Test 4 Content
1/131
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
132 Terms
diabetes mellitus
DM, is a metabolic disorder characterized by chronic hyperglycemia. people with it will have blood samples taken at any time and those levels will be far higher than what physiological norm should be. insulin, the key regulator of blood glucose levels can’t effectively maintain blood glucose levels in range
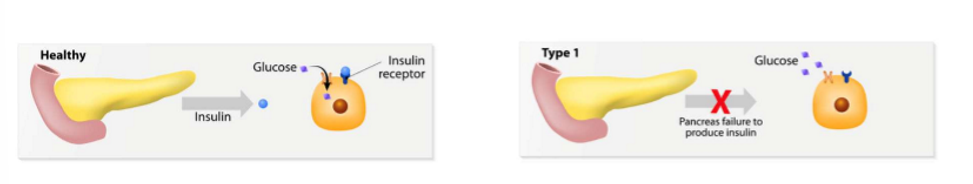
type 1 DM
insulin producing pancreatic beta cells are progressively destroyed by an autoimmune process. this is insufficient productions
type 2 DM
combination of genetic, metabolic, and environmental factors lead to an altered production and response to insulin with increased glucose production. this is very common
gestational DM
metabolic and hormonal changes in the later stages of pregnancy induce insulin resistance and increase the risk of developing type 2 diabetes later in life. only characteristic is during pregnancy, and will usually disappear after birth
how many pregnancies lead to gestational DM
typically affects some, not all pregnancies, usually late stages (2nd, 3rd trimester)
gestational DM if not controlled
can cause some effects on fetus, a lot of times these mothers give birth to higher weight babies than what is expected for their age. The other issue is that that baby is more at risk for developing a metabolic disease in life
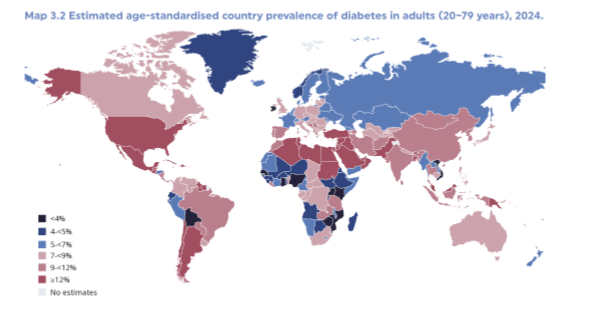
DM as a health concern
according to the international diabetes federation atlas (2025), DM affects 11.1% of the global population (already a 1% increase from few year ago), and it is estimated to rise to 13% by 2050 (but estimate will probably go up again in 2-5 years)
DM in different countries
it is estimated that 95% of this increase will occur in low and middle income countries. this is bc places w lower income often don’t have the healthiest food, and already have less resources for medicine so this is an increased burden due to high income countries
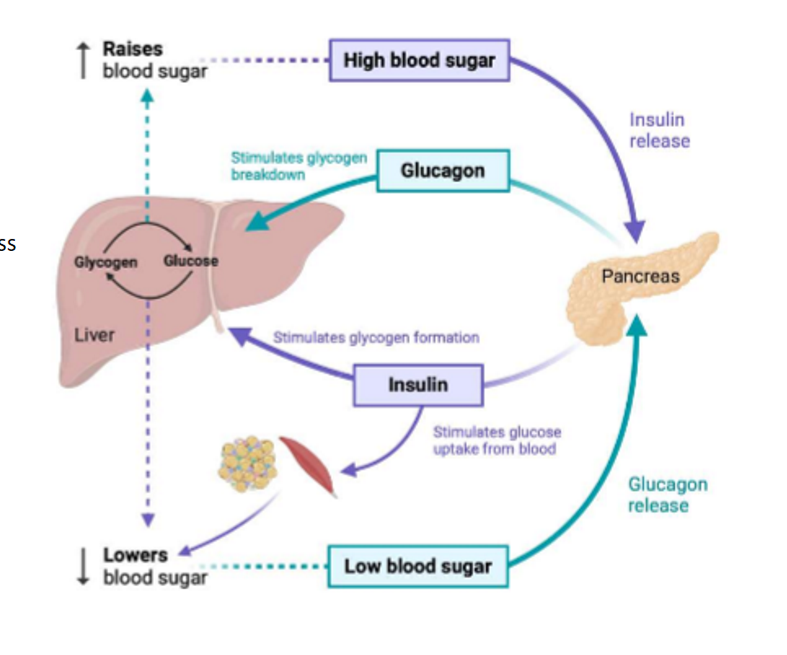
what is the problem with hyperglycemia
if not treated right away it leads to an increased risk of developing acute and chronic complication (potentially life threatning); kidney disease risk increase bc kidney damaged and over time will shut down. DM significantly increases risk of CVDs such as heart attack, stroke
link of hyperglycemia to CVDs
associated with accelerated atherosclerotic process, and it is the base of CVDs; so someone with diabetes has a higher risk of developing CVD. however they will need to be checked periodically
biological sex differences in DM and its associated CVDs
premenopausal women healthier than men their age, postmenopausal women less healthy than men their age

hypothesis for biological sex differences in DM and CVD association
large changes in hormonal levels pre and post menopause, so estrogen is possibly protecting you
rodent models of DM
helps us: recapitulate the main characteristics of diabetes (induce specific mutations to make mouse more prone to developing diabetes, and change things to make features more similar to humans w diabetes), we can study diabetes in a controlled environment, and we can test specific things (drugs, genetic factors) to learn more about diabetes (look if there’s a genetic factor by changing genes)
sex differences and preclinical research in DM
many papers about DM do not include both sexes. there is still resistance to using female animal models in basic science (sex hormone fluctuations). now papers must have both sexes unless they can give specific reason why not
how can we account for sex as a biological variable (SABV) using rodents
use male and female rodents, use specific rodent models of diabetes that present sexual dimorphism
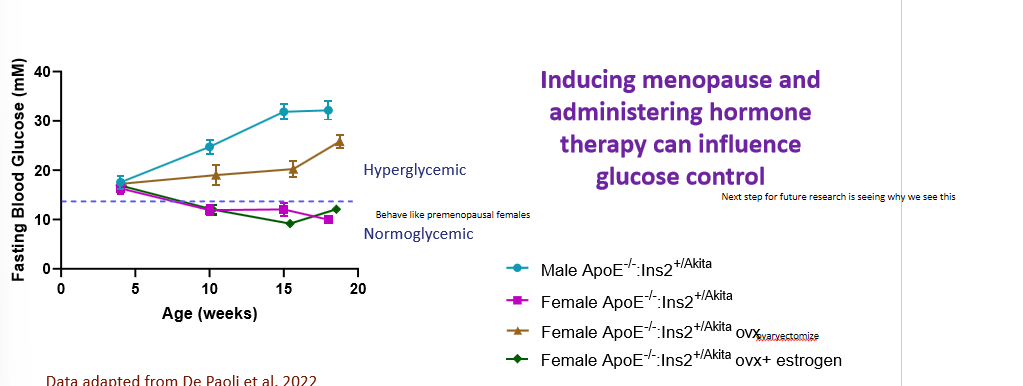
sexual dimorphism in rodent models of diabetes
male mice were found to be hyperglycemic, female mice found to be normoglycemic. not as expected. we have a transient moment in time when hyperglycemic then went down and normal again (bc at 5 weeks they go through puberty)

perceptions when contrasting results on hormone therapies
induce a negative perception in general population; many people are afraid they might increase they risk of DM, MI, and stroke; current analysis shows that hormone therapies are beneficial for cardiometabolic health when started in the early phases of menopause
how can we analyze the effects of menopause and hormone therapy in rodent models of diabetes
since the majority of estrogens are produced in the ovaries, an ovariectomy stimulates what we can see with menopause, and we can supplement hormones to mimic hormone therapies
early life
maternal and paternal; pregnancy (post conception): mother; preconception: father, sperm. egg
what kind of factors influence early life development
maternal nutrition, environment
fetal fortune telling
developing baby receives information from the mother in the form of hormones, nutrients or oxygen and uses these to predict what kind of environment it will be born to; it will then use this info to adapt its development to better its chances of survival after birth (and health into adulthood)
organisms with evidence of fetal fortune telling
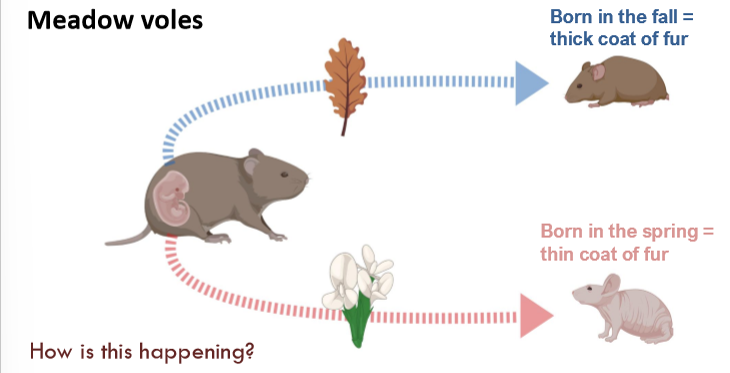
meadow vole fur, tree budding, bees
meadow vole example of fetal fortune telling
hormonal signal; melatonin due to changing photoperiod
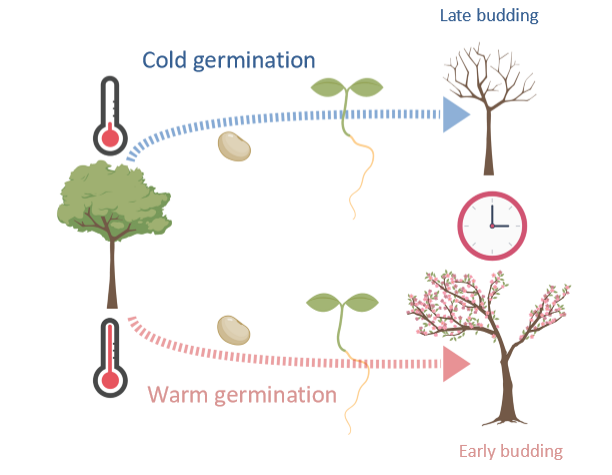
tree budding example of fetal fortune telling
temperature signal
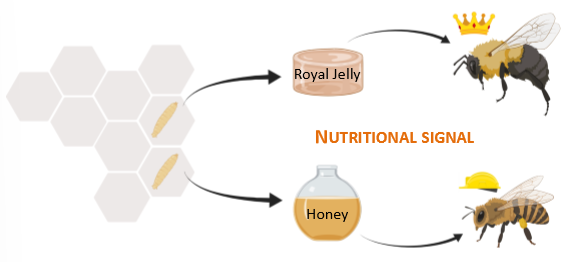
honey bee example of fetal fortune telling
nutritional signal. special royal jelly changes epigenetics of honeybee to turn it into queen bee
if fetal signaling is well vs not in the wild
good: produce healthy offpring, well suited to environment its born into. bad: contribute to poor fertility, poor survival and disease, not well suited to environment
prediction of fetal fortune telling
the developing fetus received information from the mother in the form of hormones, nutrition or oxygen and uses these to predict the external environment. may not be accurate
adaptation of fetal fortune telling
the developing fetus will then use this information to adapt its development to better its chances of survival after birth. may not necessary and thus may result in disease
misinformation that a fetus gets
stress, xenobiotics, nutrition, parental metabolism and disease. degree by which it misinforms is different (smoking and drinking is a big one). however, if parent is stressed and baby is born into stressful environment, its better for the baby bc same environment and baby uses its cortisol or something
early human studies that formed the basis for early origins science
david barker, an epidemiologist, trained in tropical medicine, asked lots of crazy questions, was interested in cvd and why some ppl got it and others did not. Looked at patterns of disease risk, What other factors were changing at that time across whole life period w cvd, Used historical data sets w birth weight of same ppl he tracked later in life, For both risk of diabetes and cad tracked w weight at birth
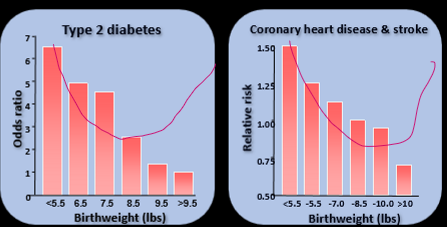
t2d and cvd and birthweight
Lower birth weight leads to higher risk with -ve correlation, These data have been replicated, new shape is u shape, So something happening w pancreas and periph vasc and heart respectively changing disease trajectory, Low weight babies grow differently, Changed field of research, Always about what you're doing currently at drs, not fetal, Weve bever considered it before, Its those first 1000 days
the first 1000 days
a period of heightened (developmental) plasticity and sensitivity to env stimuli
results and findings of david barker studies
found that birth weight acc correlated to many things, so everyone was wondering what would happen to their newborns, they posted it with no thoughts about ramifications (everyone went to check their birthweight and self diagnose), but its not acc birth weight
research that helped to see the changes seen in david barkers research
the dutch famine
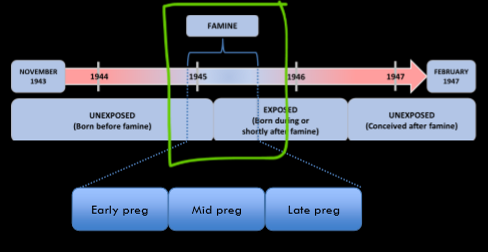
critical developmental periods → vulnerable to adversity results with dutch famine
Found distinct diff in outcomes qhether or not exposed late or early preg; All the adults exposed early in preg had normal birth weight with a bunch of other problems, which doesn’t fit right w model showed earlier; Regardless of when exposed to famine and regardless of birthweight, all had issues w metabolism

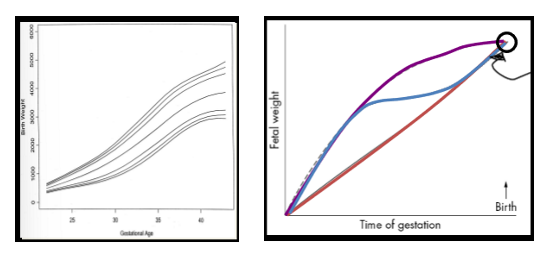
linearity of fetal development growth
not linear. Reason is its complication bc we have very long gestation, it takes rlly long to prod human; Fetal growth not linear; If exposed early or when growth rate slow, you’re gonna change stem cell formation and gene expression of specific organs but maybe not great important on growth; If done when greatest steepest amt of growth, will impact growth, HOWEVER each baby also has its specific growth rate too. so overall, terrible predictor of environment
the placenta
placenta is another big thing contributing to fetal and future adult health. when pregnant ppl go into ultrasound, they look rlly well at placenta; look if its growing as it should bc its nutrient exchange bw mother and baby. contributes to: maternal-fetal exchange, protective barrier, and endocrine signaling. placenta responds to changes that mother goes through
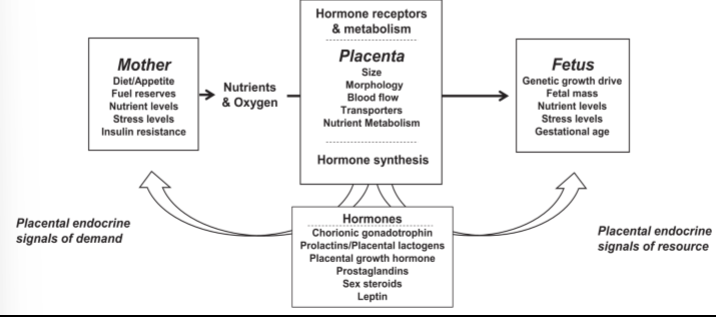
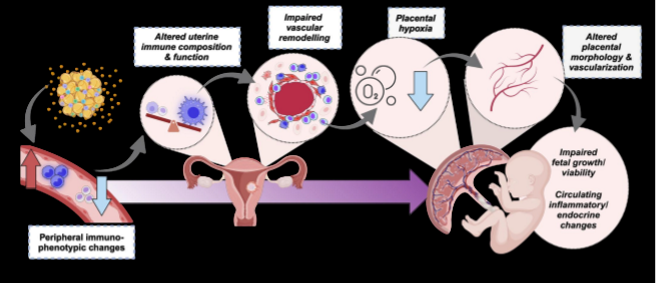
changes in placental vasculature
one major pathway that adversity can change fetal development. changes in each of these or things going wrong can change baby: peripheral immunophenotypic changes, altered uterine immune composition and function, impaired vascular remodeling, placental hypoxia, altered placental morphology and vascularization →→→ impaired fetal growth/viability + circulating inflammatory/endocrine changes
evidence of paternal programming of offspring disease
one of the first studied published w evidence of paternal risk was in 2000s (compared to maternal, this is very late); nutrition in Sweden of group of ppl that were either exposed to high or low levels of nutrition, their sons looked at; if g-pa was given lots of food early on, they found that offspring had increased risk of dying of diabetes (4x). NOT CAUSATION, JUST CORRELATION
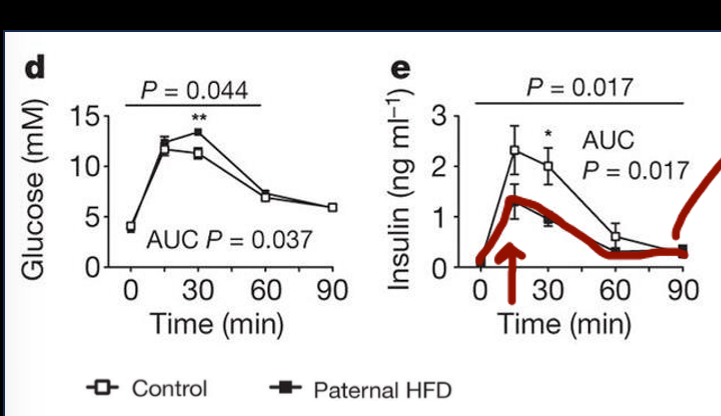
early study results of paternal hf diet to metabolic function of offspring
weak study initially, very first preclinical model that took males, gave them hfd, let them mate, and looked at offspring; offspring grown w controlled diet; then gave those mice glucose tolerance test which showed that it was higher at one time point, and insulin lower
fruit fly paternal studies vs humans
flies: paternal sugar intake controls offspring adiposity in drosophila (reprograms chromatin in offspring); humans: found same genes associated w chromatin regulation were differentially expressed in obese indiv. so the chromatin modifications obs in flied might also be relevant to human obesity susceptibility
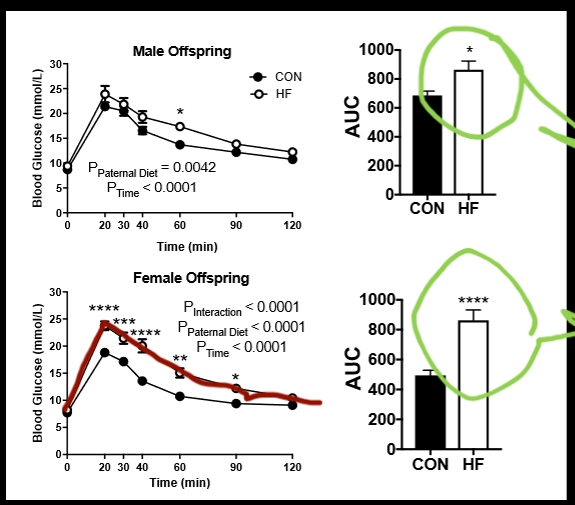
male vs female offspring paternal induced obesity in mice
paternal diet induced obesity induces metabolic dysfunction. male offspring of these obese males display impaired clearance, female offspring of these obese males more vulnerable than males
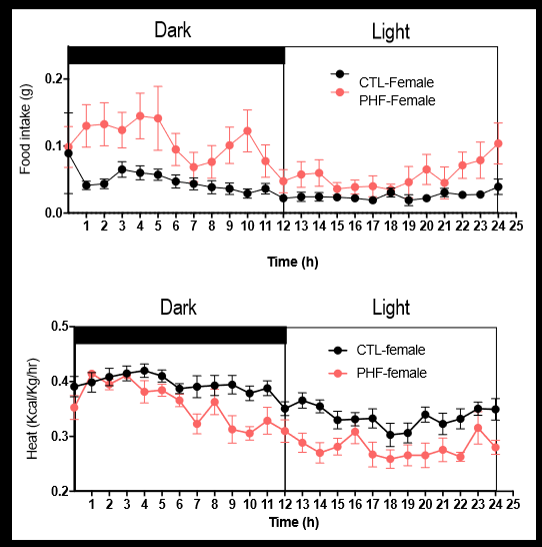
young females born to obese fathers results
paternal obesity results in impaired whole body metabolism in offspring. young female offspring born to obese fathers showed: increased food intake particularly during their “active” dark cycle; and an overall result in heat production: suggesting lower energy consumption so lower metabolic rate (glucose intolerant)
how are father affecting their offspring
sperm and seminal fluids are impacted. pretty much all the same factors that are affected from maternal side affect paternal side
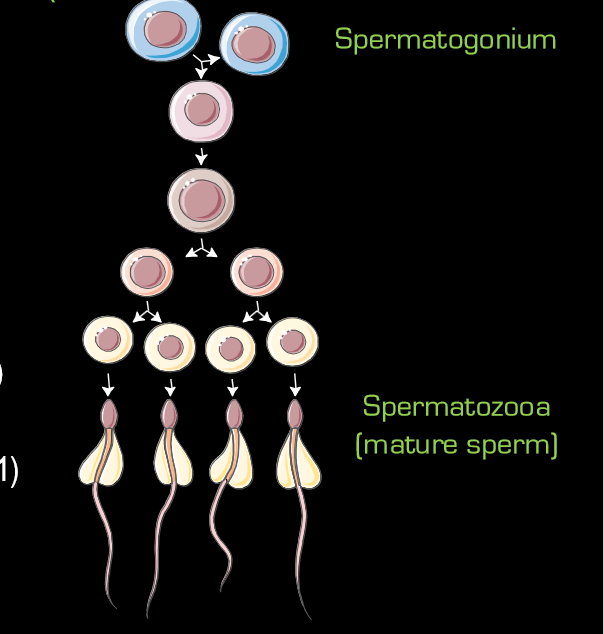
how do obesity, metabolic disease, and substance abuse affect sperm quality
reduce sperm conc, reduce sperm motility, increase sperm DNA damage, and increase sperm oxidative stress
paternal substance abuse effect on offspring
effects on sperm motility and #, increase risk of miscarriage and preterm birth, incidence of birth defects and low birth weight, risk of neurodevelopmental issues in offspring
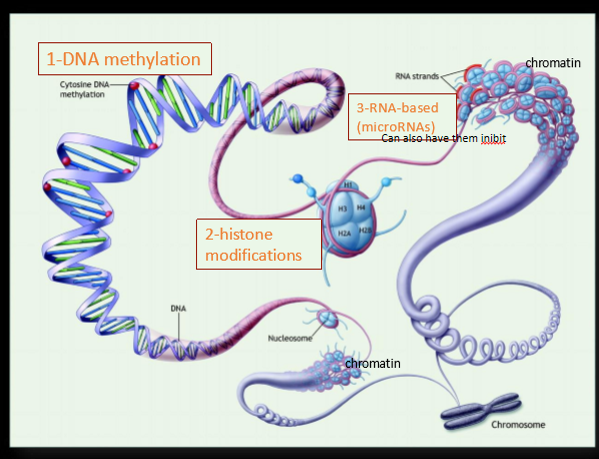
sperm epigenetics
reason we see these changes. process by which phenotypic trait variations that are caused by external or environmental factors result in functionally relevant changes to the genome that do not involve a change i n the nucleotide sequence: DNA methylation → histone modifications
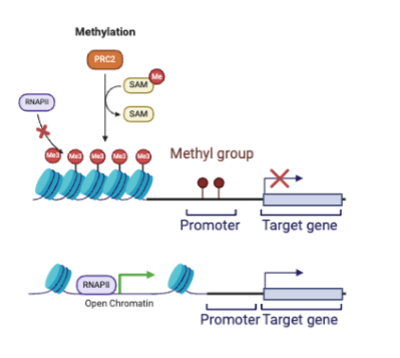
DNA methylation
addition of methyl group. high methylation=low gene expression; low methylation=high gene expression
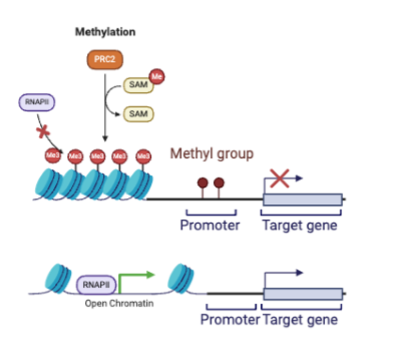
high methylation
low gene expression bc DNA is packed tightly into nucleosomes and cannot be accessed by RNA polymerase or transcription
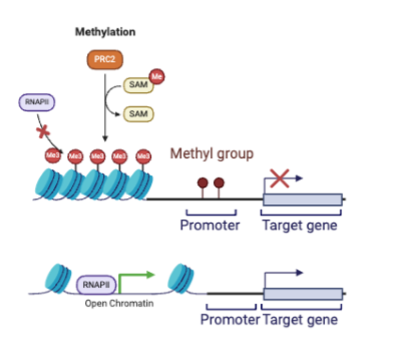
low methylation
high gene expression bc DNA wrapped nucleosomes are spread apart so that RNA polymerase can transcribe the gene
histone modifications
histone are essential for tightly packing sperm DNA into nucleosomes, necessary for reducing nuclear volume, protects the DNA during transit before fertilization. Histone rlly important in sperm bc sperm have very small space to put DNA so histone rlly important in how sperm transmit and transport DNA. this is usually what happens when sperm changes
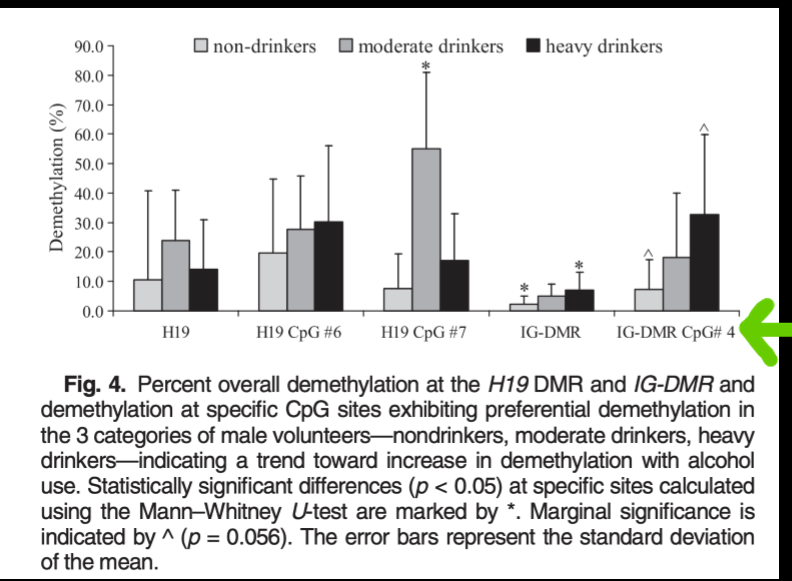
paternal alcohol intake
alters DNA methylation in sperm in genes that regulate fetal growth
paternal diet on sperm epigenome
alters it: altered sperm methylome, histone methylation, and microRNA profile

how does our diet influence epigenetics
dietary components from our food are funneled into a biochemical pathway (1-c metabolism)that extracts methyl groups and then attaches them to DNA, RNA, and proteins
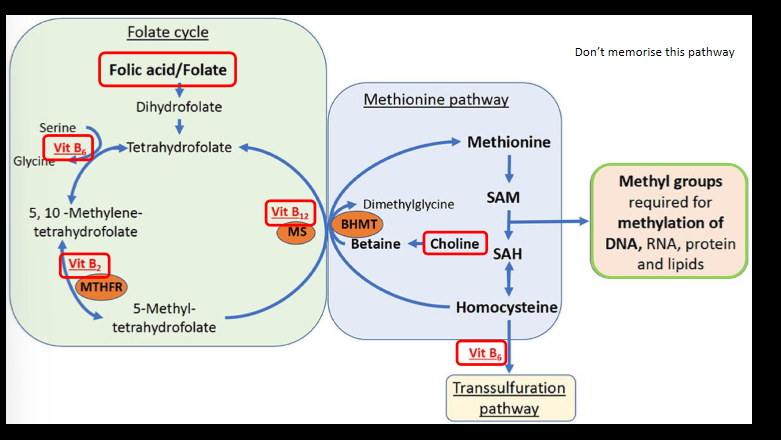
paternal folate effect on fetal growth
low paternal dietary folate alters the mouse sperm epigenome and is associated with -ve pregnancy outcomes. showed how diet affects sperm and how it relates to fetal growth and dvlpmt thru mostly histone changes. 1) differential methylation in sperm of genes implicated in dvlpmt, chronic diseases (cancer, diabetes, autism, schizophrenia); 2) show that epigenetic transmission may involve sperm histone H3 methylation or DNA methylation; 3) paternal folate deficiency increased birth defects in offspring (craniofacial and musculoskeletal malformations)
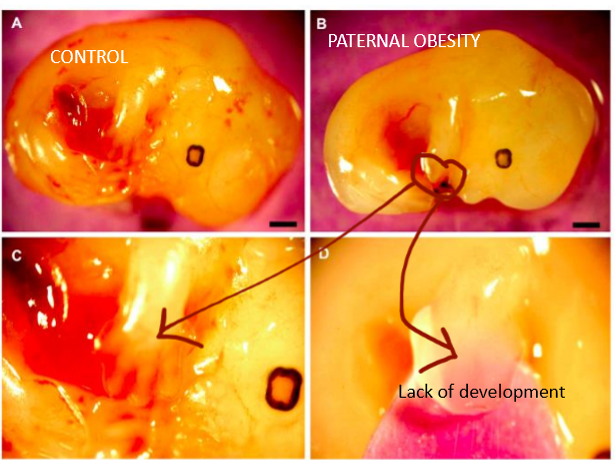
speed of development of fetus in utero when paternal side has diet-induced obesity
slow development, shown is lack of interdigitation
placenta and male lineage
the placenta is an embryonic/fetal tissue; since sperm come from the paternal lineage and contribute to the formation of the fetus and placenta is fetal tissue, sperm contribute to the placenta, so paternal factors lead to placental factors
seminal fluid on embryo
seminal fluid equally important for development of uterine environment, stimulation of uterine immune cells by seminal fluid leads to immune control
sperm epigenome on placenta
carries DNA methylation marks and histone modifications that influence early placental cell differentiation and placental formation
paternal miRNA and ncRNAs on fetal dvlpmt
delivered by sperm, affect pathways involved in placental angiogenesis, immune function, and nutrient transport
sperm-derived epigenetic marks on fetus
affect imprinted genes; paternally inherited imprinted genes (like IGF2, H19) play a crucial role in placental growth and nutrient exchange
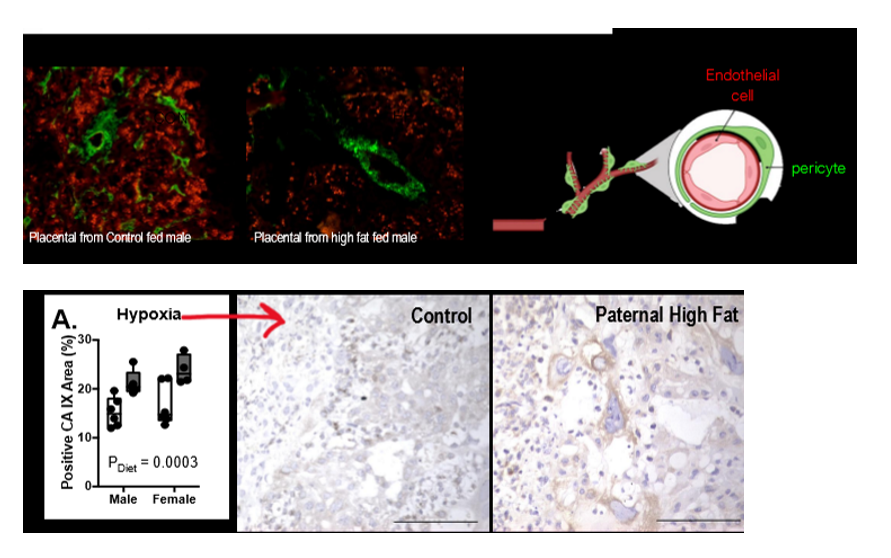
results on Jazwiec lab on paternal obesity inducing placental changes
induces placental hypoxia and sex-specific impairments in placental vascularization and offspring metabolism. paternal hf diet induced obesity contributes to poor vessel integrity (lack of pericytes) in the placenta. paternal obesity → bad placental vasculature → hypoxia. bad bc then fetus doesn’t get its oxygen to live and grow
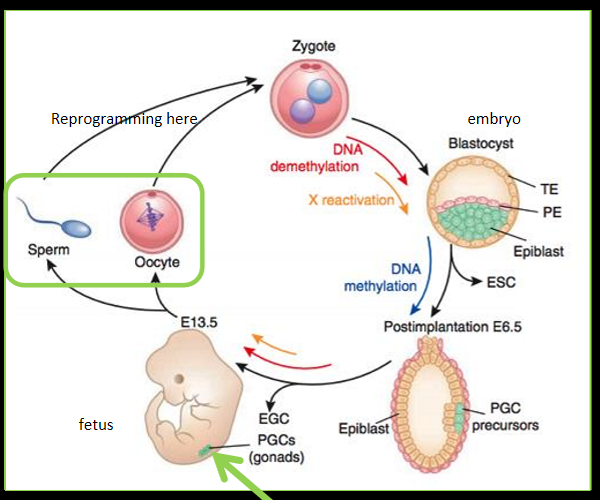
transgenerational vulnerability and epigenetics
epigenetics modifications regulate cell destiny, important in primordial germ cells (sperm and oocyte), since germ cells develop in utero = early life factors can alter fetal gonadal development = changes in primordial germ cells = transgenerational impacts
what determines out health and disease risk
preconception health, early life, diet, physical activity, disease risk
why do we need to disrupt the vicious cycle of obesity
1) modern diets involve the overconsumption of foods rich in lipids and carbs, and this positive energy balance is associated with the development of obesity and metabolic disease
IF
intermittent fasting; a dietary strategy that alternates bw periods of fasting and eating, IF has shown beneficial effects on metabolism in humans and mice: reducing body weight, improving insulin sensitivity, improved plasma cholesterol and blood pressure
types of IF
ADF, 5:2 diet, and TRE
ADF
alternate day fasting.

5:2 diet
type of IF.
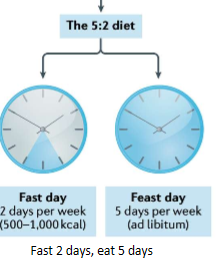
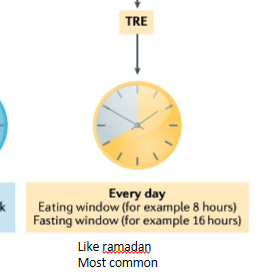
TRE
time-restricted eating
wolves and IF
wolves can go for several days or weeks without eating, and will then consume massive amts of meat in a single sitting (called feast famine cycling); a single wolf can eat up to 20-22 pounds of meat in one meal (roughly 20% body weight)
euglycemia
the normal amount of glucose in the body
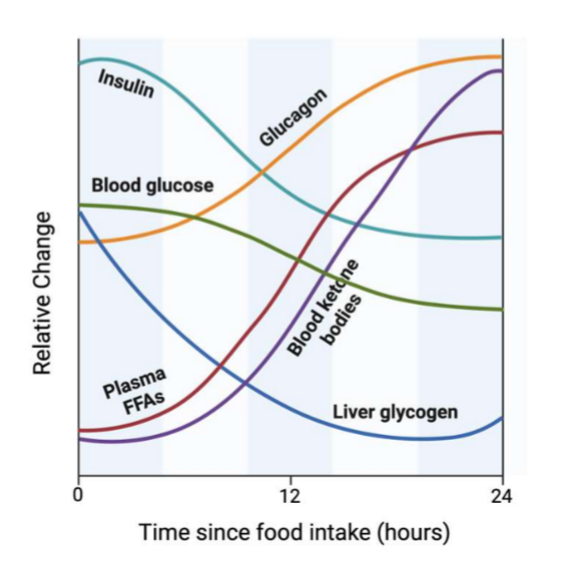
euglycemia during fasting
there is a transition in the relative use of diff substrates to maintain it: 1) glycogenolysis (liver and muscle); 2) lipolysis (WAT, FAs converted to ketone bodies via beta ox, and glycerol converted to gluconeogenic substrate); 3) proteolysis (muscle proteins converted to AA for liver gluconegenesis) sort of memorzie graph. liver-derived, glucose-based energy → adipose-derived, FA based energy
fasting and blood glucose
fasting alters cellular energy status, triggering adaptive metabolic responses. it reduces bg and insulin levels, increasing energetic stress at cell level; decreasing ATP levels in cell and activate AMPK (energy sensor). cells shift from growth and synthesis → maintenance and repair
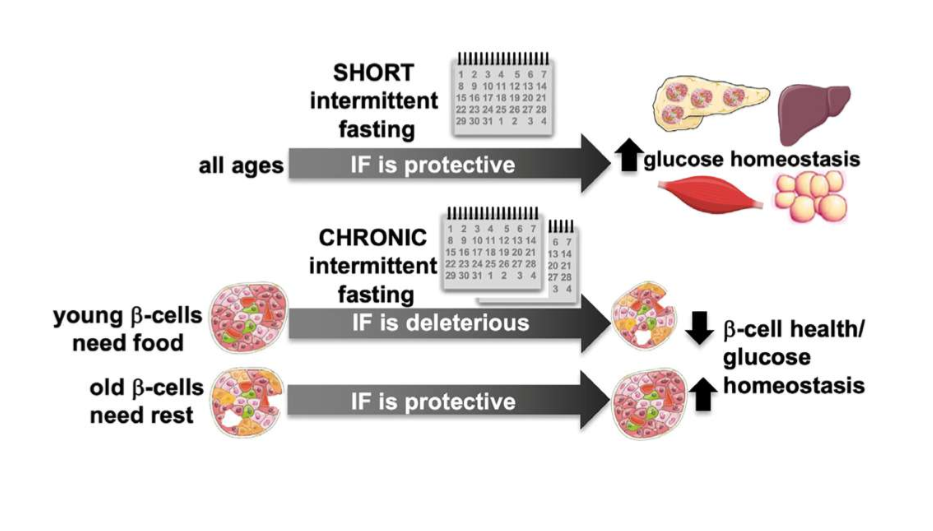
what is currently known about mice and IF
alternating bw 2 fasting day and 2 feeding days improves glucose homeostasis, and this is largely independent of changes in body weight (ability for glucose homeostasis to improve where there isn’t weight loss) IF improves metabolic and islet dysfunction obs in t2d which is characterized by insulin resistance, beta cell dysfunction, and hyperglycemia
what is currently unknown about IF and mice
very little research has been conducted on IF and t1d (not metabolic, autoimmune). beta cells have limited regenerative capacity, resulting in beta cell atrophy, reduced insulin content, and highly inflamed islets in t1d
what journal 3 hoped to prove
if IF can help w t1d, and if it can help, how young and how old
grouping in experiment
male mice split into 3 groups= young (2 mos), middle-aged (8 mos), and old (18 mos) mice did short term (5wk) or long-term (10wk) IF.
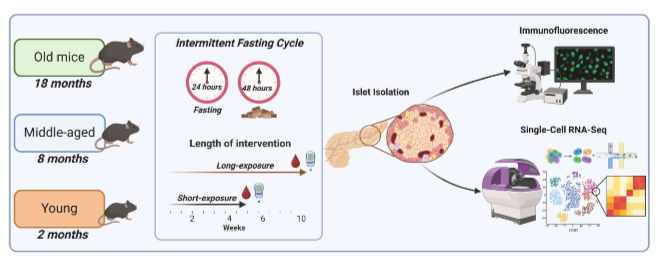
type of IFs measured in mice
half of mice followed a 1:2 IF protocol, which consists of 24 hr of food starvation and then 48 hr free access to food. mice in control group had unrestricted access to food (ad libitum)

glucose tolerance test
for journal 3, they did a GTT which measures how well the body handles glucose. if bg levels remain high throughout the test, it indicates that the body cannot process and/or store glucose efficiently. 1) fasting. 2) baseline-fasting blood sample taken. 3) glucose administration. 4) monitoring- blood is taken at intervals
GTT vs ITT
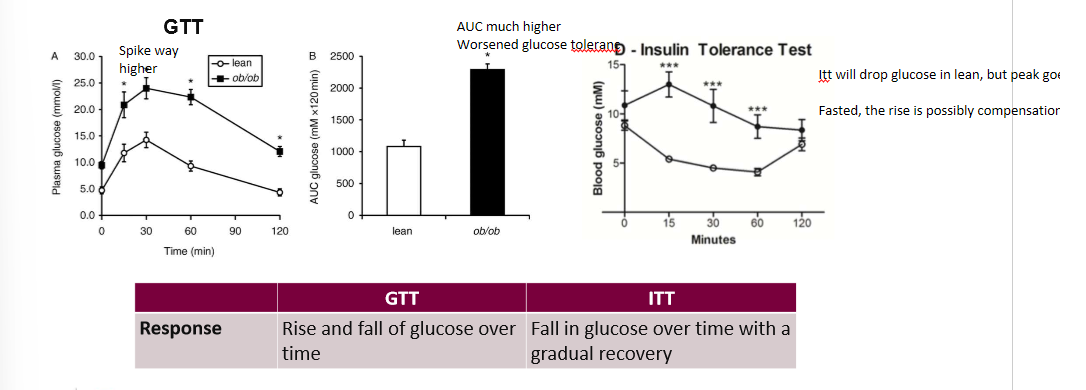
results of GTT and ITT in journal 3
ob/ob mice showed much higher spikes on GTT and musch higher spike in ITT
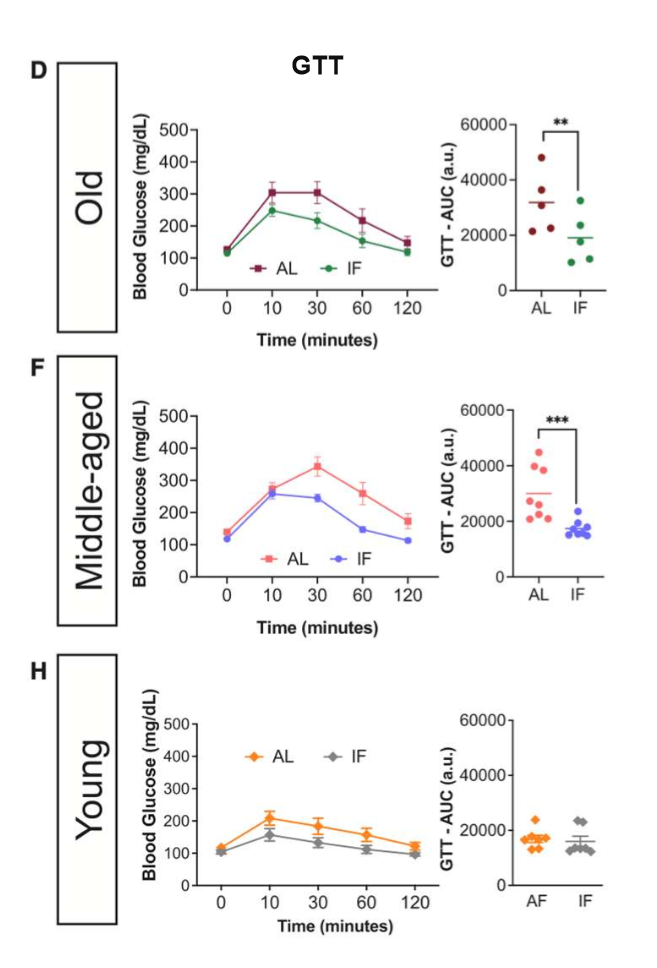
glucose tolerance results in journal 3
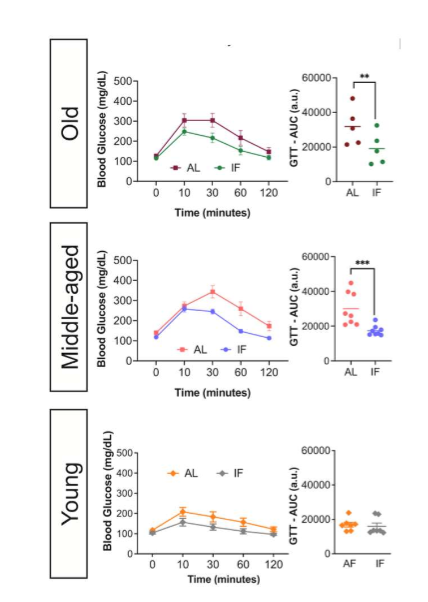
in old and middle-aged mice subjected to LT IF, glucose tolerance improved; glucose tolerance of young mice did not differ bw the LT IF and control groups
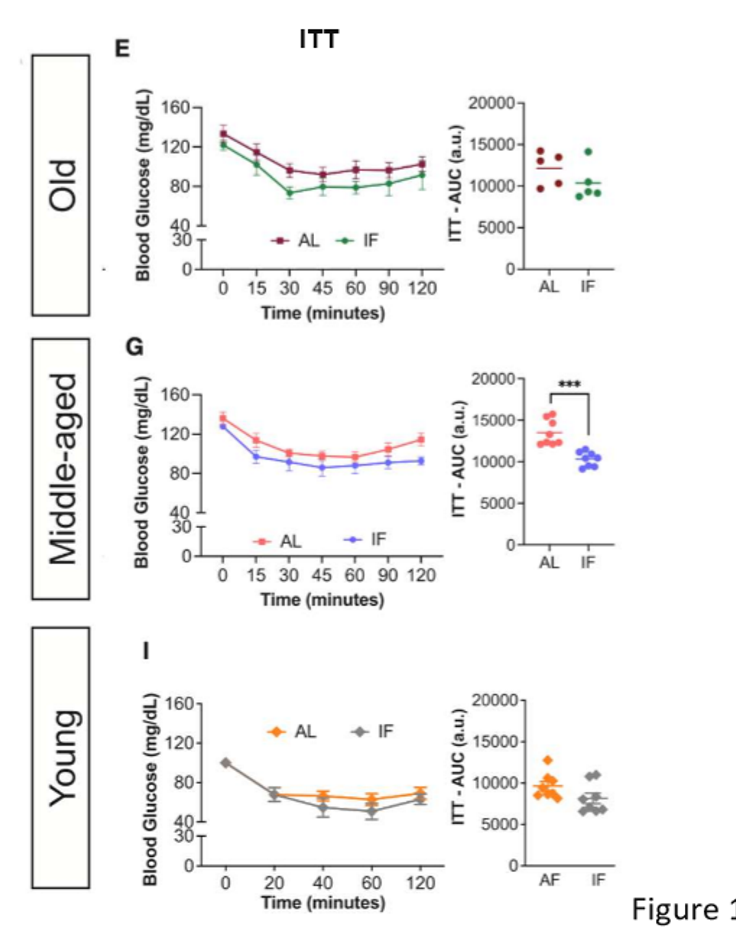
insulin tolerance results in journal 3
in old mice subjected to LT IF, insulin tolerance did not differ; in middle-aged mice, LT OF improved insulin tolerance; ITTs of young mice did not differ bw the LT IF and control groups
glucose homeostasis conclusion of journal 3
LT IF improves glucose homeostasis in middle-aged and old mice but not in young mice. scientists then wanted to know if this is bc pancreatic beta cells behave diff w age
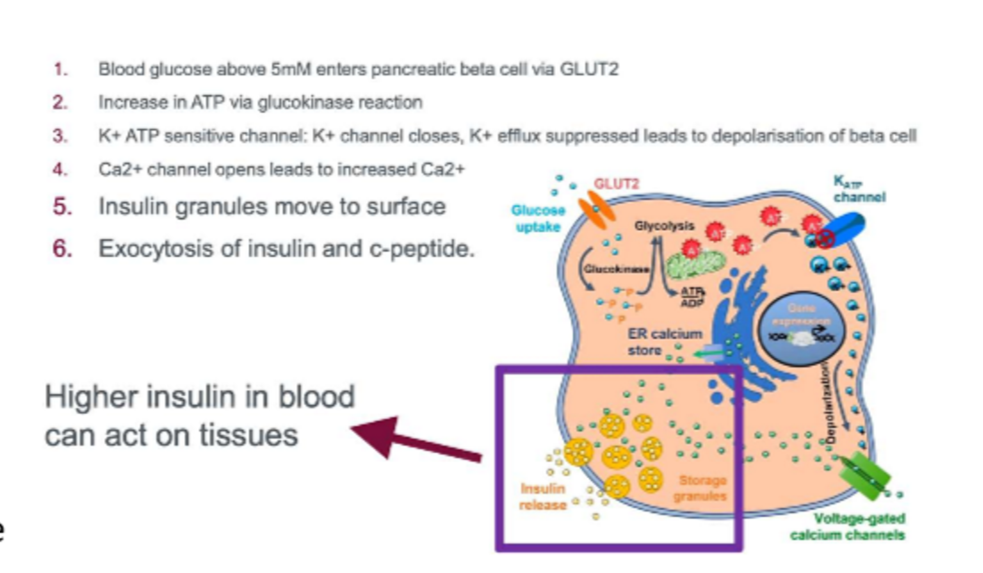
GSIS
glucose stimulating insulin secretion to assess beta cell function. scientists isolated pancreatic islets to do this. islets first placed in low-glucose env to mimic fasting conditions, high glucose is added so the islets release insulin as they would after a meal. KCL is then added to determine max secretion of insulin since it depol the beta cell membrane, causing Ca influx that riggers insulin release
results of GSIS in journal 3
at LG, islets from the LT IF and control groups secreted similar insulin levels; LT IF improved GSIS in old mice in HG conditions, but had no effect in middle-aged mice; in young mice, LT IF reduced GSIS in HG conditions compared to control feeding
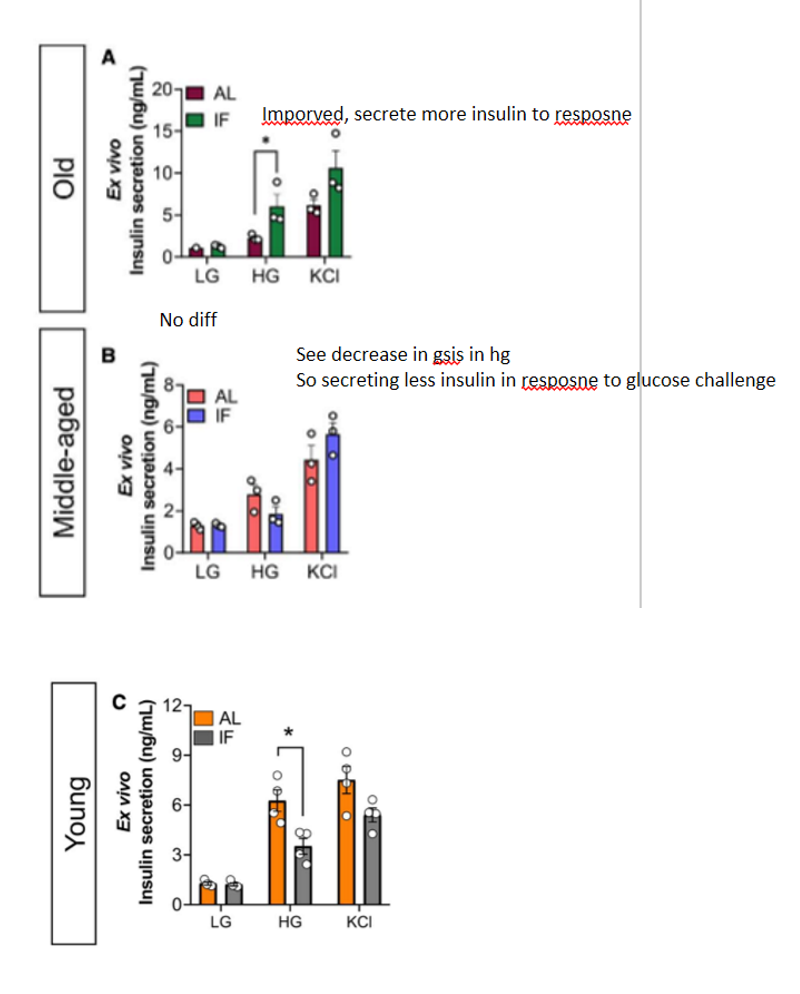
islet quantity results in journal 3
unchanged in old mice (more insulin content in islets of old mice that underwent IF); decreased in middle-aged and young mice compared to control, also less insulin content in islets of young mice that underwent IF
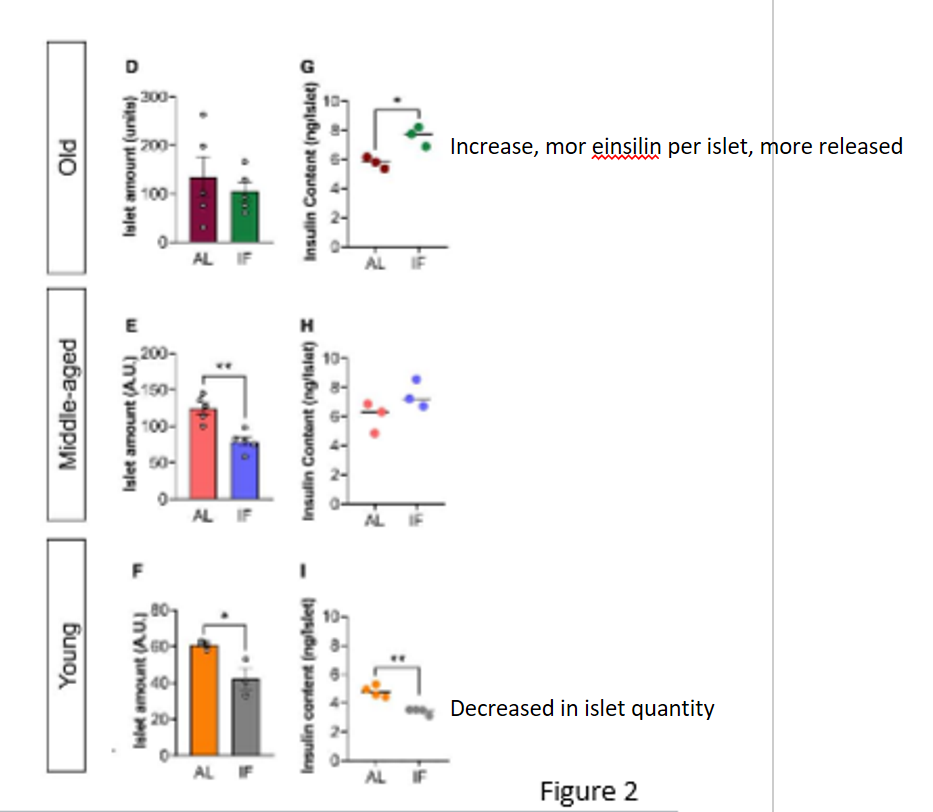
conclusion on mouse islet functions in journal 3
LT IF improved ex vivo islet function in old mice and impaired islet function in young mice
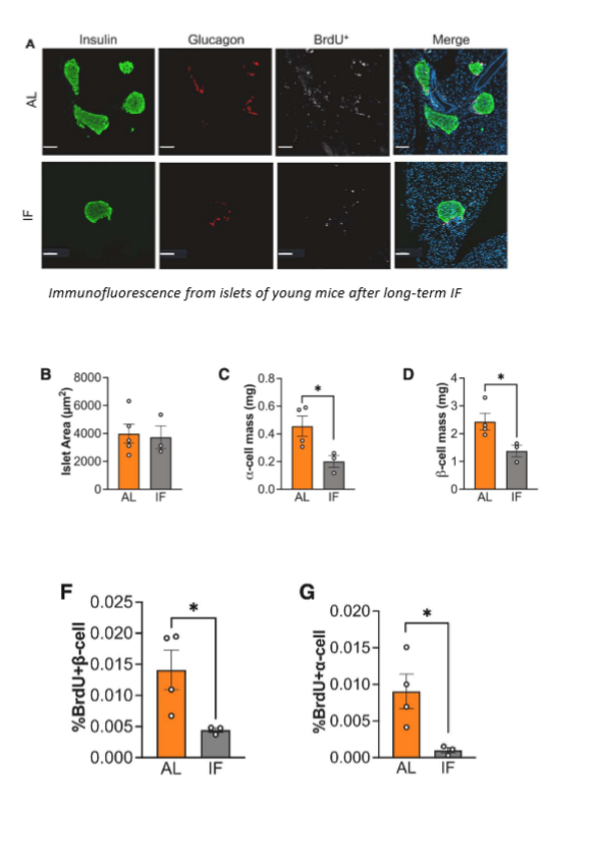
immunofluorescence imaging of pancreatic iselts
mice were injected w BrdU for 5 days after 10 wks of IF (used to detect cell prolifiration); dissected pancreas was fixed and sectioned; primary antibodies targeting insulin and glucagon were used, w secondary antibodies for detection; tissue sections were imaged
results of immunofluorescence imaging of pancreatic islets
in young mice, LT IF was associated w less BrdU+ labelling in insulin and glucagon +ve cells, pointing to reduced proliferation of alpha and beta cells. in young mice that underwent IF, islets had less proliferation and total numbers of alpha and beta cells, suggesting that both cell types need constant access to nutrients to maintain proper survival and fucniton
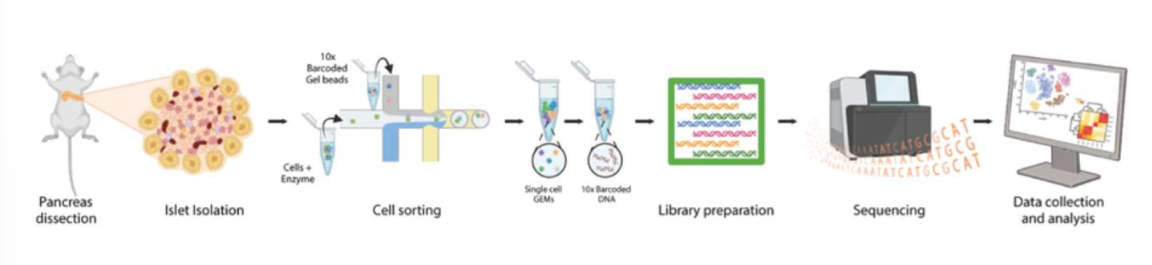
scRNA-seq as a method for journal 3
thhe researchers peformed this of isolated islets from the three ages after LT IF. this technique analyzes gene expression at the single cell level (useful for identifying diff cell types, understanding developmental processes, and studying disease mechanisms
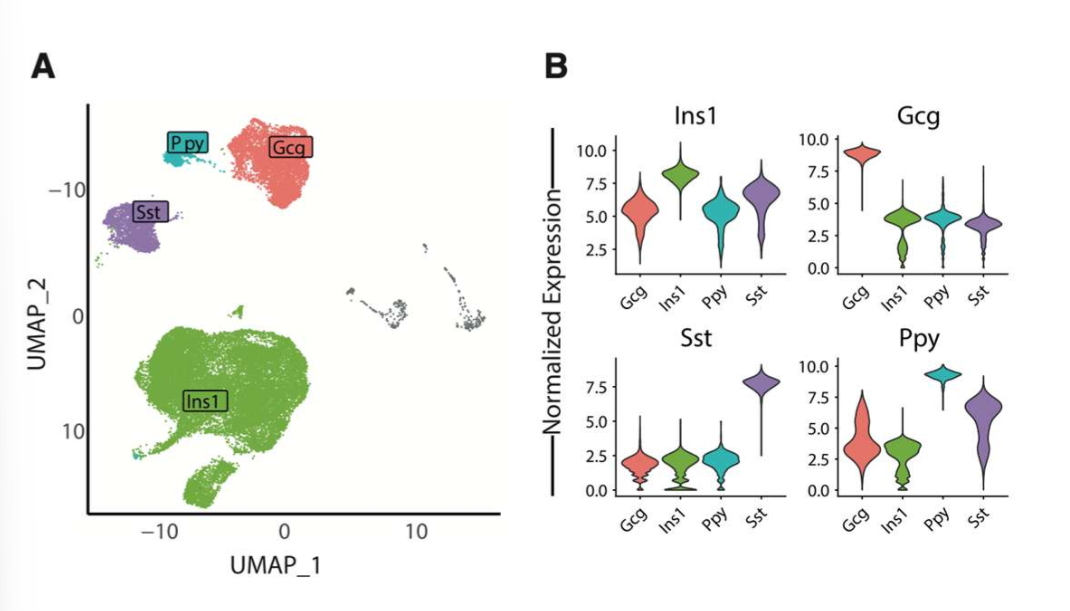
known marker genes for endocrine cell types found in journal 3
gcg: glucagon found in alpha cells; Ins1: insulin found in beta cells; Ppy: pancreatic polypeptide; Sst: somatostatin in delta cells
bulk RNA-seq
measures the average gene expression level across a pop of cells or tissues, rather than individual cells. combines RNA from all cells into a single measurement
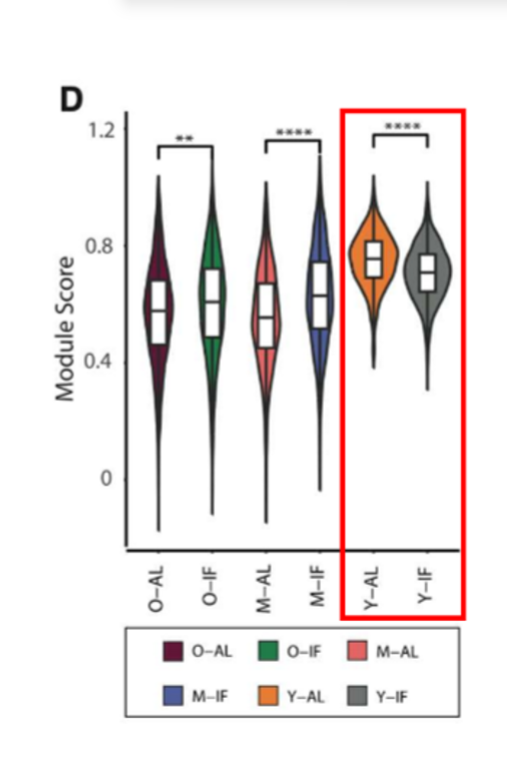
beta cell maturation results of journal 3
analysis focused on beta cells to determine age and IF dependent transcriptomic responses. the most significant diff bw control and LT IF groups was the beta cell maturation gene set; in old and middle aged mice, beta cell maturation scores were higher after LT IF but they were lower in young mice compared to controls (not grown enough)
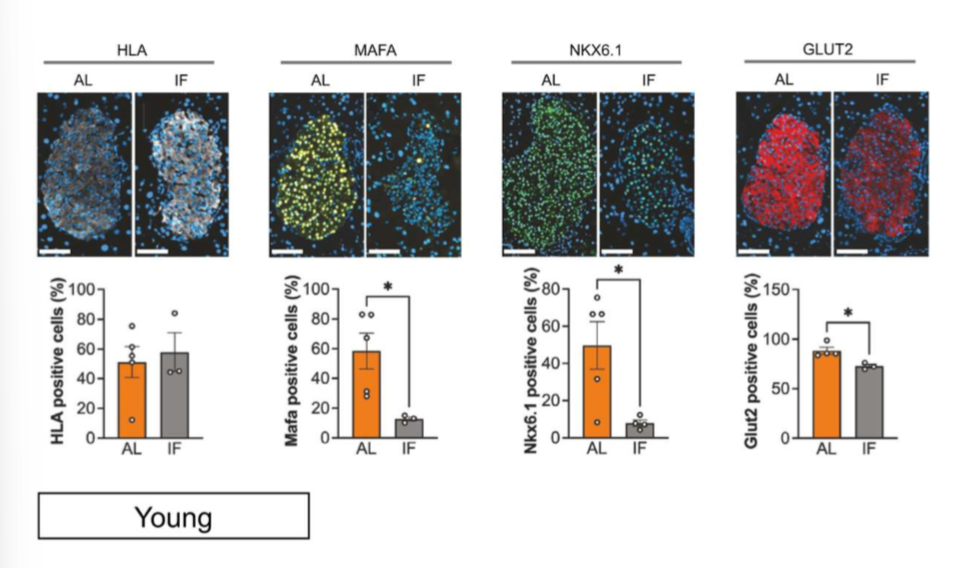
results of key markers of glucose transport, and insulin synthesis and secretion
the authors measured if transcriptional changes translated into pt levels by histoimmunofluorescence (ABs tagged w fluorescent dyes).
results of scRNA-seq in journal 3
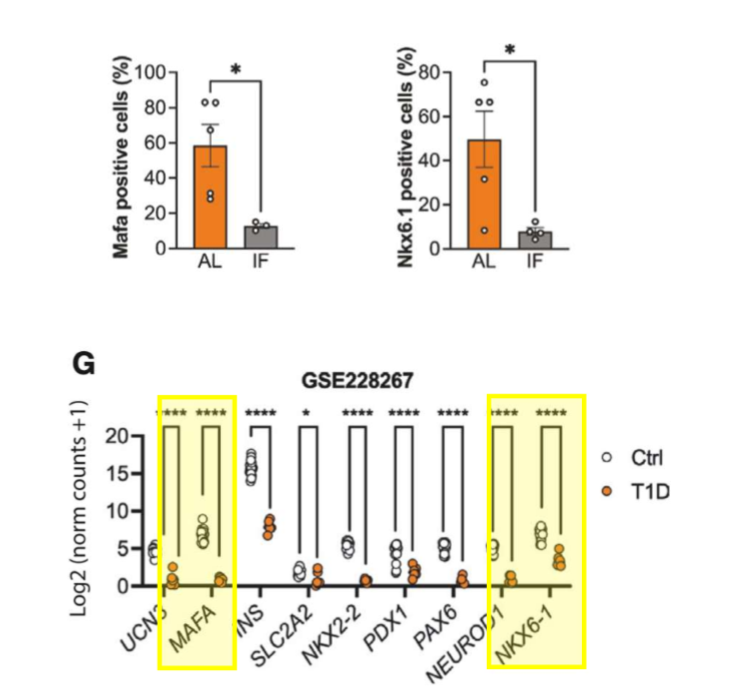
scRNA-seq reveals impaired beta cell maturations and development in young mice. they compared their findings to bulk RNA-seq datasets from human islets of donors w t1d and t2d; the genes downregulated in the young mice that did LT IF showed a similar pattern of downreg in smaples from pts w t1d but not those w t2d
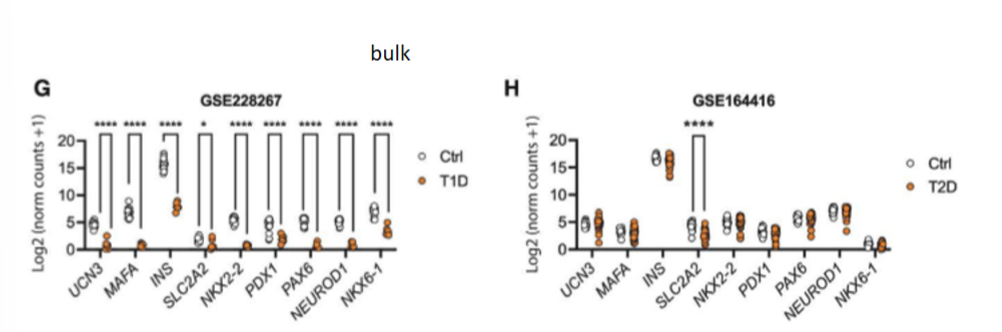
key idea from results of journal 3
scRNA-seq analysis demonstrated impaired beta cell maturation and development in young IF mice w similar pattern of transcriptional downreg to that seen in t1d pts
ST IF and glucose homeostasis
improves glucose homeostasis without altering islet function across all age groups
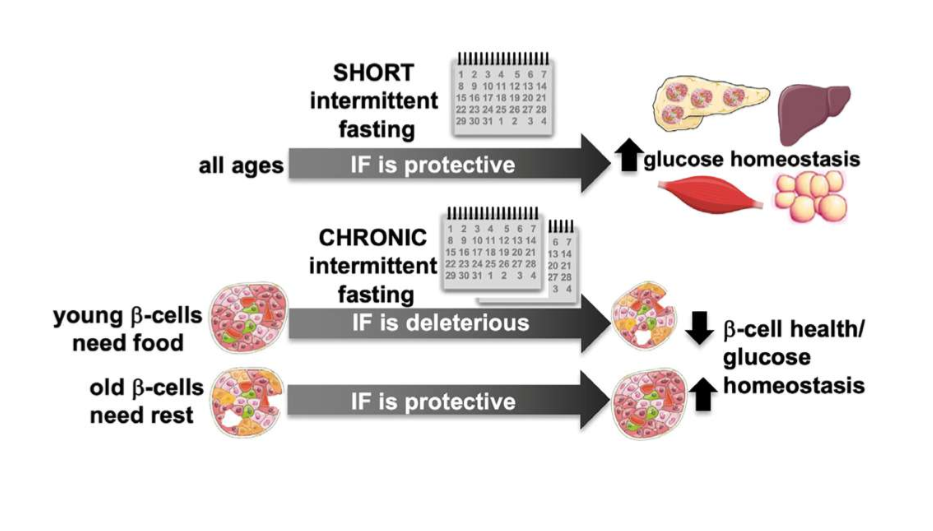
LT IF and glucose tolerance and beta cell function
improved glucose tolerance and beta cell function in old mice, but impaired beta cell function and maturation in young mice