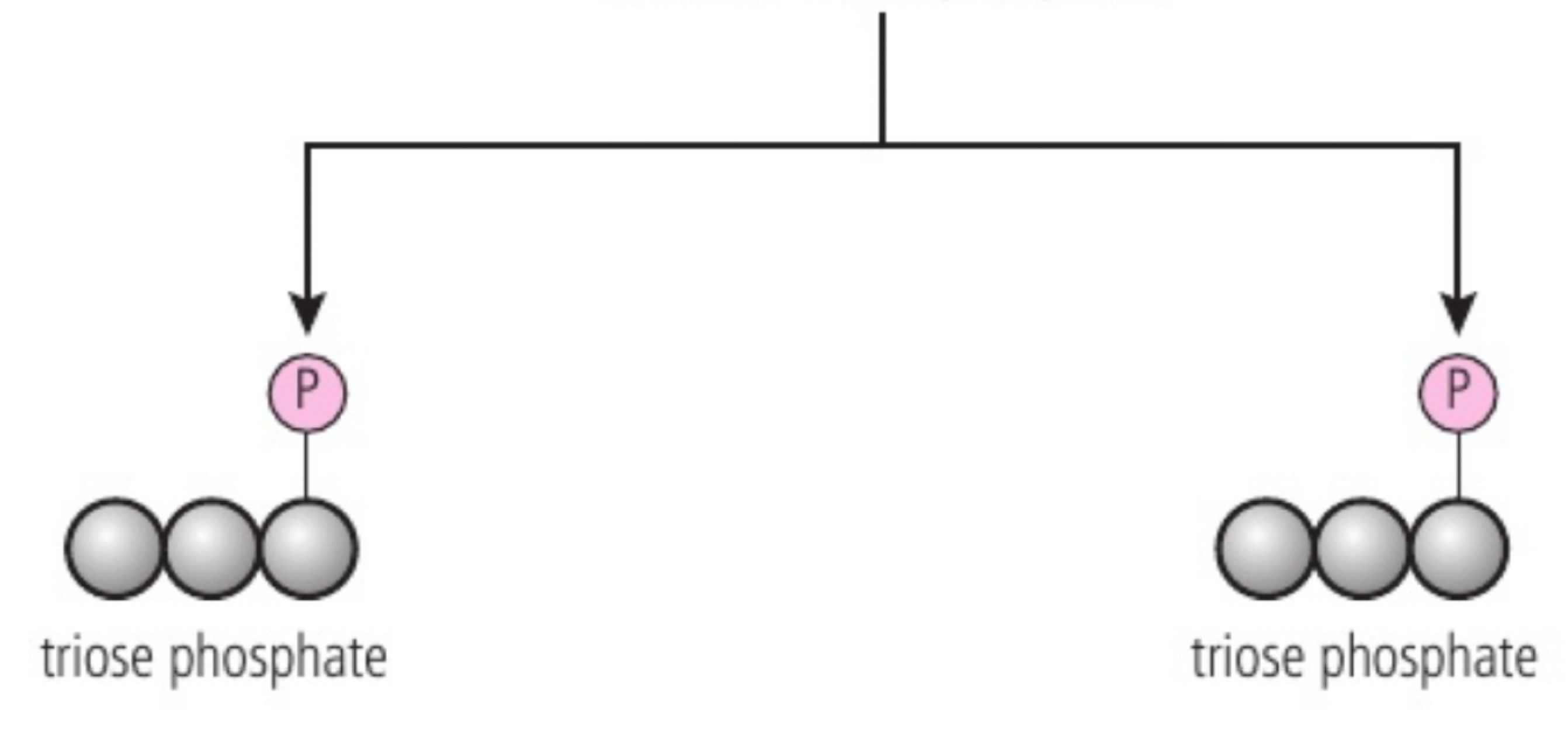
a. glycolysis is the first step of both aerobic and anaerobic respiration. it occurs in the cytoplasm and involves two main stages: (1) phosphorylation: glucose is phosphorylated twice to form fructose-1,6-bisphosphate (adding phosphate groups) it raises the energy level of molecules making them less stable and more reactive; 2 ATPs are used. (2) lysis: fructose-1,6-bisphosphate is split into two triose phosphate molecules.
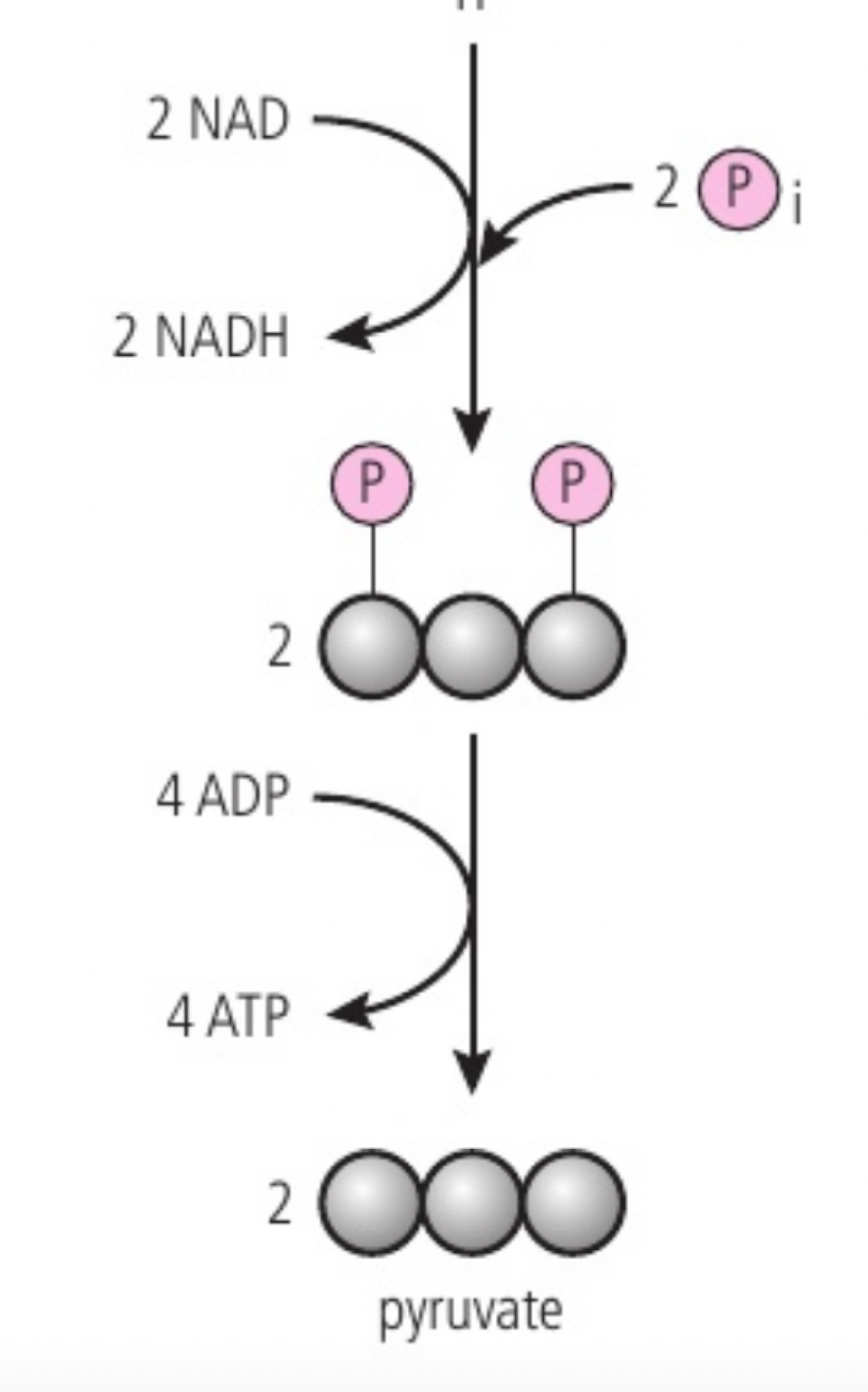
b. each triose phosphate is oxidised, as hydrogen atoms are removed and each binds to NAD+ to form reduced NAD (NADH).
c. ATP formation while 4HADH is being formed, substrate level phosphorylation is the addition of inorganic phosphate to a carbon compound, used to add an inorganic phosphate to ADP to produce ATP. this results in the formation of 4 ATP molecules, 2 NADH molecules and 2 pyruvate molecules
Conversion of pyruvate to lactate as a means of regenerating NAD in anaerobic cell respiration
a. once pyruvate is obtained, the next pathway depends on the presence of oxygen. In not great anaerobic respiration occurs as in the cytoplasm the pyruvate is converted to lactate in animals and ethanol and carbon dioxide in plants/anaerobic respiration.
b. sensation is lactate (exercise exhaustion) and not be late at higher concentrations.
c. to glycolysis can continue in a cell long as long glucose does and ADP and NAD are regenerated. The NAD is regenerated by transferring hydrogen atoms from reduced NAD to pyruvate to create lactate.
d. this regeneration allows glycolysis to continue, but it yields a net of 2 ATP molecules per glucose molecule. This is useful where large amounts of ATP are needed for a short period of time, but lactate does leave the cell or cytoplasm and blood so not sustainable for long.
Anaerobic cell respiration in yeast and its use in brewing and baking
a. the metabolic pathway is used for anaerobic respiration in yeast as in humans apart from the method used to regenerate NAD.
b. yeast converts pyruvate to ethanol by transferring hydrogen atoms from reduced NAD producing NAD allowing glycolysis to continue.
c. in yeast when baking or wine is up as the oxygen depletes the dough rises and then produces ethanol and CO2, the CO2 produces while baking.
d. yeast can be used to produce ethanol by fermentation by it being cultured in a liquid containing sugar and no oxygen so ethanol is produced and the CO2 is lost to the atmosphere.
Oxidation and decarboxylation of pyruvate as a link reaction in aerobic cell respiration
a. if oxygen is available the pyruvate produced is processed further to release more energy but the pyruvate has first to be converted into the acetyl group (CH3CO-) that can be fed into the krebs cycle.
b. this is the link reaction. First carbon dioxide is removed (decarboxylation) and then a pair of hydrogen atoms is removed (oxidation) reducing (NAD+) (which is reduced to form NADH).
c. to produce acetyl-CoA, it is then used to the krebs cycle in mitochondrion. There are also enzymes from which accept 2-carbon fragments of pyruvate to form acetyl groups.
Oxidation and decarboxylation of acetyl groups in the Krebs cycle with a yield of ATP and reduced NAD
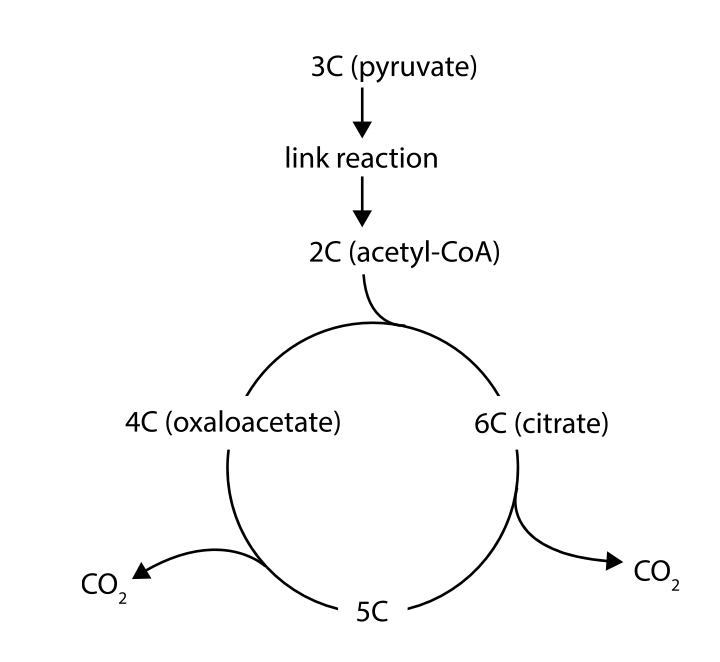
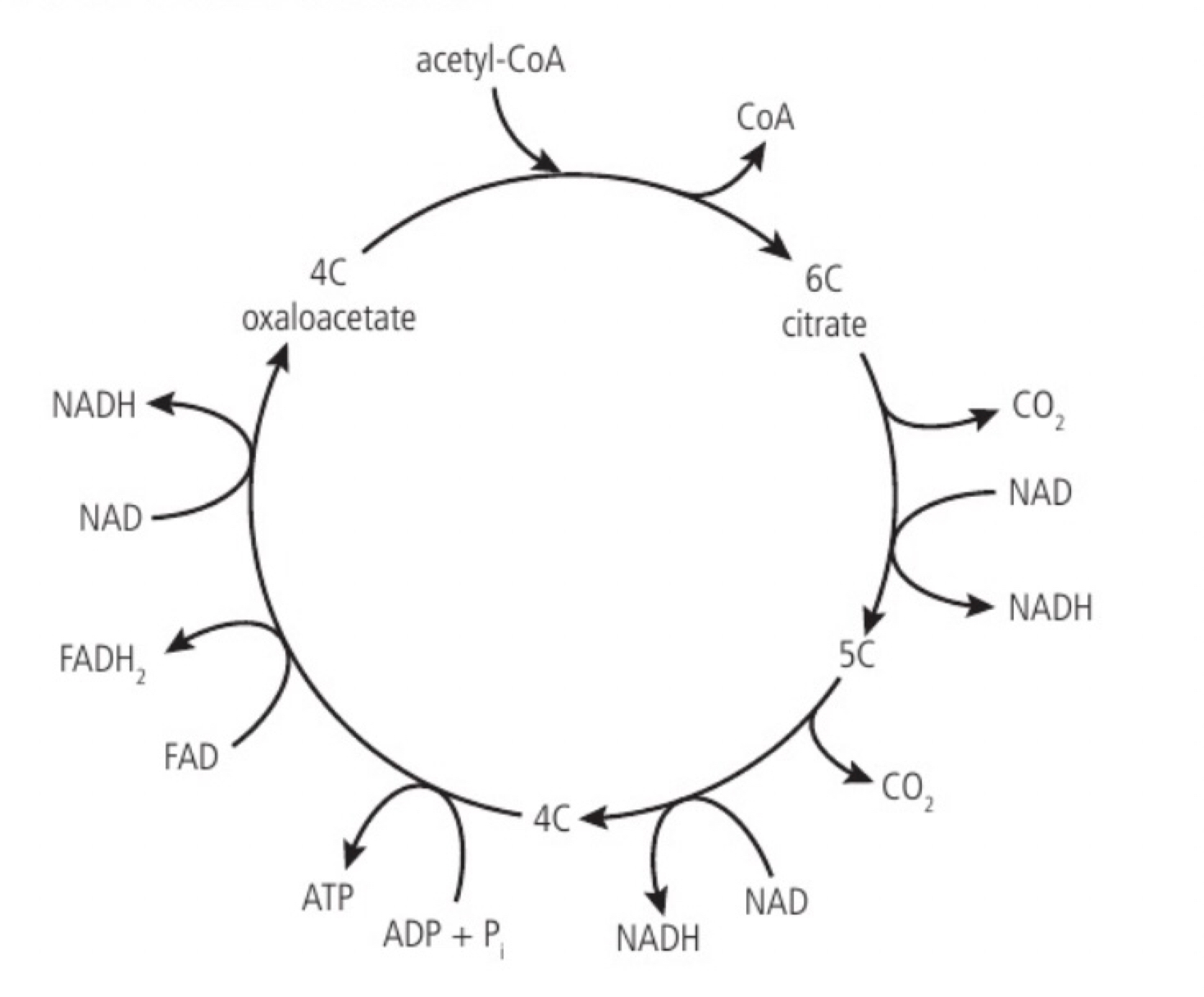
a. the acetyl group produced in the link reaction can be fed into the krebs cycle by transfer from acetyl CoA to a four-carbon organic acid (oxaloacetate), this produces a six carbon organic acid (citrate). Citrate is broken down back to oxaloacetate while producing intermediates, this regeneration allows the cycle to continue processing more acetyl groups.
b. the acetyl (cc) from acetyl-CoA combines with oxaloacetate (4c) to form citrate (6c). citrate is converted into a 5-carbon compound at least as CO2 (decarboxylation) and hydrogen is removed and accepted by NAD+ to produce NADH (oxidation). the 5C compound is converted into a 4C compound and another decarboxylation occurs. the 4C compound is rearranged and another oxidation reduces NAD+ to NADH and a phosphate group is transferred to ADP forming ATP and additional hydrogen atoms are removed, reducing FAD to FADH2 (oxidation) resulting in the regeneration of oxaloacetate (4C).
Transfer of energy by reduced NAD to the electron transport chain in the mitochondrion
a. the electron transport chain is a series of carriers located in the inner membrane of the mitochondrion. reduced NAD supplies its electrons to the first carrier in the chain. the electrons (as part of the hydrogen atoms) come from oxidation reactions in glycolysis, the link reaction and krebs cycle (the hydrogen atoms are split). the electrons have a high energy level and pass into the electron transport chain. the chain occurs in the inner mitochondrial membrane and at the membranes of the cristae; embedded in these membranes are electron carrier molecules that are easily reduced and oxidised. the reduced NAD is converted back into NAD.
Generation of a proton gradient by flow of electrons along the electron transport chain
a. as electrons pass along the chain from one carrier to the next they release energy; the electron carriers act as proton pumps and use the energy released by electron flow to pump protons (H+) from the matrix to the intermembrane space. this causes a proton concentration gradient to develop across the inner mitochondrial membrane (the relatively small space allows it to become steep quickly). this gradient provides the energy for the formation of ATP from ADP and inorganic phosphate (Pi) through chemiosmosis
oxygen is the final electron acceptor in the electron transport chain and protons from the matrix reducing metabolic water and allowing continued flow of electrons along the chain as if no oxygen is available electrons flow stops and reduced (NAD cannot be converted back to NAD) and so the link reaction and krebs cycle cannot continue.
Chemiosmosis and the synthesis of ATP in the mitochondrion
a. the proton gradient established across the inner mitochondrial membrane by electron flow through the electron transport chain stores potential energy which is used by enzyme ATP synthase to generate ATP by phosphorylation of ADP. this is called chemiosmosis. ATP synthase has a rotor that can spin and other parts which are not mobile. the rotor is embedded in the inner mitochondrial membrane and allows protons to diffuse across the membrane in the matrix where the concentration is depleted. ATP synthase has binding sites on the sides of the rotor which collect protons and proton addition for rotation (potential energy from gradient, transferred to kinetic energy of rotation). the rotor has 3 central stalks (requires rotation) bound to separate parts of the active site. a conformational change is induced in the central stalks that forces ADP and phosphate together to produce ATP; after this returns to its original conformation releasing the ATP. (one 360° turn of the rotor/central stalk results in 3 ATP molecules and needs nine protons to pass through the ATP synthase).
Role of oxygen as terminal electron acceptor in aerobic cell respiration
a. oxygen is the final electron acceptor in the electron transport chain and protons from the matrix reducing metabolic water and allowing continued flow of electrons along the chain as if no oxygen is available electrons flow stops and reduced (NAD cannot be converted back to NAD) and so the link reaction and krebs cycle cannot continue.
Differences between lipids and carbohydrates as respiratory substrates
a. lipids (triglycerides): CH2O : ratio 1:2:less 0.65 (so relatively low carbohydrate, so higher yield of energy), energy released is 39 kJ per gram, metabolic pathway is β-oxidation (beta oxidation to produce acetyl groups that feeds cycle and chemiosmosis)
b. carbohydrates: CH2O : ratio 1:2:1 (so relatively more oxidised, so lower yield of energy), energy released is 17 kJ per gram, metabolic pathway is (i) glycolysis: the reaction to produce acetyl group then krebs cycle and chemiosmosis
c. carbohydrates are preferred because energy pathways are faster and automatically as glucose is soluble and enzymes easily available for easier transport.
Transformation of light energy to chemical energy when carbon compounds are produced in photosynthesis
Photosynthesis is the process by which plants, algae, and some bacteria convert light energy from the sun into chemical energy stored in the form of organic compounds like glucose.
During photosynthesis light energy is absorbed by pigments and is transformed into carbon compounds which contain chemical energy (the energy is stored within the bonds of the organic compounds). This transformations fuels the life processes in ecosystems (Producers, such as plants, algae and cyanobacteria, absorb light and produce glucose and other carbon compounds. Primary consumers eat the producers, absorbing the energy they have stored. Every organism, regardless of the trophic level it belongs to, relies upon the energy from the sun that was converted using photosynthesis.
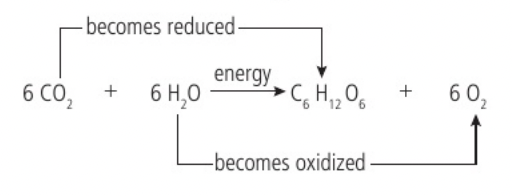
Conversion of carbon dioxide to glucose in photosynthesis using hydrogen obtained by splitting water
Carbon dioxide + water —light→ glucose + oxygen
To convert carbon dioxide (CO2, 1 carbon : 2 oxygens) into glucose (C6H12O6, 1 carbon : 2 hydrogens : 1 oxygen), half of the oxygen must therefore be removed and hydrogen must be added. The hydrogen is obtained by splitting water molecules into hydrogen and oxygen with the presence of light (photons) for energy (photolysis) (12H2O → 24H + 6O2). Half the hydrogen atoms become part of a glucose molecule and the other half are used to reduce carbon dioxide (remove one oxygen atoms from carbon dioxide and convert it to water) and form glucose (6CO2 + 24H → C6H12O6 + 6H2O)
Oxygen as a by-product of photosynthesis in plants, algae and cyanobacteria
Oxygen is a by-product of splitting water to release hydrogen in plants, algae and cyanobacteria. These organisms use some of the oxygen that they produce in aerobic cell respiration but most of it is excreted into the environment.
Photolysis increases the concentration of oxygen inside chloroplasts. This causes oxygen to difuse out of chloroplasts and then out of leaf cells to air spaces inside the leaf. The oxygen then difuses through stomata to the air outside the leaf.
Separation and identification of photosynthetic pigments by chromatography
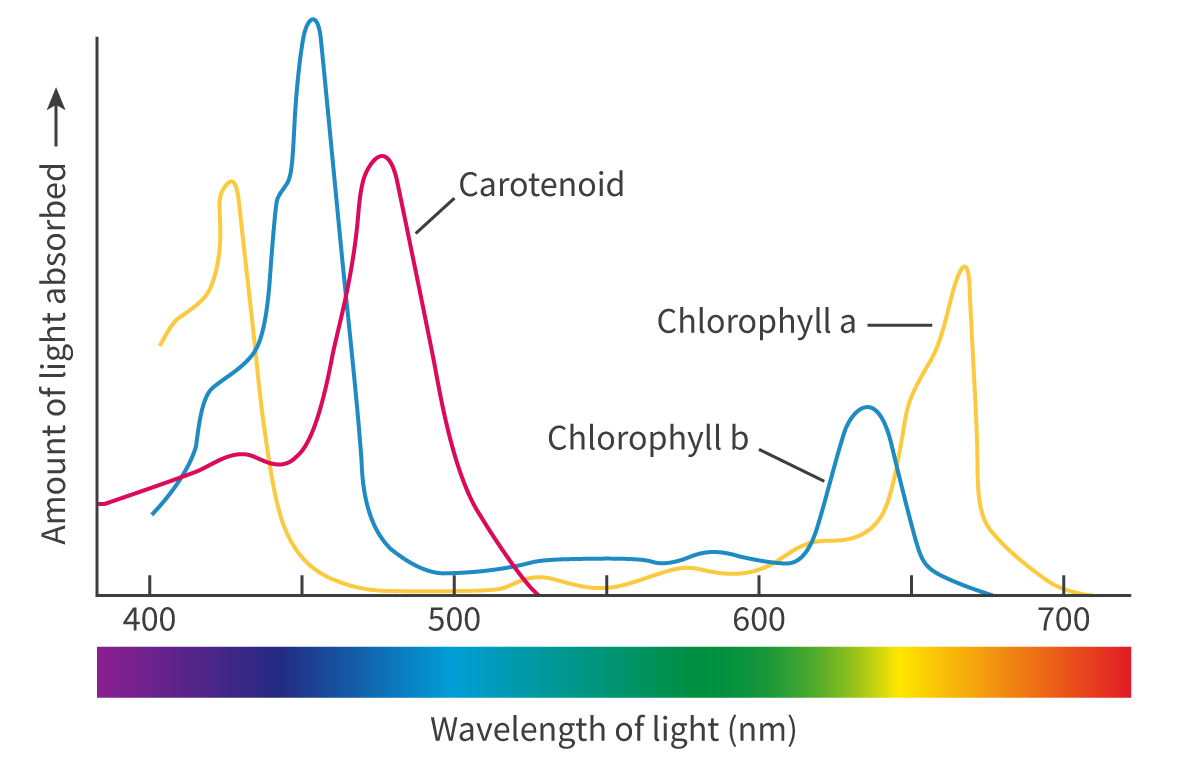
Photosynthesis takes place in chloroplasts. chlorophyll a and chlorophyll b are the main pigments that capture the photons which reflect green light and absorb most of the other wavelengths in the visible light spectrum. There are other accessory pigments involved in photosynthesis that assist chlorophyll in capturing light energy during photosynthesis. These pigments broaden the absorption spectrum, allowing plants to capture light energy from a wider range of wavelengths.
Paper chromatography is a technique used to separate mixtures of substances based on the movement of the different substances on a piece of paper by capillary action. The paper is the stationary phase while the solvent used to develop the chromatogram is the mobile phase (that carries the sample components through the system facilitating the separation and migration of the sample components). The pigments are commonly separated using an alcohol/solvent mixture.
Tear up a leaf into small pieces
Grind the leaf pieces with sharp sand and propanone to extract the leaf pigments
Transfer a sample of extract to a watch glass
Use a hairdryer to evaporate of the propanone and the water from the cells’ cytoplasm.
Add a few drops of propanone to dissolve the pigments
Build up a concentrated spot 10 mm from the end of a strip of chromatography paper (or TLC) by transferring tiny drops of solution allowing it to dry before adding another drop
Suspend the strip in a tube with the base dipping into running solvent
Remove the strip from the tube when the running solvent has nearly reached the top. Draw a pencil line to show how far the solvent moved
Use a ruler with millimetre markings to measure the distance moved by the running solvent (the distance between the two lines) and the distance moved by each pigment (the distance between the lower line and the cross in the centre of the circle).
the pigment in each spot can be identified from its colour and its Rf calue. Calculate the Rf value for each pigment, using the equation: Rf = distance run moved by pigment / distance run moved by solvent (pigments with higher Rf values are more soluble in the solvent being used and are often smaller in size and have lower affinity for the aper used)
Absorption of specific wavelengths of light by photosynthetic pigments
Photosynthetic pigments are specialized molecules that absorb light energy and convert it into chemical energy during photosynthesis.
The electromagnetic (EM) spectrum is the range of frequencies and wavelengths of electromagnetic radiation emitted from the sun. The different photosynthetic pigments, such as chlorophyll a and b as well as xanthophyll and the carotenoids, all absorb and reflect different wavelengths of light. The wavelengths reflected give the pigments their colour. the structure of the pigments allows an electron within the molecule to jump form one energy level up to a higher level (excited) using energy obtained by absorbing a photon of light which are then used to power the light-dependent stage. Those electrons are replaced with the electrons released from the photolysis of water.
only specific wavelengths of light have the amount of energy needed to raise an electron to a higher energy level and only those wavelengths are absorbed and the others are reflected.
The wavelengths of light absorbed by each pigment can be represented in a graph known as an absorption spectrum (light absorbed plotted against the wavelength of light). it also indicates the ware of photosynthesis are the different wavelengths of light (high percentage of light absorption meaning photosynthesis is also high)
Similarities and differences of absorption and action spectra
An action spectrum is graph like that shows the photosynthetic activity at each wavelength and can be generated using a variety of measurements from oxygen production to carbon dioxide absorption. An action spectrum is a good indicator of which wavelengths are most efficient for photosynthesis. Some wavelengths cause a higher photosynthetic rate than others.
Difference: The absorption spectrum is derived from individual pigments like chlorophyll a, chlorophyll b, and carotenoids. Whilst the action spectrum reflects the combined effect of all pigments and the photosynthetic machinery.
The absorption spectrum shows what light is captured, while the action spectrum shows how effectively that light drives photosynthesis.
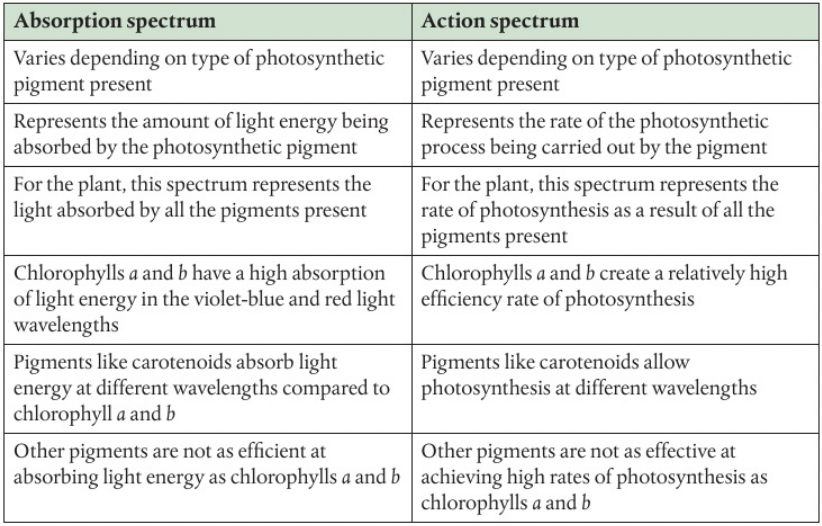
Techniques for varying concentrations of carbon dioxide, light intensity or temperature experimentally to investigate the effects of limiting factors on the rate of photosynthesis
Photosynthesis is affected by various factors but usually just one is limiting the rate at a particular time. this is the factor that is nearest to its minimum called the limiting factor. the three possible limiting factors are temperature, light intensity and carbon dioxide concentration
temperature: use a water plant (pondweed) in a water bath or on a hot plate to vary the temperature. range should be between 5 to 45 C. and to kee it controlled use a thermostat at 25C and keep it there throughout the experiment
light intensity: move light source to difference distances and measure light intensity with a lux meter (light intensity = 1/distance^2) range 4-14 cm and no light. to keep controlled keep the light source at a constant distance.
carbon dioxide concentration: start with boiled, cooled water (no CO2) then add measure quantities of NaHCO3 to increase the CO2 concentration. range 0 to 50 mmol dm^-3. to keep controlled add enough NaHCO3 to give a high CO2 concentration
when writing an experiment: one limiting factor at a time remaining should be controlled and a suitable range is chosen.
measuring the rate of oxygen production of carbon dioxide intake (or changes in biomass) (for example through pH to measure CO2 consumed, less acidic is more consumed, or water bubbles per unit time or volume of oxygen produced (oxygen probe)
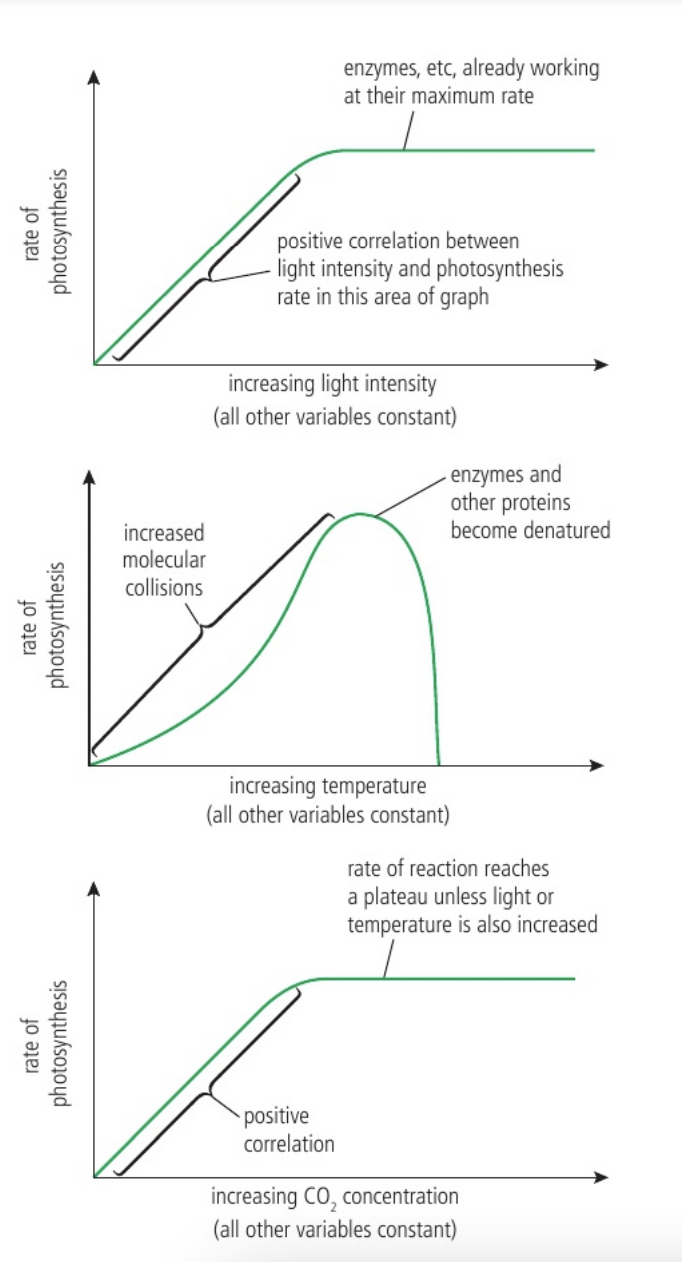
Carbon dioxide enrichment experiments as a means of predicting future rates of photosynthesis and plant growth
Mainly due to human activity, the concentration of carbon dioxide in our atmosphere is increasing. Understanding how plants will respond to these higher levels of CO2 is important. It will allow us to better predict future food production. This can be investigated using carbon dioxide enrichment experiments.
There are two main methods for this: Enclosed Greenhouse Experiments and Free-Air Carbon Dioxide Enrichment (FACE) Experiments
Using an enclosed greenhouses where the concentration of CO2 can be carefully monitored and controlled. Elevated levels of CO2 can be created by burning fuels or other sources and there should be a control greenhouse without enriched CO2, the effect of CO2 enrichment can be measured via total biomass produced or the yield of the fruits or vegetables grown. The limitations of these studies are that, as they are done in controlled conditions, there are natural factors and variables that are not able to be taken into account.
Free-air carbon dioxide enrichment experiments (FACE) is carried out in a natural ecosystem. Pipes surround the area (between 1 m and 30 m in diameter) and release CO2 continuously. Sensors within the area monitor CO2 levels to ensure elevated concentrations are maintained. This allows for a more natural way of measuring the impact of higher levels of CO2 in the atmosphere. However, this method is usually very expensive to carry out.
carbon dioxide is usually a limiting factor so increased CO2 levels mean higher rate of photosynthesis
Photosystems as arrays of pigment molecules that can generate and emit excited electrons
Photosystems are used in the light-dependent reactions of photosynthesis which capture light energy which is then needed for the synthesis of carbohydrates in the Calvin cycle.
Photosystems are molecular arrays of chlorophyll and accessory pigments with a special chlorophyll as the reaction centre from which an excited electron is emitted. Photosystems are always located in membranes (in photosynthetic eukaryotes the photosystems are in the thylakoid membranes, also exist in cyanobacteria). Their presence in membranes allows for the efficient capture and utilisation of light energy for photosynthesis.
The centre of a photosystem is the reaction centre which is surrounded by various pigment molecules bound to proteins called light-receiving complexes (composed of chlorophyll and other accessory pigments) which allows energy to be captured and passed to the central molecule. when a light-receiving pigment absorbs a photon the energy is transferred from pigment molecule to pigment molecule until it reaches the reaction centre complex which includes a pair of special chlorophyll molecules whose electrons can move away from the molecules when energy reaches them (photoactivation). these energised electrons are then received by a primary electron acceptor.
Plants and cyanobacteria utilise 2 photosystems each absorbs light most efficiently at a different wavelength. PS I is most efficient at 700nm so labelled P700 and PS II is most efficient at 680nm and so is labelled as P680
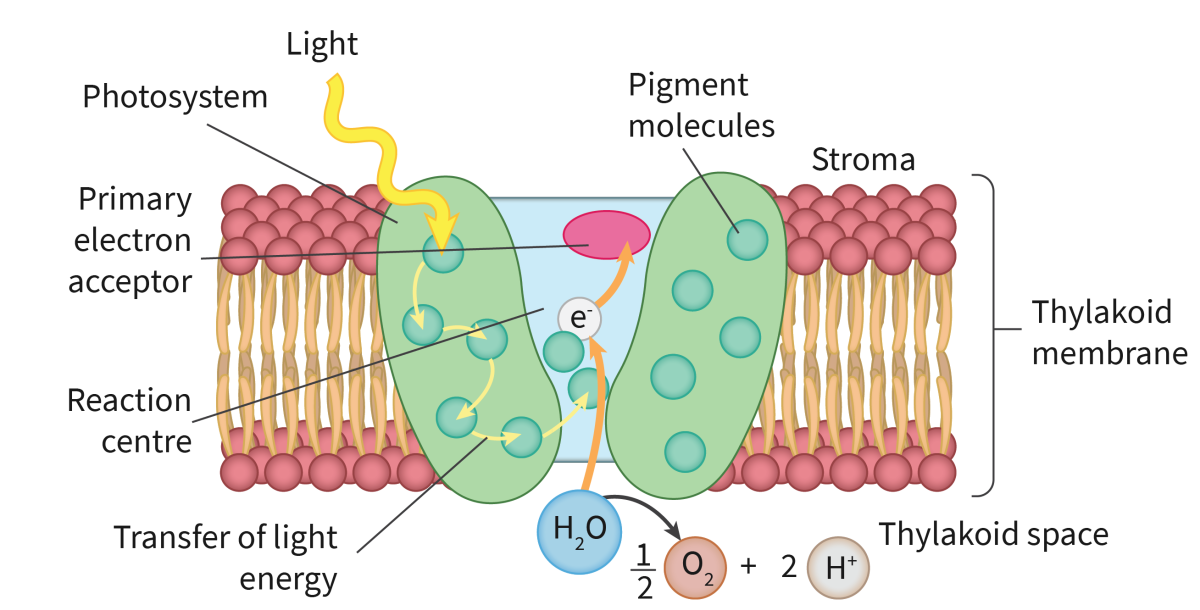
Advantages of the structured array of different types of pigment molecules in a photosystem
The structured ai.Tay of clifterent types of pigments and accessory pigments enables light energy to excite electrons in a controlled ,vay and direct these electrons along electron transport chains.
Accessory pigments exist as different types and in large numbers in a photosystem so that all the parts of the light reactions of photosynthesis can occur. Only through the use of many pigment molecules can enough light energy be absorbed to photoactivate the central chlorophyll molecule. This causes the excitation and release of the electrons that will provide the energy for the rest of the light-dependent phase of photosynthesis. If only one pigment existed in a photosystem, photosynthesis would not proceed beyond the light-dependent reactions. Energy is only transferred from one pigment molecule to another when the molecules are in a close and precise orientation. Otherwise, light energy is lost by fluorescence. The structured array also ensures that absorbed energy is funnelled to the reaction centre of the photosystem. Chlorophyll a must be present (primary pigment).
Generation of oxygen by the photolysis of water in photosystem II
The first steps of the light-dependent reactions:
A photon of light is absorbed by a pigment in photosystem Il and is transferred to other pigment molecules until it reaches one of the chlorophyll a (P680) molecules in the reaction centre. The photon energy excites one of the chlorophyll a electrons to a higher energy state.
This electron is captured by the electron acceptor of the reaction centre.
Water is split by an enzyme to produce electrons, hydrogen ions and an oxygen atom (in the oxygen-evolving complex (OEC)). This process is driven by the energy from light and is called photolysis. The electrons are supplied one by one to the chlorophyll a molecules of the reaction centre. These electrons replace the electrons lost from chlorophyll a to the electron acceptor.
When photolysis occurs during the light-dependent reactions, the protons and electrons are used in photosynthesis. The oxygen is given off as a waste product.
The OEC binds a total of two water molecules and splits them into four electrons, four protons and one molecule of oxygen.
2H2O → 4H+ + O2 + 4e−
The electrons pass to the P680. The protons are released into the thylakoid space, contributing to a proton gradient across the thylakoid membrane. The oxygen molecules are a waste product and diffuse out of the thylakoids. It is this waste oxygen that altered the atmosphere of planet Earth, allowing organisms to respire aerobically and causing oxidation of dissolved iron and other minerals in the oceans.
ATP production by chemiosmosis in thylakoids
The production of ATP in photosynthesis is called photophosphorylation.
After photolysis has occurred to replace the excited electron:
The excited electrons from the chlorophyll a pass from the primary acceptor down an electron transport chain, losing energy at each exchange. The first of the three carriers is plastoquinone (PQ). The middle carrier is a cytochrome complex.
The energy lost from the electrons moving down the electron transport chain drives chemiosmosis to bring about phosphorylation of ADP to produce ATP
ATP synthase is located in thylakoid membranes. A proton gradient is generated by chains of electron carriers in thylakoid membranes. Excited electrons pass along these chains, releasing energy (energy is released when electrons are exchanged from one carrier to another) which is used to pump protons from the stroma to the thylakoid space. Hydrogen ions diffuse back into the stroma through the channels of ATP synthase.
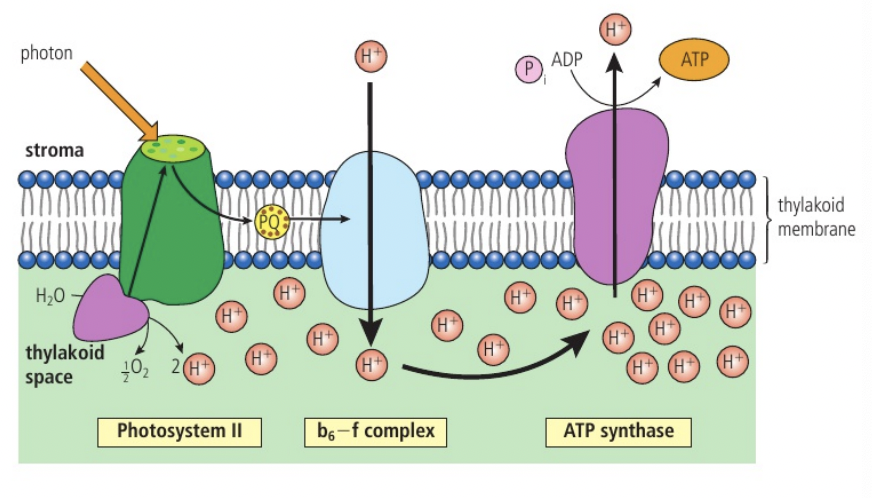
Cyclic photophosphorylation
Cyclic photophosphorylation involves photosystem I (PSI) only
Light is absorbed by photosystem I (located in the thylakoid membrane) and passed to the photosystem I primary pigment (P700)
An electron in the primary pigment molecule (i.e. the chlorophyll molecule) is excited to a higher energy level and is emitted from the chlorophyll molecule in a process known as photoactivation
This excited electron is captured by an electron acceptor, transported via a chain of electron carriers known as an electron transport chain before being passed back to the chlorophyll molecule in photosystem I
As electrons pass through the electron transport chain they provide energy to transport protons (H+) from the stroma to the thylakoid lumen via a proton pump
A build-up of protons in the thylakoid lumen can then be used to drive the synthesis of ATP from ADP and an inorganic phosphate group (Pi) by the process of chemiosmosis
Chemiosmosis is the movement of chemicals (protons) down their concentration gradient, the energy released from this can be used by ATP synthase to synthesise ATP
The ATP then passes to the light-independent reactions
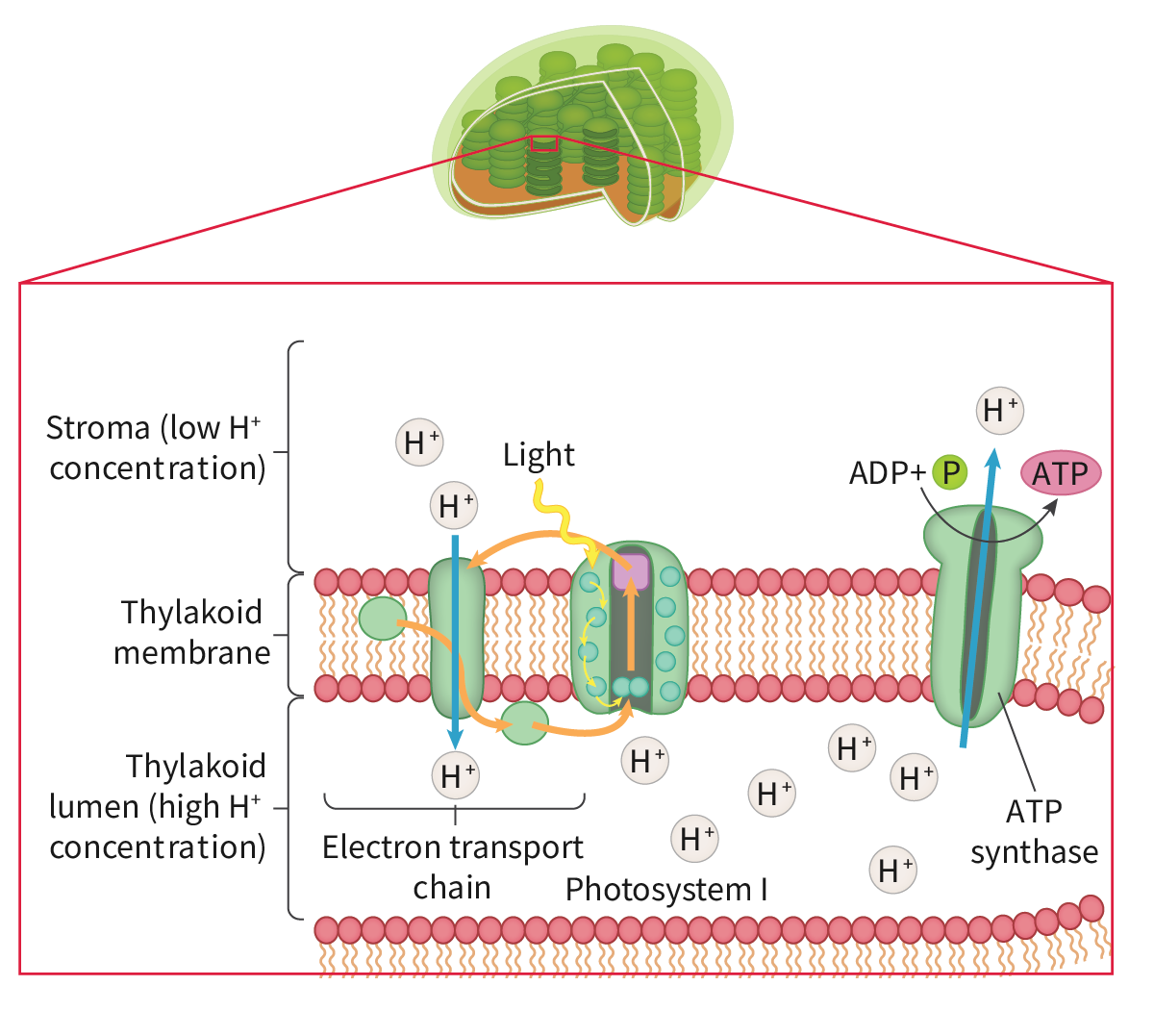
Non-cyclic photophosphorylation
In non-cyclic photophosphorylation, light energy excites electrons in PSII, which are then passed along the ETC, pumping H+ and generating ATP. The electrons then enter PSI where they are re-energised with light energy, passed to ferredoxin and then used to reduce NADP.
light is absorbed by both photosystem I and photosystem II
photoactivation - electron in the primary pigment molecule of each photosystem are excited to a higher energy level.
excited electrons from photosystem II are passed to photosystem I via an electron transport chain where they are re-energised with light energy, releasing sufficient energy to synthesise ATP
electrons form the photolysis of water replace those lost from photosystem II
excited electrons from photosystem I and hydrogen ions from the photolysis of water both combine with NADP to form reduced NADP
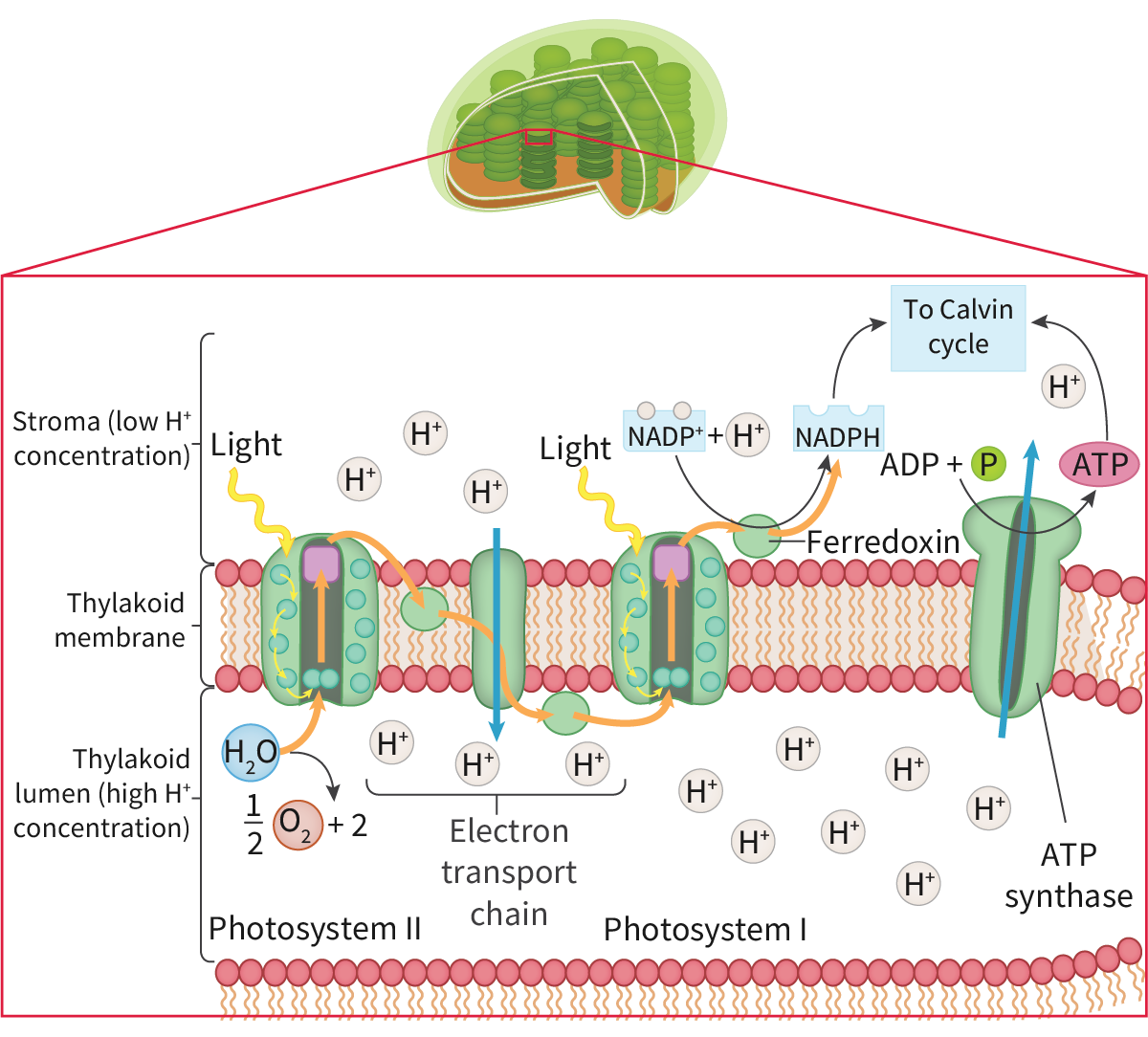
Reduction of NADP by photosystem I
A photon of light is absorbed by a pigment in photosystem I. This energy is transferred through several accessory pigments until received by a chlorophyll a (P700) molecule. This results in an electron with a higher energy state being transferred to the electron acceptor. The energised electron from photosystem Il fills the void left by the newly energised electron.
The electron with the higher energy state is then passed down a second electron transport chain that involves the carrier ferredoxin.
The enzyme NADP reductase catalyses the transfer of the electron from ferredoxin to the energy carrier NADP. Two electrons from photosystem I and one hydrogen ion from the stroma are required to reduce NAP fully to NADPH
Thylakoids as systems for performing the light-dependent reactions of photosynthesis
the light- dependent reactions take place in the thylakoid
photolysis of water: thylakoid lumen (inside the thylakoid space), uses enzymes embedded in the thylakoid membrane
synthesis of ATP by chemiosmosis: across the thylakois membrane via ATP synthase (protons build up in the lumen and diffuse back into the stroma through ATP synthase embedded in the thylakoid membrane)
reduction of NADP+: stromal side of the membrane, uses NADP+ reductase in the thylakoid membrane facing the stroma
The thylakoid membrane has components:
Impermeable to protons so a proton gradient can develop
Encloses a small volume of fluid so a gradient develops rapidly
Made of phospholipids so can hold photosystems composed of hydrophobic pigment molecules
Holds other components in the correct relative positions--ATP synthase, electron carriers and NADP reductase.
Carbon fixation by Rubisco
The light-independent reactions, also known as the Calvin cycle, occur within the stroma of the chloroplast. Although the Calvin cycle is not directly dependent on light, it is indirectly dependent on light because it requires the energy carriers ATP and reduced NADP (NADPH) which are produced by the light-dependent reactions.
Carbon dioxide diffuses into the stroma of the chloroplast
Ribulose bisphosphate (RuBP), a 5-carbon compound, binds to an incoming carbon dioxide molecule in a process called carbon fixation (the conversion of inorganic carbon (carbon dioxide from the atmosphere) to organic carbon by a living organism). This fixation is catalysed by an enzyme called RuBP carboxylase (Rubisco). The result is an unstable 6-carbon compound.
The unstable 6-carbon compound breaks down into two 3-carbon compounds called glycerate 3-phosphate (GP).
The Calvin cycle takes place in the stroma of the chloroplasts, where there is a high concentration of Rubisco. There are two reasons for this: Rubisco is a ‘slow’ enzyme and the Calvin cycle is rather inefficient due to its high energy requirement. Additionally, Rubisco can be competitively inhibited by oxygen, binding to it in preference to carbon dioxide, making it very inefficient at low concentrations of carbon dioxide. To speed up the process, many molecules of Rubisco enzyme are needed causing it to be the most abundant enzyme on Earth.
he 3-carbon molecules of GP form two other 3-carbon molecules called triose phosphate (TP) via a reduction reaction. The hydrogen needed for this is supplied by reduced NADP. Energy is also needed and is supplied by ATP.
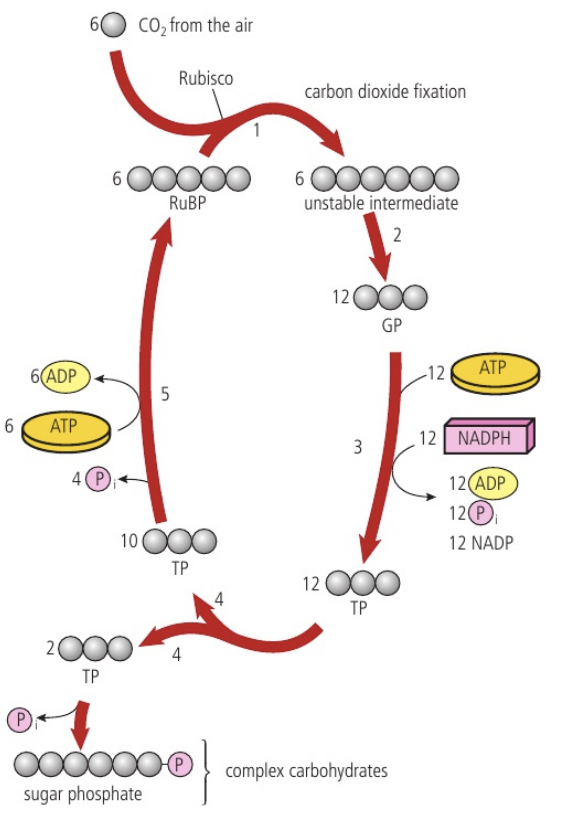
he molecules of TP may then go in either of two directions. Some leave the cycle to become sugar phosphates, which may then become more complex carbohydrates. Most, however, continue in the cycle to reproduce the original molecule in the cycle, RuBP.
For the Calvin cycle to continue, every RuBP consumed must be replaced. Five molecules of triose phosphate are converted by a series of reactions into three molecules of RuBP. This process requires the use of energy in the form of ATP. If glucose is the product of photosynthesis, for every six molecules of triose phosphate made by the light-independent reactions, five are used to regenerate RuBP and one exits the cycle.
Synthesis of triose phosphate using reduced NADP and ATP
The 3-carbon molecules of GP form two other 3-carbon molecules called triose phosphate (TP) via a reduction reaction. The hydrogen needed for this is supplied by reduced NADP. Energy is also needed and is supplied by ATP.