AQA Chemistry 2: Bonding, structure and the properties of matter
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What are the three types of strong chemical bonds?
ionic, covalent, metallic
What is ionic bonding?
Particles are oppositely charged ions
Form with a metal and a non metal
Electrons are transferred
Metals lose electrons and become positively charged
Non-metals gain electrons and become negatively charged
What is an ionic compound and how are they held together?
An ionic compound is a giant structure of ions.
They are held together by strong electrostatic forces of attraction between oppositely charged ions.
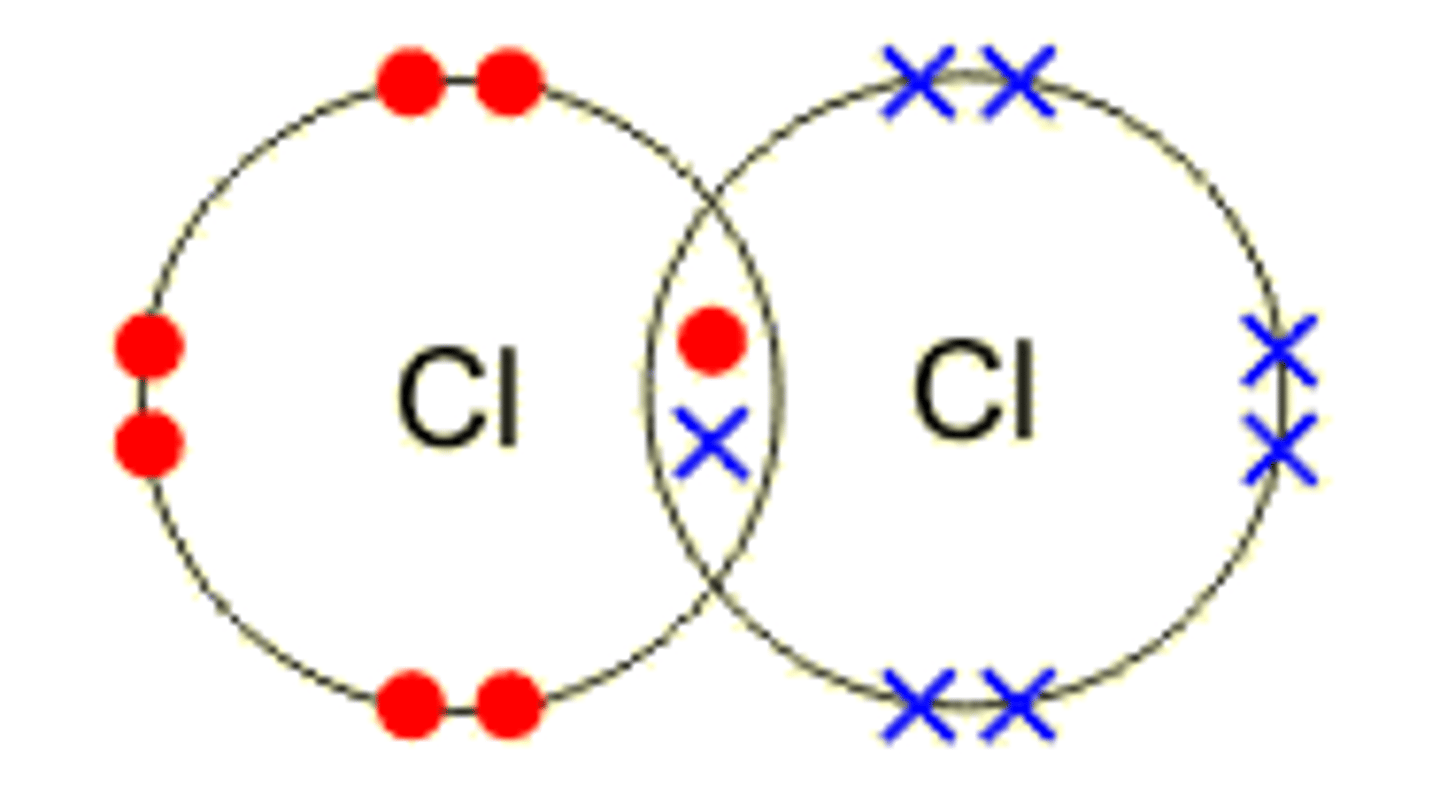
What are the advantages and disadvantages of using dot and cross diagrams?
+
Useful for illustrating electron transfer
Shows which atom the electrons came from
-
Does not show the 3D arrangement
Does not show relative sizes of the atoms
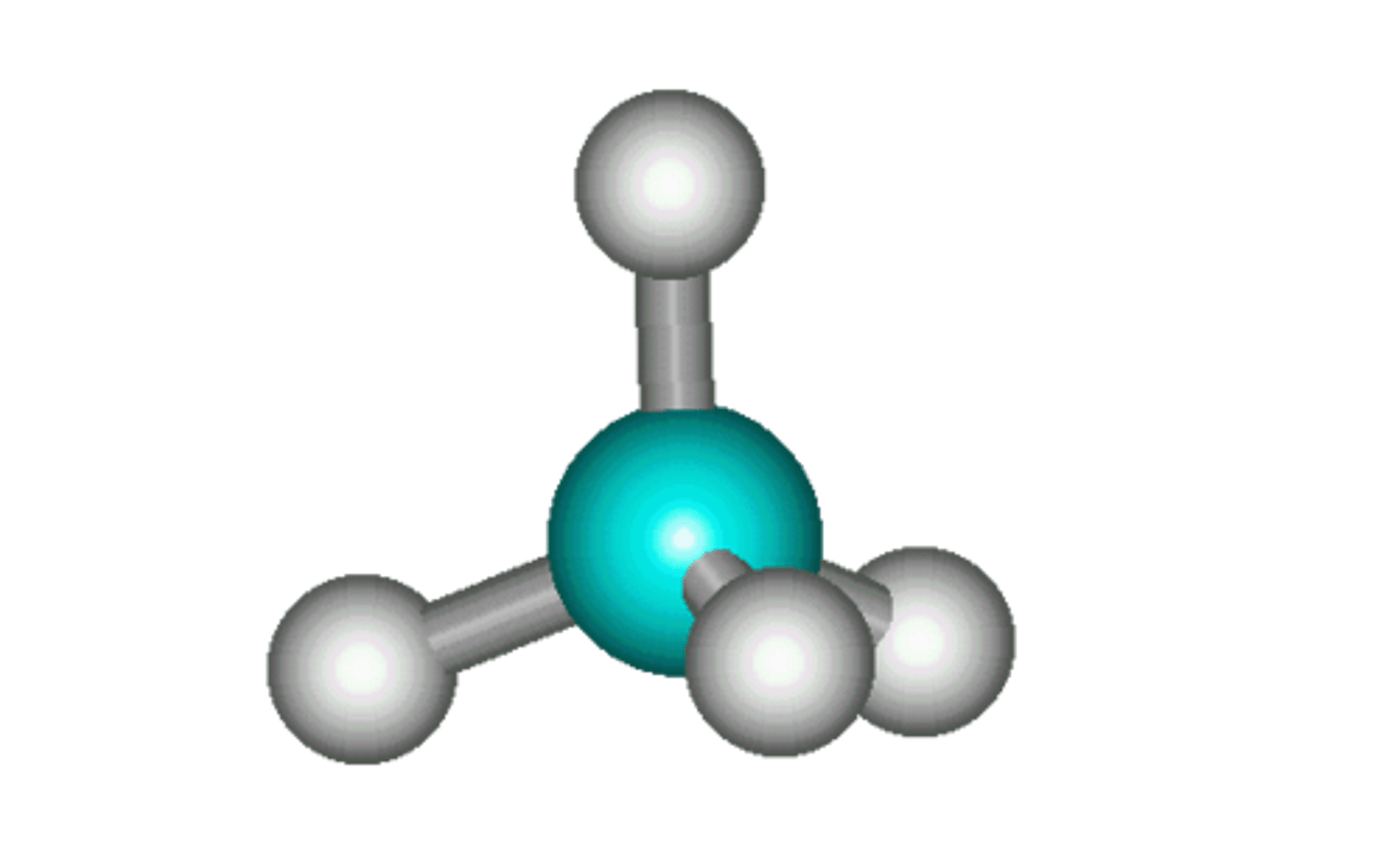
What are the advantages and disadvantages of using ball and stick diagrams?
+
Useful for showing atoms in 3D
Useful for showing shape
-
Does not show movement of electrons
Shows atoms as far apart (with 'sticks'), this is not really the case, atoms are closer together
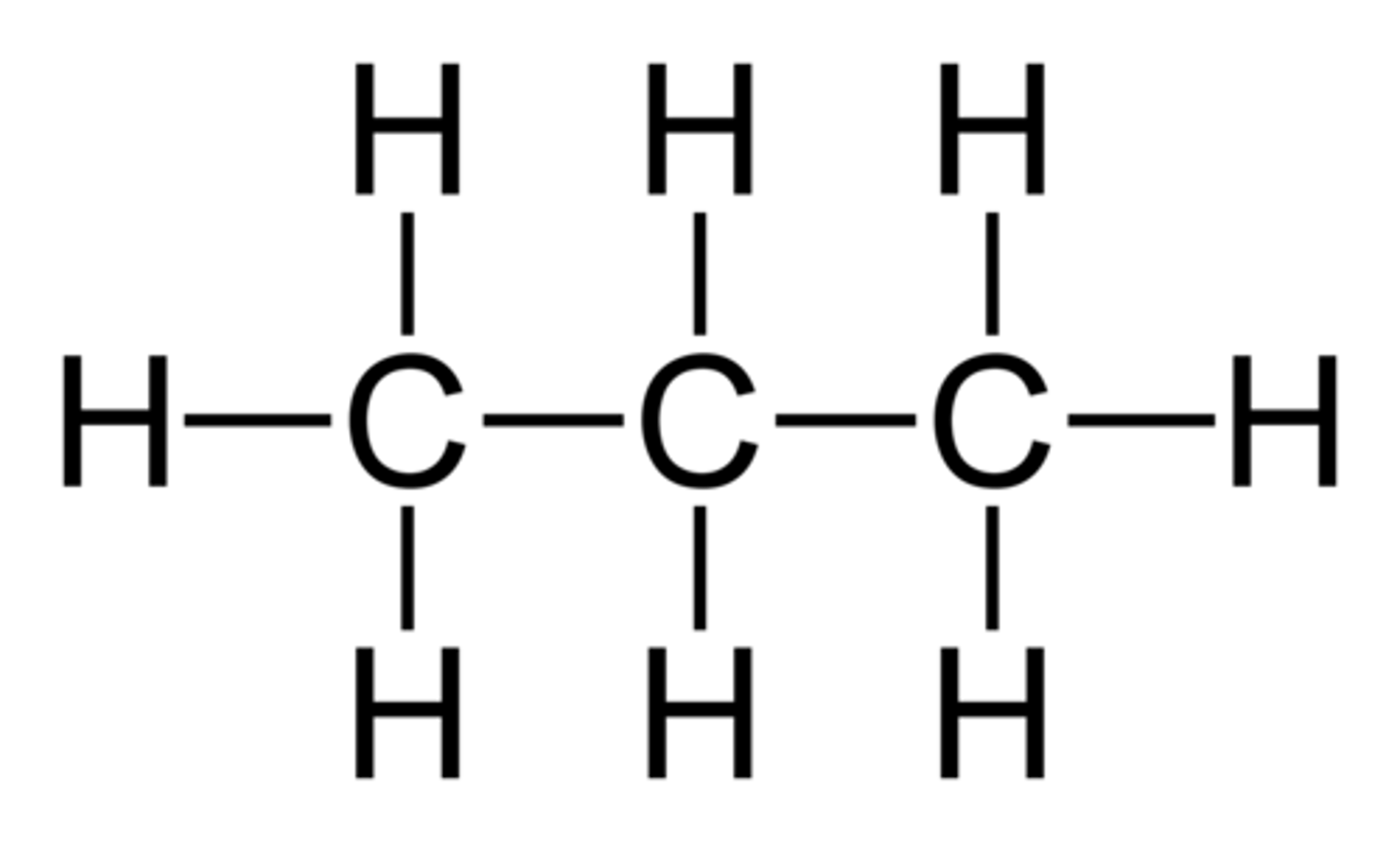
What are the advantages and disadvantages of using 2D representations of molecules?
+
Show which atoms are in a molecule and how they connect
-
Do not show relative sizes of atoms and bonds
Does not show what the atoms are like in 3D
What are the advantages and disadvantages of using 3D representations of ionic solids?
+
Show the arrangement of ions in space
Show the repeating structure
-
Only show the outer layer
Time consuming and difficult to draw

What is covalent bonding?
Particles are atoms that share pairs of electrons
Form between non metals
Electrons are shared (Sharing is Caring... Caring... Covalent)
Mainly small molecules
Some are large - eg polymers
Some have giant covalent structures, eg diamond, silicon dioxide
What is metallic bonding?
Particles are atoms which share delocalised electrons
Form in metallic elements and alloys
Delocalised electrons are free to move through the whole structure, carrying charge
What are the three states of matter?
Solid, liquid, gas
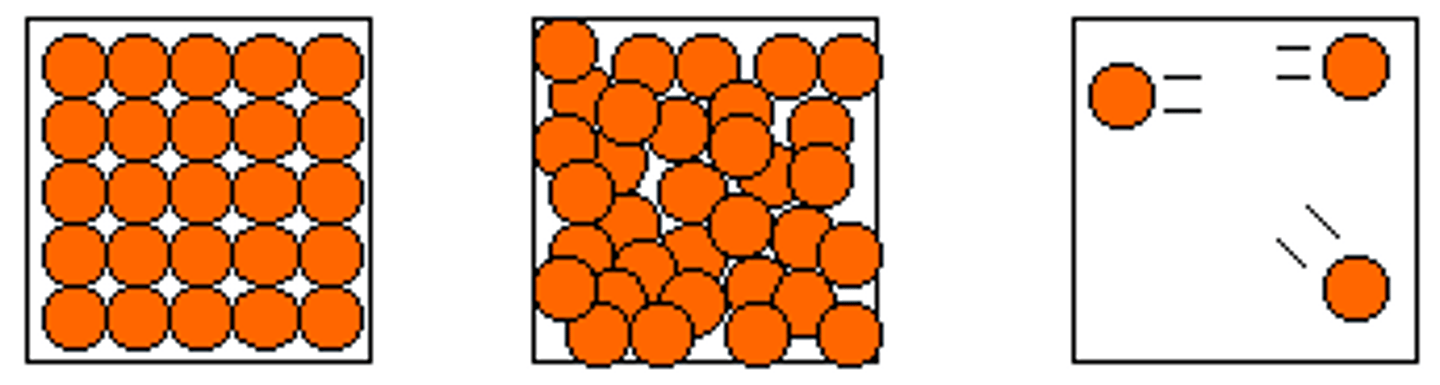
How can the particle model be represented as diagrams?
The amount of energy needed to change state from solid to liquid and from liquid to gas depends on...
... the strength of the forces between the particles of the substance.
The stronger the forces between the particles the higher...
... the melting and boiling point
[HT only] What are the limitations of the particle model?
-There are no forces represented
-All particles are represented as spheres
-The spheres are solid
What are the 4 state symbols?
s (solid), l (liquid), g (gaseous), aq (aqueous)
What are the properties of ionic compounds?
- Regular lattice structure
- Very strong electrostatic forces of attraction between ions in all directions
- High melting and boiling points due to large amounts of energy needed to break the strong bonds
- Don't conduct electricity because the ions are fixed in place.
->When molten or dissolved in water, they do as the ions are free to move, so charge can flow
What are the properties of small molecules?
-Usually gases or liquids
-Relatively low melting points and boiling points.
-Weak forces between the molecules (intermolecular forces). It is these intermolecular forces that are overcome, not the covalent bonds, when the substance melts or boils.
-The intermolecular forces increase with the size of the molecules, so larger molecules have higher melting and boiling points.
-These substances do not conduct electricity because the molecules do not have an overall electric charge.
What is a polymer?
A large molecule made of many monomers
Atoms are linked by strong covalent bonds
Strong intermolecular forces
Solids at room temperature
What are giant covalent structures?
Solids with very high melting points
Atoms are linked by strong covalent bonds
Bonds must be overcome to melt or boil
Diamond, graphite, silicon dioxide
What are the properties of metals?
Giant structures of atoms with strong metallic bonding
(most have) high melting and boiling points
Pure metals - atoms in layers, metals can be bent and shaped
Pure metals - too soft
What is an alloy?
A mixture of two or more elements, of which at least one is a metal
Makes the metal harder because the other substance distorts the layers
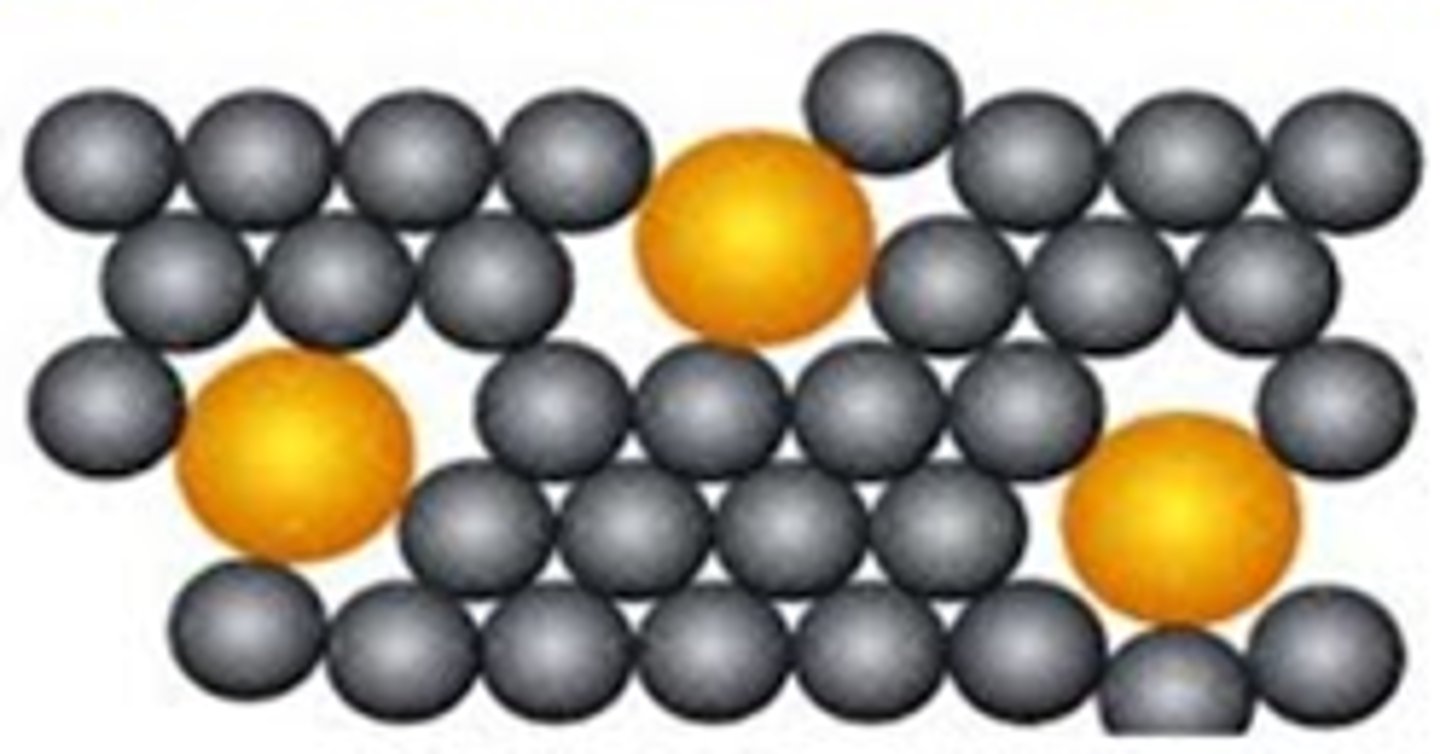
Why are metals good conductors of electricity and thermal energy store?
They have delocalised electrons that carry electrical charge
Energy is transferred by the delocalised electrons
What is the structure and bonding of diamond?
-Each carbon atom forms 4 covalent bonds
-Diamond is very hard with a high melting point
-There are no free electrons
-Diamond does not conduct electricity
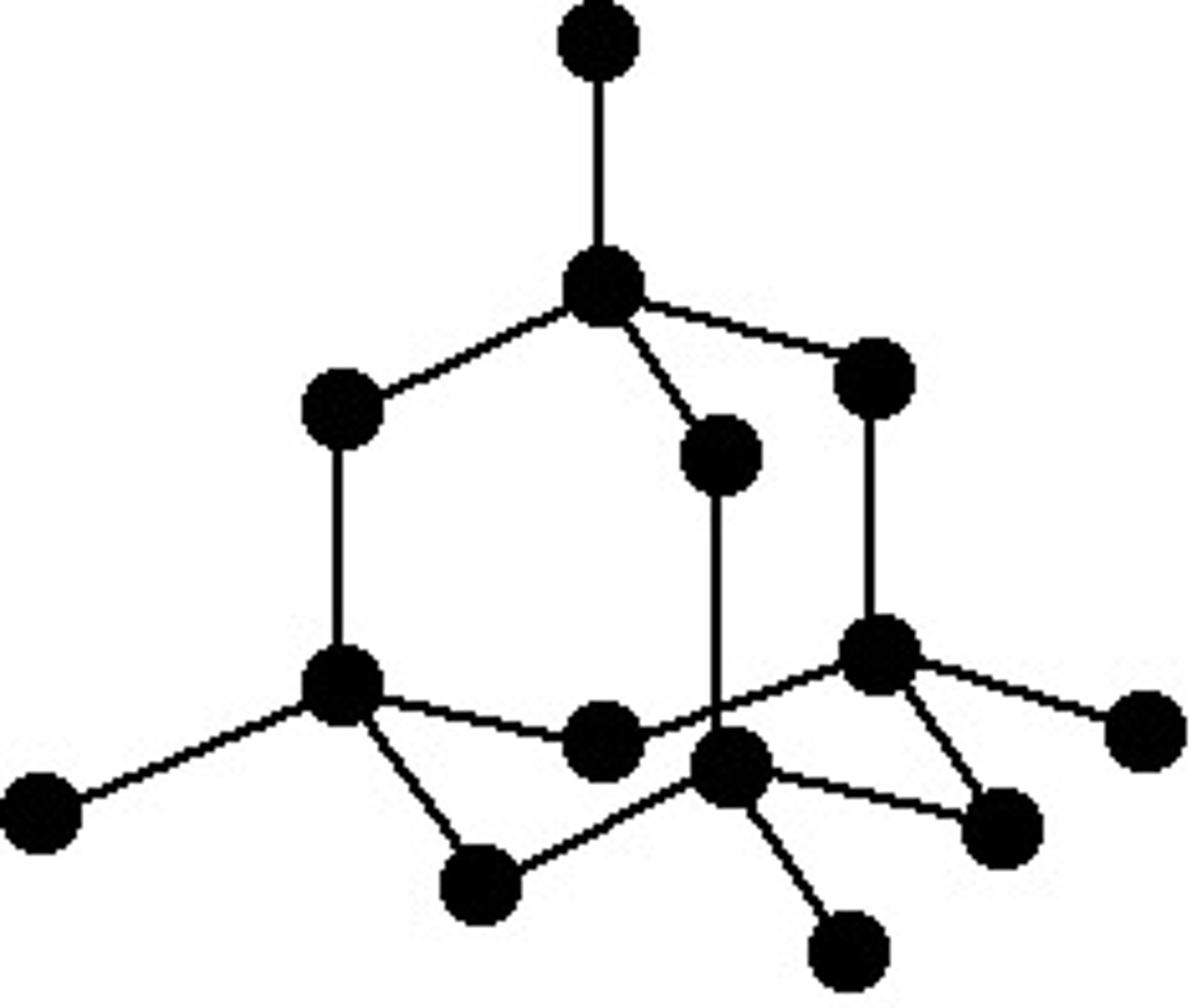
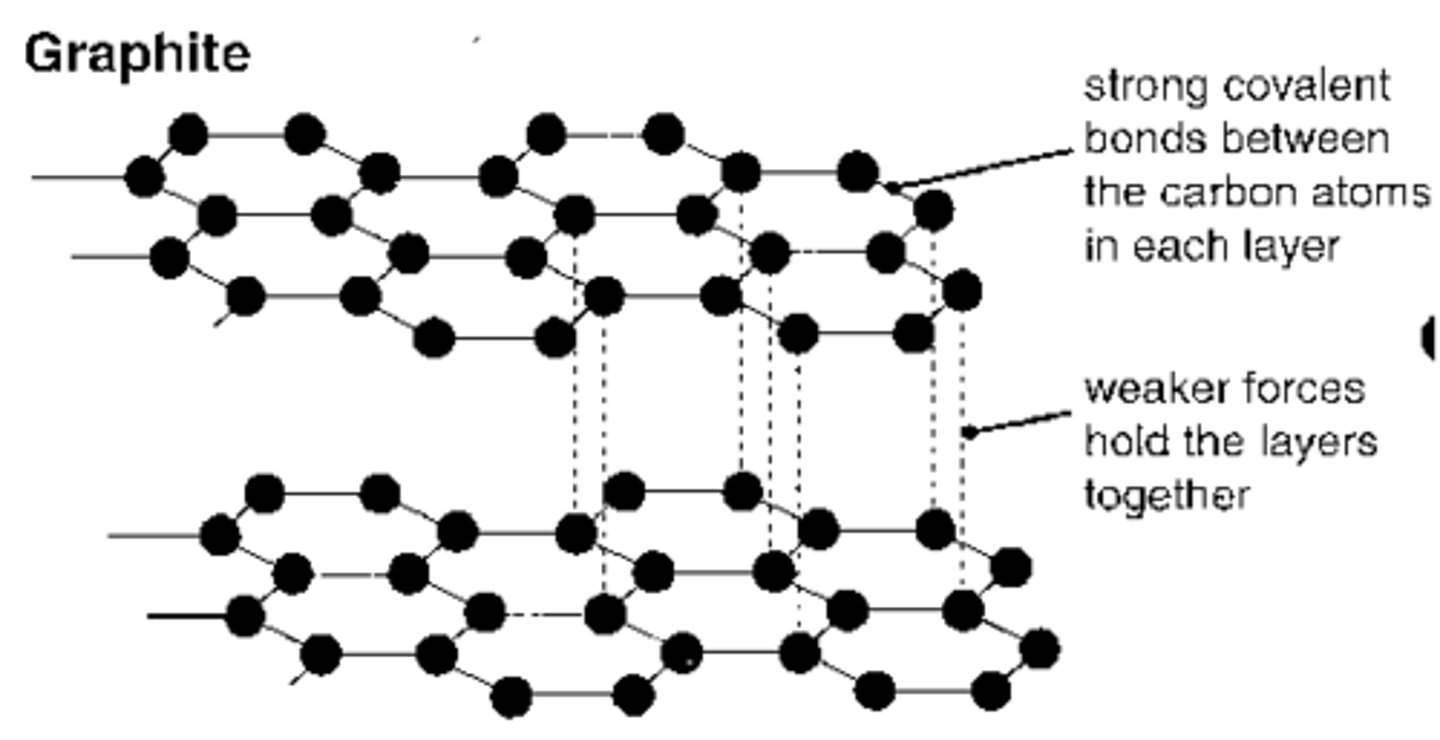
What is the structure and bonding of graphite?
-Each carbon atom forms 3 covalent bonds
-Forms layers of hexagonal rings
-No covalent bonds between layers
-One electron from each atom is delocalised
-Can conduct electricity
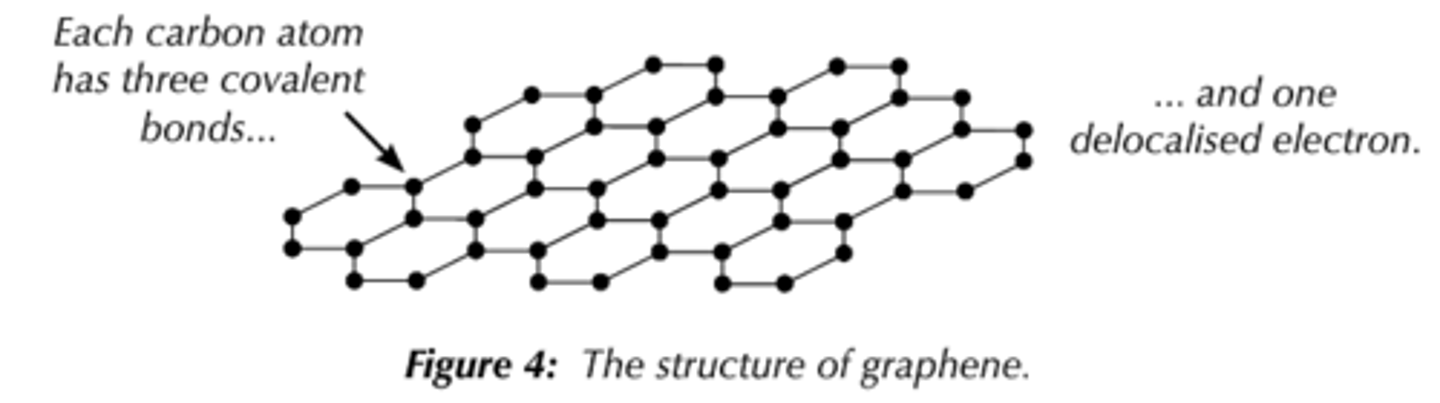
What is the structure and bonding of graphene?
-A single layer of graphite
-Each carbon atom forms 3 covalent bonds
What is the structure and bonding of fullerenes?
-Molecules of carbon atoms with hollow shapes
-Each carbon atom forms 3 covalent bonds
-Structure based on hexagonal rings, or rings of 5 or 7 carbon atoms
-First to be discovered: Buckminsterfullerene (C₆₀)
What is a carbon nanotube?
A cylindrical fullerene
High length to diameter ratio
Useful for nanotechnology, electronics and materials
[Triple only] What is nanoscience, and what are nanoparticles?
Nanoscience relates to structures that are 1-100 nm in size, of the order of a few hundred atoms.
Nanoparticles, are smaller than fine particles, which have diameters between 100 and 2500 nm.
[Triple only] What is the diameter of coarse particles?
Coarse particles - dust
1x10⁻⁵ to 2.5x10⁻⁶
[Triple only] As the side of cube decreases by a factor of 10 the surface area to volume ratio increases by a factor of...
...10
[Triple only] Why do nanoparticles have different properties to those for the same material in bulk?
High surface area to volume ratio
Smaller quantities are needed to be effective than for materials with normal particle sizes
[Triple only] What are some uses of nanoparticles?